Natural Science
Vol.4 No.10(2012), Article ID:23923,6 pages DOI:10.4236/ns.2012.410102
Biodiesel production by hydroesterification of microalgal biomass using heterogeneous catalyst
![]()
1Department of Chemical Engineering, Federal University of Rio de Janeiro (UFRJ), Rio de Janeiro, Brazil; *Corresponding Author: m.m.fortes@hotmail.com
2Research and Development Center “Leopoldo Américo Miguez de Mello” (CENPES), Petrobras, Rio de Janeiro, Brasil.
Received 14 August 2012; revised 15 September 2012; accepted 27 September 2012
Keywords: Biodiesel; Microalgae; Hydroesterification; Biomass; Heterogeneous
ABSTRACT
This paper assesses the hydroesterification process for the production of Biodiesel from Monoraphidium contortum (MORF-1) microalgae biomass, as it is a sustainable alternative not only economically, but also environmentally and ecologically to replace petroleum diesel fuel. The Biodiesel studied in this work was obtained from fatty acid esterification, a product of microalgae and methanol biomass hydrolysis reaction. CBMM’s (HY-340) niobium oxide powder was used as catalyst. The reactions were carried out in a properly closed autoclave reactor (batch), where the reagents were mixed under constant stirring at 500 rpm for hydrolysis and esterification. The products generated were submitted to gas chromatography and oxidative stability analysis. The hydroesterification process showed itself to be a promising alternative to the conventional biodiesel production process (transesterification) as it favors the use of feedstocks with any acidity and moisture content and may be performed with acid catalyst, which favors high conversions in a small range of time (30 minutes).
1. INTRODUCTION
Microalgae are a potential feedstock for biofuel production, especially biodiesel, considering that since the 50s (first mass culture project on the rooftop at MIT, USA), and afterwards officially in the 70s (Aquatic Species Program: Biodiesel from algae, National Renewable Energy Laboratory-NREL. USA) and to this day, work has been constantly done in different countries (USA, Israel, Spain, New Zealand, Australia, Germany, The Netherlands, etc.) to optimize the benefits and minimize the disadvantages associated to the production of biofuels by using microalgae as feedstock.
In this paper, the in situ Monoraphidium contortum (MORF-1) microalgae biomass biodiesel production by the hydroesterification method (hydrolysis followed by esterification) is studied. In addition, a chromatographic approach on the structural variations observed in the products formed is presented.
2. MATERIALS AND METHODS
2.1. Feedstocks
For the production of fatty acids (hydrolysis reaction), a species of freshwater microalgae biomass (Monoraphidium contortum) grown in a photo-bioreactor mounted in the Pilot GREENTEC/School of Chemistry/UFRJ area was used as feedstock; as hydrolasing agent, distilled water.
Biomass harvesting took place in the exponential growth phase by using flocculation, followed by centrifugation (Figure 1).
For the production of biodiesel (esterification reaction), the fatty acids of these microalgae, which were obtained as the biomass hydrolysis product, were used as feedstock. Absolute methyl alcohol, with 99.9% purity, supplied by TEDIA.
2.2. Catalyst-Esterification
The catalyst system used in the esterification reaction was niobium oxide powder (HY-340), which presents 90m2/g surface area, given by CBMM (company dedicated to the extraction, processing, manufacturing and marketing products based on niobium).
2.3. Equipment
All the reactions were performed in batch reactor (Parr Instruments Inc.-4842 Model), autoclave type, made of

Figure 1. Paste resulting from centrifugation.
stainless steel, with 300 mL useful volume and 3.000 psi maximum working pressure. Such reactor has a tube for withdrawing samples, as well as a stirring system and external heating mantle (Figure 2).
The operating conditions used in the reactions are shown in Table 1.
2.4. Dry Weight of Microalgal Biomass
To obtain the dry weight of the centrifuged microalgal biomass, watch-glasses were dried in an Icamo furnace at 105˚C for 1 hour and cooled in a desiccator for 20 minutes. Cellulose nitrate filter paper with 0.8 μm porosity, Stedium Sartorius Biotech brand, was used. The filter was pretreated in the furnace, as directed by the manufacturer. Then, the watch-glass with the paper filter were weighed in a Shimadzu balance, AY220 model, with the aid of tweezers, to avoid hand contact and weighing errors. The watch-glass with the filter was reserved in dessicator up to the moment of the vacuum filtration of the concentration, as shown in Figure 3. The analysis was performed in triplicate.
Filter paper was placed on the vacuum filter holder (a). Unit sealing (b). Addition of 10 mL concentrated medium into the filter (c). Addition of 40 mL MilliQ water for filtration (d). Addition of the filter with the filtered cells mass on the specific watch-glass (e) and furnace drying at 60˚C until constant weight, for the dry weight attainment (f).
2.5. Lipid Content Determination
To perform the quantitative analysis of the lipids, 0.5 g or 500 mg of dry weight cells of the studied microalgae were used, in triplicate. The mass of the sample used in the analysis was estimated from the dry mass of cells obtained as in the previous item.
To perform the extraction of lipids, the sample was submitted to two extraction stages. It was weighed in a centrifuge tube on a digital scale. With the aid of a pipette,

Figure 2. Autoclave reactor.

Figure 3. Simplified scheme to attain dry weight by vacuum filtration.

Table 1. Operating conditions.
40 mL (2:1) of chloroform/methanol [1] were added. The tube was closed, agitated in vortex at 1400 rpm for 20 min, allowed to settle for 30 min and submitted to the a new agitation for 20 min. Next, the sample was centrifuged at 3000 rpm for 15 min at room temperature.
After the centrifugation, the liquid phase of the sample was collected in a beaker whose weight was previously known, which was taken to the furnace at 40˚C for the evaporation of the solvents. The lipid quantitative was estimated by gravimetric analysis, relating the dry mass cells with the obtained lipid dry weight.
2.6. Lipid Profile of the Microalgal Biomass
To perform the lipid profile analysis, the sample obtained during the lipid content determination (as in theprevious item) was submitted to methanolysis reaction, according to the described methodology [2], with modifications.
300 mg of the lipid sample were used. Then, saponification was carried out with 1 mL of KOH-CH3OH (alcoholic potash) saturated solution at 75˚C for 10 minutes in Nova Ética’s Water-bath. Afterwards, the sample was submitted to methanolysis with 2 ml methanol with 5% HCl at 75˚C for 10 minutes. Such stages were performed in closed flask to avoid evaporation. Subsequently, the phase containing the fatty acids was set apart with the addition of 2 ml distilled water and 2 mL PA hexane. The upper phase of the hexane was collected with an automatic pipette and transferred to a flask that was placed in a furnace at 60˚C for evaporation of the solvent and sample concentration.
The dried and reacted lipid sample had around 150 mg. 300 μL P.A. heptane was added to it. The sample was manually shaken until it was homogeneous; then, it was eluted by gas chromatography (GC).
Analyses were performed in a Shimadzu GC, model 2014, with Split injector at 250˚C, with a 20 mL/min flow, FID detector at 250˚C and isothermal oven at 200˚C. A Quadrex Carbowax 20 M capillary column, with polyethylene glycol stationary phase and 30 m × 0.32 mm × 0.25 μm dimensions was used.
The lipid profile was identified by comparison with the standard sample and determined by the integrated areas of each peak.
2.7. Acids Composition
Determination-Hydrolysis
The products obtained by hydrolysis reactions were analyzed according to the fatty acids composition of the sample. To identify the fatty acids, different fatty acid patterns were injected to identify the peaks areas and retention times. Heptadenoic acid was used as internal standard.
This method was developed, with the purpose of predicting both qualitatively and quantitatively, by observing the chromatograms, how many triacylglycerides can be transformed into fatty acid and thus, observe the performance of the reaction in the suggested conditions.
The dilution of the sample (hydrolyzed product) in heptadenoic acid at a 0.05:1 (m/m) ratio was necessary. Then 1 μL of the sample was injected into the Shimadzu chromatograph, model GC-2010, with split/splitless injector, flame ionization detector (FID), Quadrex Carbowax column (30 m × 0.32 mm × 0.25 μm), under the following conditions : 200˚C isothermal, Injector: 250˚C, Detector: 250˚C, Carrier gas pressure: 1.9 mL/min. Analyses were performed for 1 hour.
2.8. Biodiesel Characterization
The final product of the hydroesterification process was characterized according to ANP’s (Brazil’s oil regulator) No. 42 resolution, “Specification for B100 biodiesel”.
Percentage Determination of FAME (Fatty Acid Methyl Esters-Gas Chromatography (GC-FID)
The reaction products obtained by hydroesterification reactions (hydrolysis and esterification) were quantified by gas chromatography (GC) according to the content of fatty acid esters. In this methodology it is possible to identify both qualitatively and quantitatively the esters present in biodiesel. Methyl heptadecanoate was used as internal standard.
The dilution of 0.25 g of the sample (biodiesel) in 5 ml methyl heptadecanoate solution was necessary. Next, 1 μL of the sample was injected into the Shimadzu chromatograph, model GC-2014 with split/splitless injector, Flame ionization detector (FID), Quadrex Carbowax 20M capillary column (30 m × 0.32 mm × 0.25 μm), with the following conditions: 200˚C isothermal, Injector: 250˚C, Detector: 250˚C, Gas carrier: Helium or Hydrogen, 30 kPa to 100 kPa pressure. 1 mL to 2 mL flow, Total time of analysis: about 25 min. A computer, equipped with the GC SOLUTION software was used for the capture and analysis of the chromatograms.
3. RESULTS AND DISCUSSION
3.1. Lipid Profile
The lipid profile of the studied microalgal biomass, obtained by the esters analysis in the GC is shown in the chromatogram of Figure 4.
According to these data, the studied species showed the following fatty acids distribution: SAFA (saturated fatty acid) > PUFA (polyunsaturated fatty acid) > MUFA (monounsaturated fatty acid) content. The higher values refer to SAFAs, being C16:0 (palmitic acid) and C11:0 (undecanoic acid) the ones that stand out.
Concerning the PUFAs, high content of C18:3 (lino lenic) was observed. The second highest content of

Figure 4. Chromatogram-Lipid profile of the microalgal biomass. Identification: C11:0 (9.23%), C12:0 (1.18%), C14:0 (0.41%), C16:0 (29.80%), C18:0 (1.14%), C18:1 (23.61%), C18:2 (4.75%), C18:3 (26.17%), C20:0 (0.31%), C20:1 (0.53%), C22:0 (0.10%).
PUFA was the linoleic (C18:2) one.
As for the MUFAs, the oleic acid (C18:1) stands out.
The studied microalgal biomass had high levels of SAFA, mainly palmitic acid (C16:0) with concentrations near 30%, a feature similar to palm oil and tallow, which are high in saturated fat.
On the other hand, the species had a high concentration of monounsaturated fats, mainly represented by C18:1 (oleic acid) ≈23%, highly concentrated in tallow and mainly in olive oil.
C18:3 (linolenic acid) was also identified in high amounts (≈ 26%), as well as C18:2 (linoleic acid) in (≈ 5%); they appear at higher concentrations in soy.
These results were consistent with those found in previous papers, in which palmitic acid was determined as prevalent in most of the microalgae species grown in freshwater [3-5].
The Scenedesmus obliquus microalgae showed palmitic acid (C16:0) concentration-between 35.86% and 43.06% [6-7].
This amount of saturated fatty acids in the microalgae was also observed by [2], when he studied the lipid profile of the microalgae Chlorella vulgaris, Scenedesmus and Botryococcus sp braunni. In that study, the researchers achieved 36.3% palmitic acid for Scenedesmus sp.
3.2. In Situ Hydrolysis of the Microalgal Biomass
To perform the in situ hydrolysis of the microalgal biomass, 300g of the centrifuged sample, consisting of 5% biomass and 95% water, was used. The sample was hydrolized at 250˚C temperature for one hour reaction. Since the lipid content of such biomass is around 27.79% (dry weight), a definitive oil phase was not observed as expected (Figure 5). Thus, it was necessary to perform a hexane extraction to collect the fatty acids resulting from hydrolysis. After extracting and drying in a furnace at

Figure 5. Product of the microalgal biomass hydrolysis.
60˚C, a sample of 60 mg (fatty acids mixture) was obtained in each hydrolysis. Five hydrolysis experiments were carried out, until a significant amount of sample was obtained to allow the esterification stage.
Figure 6 shows the chromatogram of the fatty acids profile present in the hydrolysis product. Such result confirms the in situ occurrence of microalgal biomass hydrolysis.
This is a very interesting result; it represents a signifycant economic benefit, considering the possibility of producing biodiesel directly from wet biomass, eliminating the lyophilization and oil extraction processes.
3.3. Esterification—The Generation of Methyl Esters
For the esterification reaction, niobium oxide powder was used as catalyst.
The reactions were carried out by using excess alcohol and the mixture of fatty acids resulting from the hydrolysis process, MM = 256.42 g/mol, according to the following protocol: alcohol molar ratio: 30:1 fatty acid, 200˚C, 10% catalyst, 1 hour reaction and 500 rpm agitation, for as observed by [8], above such rotations, no significant conversions were found, despite knowing that 80% of the vegetable oil is converted into biodiesel in the first 10 to 20 minutes contact among the reactants [8,9]. 60 minutes was the response time established for all the experiments since, as described by [8], times over 20 minutes did not cause significant increases in the reaction conversion; however, they may favor the observation of reaction kinetics as a whole, as they are scarcely known reactions.
In the esterification reaction there is no glycerol formation, so the product was directly submitted to drying to remove the residual water and methanol. The catalyst could be recovered by filtration.
The final product of the reaction was submitted to chromatographic analyses so as to identify the product formed in the reaction. The results confirm the occur-
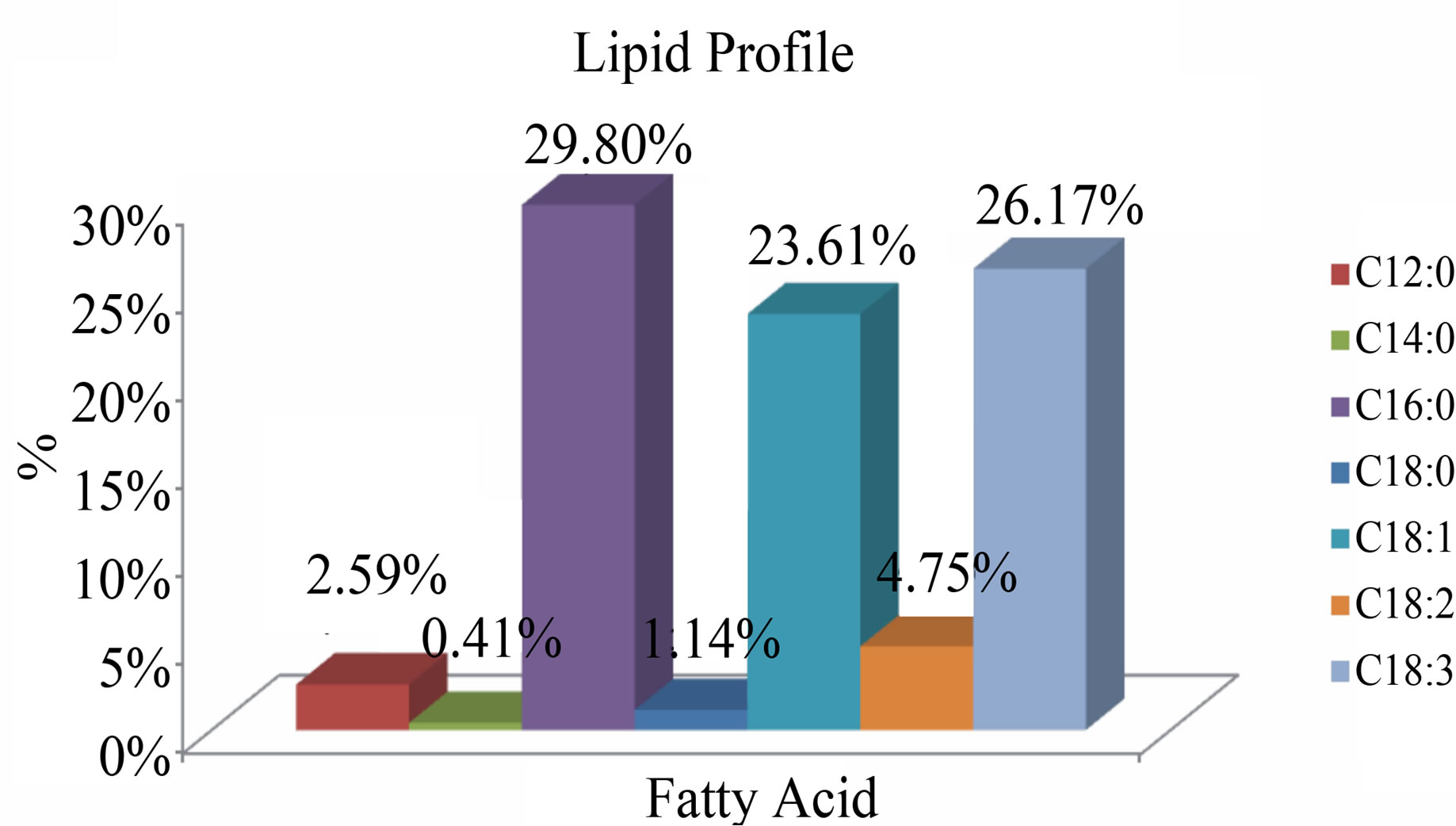
Figure 6. Chromatogram-Fatty acids obtained by in situ microalgal biomass hydrolysis. Identification: C12:0 (2.59%), C14: 0 (0.88%), C16: 0 (39.32%), C18: 0 (8.49%), C18:1 (29.35%), C18:2 (4.36%), C18:3 (14.99%).
rence of the fatty acids esterification process. The behavior follows the same pattern previously discussed in the microalgal biomass lipid profile determination and can be seen in Figure 7.
It is important to observe that the total ester percentage was of 94.27%. Similar results have been found by [9], studying the production of fatty esters in the presence of methanol with Chlorella vulgaris microalgae by using acidic transesterification process. The lipids rate of conversion into methyl esters obtained in this study was, in average, of 91.7%.
3.4. Biodiesel Characterization
The methyl esters obtained by using the proposed technology were characterized in relation to some distinctive parameters required by ANP. While still preliminary, they give an idea of the microalgae biodiesel quality. However, there is still a long way to go in the process of optimization.
It is known that among vegetable oils, fatty acid composition varies and so do their physicochemical properties (for instance, the oxidative stability). The same occurs with the oil extracted from different microalgae species and also with the same species grown under different conditions.
Biodiesel and biodiesel blends are exposed to air, acids begin to form, which are transferred to a conductivity cell containing deionized water. Once the acid concentration in the water is high enough, the conductivity undergoes a rapid increase that is called the induction period.
The oxidative stability of the sample obtained in this experiment was around 4.77 hours (Figure 8), according to the EN14112 trial performed in the Rancimat device. This result is consistent with the previous ones, in other words, high content of saturated fatty acids in the feedstock; therefore, a high content of saturated esters in the biodiesel composition—an aspect earlier confirmed by chromatography. The value obtained is not very different

Figure 7. Chromatogram-Methyl esters resulting from the microalgal biomass hydroesterification. Identification: C12:0 (0.63%), C14:0 (0.52%), C16:0 (30.62%), C16:1 (2.58%), C18:0 (4.01), C18:1 (34.70%), C18:2 (6.54%), C18:3 (18.03%), C20:0 (0.61%), C20:1 (0.65%) C22:0 (0.48%), C24:0 (0.08%), C24:1 (0.49%).

Figure 8. Oxidative stability of the studied microalgal biomass methyl esters.
from the esters profile normally observed in the biodiesel derived from conventional oleaginous (6 hours time for the standard soy biodiesel).
As already widely discussed, palmitic acid, oleic acid and linolenic acid were the dominant constituents in the lipid composition of the studied microalgal biomass. These medium chain fatty acids (C16 and C18) are considered ideal for producing high quality biodiesel, as mentioned in [10-11]’s work and are the most direct and accurate parameter to assess microalgae’s potential as feedstock for the production of biodiesel, since not all of the soluble compounds in organic solvents used in extraction may be converted into biodiesel.
There are other parameters that are considered very important when studying the quality of biodiesel, such as the amount of mono-, diand tri-glycerides present in biodiesel. These data allow the observation of the reaction performance in the assessed reaction conditions evaluated. The values obtained in this study are presented in Table 2. They are under the maximum limits required by ANP, a fact undoubtedly associated to the obtainment of biodiesel from free fatty acids. Although these results are still embryonic, they hold an enormous promise concerning

Table 2. Preliminary characterization of the biodiesel made from Monoraphidium contortum (MORF-1) microalgae.
microalgae’s potential as a future source of biofuel.
4. CONCLUSIONS
1) It is possible to obtain fatty acids through in situ microalgal biomass hydrolysis.
2) The analysis of the fatty acids in the studied microalgae displayed saturated fatty acids predominance, which was considered ideal for producing high quality biodiesel.
3) By using the proposed technology, the characteristics of the biodiesel obtained from the studied microalgal biomass are similar to the biodiesel produced from conventional feedstock, such as palm oil, soy, etc.
REFERENCES
- Folch, M., Lees, M. and Stanley, G. (1957) A simple method for the isolation and purification of total lipids from animal tissues. The Journal of Biological Chemistry, 226, 497-509.
- Yoo, C., Jun, S. and Lee, J. (2010) Selection of microalgae for lipid production under high levels carbon dioxide. Bioresource Technology, 101, 71-74. doi:10.1016/2009.03.030
- Colla, L., Bertolini, T. and Costa, J. (2004) Fatty acids profile of Spirulina platensis grown under different temperatures and nitrogen concentrations. Zeitschrift fur Naturforschung, 59, 55-59.
- Deshnium, P., Paithoonrangsarid, K. and Suphatrakul, A. (2000) Temperature-independent and dependent expression of desaturase genes in filamentous cyanobacterium Spirulina. FEMS Microbiology Letters, 184, 207-213. doi:10.1111/1574-6968.2000.09015
- Olguín, E., Galicia, S. and Angulo-Guerrero, O. (2001) The effect of low light flux and nitrogen deficiency on the chemical composition of Spirulina sp., Bioresource Technology, 77, 19-24. doi:10.1016/0960-8524(00)00142-5
- Makulla, A. (2000) Fatty acid composition of Scenedesmus obliquus: Correlation to dilution rates. Limnology, 30, 162-168. doi:10.1016/0075-9511(00)80011-0
- Willis, M., Lencki, R. and Marangoni, A. (1998) Lipid modification strategies in the production of nutritionally functional fats and oils. Criticals Reviews in Food Science and Nutrition, 38, 639-674. doi:10.1080/10408699891274336
- Rodrigues, B., Constantino, A. and Carvalho, L. (2005) Palm fatty acid esterification using heterogeneous catalysts. 13 Brazilian Congress of Catalysis, 4, 1-4.
- Veljkovic, V.B., Lakicevic, S.H., Stamenkovic, O.S., Todorovic, Z.B. and Lazic, M.L. (2006) Biodiesel production from tobacco (Nicotiana tabacum L.) seed oil with a high content of free fatty acids. Fuel, 85, 2671-2675. doi:10.1016/2006.04.015
- Marchetti, J. and Miguel, V. (2007) Heterogeneous esterifications of oil with high amount of free fatty acids. Fuel, 86, 906-910. doi:10.1016/2006.09.006
- Furuta, S., Matsuhashi, H. and Arata, K. (2004) Biodiesel fuel production with solid superacid catalysis fixed bed reactor under atmospheric pressure. Catalysis Communications, 5, 721-723. doi:10.1016/2004.09.001

