Applied Mathematics
Vol.05 No.12(2014), Article ID:47375,12 pages
10.4236/am.2014.512168
Assessment of Myogenic Power Expenditure Due to Arterial Wall Smooth Muscle Contraction Based upon the Fractal Nature of Vascular Trees
Akira Kamiya*, Masahiro Shibata, Kimiko Yamamoto
Research Laboratory for Interdisciplinary Sciences, Tokyo, Japan
Email: *akamiya@kamakuranet.ne.jp
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 March 2014; revised 30 April 2014; accepted 7 May 2014
ABSTRACT
The purpose of this study is 1) to present a biomechanical model for evaluating the myogenic power expended in an arterial segment due to vascular smooth muscle contraction (VSMC) and 2) to assess the total power expenditure in the entire systemic arterial tree by utilizing the fractal nature of the branching architecture. The model is based on the mechanical equilibrium between the stretch stress exerted by blood pressure inside the vessel lumen and constricting stress elicited by VSMC in the vascular wall. An expression for myogenic power expenditure is formulated for a unit wall mass as a function of the internal vessel radius and extent of strain. This expression was then integrated over selected range of vessel radii, by taking into account of the fractal nature of the branching structure. When the total myogenic power expended in the systemic arterial tree in rat at the moderate strain level is converted to the oxygen consumption rate, it amounts to approximately 18% of the whole body oxygen consumption rate. This suggests that the mechanical power expenditure due to VSMC is a significant factor that should not be ignored in studies of vascular energetics.
Keywords:
Myogenic Active Stress, Biomechanics, Fractal Integral, Oxygen Consumption, Allometric Law

1. Introduction
The energetics of the vascular system has been explored, mainly with respect to the optimum models of its branching system, e.g., the minimum work model by Murray [1] [2] or the minimum volume model by Kamiya and Togawa [3] and Kamiya et al. [4] . In most analyses of these models, the cost function to be minimized has been defined as the sum of the mechanical power loss due to viscous resistance against blood flow through narrow vessels and chemical energy demand to sustain massive volume of blood fresh and active in large vessels. However, one problem with these models is that the mechanical energy expenditure due to vascular smooth muscle contraction (VSMC), which consistently regulates the vascular tone and radius, has not been included in the cost function; this is in contrast with cardiac ventricular energetics [5] in which myocardial contraction is treated as an essential element in exerting mechanical power for pumping blood. VSMC has been neglected because, as demonstrated by Johnson [6] , the oxygen consumption rate of VSMC per unit of mechanical power is astonishingly low, in comparison with those of cardiac and skeletal muscle contractions (interlocking mechanism). However, the significant role of VSMC in the vascular energetics should not be ruled out, until we accurately determine the magnitude of its mechanical power expenditure and assess the amount of oxygen (O2) it consumes.
As a matter of fact, in vivo measurements of O2 tension  in peripheral arterial blood in rat cremaster muscle by Shibata et al. [7] have revealed that the level of
in peripheral arterial blood in rat cremaster muscle by Shibata et al. [7] have revealed that the level of 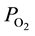 in arterial blood ordinarily declines toward terminals and that the descending rate significantly diminishes when the terminal arterial VSMC is eliminated by topical application of vasodilator. These findings suggest that one of the factors inducing
in arterial blood ordinarily declines toward terminals and that the descending rate significantly diminishes when the terminal arterial VSMC is eliminated by topical application of vasodilator. These findings suggest that one of the factors inducing 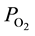 reduction in arterial blood toward the periphery is the active consumption of O2 within the vascular wall due to VSMC [7] .
reduction in arterial blood toward the periphery is the active consumption of O2 within the vascular wall due to VSMC [7] .
To evaluate the total mechanical power due to VSMC in the entire vascular system, we need to utilize the fractal nature of the vascular branching structure introduced by Mandelbrot [8] . In our preceding study [9] , we confirmed that various morphological and functional properties of the vascular system can be quantified by fractal-based integrals and their derivatives (see Appendix). Accordingly, the most urgent task to be done in this study is to construct a theoretical model for evaluating the mechanical power expended by VSMC per unit vascular wall mass and to assess its total amount in relevant vessel region using fractal integration. The outcomes of the analyses will be compared with conventionally physiological findings to verify the validity of the assessment and to substantiate the significance of this type of approaches in system physiology.
2. Methods
1) Theoretical assessment of mechanical power elicited by VSMC.
Figure 1 illustrates the circumferential stress-strain  relationship in a vascular wall under the condition that the internal hydrostatic pressure (blood pressure,
relationship in a vascular wall under the condition that the internal hydrostatic pressure (blood pressure, ) remains constant. It is known that in such a
) remains constant. It is known that in such a  diagram, the area enclosed by the trajectory of one cycle represents the amount of energy exerted in the cycle per unit mass.
diagram, the area enclosed by the trajectory of one cycle represents the amount of energy exerted in the cycle per unit mass.
The average circumferential stress 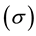 in the wall can be expressed, according to Laplace’s law, as
in the wall can be expressed, according to Laplace’s law, as
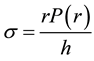 (1)
(1)
where  is the internal radius of a cylindrical vessel and
is the internal radius of a cylindrical vessel and 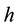 is its wall thickness. The strain
is its wall thickness. The strain  on the vessel wall is ordinarily expressed with the unstressed radius
on the vessel wall is ordinarily expressed with the unstressed radius 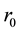 as,
as,

In the present analyses, the standard state for any vessel segment is set at its maximally vasodilated state, which is the point indicated by s in Figure 1. The suffix s attached to parameters in the text designates those at the standard state. The radius 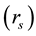 in this standard state is given in terms of the corresponding strain
in this standard state is given in terms of the corresponding strain  by,
by,
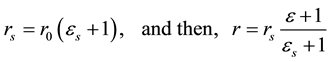 (2)
(2)
We presume that the physiological values of 

For the relation between the wall thickness 





Figure 1. Stress-strain 



0.2 [11] . Accordingly, we have,

According to Schmidt-Nielsen [12] , a number of morphological and functional properties in mammals can be expressed as the power functions of their body weights (the allometric law). The radius of the aorta 




In Figure 1, we consider a quasi-static constricting process of a vessel due to VSMC under the condition of constant internal hydrostatic pressure. In this process, which starts from the maximally dilated (standard) state, substantial changes in the entire stress 












The curvature of the elastic stress 






Table 1. Values of constant parameters used in present simulation studies.
Using Equations (5) and (6), the active myogenic stress 

In a steady state of the wall, the constricting force due to the circumferential stress 














The expanding rate 







Here, 




where,
With respect to the blood pressure profile 
Equation (10) gives the magnitude of mechanical power generated by VSMC at a branch of radius 


2) Fractal integration of the mechanical power expenditure by VSMC.
Based upon the fractal nature of the vascular tree [8] , we formulate the aggregated magnitude of the mechanical power generated by VSMC for any given range of vessel radii, under several assumptions. As shown in the Appendix, our mathematical model for fractal trees [9] introduces a variable called the “aggregated branch length





Here, 







From Equations (10) and (11), the magnitude of mechanical power elicited by VSMC in branches having radii within a minute range, 




provided the value of 
The integral in Equation (12) may not be solved analytically but when appropriate data for the involved parameters are available, a numerical solution may be found. In such numerical integrations, we need to employ a logarithmic transformation of the variable 

3. Results
Figure 2(a) shows distributions of myogenic power 







Figure 2(b) contains the results of the fractal integrals for 


Figure 2. (a) Distributions calculated from Equation (10) for myogenic power expenditure per unit wall tissue mass 


Table 2. Values of body weight 


data as that used for Figure 2(a). The radius ranges for the integrals 


Results corresponding to those in Figure 2 are also calculated using the rat data in Table 2 with 


Based on present and reported data, we now try to estimate the coefficient between oxygen consumption rate and myogenic power expenditure due to VSMC. Using the fluorescence quenching method, Shibata et al. [7]


Figure 3. (a) Distributions from Equation (10) for myogenic power expenditure per unit wall tissue mass 


measured the oxygen tension 






The differences in the two 







4. Discussion
In evaluating the myogenic power expenditure due to VSMC and performing its fractal integration leading to the results in Figure 2 and Figure 3, a number of assumptions were introduced. One of the major hypotheses, which was used being in the quasi-static constriction of vascular wall in Figure 1, was that the hydrostatic pressure inside the vessel was assumed constant regardless of the extent of strain,




The other major hypothesis used in the present calculations is that not only such univocal parameters as 



Figure 4. Profiles of blood pressure, 




large and small vessels is a very bold assumption that apparently over-simplifies the physiological situations.
Nevertheless, we try to assess the total amount of oxygen consumption rate, 










We now compare the result in Equation (14) with the total oxygen consumption rate for the whole body in rat, 





Acknowledgements
This study was supported by a Grand-in-Aid from Japan Society for Atherosclerosis Research Foundation.
References
- Murray, C.D. (1926) The Physiological Principle of Minimum Work I. The Vascular System and the Cost of Blood Volume. Proceedings of the National Academy of Sciences, 12, 207-214. http://dx.doi.org/10.1073/pnas.12.3.207
- Murray, C.D. (1927) A Relationship between Circumference and Weight in Trees and Its Bearing on Branching Angles. The Journal of General Physiology, 10, 725-729. http://dx.doi.org/10.1085/jgp.10.5.725
- Kamiya, A. and Togawa, T. (1972) Optimal Branching Structure of the Vascular Tree. Bulletin of Mathematical Biophysics, 34, 431-438. http://dx.doi.org/10.1007/BF02476705
- Kamiya, A., Wakayama, H. and Baba, K. (1993) Optimality Analysis of Vascular-Tissue System in Mammals for Oxygen Transport. Journal of Theoretical Biology, 162, 229-242. http://dx.doi.org/10.1006/jtbi.1993.1084
- Suga, H. (1990) Ventricular Energetics. Physiological Reviews, 70, 247-277.
- Johnson, P.C. (1984) The Myogenic Response. In: Handbook of Physiology, Sect. 2 (The Cardiovascular System), American Physiological Society, Bethesda, Chapter 15, 409-442.
- Shibata, M., Ichioka, S. and Kamiya, A. (2005) Estimating Oxygen Consumption Rates of Arteriolar Walls under Physiological Conditions in Rat Skeletal Muscle. American Journal of Physiology―Heart and Circulatory Physiology, 289, H295-H300. http://dx.doi.org/10.1152/ajpheart.00830.2004
- Mandelbrot, B.B. (1977) The Fractal Geometry of Nature. In: Trees and the Diameter Exponent, Freeman, New York, Chapter 17, 156-165.
- Kamiya, A. and Takahashi, T. (2007) Quantitative Assessments of Morphological and Functional Properties of Biological Trees Based on Their Fractal Nature. Journal of Applied Physiology, 102, 2315-2323. http://dx.doi.org/10.1152/japplphysiol.00856.2006
- Suwa, N. and Takahashi, T. (1971) Morphological and Morphometrical Analysis of Circulation in Hypertension and Ischemic Kidney. Urban & Schwarzenberg, Munchen.
- Folkow, B. and Neil, E. (1971) Circulation. In: Vascular Length and Radius, Oxford Univ. Press, Oxford, Chapter 5, 36-56.
- Schmidt-Nielsen, K. (1984) Scaling: Why is Animal Size So Important? In: Heart and Circulation, Cambridge Univ. Press, Cambridge, Chapter 11, 126-142.
- Abe, H., Hayashi, K. and Sato, M. (Eds.) (1996) Data Book of Mechanical Properties of Living Cells, Tissues, and Organs. In: Blood Vessels, Springer-Verlag, Tokyo, Chapter 2.2, 25-114.
- Rosen, R. (1967) Optimality Principles in Biology. In: The Vascular System, Butterworths, London, Chapter 3, 41-60.
- Haynes, R.H. (1960) Physical Basis of the Dependency of Blood Viscosity on Tube Radius. American Journal of Physiology, 198, 1193-1200.
- Zweifach, B.W. and Lipowsky, H.H. (1984) Pressure-Flow Relations in Blood and Lymph Microcirculation. In: Handbook of Physiology, Sect. 2 (The Cardiovascular System), American Physiological Society, Bethesda, Chapter 7, 231-307.
Appendix
A summary of our preceding study [9] on fractal-based assessments of morphological and functional properties of the vascular system.
For a vascular system characterized by a fractal branching architecture, the probability 





Here, 



To intuitively perceive the fractal-based integrations of the morphological properties in the vascular system, we introduce a term “aggregated branch length” which is defined as the sum of branch lengths of vessels in a group sorted by radius around 



where 
From (A-1) and (A-2), the aggregated branch length






It is now apparent that a segmental integral, 

can be expressed as,
The expectation value of the aggregated branch length 



On the other hand, Suwa and Takahashi [10] have established that the relationship between branch length 


In various vascular systems, the values of the exponent 


where 

Since the radii of terminal branches 






This widely substantiated relationship is known as the “empirical power law of the vascular branching” [1] [8] [14] and the value of 

By comparing (A-9) and (A-10), we have,

Thus, the exponent 


In addition, the expression 







Here, 







The profile of the blood pressure 





Here, 

where 

(see Table 1). Since we have 

13), the pressure gradient in the vascular system against branch radius, 

The symbol ± corresponds to the arterial and venous sides, respectively. In general, the pressure profile 








By integrating the approximation (A-16), we have a simple analytical equation for blood pressure profile 

where 




Another issue to consider about the blood pressure profile 



In relatively large vessels where

In small vessels in which the tube radius-dependency of viscosity is more evident (



NOTES
*Corresponding author.












