Green and Sustainable Chemistry
Vol.3 No.4A(2013), Article ID:39912,9 pages DOI:10.4236/gsc.2013.34A004
Oligoalkylnaphthenic (C6-C12) Oils Obtained in the Presence of Ti-Containing Ionic-Liquid Catalysts
The Y.H. Mamedaliyev Institute of Petrochemical Processes of Azerbaijan National Academy of Sciences, Baku, Azerbaijan
Email: *xayala_550@rambler.ru
Copyright © 2013 Akif Hamid Azizov et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received August 5, 2013; revised September 5, 2013; accepted September 12, 2013
Keywords: Ionic Liquid; Oligomerization; PAO
ABSTRACT
In this present paper, the result of C6-C12 α-olefins oligomerizaton in the presence of recyclable chloroaluminate type ionic-liquid catalysts and their combination with Ti-containing complexes is presented. By different methods of analysis the composition and structure of oligoalkylnaphtenic products obtained in the presence of ionic-liquid catalysts and Ti-containing modifiers have been studied.
1. Introduction
The continued use of large quantities of organic solvents as liquid media for chemical reactions, extractions and formulations is a major problem in nowadays chemical industry. Here E-factor, as it was defined by Roger Sheldon, is the environmental factor of a process, e.g. the ratio (by weight) of the by-products to the desired product(s). As can be seen, the oil refining and bulk chemicals industries are the least polluting industries, while thee fine chemicals and pharmaceutical companies, using inefficient and dirty processes, albeit on a much smaller scale, are polluting the environment severely. In addition to the problematic volatility of these solvents, they often present additional risk due to toxicity and/or flammability [1]. Thus, the discovery of surrogates to traditional organic solvents which offer reduced environmental impact and safer processes is of great importance.
J. D. Holbrey and K. R. Seddon consider four main alternate strategies for this: solvent-free synthesis, the use of water, the use of supercritical fluids as solvents and the use of ionic liquids as solvents.
The option of the use of ionic liquid as solvents seems to be one of the most promising methods in green chemistry. The unique peculiarities of low temperature ionic liquids such as non-volatility, non-flammability, low melting points, high thermal stability in a wide temperature range, relatively low viscosity, inexpensivity and diversity to manufacture and so on, have attracted interests of investigators from industrial and academic research centers from all around the world. They are highly solvating liquids, composed of bulky cations and anions, which dissolve many organic and inorganic substances. In the last few years there appeared a lot of papers and patents, devoted to preparation methods, solvent properties and application of ionic liquids as the solvents in different processes. Some of them have been overviewed in [1-10].
Low temperature ionic liquids are widely investigated as replacement for environmentally hazardous volatile organic solvents in the processes of organic synthesis and petrochemistry.
They have been used primarily as replacements for conventional media in green chemical processes or as solvent catalysts. The first use of ionic liquids in homogeneous catalysis was reported in 1990 for dimerisation and polymerization reactions [11,12]. Since then, a rapidly growing number of transition metal catalysed reactions have been described in various ionic liquids.
Ionic liquids are often called “green solvents”, and indeed the link between ionic liquids and green chemistry is mainly related to the characteristic of low volatility. The review presents C6-C12 α-olefins oligomerizaton in the presence of recyclable chloroaluminate type ionicliquid catalysts and their combination with Ti-containing complexes. Many of methods of analysis are used to study the composition and structure of oligoalkylnaphtenic products.
2. Experimental
All operations on the catalyst synthesis and oligomerization reactions were performed in an inert gas (nitrogen, argon) atmosphere using anhydrous solvents (benzene, toluene) and freshly distilled C6-C12 α-olefins.
α-olefins—before the use was dried over calcinated Al2O3 or CaCl2 and purified by distillation in atmospheric pressure.
Waterless AlCl3 was stored in an inert atmosphere in the hermetic vessels.
Diethylamine hidrocloride (DEAHC) used for complex ammonium salts preparation were purified by distillation at atmospheric pressure or by recrystallization.
Chloroaluminate ionic—liquids were prepared by interacting AlCl3 with the corresponding complex ammonium salt taken at molar ratios 1.5 - 2.1.
Synthesis of the ionic-liquid catalytic systems and also oligomerization of α-olefine in their presence were carried out in the thermostarted dried up glass reactor in the atmosphere of dry nitrogen or argon.
With that end in view the reactor was preliminary carefully vacuumized at warming up in a nitrogen current before preparation of the complex catalyst. Then after cooling to a room temperature the corresponding amine were entered into it.
The needed quantity of AlCl3 was added to the obtained complexes in a nitrogen current at intensive mixing. Catalyst preparation was accomplished at 30˚C - 35˚C during 10 - 30 minutes.
The ionic liquids obtained represented themselves viscous liquids at room temperature with a yellowish shade.
After termination of chloroaluminate ionic-liquid catalysts (ILC) synthesis α-olefin was introduced into the reactor at mixing through a drop funnel.
TiCl4 was applied in the form of a mobile colourless liquid smoking strongly on air. Molecular weight (M) 189.71, density (D25) 1.726, fusion temperature (Tm)- 23˚C, boiling temperature (Tb) 135.8˚C (760 mmHg).
2,6-di-(2’,6’-diisopropyl-1-aminomethyl)-4-(1-methylc yclo-pentile)-phenol (M = 554, Тboiling point = 205 - 207˚C, Тmelting point = 91˚C) has been prepared using the known method by interaction of para-(1-methylcyclopenthyl)-phenol
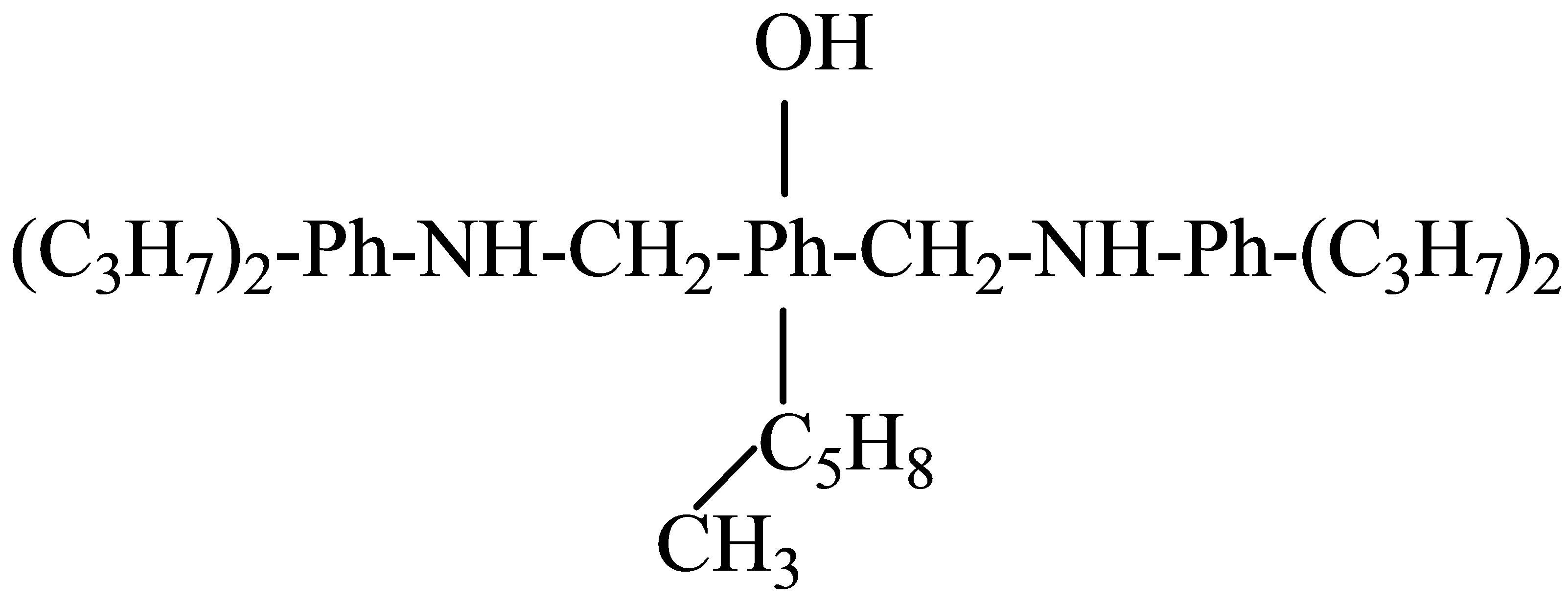
with formaldehyde and 2,6-diisopropylaniline according to Mannich reaction.
The titanium-phenolate complex (TF) was synthesized by the simple one-step method of interacting 2,6-di-(2’ 6’-diisopropyl-1-aminomethyl)-4-(1-methylcyclopentile)phenol with TiCl4:
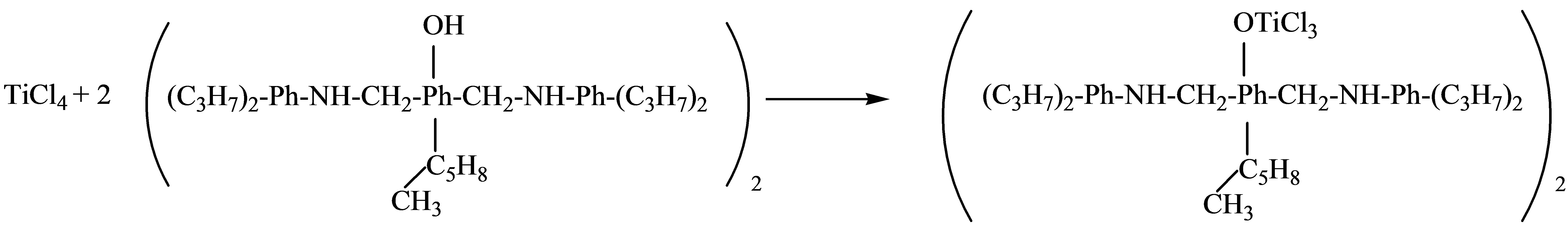
HCl released from the reaction was bound to the amine functionality of the phenolic compound in situ and remained as a part of the formed Ti-containing complex, which, after removal of the solvent, was represented as the brown colored powder, supposedly, sof zwitterionic type complex of the following formula:
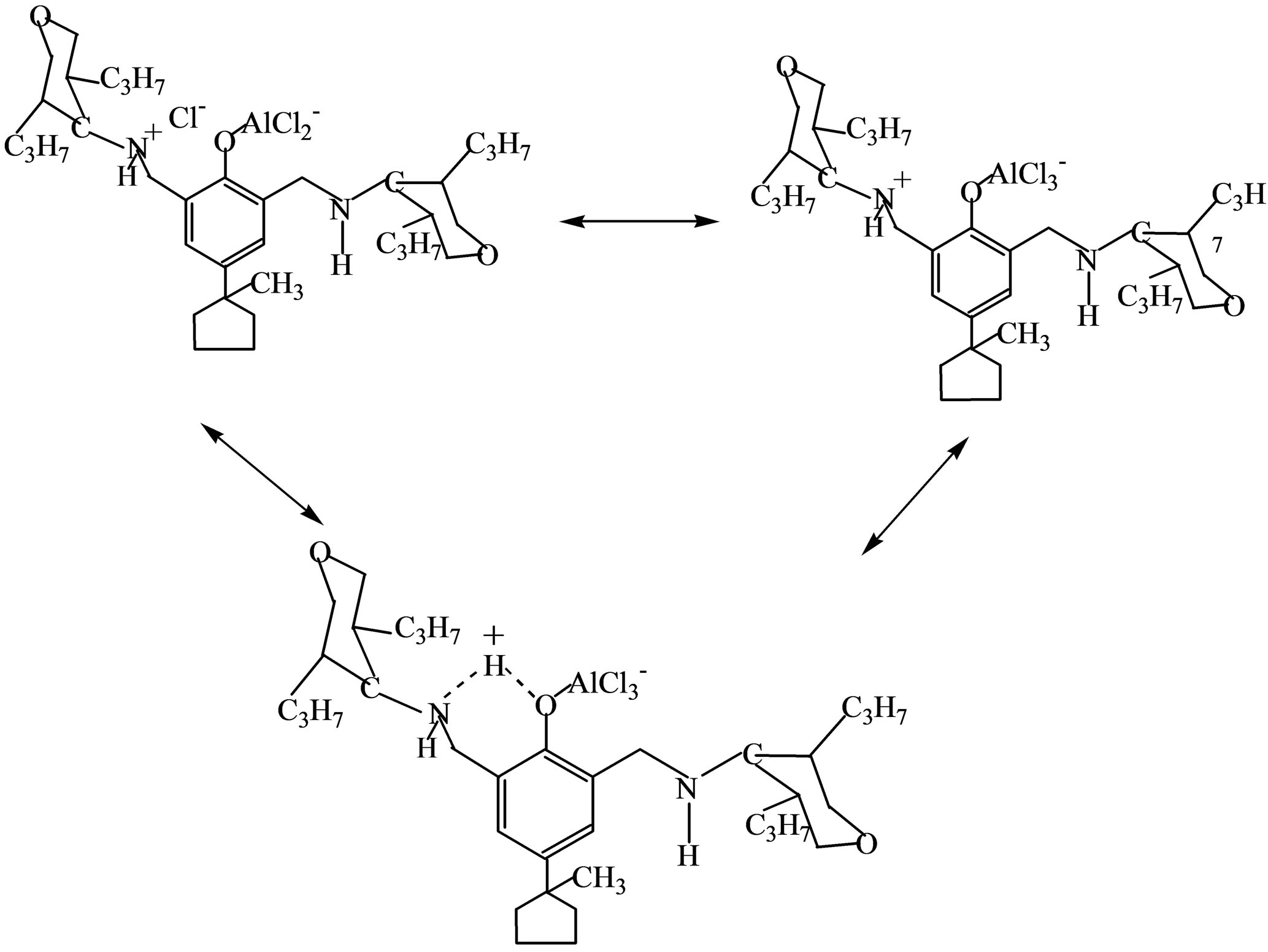
The following ionic-liquid catalysts were prepared by this method:
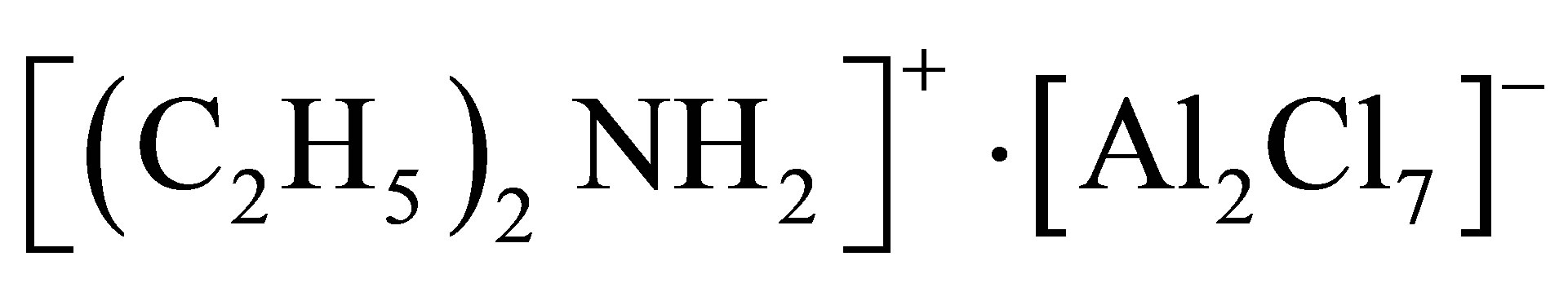 (ILC-I)
(ILC-I)
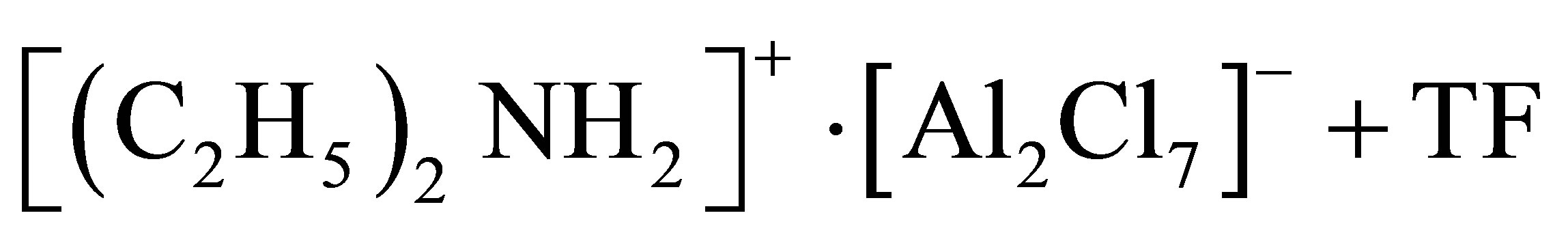 ILC-II)
ILC-II)
For the preparation of ТF was dissolved in ILC-I at the weight ratio TF:ILC-I = 0.001:1.
The experiments on oligomerization were conducted at the temperatures 60˚C over 3 h with intensive agitating of reaction mass by a electromagnetic stirrer. After the termination of oligomerization the reaction products were separated from the catalyst by washing off it with 10% solution of NaOH and then by the distilled water till the neutral reaction or by decantation. After drying the unreacted monomer was distilled off from the obtained oil in vacuum and synthetic oil fraction having b.p. 250 - 350˚C or >350˚C was isolated.
In case of application in the quality of ionic-liquid catalyst of the catalytic system, prepared on aluminum chloride basis, the oligomerization process was conducted at the fusion temperature of the specified ILC. In this case, upon termination of reaction the received reaction mixture was divided into two layers at cooling: the bottom layer represented ILC layer, the top layer – the oligomerizate layer. After separation of ILC from oligomerizate by decantation it was stored in an inert gas atmosphere for further reusing it repeatedly.
Oligomerization of C6-C12 α-olefins was carried out with application of the synthesized ionic-liquid catalytic systems in quantities, in which AlCl3 content was within 2% mass, counted on the olefin. The received results are presented in Table 1.
Oligomerization of C6-C12 α-olefins was conducted in the presence of ILCS-I and ILCS-II. A way of preparation of ILCS, a course of carrying out of the oligomerization processes and the analysis methods.
The results of researches on oligomerization of α-olefin in the presence of ILCS-I and ILCS-II at 60˚C, identical concentration of catalyst (0.2% AlCl3 masses, counted on olefin) are resulted in Table 1.
Apparently, the total amount of ILCS-I and ILCS-II in all cases of oligomerization makes 6% of mass., and a yield of oligomer amounts 85 - 103.92 gr oligomer/gr Al × hour, 77.1 - 90.3 × 103 gr oliqomer/gr Ti × hour, correspondingly. After the end of reaction, oligomerizate easily separates from catalyzate because of diphasic character of the process. ILCS is kept in the form of uncolored bed in an inert medium. A yield of products, within 3 h. amounts 68% - 85%.
3. Methods of Analysis
Oligomerization products were analyzed by exclusion chromatography, DSC, IRand PMR-spectroscopy.
Molecular-weight distribution (MWD) of the obtained products was studied by size exclusion chromatography method using high performance “Kovo” (Czech Republic) liquid chromatograph with a refractive index detector. Two 3.3 mm - 150 mm columns packed with the “Separon-SGX” stationary phase with a particle size of 7 mm and a porosity of 100 A˚ were used. Dimethylformamide was used as an eluent (flow rate 0.3 ml/min, temperature 20 - 25˚C). A calibration plot of log M versus VR in the range M = 2 – 100 - 102 was obtained using polyethylene glycol standards and transformed to the common dependence of fraction (%) of chains from their molecular weights (Figure 1). Calculations of the average molecular weights and MWD characteristics were made from the data of size exclusion chromatographic analysis in accordance with the procedure described in [14]. Average molecular weights (Mw and Mn) were calculated using the following formulas:
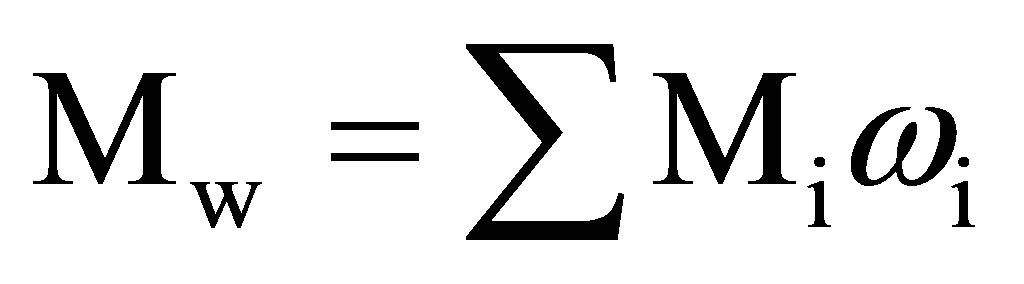 ,
, 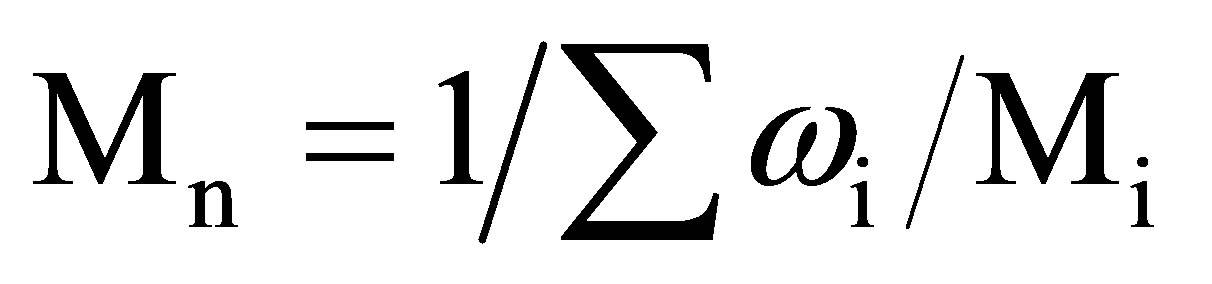
where Mi is the molecular weight, corresponding to the ith slice of the chromatogram; ωi is the area fraction of the ith slice.
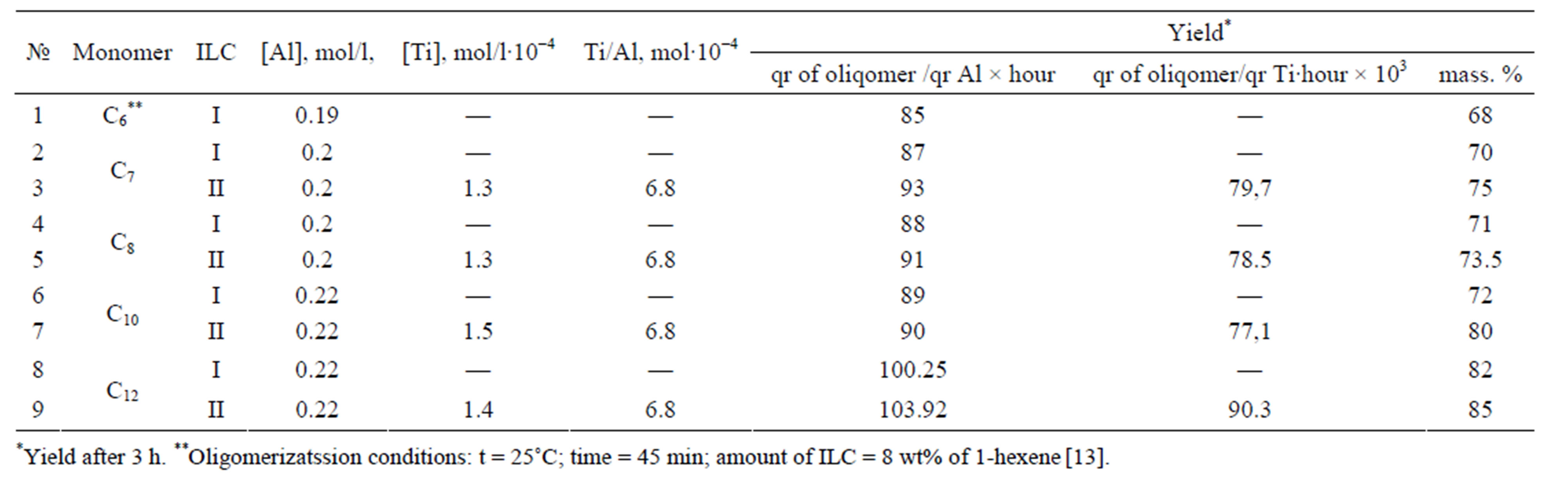
Table 1. Oligomerization of α-olefin depending on composition of ILCS [Conditions: aminophenol: TiCl4 = 1:1 mol., medium: argon].
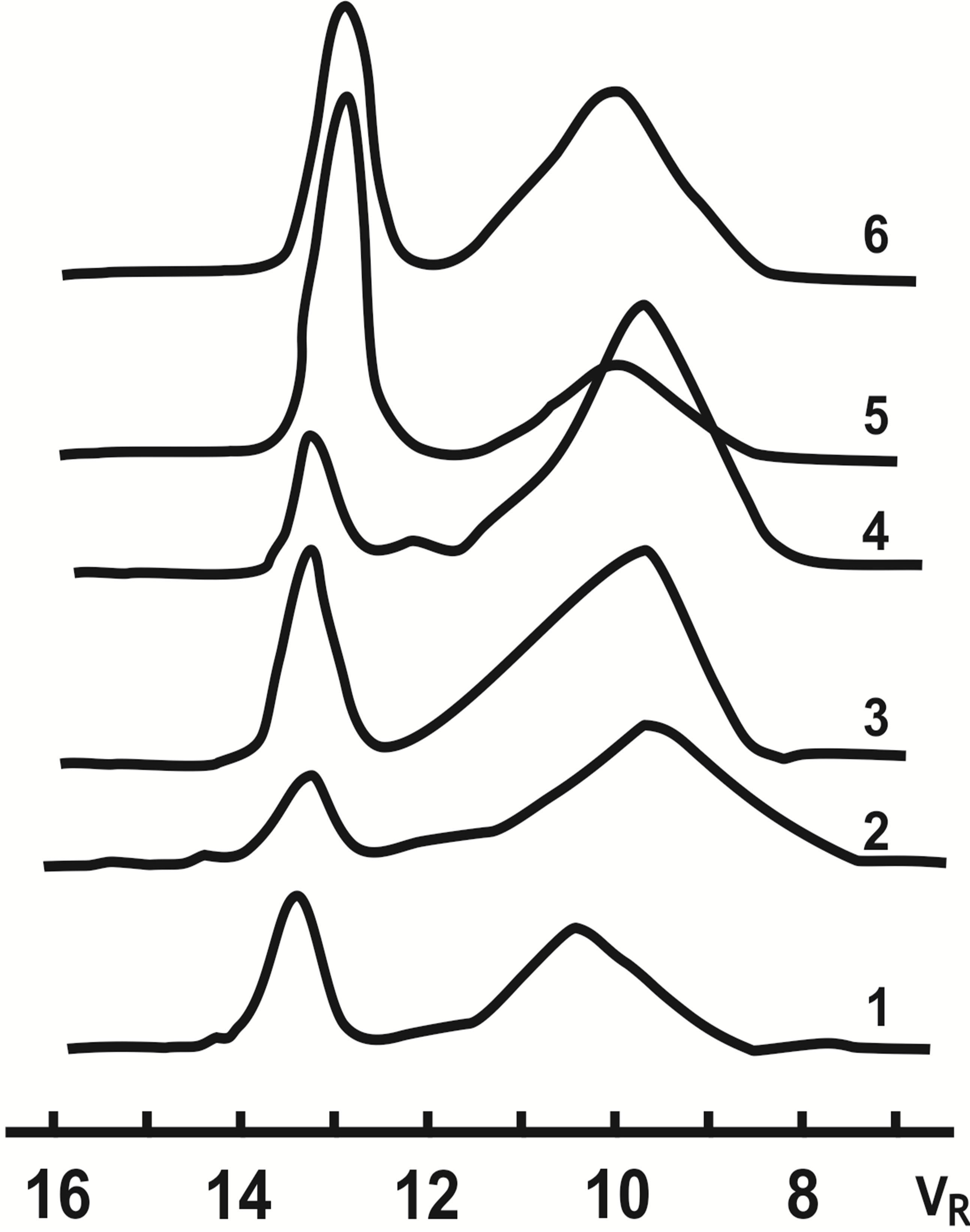
Figure 1. Exclusion chromatograms of oliqoalkylnaphtenic (C6-C12) fraction obtained in the presence of ILCS-I and ILCS-II.
DSC analysis was carried out on a Thermoelectron Q- 20 Differential Scanning Calorimeter, USA (Joint Grant of NASA and CRDF no. SIP-03), at a heating rate of 10˚C/min in the air or nitrogen atmosphere.
IR-spectroscopy analysis of the samples was performed on a Spectrum ВХ, FT-IR system “Perkin-Elmer” (USA) in the range 650 - 4400 sm−1.
PMR spectra were recorded on a Brunker pulsing Fourier spectrometer (Germany) operating at the frequency of 300 MHz and at room temperature. Deuterated acetone was used as a solvent. The relative amount of protons in various structural fragments was calculated by the integration of the corresponding resonance absorption bands.
4. Result and Discussion
4.1. Molecular Weight Characteristics of the Oligomers
The results of exclusion chromatographic analysis of C6-C12 α-olefins oligomers (Fr > 350˚C) are presented on Figure 1. As is seen from Table 2, the oligomerization products of dodecene—1 received in the presence of ILCS-I and ILCS-II have the MM indexes within the limits—Mw = 1560 - 6890, Mn = 1400 - 5300, where MMD coefficient changes within Mw/Mn = 1.11 - 1.19. Anyway in each system the received MMD indexes change in the narrow limits.
4.2. Structure
The received oligomers have also been investigated by IR-spectroscopy (Figure 2). In the IR spectrums of initial monomer—α-olefin the absorption band was observed at
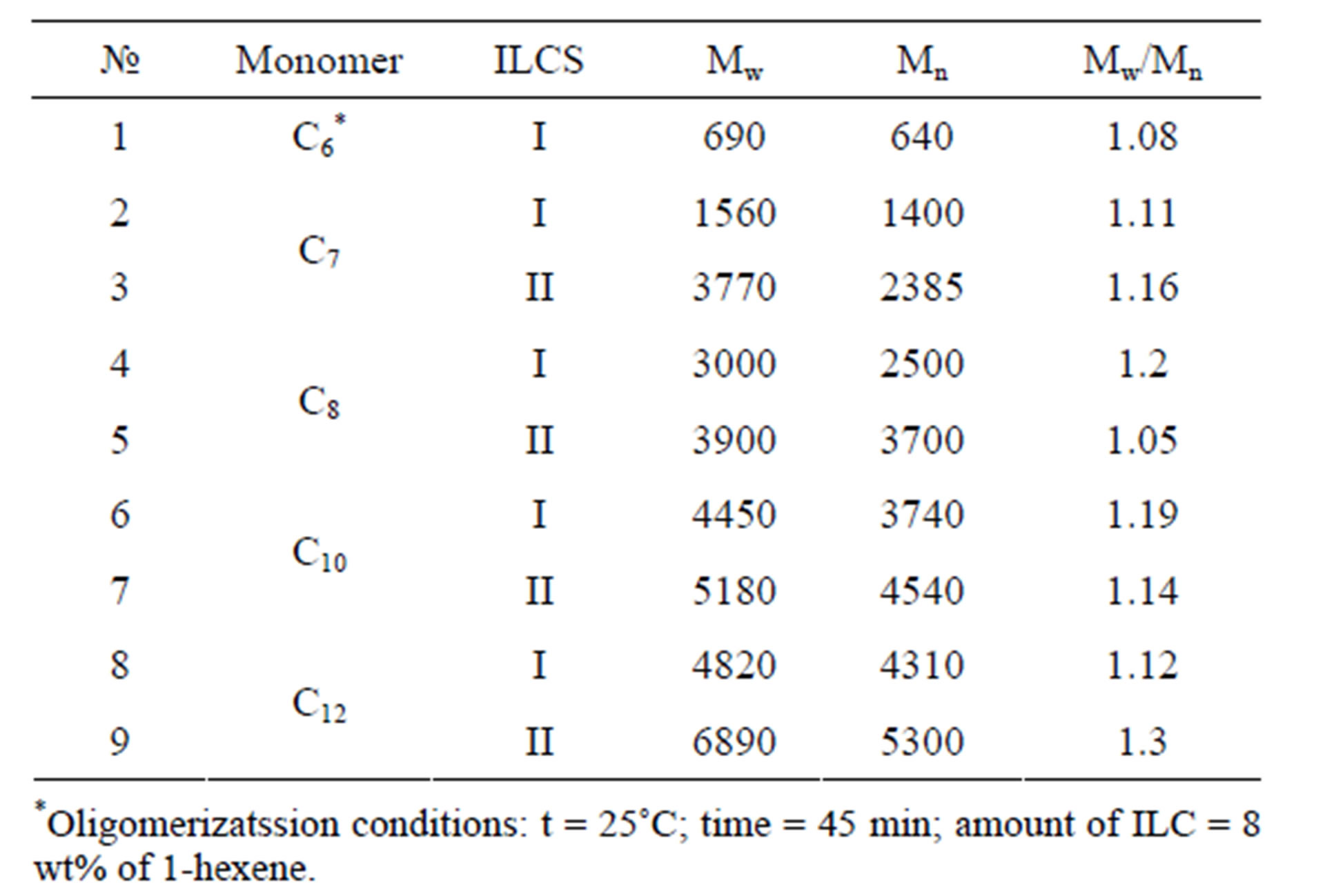
Table 2. Molecular mass indexes of oligolefin fractions received in the presence of ILCS.
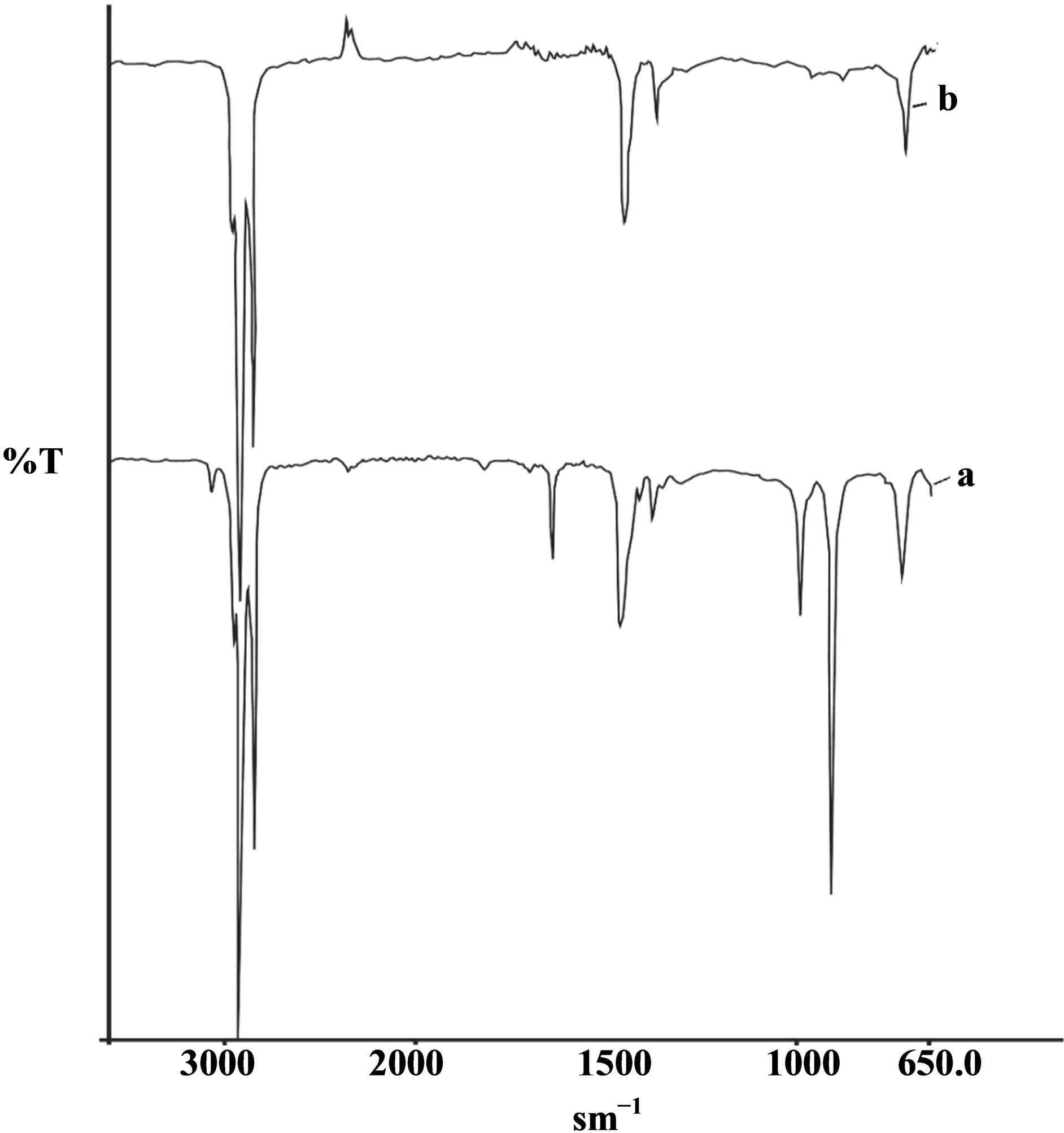
Figure 2. IR spectrum of dodecene-1 (а) and of oliqododecene fraction (b), produced in the presence of ILCS-I.
721 sm−1, responsible for the pendular fluctuations of the methylene groups (-CH2-)n with n > 4. There are also the bands of deformation at 1377.1 sm−1 and valence vibrations at 2853.5 sm−1, characteristic for methyl groups (-CH3). Deformation absorption bands at 1465.64 sm−1 and valence vibrations bands at 2922.7 sm−1 correspond also to the methylene groups (-CH2-). Except the listed absorption bands, there are in the spectrum the deformation bands at 1300.30 sm−1 and the valence vibration bands at 2955.60 sm−1, characteristic for -CH groups. Besides, there are the absorption bands at 908.3 sm−1, 992.05 sm−1 corresponding to deformation fluctuations of the end vinyl groups. And also the bands s with maxima at 1821.1 sm−1 are displayed, responsible for an overtone of nonplanar deformation fluctuations of the end vinyl groups. Absorption band with a maximum at 1641.6 sm−1 is characteristic for the valence vibrations of С=С bonds of nonsaturated hydrocarbons, and at 3076.4 sm−1 for the valence vibrations of -CH = group in an alkene molecule.
Analysis of the IR spectrums of C6-C12 oliqoalkyl fraction (OAF) received in the presence of ILCS has shown that intensity of absorption bands in them with maxima at 908.3 sm−1, 992. sm−1, 1641.6 sm−1, 1821.4 sm−1, characteristic for nonsaturated hydrocarbons is much less than in the initial monomer. There are in the spectrums the absorption bands at 980.3 sm−1, 1020.2 sm−1, characterizing the oscillating motions of the carbon atoms in the naphthenic rings, and at 900.2 sm−1, responsible for hydrogen atom of the isolated naphthenic ring [15,16].
For α-olefins and oliqoalkyl fraction, received in the presence of ILCS were calculated the optical densities of the bands characterizing oscillating motions of paraffinic, naphthenic, nonsaturated fragments. The obtained data are presented in Table 3.
The results of definition of structure oligoalkylfraction synthesized in the presence of ILCS (Tables 4, 5 and Figure 3) by NMR-spectroscopy method have also confirmed the data of the IR-spectral analysis. The PMRSPECTRA of oligoalkylfraction samples, received in the presence of ILCS-I and ILCS-II, are also characterized by the presence of the resonance absorption bands of the protons of methyl (CH3-) (0.89 - 0.98 ppm) and methylene (-CH-)n (1.28 ppm) groups, and also resonance absorption bonds in the field of 1.50 - 1.80 ppm, corresponding to chemical shifts of the protons in the naphthenic frames.
After strong amplification (16 times) the signals in the field of 4.8 - 5.5 ppm, corresponding to nonsaturated structures, very weak absorption bands that specified the insignificant presence of olefinic linkages have been found. At significant intensifying of resonance absorption by accumulation in the PMR spectra of oligodecene
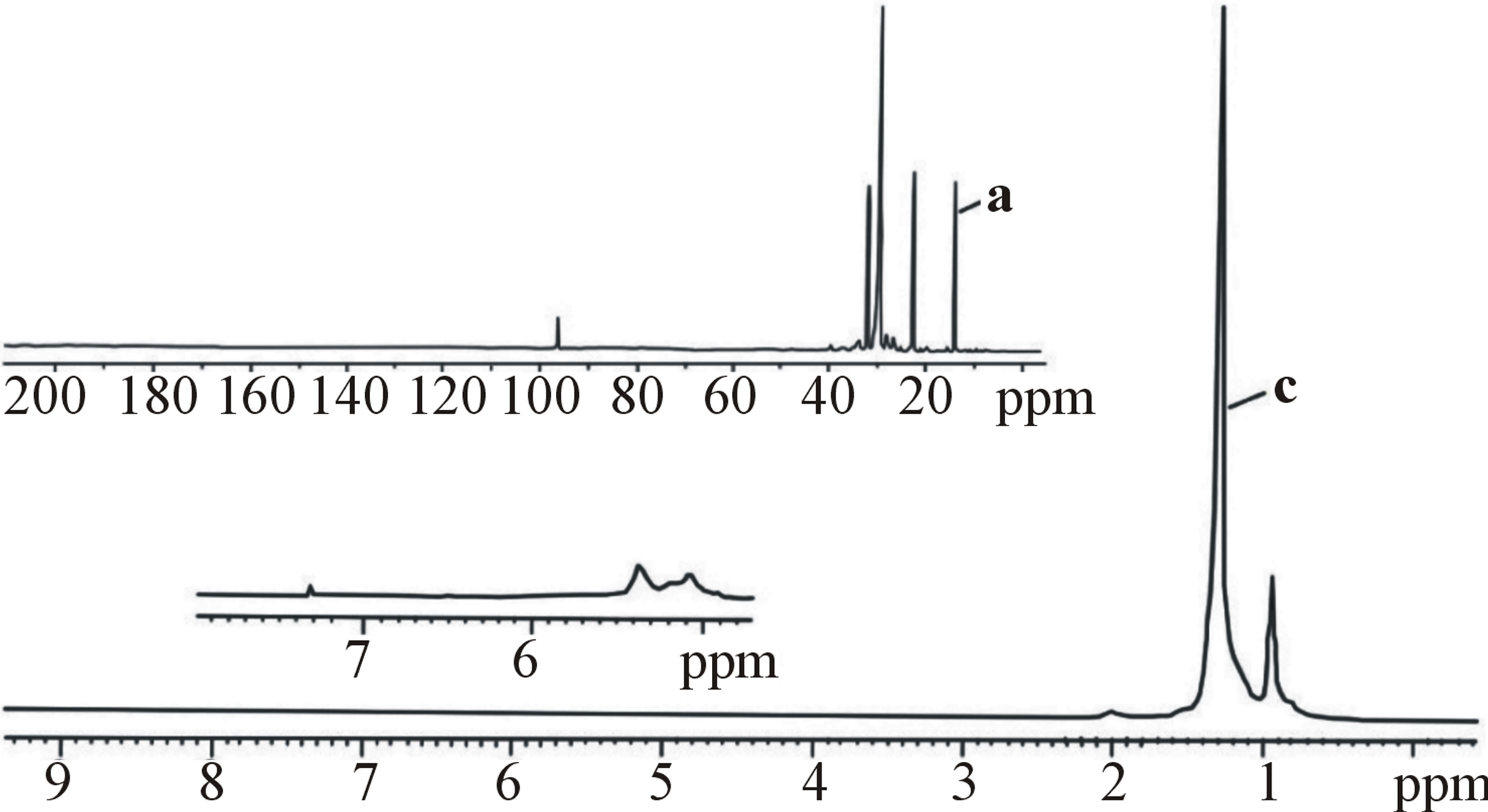
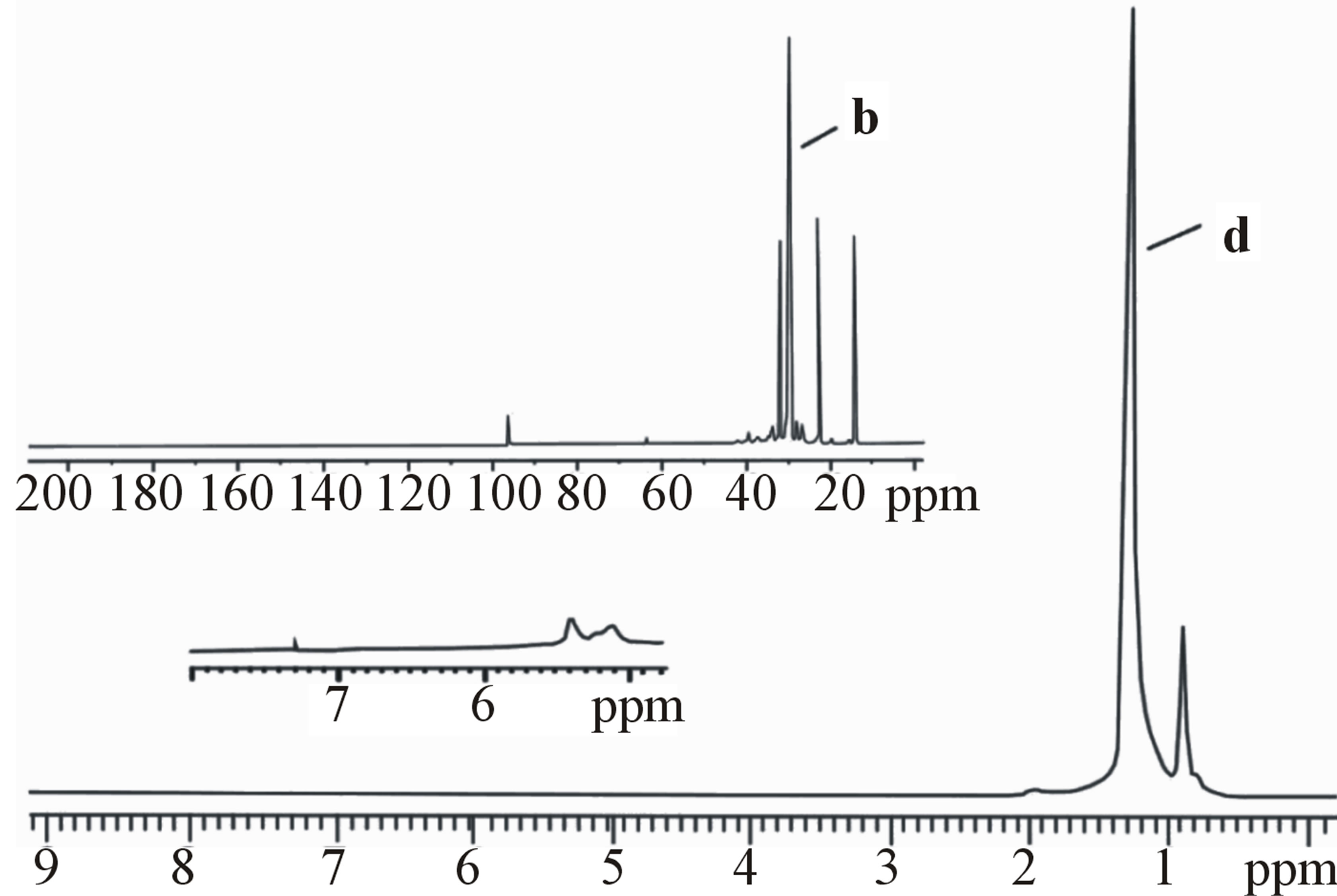
Figure 3. 13С NMR (а, b) and PMR (с, d) spectra of oligododecene fraction produced in the presence: ILCS-I (а, с) and ILCS-II (b, d).
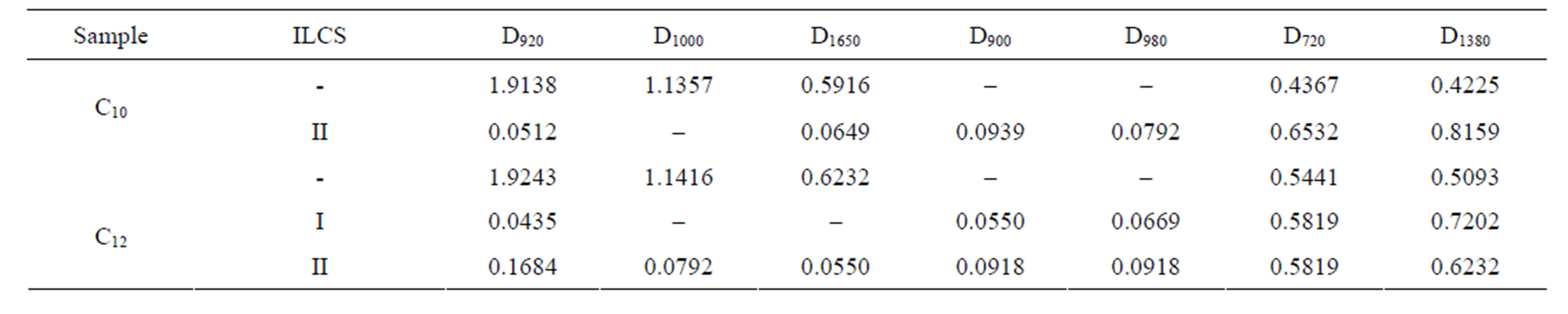
Table 3. Optical densities calculated from IR spectra.

Table 4. Structural parameters determined from PMR spectral data of dodecene-1in the presence of ILCS oligomerization products.
Table 5. Assignment of the signals in the 13С NMR spectra of dodecene-1 oligomerization products.
weak absorption bands of protons of the СН = СН2 methylene groups (at 5.0 - 5.5 ppm), and of vinylene CH = СН groups at 5.7 - 5.7 ppm) are observed also [17].
Research of the structural parameters of C6-C12 α-olefins oligomerization products, specify passing of the process with formation of the oligomers enriched with the fragments of (iso) paraffinic and naphthenic nature which concentration changes within 96.3 - 97.3 and 2.4% - 3.0% mass depending on the composition of catalytic system. Accordingly, concentration of the fragments containing multiple bonds make only 0.3% - 1.0% mass., which is much more low than in initial olefine (Table 5).
This fact once again obliquely testifies to passing of ring formation in the last part of a growing chain. With growth in the length of an alkyl radical in the olefin molecule the probability of bigger naphthenic ring formation increases.
The results of IR-, PMR and NMR spectral analyses have shown identity of frames of all investigated oligoalkylnaphtenic samples, synthesized in the presence of chloroaluminate ILCS. Though, the weak absorption bands observed in both spectra, characterizing multiple bonds, testify to the presence in the synthesized products of the trace amounts of nonsaturated fragments.
4.3. Some Physical and Chemical Properties Oligoalkylnaphtenic Oils
Some physical-chemical indexes of oligoalkylnaphtenic product, produced in the presence of ILCS-I and ILCS-II, have also been estimated. The results are presented in Table 6. The values of viscosity index (VI) have been calculated in accordance with GOST 25371-97. Physical - chemical properties of the synthesized oligoalkylnaphtenic product were defined according to the procedures [18,19].
From the data of Table 6 it is evident that the samples of the synthesized oligoalkylnaphtenic oils have low density values, high viscosity and viscosity index (≥108), low freezing point (−33˚C - −45˚C), high ignition temperature (≥172˚C), good antioxidative stability, detergency and low vapor ability, do not contain in their composition sulphurous compounds and metal-complexes.
Thus the results of conducted researches show that upon oligomerization of C6-C12 α-olefins with participation of the chloroaluminate type ionic liquid catalysts may be synthesized the oligomeric molecules having predominantly hybridized paraffinic-naphthenic structures, free of unsaturated fragments. This may favor to use them as a component for preparation of synthetic and semisynthetic base oil compositions without use of hydrogenation step or providing it in a mild condition.
With that end in view hydrogenation of oligododecenenaphtenic oil was conducted using industrial Ni/Cr/ Pd catalyst at Р = 30 атм., Т = 120˚C, time = 4 h. It has been found that even in that comparatively hard conditions the VI indexes of the tested samples do not change practically after hydrogenation (VI = 159 before, and 158 after hydrogenation). But some destruction of the high molecular weight molecules takes place during hydrogenation.
In the DSC curves the shift in the position of high molecule peak at VR = 10 - 12 to VR = 14 takes place. After hydrogenation the share of the oligomer molecules with VR = 14 decreases. The destruction process leads to some changes in the thermal indexes of oligomerization product oligododecenenaphtenic defined by DSC method (Termoeleсtron Corporation Q-20, USA). After hydrogenation of oligododecenenaphtenic the heat flow value (Wt/g) decreases a little (Figure 4). The hydrogenated product characterizes by less induction period and more complicated course of its oxidation (Figure 5), which confirms occurrence of destruction process taken place in the hydrogenation conditions.
5. Conclusions
C6-C12 α-olefins oligomerization in the presence of chloroaluminate ionic liquids and their combination with Ti-containing complexes used as modifiers have been studied. The structure, molecular-weight characteristics and low-temperature properties of produced oligomers have been examined by means of IR, PMR spectroscopy, size exclusion chromatography and the DSC.
It was shown that in all cases of using ILCs, the obtained oligomers contain predominantly hybrid hydro-
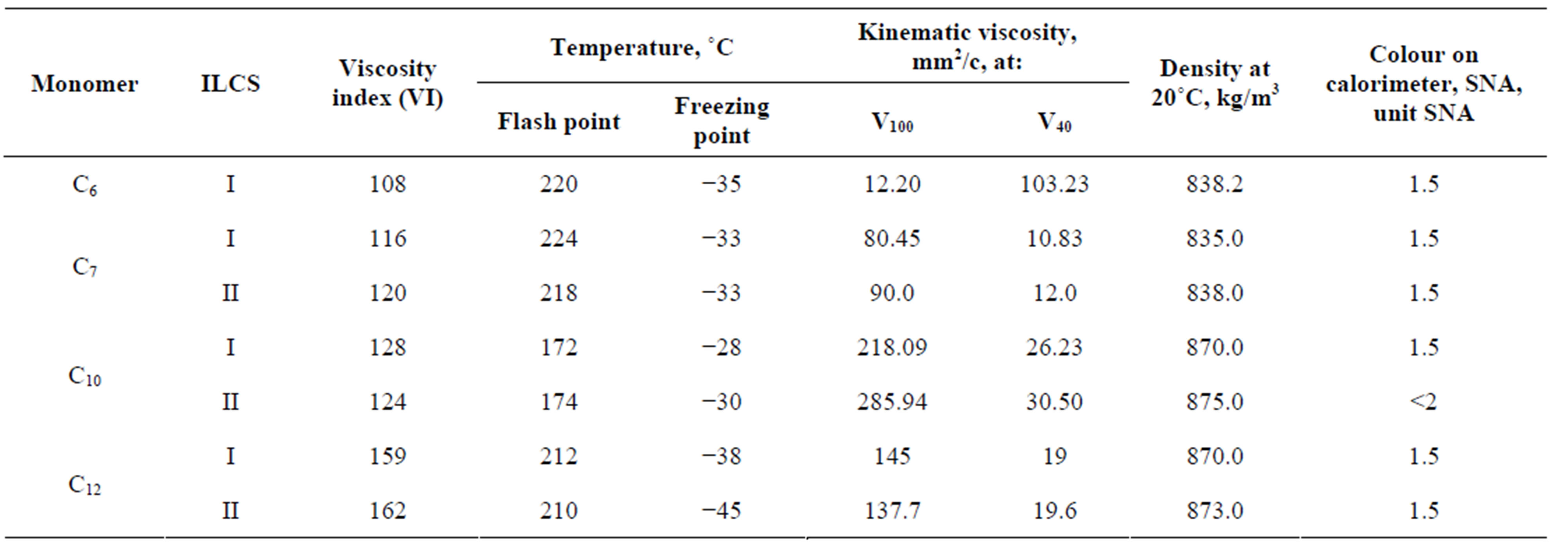
Table 6. Physical - chemical properties of the synthesized oligoalkylnaphtenic oils.
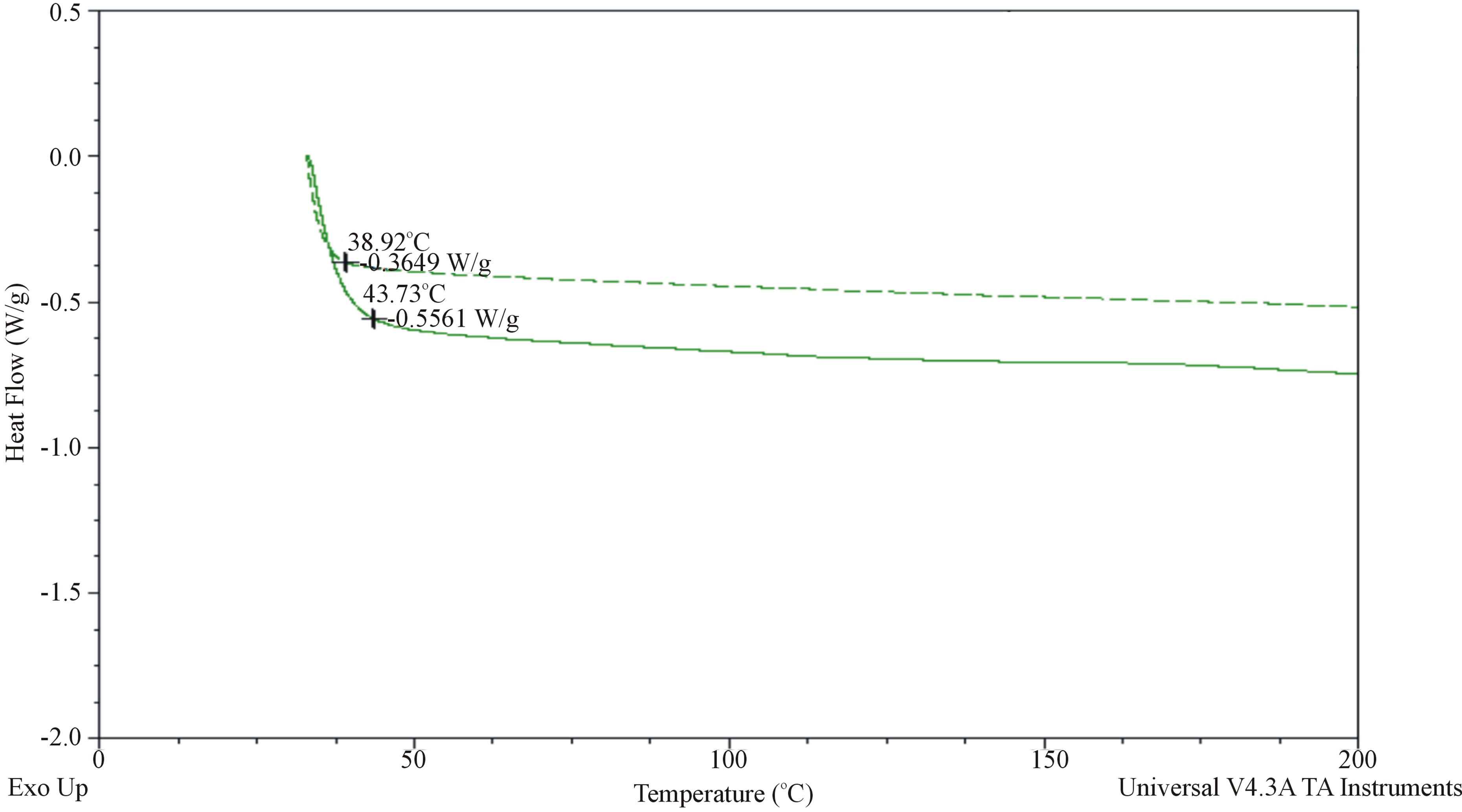
Figure 4. DSC thermo-stability curves (in nitrogen atmosphere) of oligododecenenaphtenic ODF before (---) and after (-) hydrogenation.
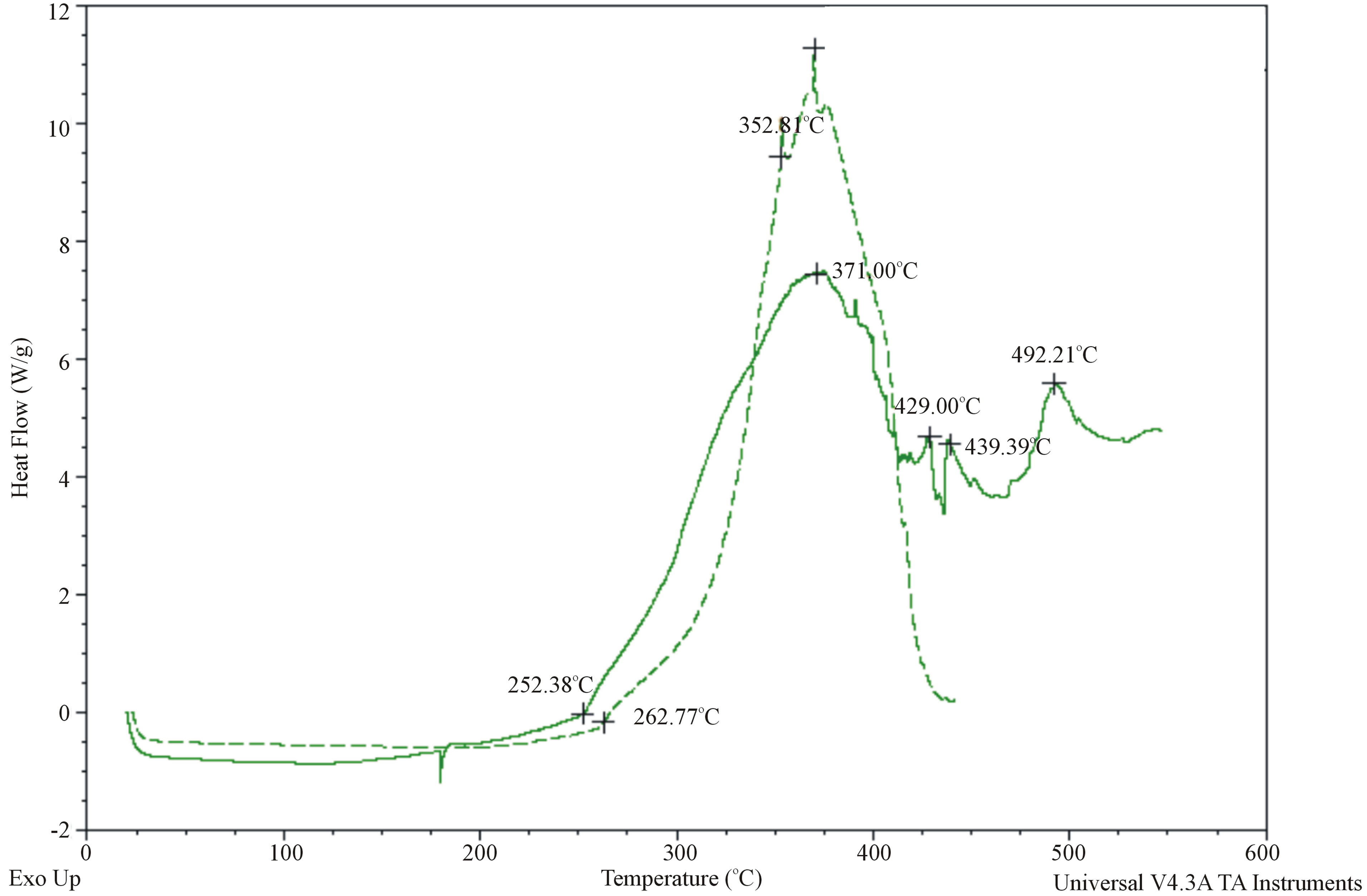
Figure 5. DSC thermo-oxidation (in oxygen atmosphere) curves of non-hydrogenated(---) and hydrogenated (-) oligododecenenaphtenic.
carbons of paraffinic-naphthenic structure with long alkyl radical branching termed as oligoalkylnaphthenic hydrocarbons.
The synthesized oligoalkylnaphtenic oils are characterized by low density values, high viscosity and viscosity index (≥108), low freezing point (−33˚C - −45˚C), high ignition temperature (≥172˚C), good antioxidative stability, detergency and low volatility. They may be used as the high viscosity base oils with good thermooxidative stability or as a component for preparation of the synthetic and semisynthetic base oil compositions without use of additional hydrogenation step or by conducting it in a mild condition.
REFERENCES
- J. D. Holbrey and K. R Seddon, Clean Product and Processes, Vol. 1, 1999, pp. 223-236.
- A. H. Azizov and Z. H. Asadov, “Production and Usage of α-Olefins and Advantages in the Metal-Complex Catalyzed Selective Oligomerization of Ethylene,” Processes of Petrochemistry and Oil Refining, Vol. 1, 2003, pp. 44- 67.
- T. Welton, “Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis,” Chemical Reviews, Vol. 99, No. 8, 1999, pp. 2071-2083. http://dx.doi.org/10.1021/cr980032t
- A. H. Azizov, “Preparation Methods and Solvent Properties of Low Temperature Ionic Liquids,” Process of Petrochemistry and Oil Refening,” Vol. 1, No. 8, 2002, pp. 90-115.
- A. H. Azizov, “Low Temperature Ionic Liquids in Organic and Petrochemical Synthesis,” Processes of Petrochemistry and Oil Refining, Vol. 2, No. 9, 2002, pp. 6-48.
- R. V. Aliyeva, “Ionic Liquids in Catalysis Petrochemical Processes,” Processes of Petrochemistry and Oil Refining, Vol. 1, No. 16, 2004, pp. 26-46.
- R. V. Aliyeva, “Ionic Liquids during Separation and Extraction,” Processes of Petrochemistry and Oil Refining, Vol. 2, No. 25, 2006, pp. 30-39.
- A. A. Xanmetov, Kh. H. Asgerova, R. V. Aliyeva and A. H. Azizov, “Ionic Liquids to Oligomerization and Polymerization Processes of Olefins,” Processes of Petrochemistry and Oil Refining, 11, 42, 2010, pp. 309-328 (in Russian).
- P. Wasserscheid, “Green Industrial Applications of Ionic Liquids,” Kluwer Academic Publishers, Boston, 2003, pp. 29-47.
- K. R. Seddon, “Ionic Liquids for Clean Technology,” Journal of Chemical Technology and Biotechnology, Vol. 68, No. 4, 1997, pp. 351-356. http://dx.doi.org/10.1002/(SICI)1097-4660(199704)68:4<351::AID-JCTB613>3.0.CO;2-4
- H. Olivier, “Recent Developments in the Use of NonAqueous Ionic Liquids for Two-Phase Catalysis,” Journal of Molecular Catalysis A, 146, 1999, pp. 285-289.
- H. Olivier, J. A. Chodorge and P. Travers, “Olefin Oligomerization with Homogeneous Catalysis,” Petroleum Technology Quarter (PTQ), Autumn, 1999, p. 141.
- A. H. Azizov, et al., “Selective Synthesis and the Mechanism of Formation of the Oligoalkylnaphthennic Oils by Oligocyclization of 1-Hexene in the Presence of IonicLiquid Catalysts,” Applied Catalysis A: General, Vol. 375, 2010, pp. 70-77.
- N. R. Bektashi, A. M. Mustafayev, Qusyynov, et al., “Chromatographic Investigation of Regularities of Synthesis of Halogen-Containing Unsaturated Polyesters,” Journal Prikladnoy Chimii, Vol. 84, No. 7, 2011, pp. 1211- 1217 (in Russian).
- К. Nakanisi, “Infrakrasnie Sprctri I Stroenie Organi- çeskixch Soedineniy, М.,” 1965, p. 210 (in Russian).
- L. Bellami, “The Infra-Red Spectra of Complex Molecules,” Publishing House of Foreign Literature, Moscow City, 1963, p. 25 (in Russian).
- E. Breitmayer, “Carbon-13 NMR Spectrocopy,” 3rd Edition, VCH, Weinheim, pp. 183-196.
- “Nefteprodukti Metodi Ispitaniy Part 1, İzdatelstvo Standartovç Moskva,” 1977 (in Russian).
- B. V. Belyanin and V. N. Erikh, “Tekhniçeskiy Analiz Nefteproduktov i Qaza,” Leninqrad, Chimiya, 1979, pp 111-112 (in Russian).
NOTES
*Corresponding author.

