World Journal of AIDS
Vol. 2 No. 1 (2012) , Article ID: 18021 , 9 pages DOI:10.4236/wja.2012.21004
Impact of Switch to Fosamprenavir and Addition of Lovaza® for Treatment of Hypertriglyceridemia in HIV-Infected Subjects on Antiretroviral Therapy
![]()
1Private Practice, Bakersfield, USA; 2Pacific Oaks Medical Group, Beverly Hills, USA; 3AIDS Healthcare Foundation, Los Angeles, USA; 4Statworks, Research Triangle Park, Durham, USA; 5GlaxoSmithKline, Research Triangle Park, Durham, USA.
Email: *felizarta@pol.net
Received August 22nd, 2011; revised October 29th, 2011; accepted November 14th, 2011
Keywords: Fish Oil; Fosamprenavir; HIV; Hypertriglyceridemia; Lovaza; Switch; Triglycerides
ABSTRACT
Background: Managing hypertriglyceridemia in HIV-infected patients often requires multiple pharmacologic strategies. Many protease inhibitors (PIs), one of 6 classes of drugs used to treat HIV, have been associated with hypercholesterolemia and drug interactions. For this study, we examined a dual strategy to manage hypertriglyceridemia in HIVinfected patient taking PIs: 1) switching patients to fosamprenavir (FPV), a PI with fewer drug interactions, and 2) adding prescription fish oil (LOVAZA®), which has been shown to reduce triglycerides. Methods: This multicenter, 24-week study enrolled 36 patients virologically suppressed (HIV-1 RNA < 50 copies/mL) on PI-containing therapy with screening triglyceride levels of 200 - 1200 mg/dL and LDL cholesterol levels ≤ 160 mg/dL. At baseline, patients were switched to ritonavir (RTV)-boosted fosamprenavir (FPV 1400 mg/RTV 100 mg QD) and any lipid-lowering agents were stopped. At Week 6, LOVAZA 4 g QD was added. Results: Five patients prematurely discontinued due to adverse events (2), non-compliance, lost-to-follow up, and protocol violation. Median triglyceride concentration was 303 mg/dL at screening, 262 mg/dL at baseline, 290 mg/dL at Week 6 (+8% from baseline), and 218 mg/dL at Week 24 (–30% from Week 6). At Week 24, 39% (12/31) of patients had triglycerides < 200 mg/dL. Among patients reaching Week 24, 100% (31/31) and 90% (28/31) had HIV-1 RNA < 400 and < 50 copies/mL, respectively. Conclusions: In this study, a switch to FPV/RTV followed by LOVAZA decreased median triglyceride levels and modestly increased the percentage of patients with triglyceride levels < 200 mg/dL while maintaining virologic suppression in HIV-infected subjects with hypertriglyceridemia. Our data suggest that baseline PI may affect the likelihood of achieving triglycerides < 200 mg/dL after 18 weeks on study. A larger study would be needed to understand the relative contributions of choice of protease inhibitor and LOVAZA to triglyceride concentrations in HIV-infected patients.
1. Introduction
Ritonavir (RTV)-boosted protease inhibitor (PI)-containing regimens are favored by many clinicians because of their virologic potency, strong immunologic response, durability, and high barrier to resistance [1]. However, boosted PI regimens are also associated with gastrointestinal side effects, insulin resistance, and dyslipidemia, particularly hypertriglyceridemia (triglyceride blood concentration ≥ 150 mg/dL) [1].
Hypertriglyceridemia is an independent risk factor for coronary heart disease in both the general population [2] and among people living with HIV [3]. While first-line therapy for hypertriglyceridemia involves lifestyle changes (e.g., diet, exercise, weight management) [2], such interventions often require significant effort from the patient and are unlikely to be sustained long-term [4]. The use of lipid-lowering agents is increasing among the HIV-infected population [5], but the choice of medication is limited by drug-drug interactions and overlapping toxicities between the lipid-lowering agents and antiretroviral therapy (ART) [3,6].
In particular, there are known pharmacologic interacttions between many PIs and HMG-CoA reductase inhibitors (statins) that increase the plasma concentrations of the statins to unacceptably high levels [1]. Fibrate therapy in patients with HIV infection has been associated with 40% - 50% decreases in serum triglyceride levels [7-9], but treatment guidelines recommend fibrates only in patients with very high triglyceride levels (>500 mg/dL) or when combined with a statin [2,3]. Extendedrelease niacin has also had positive effects on triglycerides in HIV-infected patients [10], but concerns regarding insulin resistance, cutaneous flushing, gastrointestinal upset, and hepatotoxicity highlight the need for other treatment strategies for hypertriglyceridemia [2,3].
One such strategy utilizes omega-3 fatty acids (fish oils), particularly long-chain, polyunsaturated eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) [11], to reduce the synthesis and secretion of very-lowdensity lipoprotein (VLDL) particles and increase triglyceride removal from VLDL and chylomicron particles through upregulation of enzymes (i.e., lipoprotein lipase) [12]. Data from multiple studies have shown that omega- 3 fatty acids are effective in reducing serum triglycerides by 20% - 50% in the general population [13-15], particularly in patients with very high triglyceride levels (>500 mg/dL), and are associated with low rates of adverse events [11].
While omega-3 fatty acids are available over-thecounter in the United States, the number of pills required to achieve a significant decrease in triglycerides can be daunting. LOVAZA® is a prescription combination of highly purified omega-3-acid ethyl esters that contains EPA 465 mg, DHA 375 mg, and other omega-3 fatty acids for a total of at least 900 mg of omega-3 fatty acids in each 1-g gel capsule. The normal daily dose of LOVAZA is 4 g (4 capsules) per day [16].
The current study examines the therapeutic effect of two novel approaches for the management of hypertriglyceridemia: switching HIV-infected patients who are virologically suppressed on PIs to fosamprenavir (FPV), a PI with fewer drug-drug interactions; and adding LOVAZA, a purified formulation with a lower pill burden compared with non-prescription fish oil capsules. In this study, FPV 1400 mg once daily is boosted with RTV 100 mg once daily, which has been shown to provide similar efficacy, safety, and pharmacokinetic exposure as FPV/RTV 1400/200 mg daily [17-19].
2. Methods
BuLLET was a 24-week, single-arm, open-label, prospective trial examining the effect of a switch to fosamprenavir/ritonavir (FPV/RTV) and the addition of fish oil (omega-3-acid ethyl esters) in virologically suppressed HIV-infected patients with hypertriglyceridemia on other PI-containing regimens. At baseline, patients were switched from their current PI to FPV (LEXIVA®, ViiV Healthcare Limited, Research Triangle Park, NC) 1400 mg QD and RTV (Norvir®, Abbott Laboratories, North Chicago, IL) 100 mg QD. Background therapy was not changed. After a 6-week lead-in period, fish oil (LOVAZA, GlaxoSmithKline, Research Triangle Park, NC) 4 g QD was added for the duration of the 18-week treatment period. Throughout the study, patients maintained a diet low in saturated fat and an exercise program.
Enrolled patients were ≥18 years of age, had been taking a stable ART regimen consisting of a firstor secondline PI plus optimized background therapy for ≥3 months prior to screening, and had HIV-1 RNA < 50 copies/mL at screening and for ≥3 months prior to screening. At screening, patients were required to have fasting serum triglycerides ≥ 200 mg/dL (but <1200 mg/dL), fasting LDL cholesterol ≤ 160 mg/dL, CD4 cell count > 50 cells/ mm3, and be participating in a lipid-lowering diet and exercise program for ≥28 days. For females of childbearing potential, appropriate use of at least one form of non-hormonal contraception was required during and 4 weeks after the end of the study. All patients provided written informed consent to participate in the study, and the protocol was approved by the institutional review board for each study site.
Exclusion criteria included: significant laboratory abnormalities in hemoglobin, platelets, prothrombin time, absolute neutrophil count, serum creatinine, total bilirubin, alanine or aspartate aminotransferase, thyroid stimulating hormone, or lipase ≤ 28 days prior to baseline; any grade 4 laboratory abnormality at screening; current treatment with fosamprenavir or amprenavir; use of a second RTVboosted PI for reasons of virologic failure; known cardiovascular disease, congestive heart failure, uncontrolled hypertension, or risk for atherosclerotic disease; and pregnancy or breastfeeding. Use of lipid-lowering agents (either prescription or over-the-counter medications and supplements) was not permitted during the study. Any lipid-lowering agents in use prior to the study were discontinued at baseline. Patients who had previous resistance testing could not have evidence of resistance to FPV.
Patients were evaluated at screening, baseline, and at weeks 4, 6, 12, 18, and 24, and routine biochemistry, hematology, and immunology tests were conducted at each assessment. HIV-1 RNA was measured using the standard or ultrasensitive Roche Amplicor HIV-1 Monitor assay (version 1.5; Roche Diagnostics, Branchburg, New Jersey, USA). All laboratory tests were performed centrally by Quest Diagnostics (Van Nuys, California, USA). AEs and laboratory toxicities were graded using the 2004 Division of AIDS Toxicity Grading Scale. Virologic failure was defined as having confirmed virologic rebound (HIV-1 RNA ≥ 400 copies/mL).
The primary safety endpoint of the study was the percentage of patients with fasting triglycerides < 200 mg/dL at Week 24. The primary efficacy endpoint was the percentage of patients with HIV-1 RNA < 50 copies/mL at Week 24. Secondary endpoints included changes in other lipid parameters and inflammatory biomarkers, the incidence, severity, and causality of adverse events and laboratory abnormalities, and changes from baseline in CD4 cell count.
A sample size of 50 patients was planned for this study based on practical considerations. Statistical analyses for fasting triglycerides and HIV-1 RNA were performed using the intent-to-treat: exposed (ITT:E) population that included any enrolled patient who took at least one dose of study medication. Two analyses were used: missing or discontinuation equals failure analyses (MD = F), in which missing assessments or assessments taken after discontinuation from study drugs were considered failures; and observed analyses, in which missing assessments at any scheduled time point were considered unevaluable and were not imputed. The primary endpoint was the number and proportion of subjects with fasting triglycerides < 200 mg/dL at Week 24 (with 95% confidence intervals determined by Clopper-Pearson method). Results were stratified by baseline fasting triglyceride levels < 200 mg/dL, 200 - 499 mg/dL, and 500 - 1200 mg/dL.
Statistical analysis for all other secondary endpoints and safety evaluations were based on the observed analyses. Changes from baseline were assessed using the median change from baseline with 95% confidence intervals determined by the Jeffreys method [20]. Most safety results used descriptive summary statistics.
3. Results
3.1. Patient Characteristics and Accountability
Thirty-six patients with a median age of 48 years were enrolled in the study (Table 1). Almost all were white
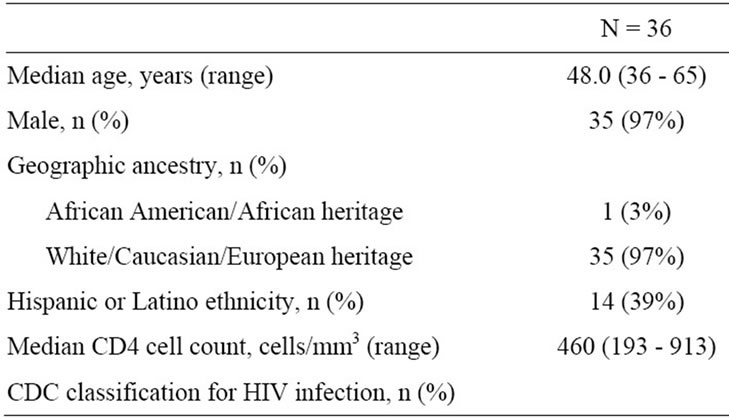



Table 1. Baseline patient demographics, characteristics, and medical history.
males, but 39% (14/36) of patients self-identified as Hispanic or Latino. Median baseline CD4 cell count was 460 cells/mm3, but 31% (11/36) of patients had CDC category C disease. At baseline, median fasting triglycerides ranged from 106 to 1004 mg/dL. Ten patients (28%) had a decrease in fasting triglycerides from ≥200 mg/dL at screening to <200 mg/dL at baseline. Most patients (28/36, 78%) were overweight or obese (body mass index ≥ 25), and 89% (32/36) had hypercholesterolemia at baseline. Approximately one-third of patients (11/36, 31%) had a history of hypertension, and one patient (3%) had type II diabetes.
Thirty-one of 36 patients (86%) completed the 24-week study. Between baseline and Week 6, 1 patient prematurely discontinued due to an adverse event. Between Week 6 and Week 24, 4 patients prematurely discontinued the study due to adverse event (n = 1), non-compliance (n = 1), lost to follow-up (n = 1), and investigator discretion (for protocol violation; n = 1).
3.2. Fasting Triglycerides
The percentage of patients with fasting triglycerides < 200 mg/dL increased during the study (Table 2). At Week 24, 33% (12/36) of patients had triglycerides < 200 mg/dL. Ten subjects (28%) had a decrease in triglyceride levels between screening (≥200 mg/dL) and baseline (<200 mg/dL). Seven of these 10 patients completed the study: 4 with triglycerides below 200 mg/dL and 3 with triglyceride levels between 237 and 277 mg/dL.
Median triglyceride concentration was 262 mg/dL at baseline (when lipid-lowering agents were stopped), and 290 mg/dL at week 6 (8% increase) when LOVAZA was initiated. By week 24, median triglycerides were 218 mg/dL (30% decrease from week 6). Between baseline and Week 24, there were no statistically significant changes in lipids (total, HDL, and LDL cholesterol, triglycerides, and total: HDL cholesterol ratio) (Table 3). However, between Weeks 6 and 24, there was a statistically significant increase in LDL cholesterol and statistically significant decreases in triglycerides and total:HDL cholesterol ratio.
Examining NMR lipoproteins, there were statistically significant increases from baseline to Week 24 in total LDL, small LDL, very small LDL, and VLDL particles and statistically significant decreases in total HDL particles, VLDL size, and large VLDL/chylomic. Between Weeks 6 and 24, there was a statistically significant increase in small LDL and statistically significant decreases in VLDL size, large VLDL/chylomic, and VLDL triglycerides.
The percentage of patients with fasting triglycerides < 200 mg/dL at each time point varied by baseline PI (Figure 1). Comparing baseline with Week 24, more patients who were switched from lopinavir/RTV had fasting triglycerides < 200 mg/dL, fewer patients who were switched from darunavir/RTV had fasting triglycerides < 200 mg/dL, and approximately the same percentage of patients who switched from atazanavir/RTV or other PI had had fasting triglycerides < 200 mg/dL. Between baseline and Week 6, median triglyceride levels increased for patients switched from atazanavir/RTV, darunavir/RTV, or other PI and decreased slightly for patients switched from lopinavir/RTV. Between Week 6 and Week 24, median triglyceride levels decreased for all baseline PIs.
3.3. Virologic and Immunologic Results
Most patients (90%, 28/31) who completed the study maintained HIV-1 RNA < 50 copies/mL (Figure 2), and all patients who completed the study had HIV-1 RNA < 400 copies/mL. There was no relationship between virologic suppression and baseline triglyceride strata or baseline PI. One non-compliant patient experienced confirmed virologic failure at Week 6. At time of virologic failure, no major treatment-emergent resistance associated mutations were detected in virus isolated from this patient. In this virologically suppressed population, median CD4 cell count increased by 55 cells/mm3 between baseline and Week 24.
3.4. Safety and Tolerability
Both FPV/RTV and LOVAZA were generally well-tolerated, with few reported adverse events (AEs). Between
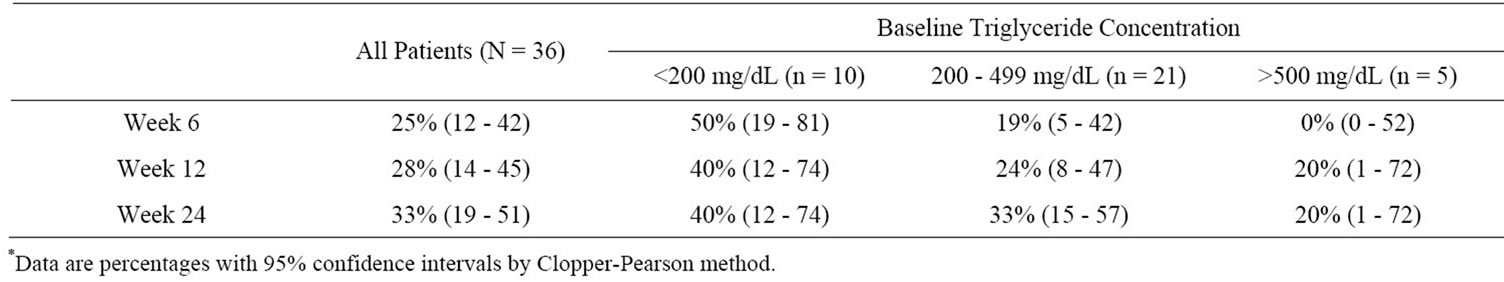
Table 2. Percentage of patients with triglycerides < 200 mg/dL at Week 24, stratified by baseline triglyceride concentration; ITT, MD = F analysis*.
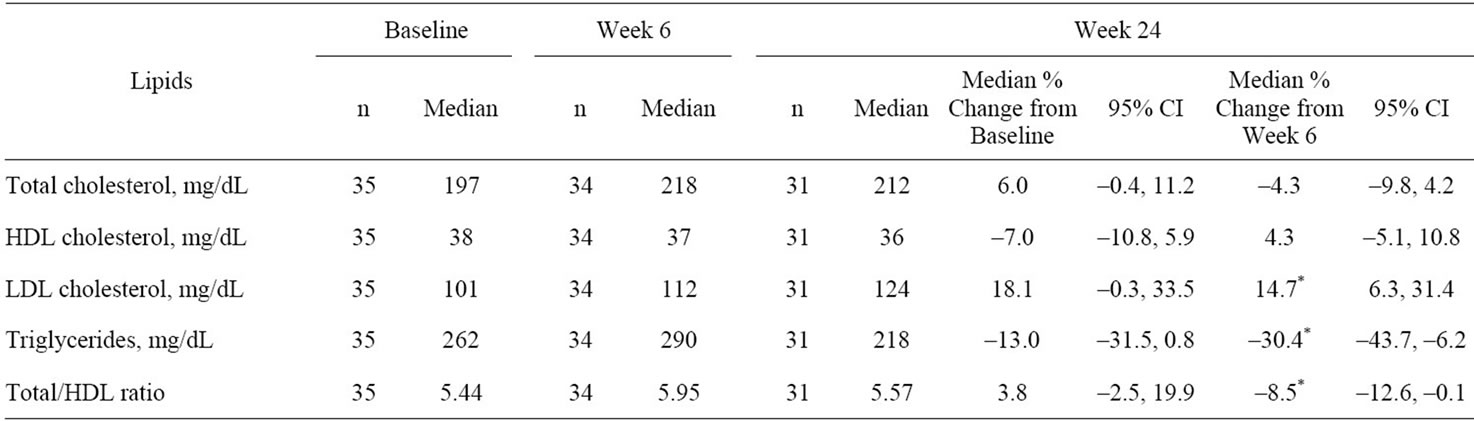
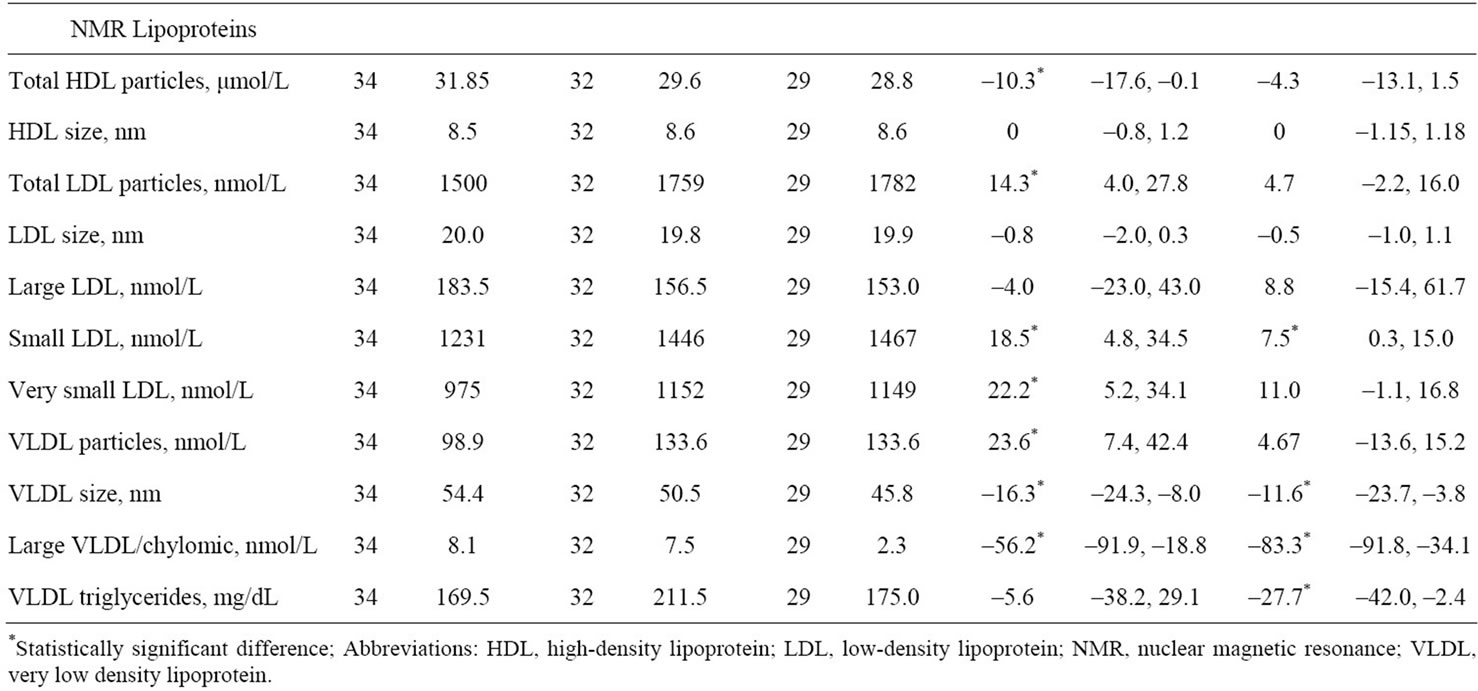
Table 3. Change from baseline in median (95% confidence intervals) in lipid and NMR lipoprotein parameters.
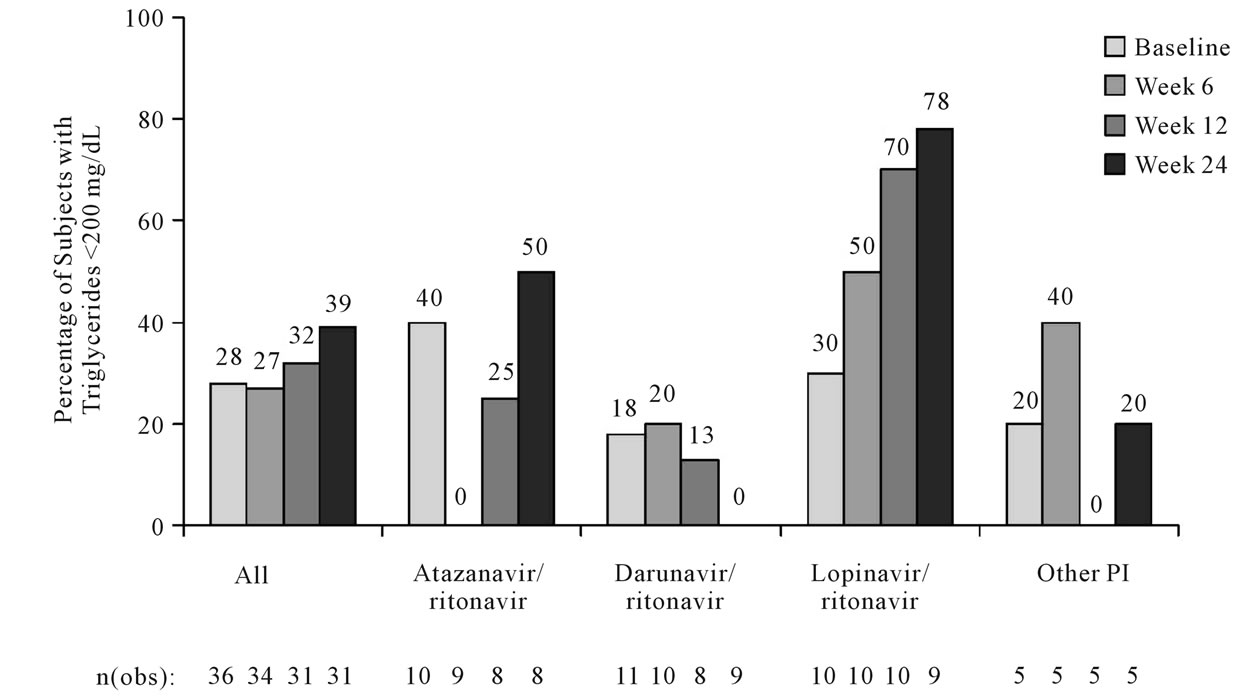
Figure 1. Percentage of patients with fasting triglycerides < 200 mg/dL over time stratified by baseline protease inhibitor (ITT-exposed population, observed analysis).
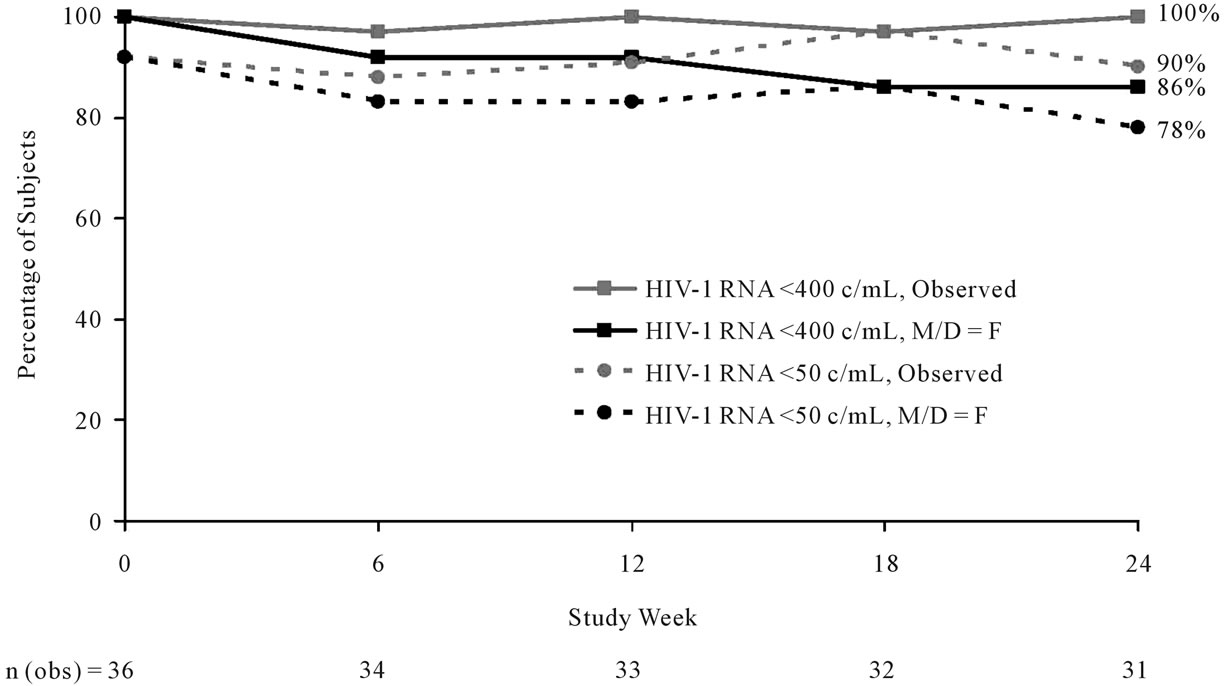
Figure 2. Percentage of patients with HIV-1 RNA < 50 copies/mL or <400 copies/mL over time (ITT-exposed population, M/D = F and observed analyses).
baseline and Week 6, one patient reported diarrhea and gastrointestinal upset and withdrew from the study. Between Weeks 6 and 24, 5 patients reported treatmentemergent grade 2 - 3 AEs. The most common AE was hypercholesterolemia (2 patients with grade 2 and 2 patients with grade 3), which led to study discontinuation in one patient. Other AEs (all reported in the same patient) were influenza, bronchitis, and vomiting. There were no grade 4 or serious AEs. One-half of patients experienced a grade 3 - 4 laboratory abnormality, the most common being increases in LDL cholesterol [7 (19%)], triglycerides [6 (17%)], total cholesterol [5 (14%)], and total bilirubin [4 (11%)].
Results with inflammatory biomarkers were mixed, and, due to the small sample size, the statistical significance of the changes was not calculated. The median percentage change from baseline to Week 24 was negative for interleukin-6 (–18%), positive for B-100/A-1 ratio (+16%), and unchanged for high sensitivity C-reactive protein (0).
4. Discussion
In this population of virologically suppressed patients taking a variety of PIs, switching to FPV/RTV at baseline and adding LOVAZA at Week 6 were associated with decreased median triglyceride concentrations in all baseline PI groups, and a larger percentage of patients achieved triglycerides < 200 mg/dL at Week 24 (39%) compared with baseline (28%) using an observed analysis. This regimen was also associated with continued virologic suppression (HIV-1 RNA < 50 copies/mL), continued immunologic improvement, and few adverse events. Consistent with studies in the general population [13,14], the decrease in triglycerides was accompanied by statistically significant decreases in VLDL size, large VLDL/chylomic particles, and VLDL triglycerides.
These results are similar to three previous trials examining the effects of fish oil in the HIV-positive population. The first trial, published by Wohl, et al., randomized 52 HIV-infected patients on ART with triglycerides > 200 mg/dL to receive nutritionist-administered dietary and exercise counseling with or without fish oil supplementation (approximately 3 g/day) [4]. Mean baseline triglyceride levels were high: 461 mg/dL for the fish oil group and 502 mg/dL for the diet and exercise group. After 16 weeks, the group receiving fish oil experienced a 19.5% mean decrease in triglycerides (a statistically significant change from baseline) compared to a 5.7% decrease in the group receiving only counseling. Mean LDL levels increased significantly (22.4%) in the fish oil group and the diet and exercise group (18.4%). These results are generally similar to our study, in which there were statistically significant changes in triglycerides (–15.5%) and LDL (+19.9%) between Week 6 and Week 24 despite a much lower mean baseline triglyceride level (312 mg/dL).
DeTruchis, et al. conducted a double-blind trial of 122 HIV-infected patients with triglyceride levels between 200 and 1000 mg/dL [21]. After a 4-week period of diet intervention alone, patients were randomized to receive 6 g of fish oil or placebo for 8 weeks. Median baseline triglyceride levels were 400 mg/dL for the fish oil group and 410 mg/dL for the placebo group. After 8 weeks, the median percent change in triglycerides was –25.5% for the fish oil group and +1.0% for the placebo group, a statistically significant difference that was similar in magnitude to the results of our trial. Data on changes in LDL cholesterol were not reported, although total cholesterol was unchanged in the fish oil group (–0.4%). Our study found a larger percent decrease in total cholesterol (–4.3% at from Week 6 to Week 24), but the change was not statistically significant.
The third previous study of fish oil in HIV-infected patients, published by Gerber, et al., was an open-label trial in patients with baseline fasting triglyceride concentrations ≥ 400 mg/dL [22]. In this study, unlike the other two studies, results were analyzed with respect to screening triglyceride values. The 100 patients, who had a median screening triglyceride level of 668 mg/dL, were randomized to 3 g of fish oil or 160 mg of fenofibrate per day. After 8 weeks, triglycerides had decreased by 46% in the fish oil group and 58% in the fenofibrate group. Both changes were statistically different from screening. LDL levels increased significantly in the same time period (+37% for fish oil group, +46% for the fenofibrate group) but total cholesterol levels were stable. However, very few patients achieved triglycerides < 200 mg/dL (8.5% of the fish oil group, 16.7% of the fenofibrate group), presumably due to the high screening concentrations. Our study included 4 patients with baseline triglycerides > 500 mg/dL, and only 1 (25%) had triglycerides < 200 mg/dL at the end of the study.
While the previous trials of fish oil in the HIV-infected population included patients with considerably higher pre-treatment triglyceride levels, the numerical results of the three previous trials are very similar to our study. All four studies found a statistically significant decrease in triglyceride concentrations, a statistically significant increase in LDL cholesterol, and no change in total cholesterol. These results are also similar to observations in the general population, where fish oil monotherapy (not combined with fibrates or statins) has been associated with an approximately 20% to 50% decrease in triglycerides and a 10% to 50% increase in LDL [23]. As reviewed by McKenney and Sica, this increase in LDL is hypothesized to occur because the presence of fish oil decreases the number of VLDL particles and increases the rate at which VLDL particles are converted to LDL particles [11].
Our study also incorporated a change in PI therapy, offering an opportunity to assess the impact of different PIs. However, these results are difficult to interpret because of two competing modifications at baseline (a change in daily RTV dose for some patients and the cessation of any lipid-lowering agents) and the small sample size for each PI.
For patients originally taking atazanavir/RTV and darunavir/RTV, the RTV dose was unchanged from 100 mg per day. The larger triglyceride increase in patients formerly taking darunavir/RTV was unexpected, as a recent study of 51 HIV-infected patients with hypertriglyceridemia on lopinavir/RTV or FPV/RTV found similar decreases in triglyceride levels over 24 weeks when switched to atazanavir/RTV (–99 mg/dL) or darunavir/ RTV (–126 mg/dL) [24]. One explanation may be the larger range of triglyceride levels at baseline for darunavir/RTV (129 - 1004 mg/dL) compared with atazanavir/ RTV (172 - 696 mg/L) in this relatively small patient population.
This study design did not capture the use of lipidlowering agents at baseline, a significant limitation of this study. Other limitations include the self-reported nature of adherence to the lipid-lowering diet and exercise program, and, although the need for fasting was stressed in the protocol, the possibility that not all patients followed the guidelines at every time point. This study was also challenging in terms of the inherent variability of triglyceride measurements, as evidenced by the large percentage of patients who had triglycerides ≥ 200 mg/dL at screening but <200 mg/dL at the baseline visit no more than 28 days later. Studies in healthy individuals have documented a 25% intra-individual variation in triglyceride measurements in the fasting state, with variations up to 65% for non-fasted blood draws [25]. In addition, triglyceride measurements are affected by sample collection and handling procedures, including storage and shipping, patient posture during collection, venous occlusion, and the use of anticoagulants [25]. Despite our efforts to standardize procedures, we cannot rule out the possibility of these issues confounding our data.
5. Conclusion
In this study of 36 HIV-infected patients with hypertriglyceridemia at screening, a switch to FPV 1400 mg/ RTV 100 mg QD followed by the addition of LOVAZA 4 g QD decreased median triglyceride levels and modestly increased the percentage of patients with triglyceride levels < 200 mg/dL. Virologic suppression was maintained in >90% of patients who completed 24 weeks on study. Our data suggest that baseline PI may affect the likelihood of achieving triglycerides < 200 mg/dL after 18 weeks on study. A larger study would be needed to understand the relative contributions of choice of protease inhibitor and LOVAZA to triglyceride concentrations in HIV-infected patients.
6. Acknowledgements
This investigator-initiated study was funded by GlaxoSmithKline. We gratefully acknowledge the patients who participated in this study. We thank the other site investigators (Clayton Barbour, MD, Daniel Coulston, MD, Peter Ruane, MB, and John Stansell, MD) and the study coordinators along with the teams from GlaxoSmithKline (Terri Becom, Harold Kong, PharmD, Sue Pippin, and Christina Frederick-Fuerlein), Medistaf (Lanny Ballard, Vicky Fantry, and Bruce Uveges) and Statworks (Kim New, Suzanne Hackett, and Thomas Clinch). We also thank Katherine A. DeBruin, PhD, of Kodiak Consulting Group, Inc., for her writing and editorial assistance in the preparation of the manuscript.
REFERENCES
- DHHS Panel on Antiretroviral Guidelines for Adults and Adolescents, “Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents,” 10 January 2011. http://aidsinfo.nih.gov/contentfiles/AdultandAdolescentGL.pdf
- National Cholesterol Education Program (NCEP), “Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III), Final Report,” National Institutes of Health Publication, No. 02-5215, 2002.
- M. P. Dube, J. H. Stein, J. A. Aberg, et al., “Guidelines for the Evaluation and Management of Dyslipidemia in Human Immunodeficiency Virus (HIV)-Infected Adults Receiving Antiretroviral Therapy: Recommendations of the HIV Medicine Association of the Infectious Disease Society of America and the Adult AIDS Clinical Trials Group,” Clinical Infectious Diseases, Vol. 37, No. 5, 2003, pp. 613-627. doi:10.1086/378131
- D. A. Wohl, H. C. Tien, M. Busby, et al., “Randomized Study of the Safety and Efficacy of Fish Oil (Omega-3 Fatty Acid) Supplementation with Dietary and Exercise Counseling for the Treatment of Antiretroviral Therapy-Associated Hypertriglyceridemia,” Clinical Infectious Diseases, Vol. 41, No. 10, 2005, pp. 1495-1504.
- J. H. Stein, Y. Wu, H. Kawabata and U. H. Iloeje, “Increased Use of Lipid-Lowering Therapy in Patients Receiving Human Immunodeficiency Virus Protease Inhibitors,” American Journal of Cardiology, Vol. 92, No. 3, 2003, pp. 270-274. doi:10.1016/S0002-9149(03)00622-2
- M. Schambelan, C. A. Benson, A. Carr, et al., “Management of Metabolic Complications Associated with Antiretroviral Therapy for Hiv-1 Infection: Recommendations of an International AIDS Society—USA Panel,” Journal of Acquired Immune Deficiency Syndromes, Vol. 31, No. 3, 2002, pp. 257-275.
- L. Calza, R. Manfredi and F. Chiodo, “Use of Fibrates in the Management of Hyperlipidemia in HIV-Infected Patients Receiving HAART,” Infection, Vol. 30, No. 1, 2002, pp. 26-31.
- A. Rao, S. D’Amico, A. Balasubramanyam and M. Maldonado, “Fenofibrate Is Effective in Treating Hypertriglyceridemia Associated with HIV Lipodystrophy,” American Journal of the Medical Sciences, Vol. 337, No. 6, 2004, pp. 315-318. doi:10.1097/00000441-200406000-00003
- J. A. Aberg, R. A. Zackin, S. W. Brobst, et al., “A Randomized Trial of the Efficacy and Safety of Fenofibrate Versus Pravastatin in HIV-Infected Subjects with Lipid Abnormalities: AIDS Clinical Trials Group Study 5087,” AIDS Research and Human Retroviruses, Vol. 21, No. 9, 2005, pp. 757-767. doi:10.1089/aid.2005.21.757
- M. P. Dube, J. W. Wu, J. A. Aberg, et al., “Safety and Efficacy of Extended-Release Niacin for the Treatment of Dyslipidemia in Patients with Human Immunodeficiency Virus Infection: AIDS Clinical Trials Group Study A5148,” Antiviral Therapy, Vol. 11, No. 8, 2006, pp. 1081- 1089.
- J. M. McKenney and D. Sica, “Role of Prescription Omega-3 Fatty Acids in the Treatment of Hypertriglyceridemia,” Pharmacotherapy, Vol. 27, No. 5, 2007, pp. 715-728. doi:10.1592/phco.27.5.715
- W. S. Harris, M. Miller, A. P. Tighe, M. H. Davidson and E. J. Schaefer, “Omega-3 Fatty Acids and Coronary Heart Disease Risk: Clinical and Mechanistic Perspectives,” Atherosclerosis, Vol. 197, No. 1, 2008, pp. 12-24. doi:10.1016/j.atherosclerosis.2007.11.008
- B. E. Phillipson, D. W. Rothrock, W. E. Connor, W. S. Harris and D. R. Illingworth, “Reduction of Plasma Lipids, Lipoprotein, and Apoproteins by Dietary Fish Oils in Patients with Hypertriglyceridemia,” New England Journal of Medicine, Vol. 312, No. 19, 1985, pp. 1210-1216. doi:10.1056/NEJM198505093121902
- E. B. Schmidt and J. Dyerberg, “Omega-3 Fatty Acids. Current Status in Cardiovascular Medicine,” Drugs, Vol. 47, No. 3, 1994, pp. 405-424. doi:10.2165/00003495-199447030-00003
- W. S. Harris, “Nonpharmacologic Treatment of Hypertriglyceridemia: Focus on Fish Oils,” Clinical Cardiology, Vol. 22, No. 6 (Supplement), 1999, pp. 40-43.
- LOVAZA Prescribing Information (Omega-3-Acid Ethyl Esters), GlaxoSmithKline, Research Triangle Park, North Carolina, 2008.
- C. Cohen, E. DeJesus, A. LaMarca, et al., “Similar Virologic and Immunologic Efficacy with Fosamprenavir Boosted with 100 mg or 200 mg of Ritonavir in HIV-Infected Patients: Results of the LESS Trial,” HIV Clinical Trials, Vol. 11, No. 5, 2010, pp. 239-247. doi:10.1310/hct1105-239
- D. A. Parks, H. C. Jennings, C. Taylor, G. E. Pakes and E. P. Acosta, “Steady-State Amprenavir, Tenofovir, and Emtricitabine Pharmacokinetics before and after Reducing Ritonavir Boosting of a Fosamprenavir/Tenofovir/Emtricitabine Regimen from 200 mg to 100 mg Once Daily (TELEX II),” HIV Clinical Trials, Vol. 10, No. 3, 2009, pp. 160-167. doi:10.1310/hct1003-160
- C. B. Hicks, E. DeJesus, L. M. Sloan, et al., “Comparison of Once-Daily Fosamprenavir Boosted with either 100 or 200 mg of Ritonavir, in Combination with Abacavir/ Lamivudine: 96-Week Results from COL100758,” AIDS Research and Human Retroviruses, Vol. 25, No. 4, 2009, pp. 395-403. doi:10.1089/aid.2008.0231
- L. D. Brown, T. T. Cai and A. DasGupta, “Confidence Intervals for a Binomial Proportion and Asymptotic Expansions,” The Annals of Statistics, Vol. 30, No. 1, 2002, pp. 160-201.
- P. De Truchis, M. Kirstetter, A. Perier, et al., “Reduction in Triglyceride Level with N-3 Polyunsaturated Fatty Acids in HIV-Infected Patients Taking Potent Antiretroviral Therapy: A Randomized Prospective Study,” Journal of Acquired Immune Deficiency Syndromes, Vol. 44, No. 3, 2007, pp. 278-285. doi:10.1097/QAI.0b013e31802c2f3d
- J. G. Gerber, D. W. Kitch, C. J. Fichtenbaum, et al., “Fish Oil and Fenofibrate for the Treatment of Hypertriglyceridemia in HIV-Infected Subjects on Antiretroviral Therapy: Results of ACTG A5186,” Journal of Acquired Immune Deficiency Syndromes, Vol. 47, No. 4, 2008, pp. 459-466. doi:10.1097/QAI.0b013e31815bace2
- H. Bays, “Clinical Overview of Omacor: A Concentrated Formulation of Omega-3 Polyunsaturated Fatty Acids,” American Journal of Cardiology, Vol. 98, No. 4A, 2006, pp. 71i-76i. doi:10.1016/j.amjcard.2005.12.029
- D. Skiest, C. Cohen, H. Khanlou, et al., “Randomized Comparison of Darunavir/r Versus Atazanavir/r on Serum Lipids in HIV-Infected Persons on Fully suppressive lopinavir/r or fosamprenavir/r with high Serum Triglycerides. Lopinavir/r or Fosamprenavir/r Switch to Atazanavir/r or Darunavir/r (LARD),” Proceedings of the 18th Conference on Retroviruses and Opportunistic Infections, Boston, 27 February-2 March 2011, poster 817.
- E. A. Stein and G. L. Myers for the National Cholesterol Education Program Working Group on Lipoprotein Measurement, “National Cholesterol Education Program Recommendations for Triglyceride Measurement: Executive Summary,” Clinical Chemistry, Vol. 41, No. 10, 1995, pp. 1421-1426.
NOTES
*Corresponding author.

