Agricultural Sciences
Vol.5 No.5(2014), Article ID:44618,20 pages DOI:10.4236/as.2014.55043
Interaction of Carbon Dioxide Enrichment and Soil Moisture on Photosynthesis, Transpiration, and Water Use Efficiency of Soybean
Madegowda Madhu1, Jerry L. Hatfield2*
1Central Soil and Water Conservation Research and Training Institute, Research Centre, Koraput, India
2National Laboratory for Agriculture and the Environment (NLAE), Ames, USA
Email: madhupmd@gmail.com, *jerry.hatfield@ars.usda.gov
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 12 February 2014; revised 16 March 2014; accepted 2 April 2014
ABSTRACT
Soybean (Glycine max (L.) Merrill) is one of the most important oil and protein sources in the world. Interactive effect of elevated carbon dioxide (CO2) and soil water availability potentially impact future food security of the world under climate change. A rhizotron growth chamber experiment was conducted to study soil moisture interactions with elevated CO2 on gaseous exchange parameters of soybean under two CO2 concentrations (380 and 800 µmol·mol−1) with three soil moisture levels. Elevated CO2 decreased photosynthetic rate (11.1% and 10.8%), stomatal conductance (40.5% and 36.0%), intercellular CO2 concentration (16.68% and 12.28%), relative intercellular CO2 concentration (17.4% and 11.2%), and transpiration rate (43.6% and 39%) at 42 and 47 DAP. This down-regulation of photosynthesis was probably caused by low leaf nitrogen content and decrease in uptake of nutrients due to decrease in stomatal conductance and transpiration rate. Water use efficiency (WUE) increased under elevated CO2 because increase in total dry weight of plant was greater than that of water use under high CO2 conditions. Plants under normal and high soil moisture levels had significantly higher photosynthetic rate (7% to 16%) favored by optimum soil moisture content and high specific water content of soybean plants. Total dry matter production was significantly high when plants grown under elevated CO2 with normal (74.3% to 137.3%) soil moisture level. Photosynthetic rate was significantly and positively correlated with leaf conductance and intercellular CO2 concentration but WUE was significantly negatively correlated with leaf conductance, intercellular CO2 concentration and transpiration rate. However, the effect of high CO2 on plants depends on availability of nutrients and soil moisture for positive feedback from CO2 enrichment.
Keywords:Elevated Carbon Dioxide, Evaporation, Interactions, Photosynthetic Rate, Soil Moisture, Soybean, Water Use Efficiency

1. Introduction
Soybean (Glycine max (L.) Merrill) is the world agricultural economy grown primarily for oil extraction and for use as a high protein meal for animal feed [1] . The world population is expected to reach nine billion people by 2050 and production of food should increase by 70% to ensure food security, which should be achieved through increase in productivity [2] . The soybean plants show a series of changes in their morphology, physiology, and biochemistry, negatively affecting their growth which can reduce productivity by 50% [3] .
The increasing atmospheric carbon dioxide (CO2) concentration have direct and indirect effects on crop plants but CO2 is often a limiting resource in plant canopies, and it’s expected to increase photosynthetic rate, plant productivity, and water use efficiency (WUE) by reducing stomatal aperture and/or number per unit leaf area and their by decrease in transpiration [4] -[30] . However, the long-term response remains uncertain due to increase in incidence of extreme weather events [29] [31] -[36] such as drought, heat waves, and heavy precipitation and floods, making crop production more unpredictable and difficult. Yields of most agricultural crops increased under elevated CO2 concentrations; productivity increases are in the range of 15% to 41% for C3 crops and 5% to 10% for C4 crops [5] [8] [29] [37] . Increased photosynthesis and associated changes in morphology [5] [20] [38] in response to elevated CO2 increased soybean yield by 24% - 37% [39] [40] and interactions of other climate change factors on plants [41] showed alterations both in physiology, growth, and development crops such as soybean [39] , cotton [42] , and many other crops [27] . Transpiration is a vital component in soilwater-plant relationship and is of particular importance in studying possible interactions of elevated CO2 and water supply in terms of plant water use and WUE. The effect of elevated CO2 concentrations on crops varies under different soil moisture regimes [43] . However, data on the interactive effects of CO2 and soil moisture on plants are scarce and often contradictory [44] . Previous studies claim that the percentage increase in plant growth due to elevated CO2 is generally not reduced by water stress [16] [45] whereas the results of many other theoretical projections and field or greenhouse experiments suggest that the relative effects of CO2 enrichment on plants are constrained by less than optimal levels of soil moisture [5] [24] [46] -[51] .
The altered physiological and gas exchange characteristics of crop plants with climate change [52] [53] and coupled with shifts in regional scale rainfall patterns leading to decreased soil water availability in some areas of the world [33] [34] has far-reaching implications particularly in arid and semi-arid regions where water is a critical consideration affecting both growth and development of crops and ultimately impacting yield and food security [54] [55] . In this study, we grew soybean under two CO2 concentrations and three soil moisture levels, and focused on the interactive effects of elevated CO2 and soil moisture levels on canopy photosynthetic CO2 uptake, canopy transpiration, water use efficiency, intercellular CO2 concentration, and growth of soybean plants during vegetative growth stages.
2. Materials and Methods
2.1. Environment
The experiment was conducted in the controlled environmental conditions under rhizotron chambers at USDAARS National Laboratory for Agriculture and Environment (NLAE) in Ames, Iowa, USA. More details of operation and control of rhizotron chambers have been described by [56] [57] . The dimension of each soil monolith is 1 by 1 by 1.5 m deep and the soil type is Monona silt loam soil (fine-silty, mixed mesic Typic Hapludoll) from southwestern Iowa. Each rhizotron growth chamber consists of three soil monoliths. Chambers are similar to a standard plant growth chamber and have microprocessor control of temperature, humidity, and lighting such that specific diurnal, weekly, and seasonal environmental patterns can be programmed. Soil water content of the monoliths also can be monitored and controlled.
Treatment consists of two levels of CO2 concentration (380 and 800 µmol·mol−1) and three soil moisture levels (Low, Normal and High) which were studied in the controlled rhizotron environmental condition. Each rhizotron chamber was assigned with a particular level of CO2 concentration where one chamber with ambient CO2 level of 380 µmol·mol−1 and another with elevated CO2 level of 800 µmol·mol−1. In each rhizotron chamber, three soil moisture regimes viz., low (5 mm), normal (7.5 mm) and high (10 mm) were imposed. Soybean genotype namely S 21-N6 planted in each soil moisture level (soil monolith) at 60 cm between two rows by opening small furrow of 5 cm depth and placing soybean seeds at 10 cm apart on 25 Oct. 2011. After sowing, each soil monolith was irrigated to 80% field water capacity (FWC) and uniform soil moisture was maintained in the entire soil profile by daily watering to all the monoliths for initial 15 days after planting (DAP) since these rhizotron monoliths were dry before. Two rhizotron chambers were maintained at maximum temperature of 25˚C and minimum of 15˚C, and at 380 µmol·mol−1 CO2 until 50% seedling emergence. Environmental variables viz., CO2 concentration, temperature and light intensity inside each chamber were continuously monitored and temperature and light were automatically adjusted by the computer to simulate diurnal variations typical of a day. Temperature and light intensity were programmed to be the same between chambers. Only CO2 was varied between chambers, one with ambient CO2 and another with elevated CO2. The environmental sensors and controlling systems of the two chambers were calibrated before the commencement of the experiment and environmental variables were continually monitored at one minute samples and 15 minutes averages during the entire course of experiment in order to minimize the variance induced by the between-chamber heterogeneity of environmental conditions. Photosynthetically active radiation inside the growth chambers was maintained approximately at 280 - 350 µ·mol·m−2·s−1 during the course of the experiment.
2.2. Imposition of Treatments
The soil moisture treatments viz., low (5 mm), Normal (7.5 mm) and High (10 mm) were imposed from 15 DAP i.e., on 08 Nov 2011 and frequency of watering was twice in a week (Friday and Tuesday). Water was measured and applied to each soil monolith by plastic rose cane as per the soil moisture treatments. Hand weeding was done on 11 Nov 2011. Elevated CO2 level was maintained in one of the chamber after 50% seedling emergence by automatically injecting CO2 into the chamber and the level in the chambers was controlled using a CO2 delivery system and chamber vents. An individual LICOR infrared gas analyzer (LI-800 Gas Hound CO2 Analyzer, LI-COR, NE, USA) was used to monitor CO2 levels at each chamber independently. Soil water content was monitored at weekly intervals with a neutron probe through an access tube positioned in the center of each monolith.
2.3. Measurements
For each growth analysis harvest, aboveand below-ground growth measurements were assessed by destructive sampling technique of two plants (first and second sampling) and five (final sampling) randomly selected from each row in each soil monolith to measure the individual plant components. Samplings were done at 29, 44 and 58 DAP. Number of leaves per plant, total leaf area (cm2·plant−1), total above-ground dry mass per plant (g·plant−1), and leaf and stem dry mass (g·plant−1) were determined for all stages. Number of pods and dry weight of pods per plant were measured at final harvest. Leaf area was measured using the LI-3100 leaf area meter (LI-COR, Lincoln, NE). All components wise fresh weights were taken and dry weights were obtained following oven-drying to constant weight at 65˚C. Total above-ground dry biomass for each plant was obtained by adding all plant components. These values were then used for statistical and growth analysis. The specific leaf area (SLA) (cm2·g−1), leaf area ratio (LAR) (cm2·g−1) and leaf weight ratio (LWR) (g·g−1) were calculated for each sampling date as the ratio of leaf area to leaf biomass, leaf area to above-ground biomass and leaf biomass to total plant above-ground biomass, respectively.
2.4. Gas Exchange Characteristics
Measurements of net CO2 assimilation rate (A), transpiration (E), stomatal conductance (gs), intercellular CO2 concentration (Ci), were made on fully expanded youngest five leaves of soybean plants using LI-6400 Portable Photosynthesis System (Li-Cor, Lincoln, NE). Two measurements were made at 42 and 47 DAP after 72 h and 24 h of watering, respectively as per the soil moisture treatments. Measurements were performed from 14.05 to 15.12 h at 42 DAP and 12.10 to 13.12 h at 49 DAP with the following specifications/adjustments, molar flow of air per unit leaf area 499.2 µ·mol·m−2·s−1, atmospheric pressure 99.7 kPa, temperature of leaf ranged from 31.77˚C to 33.39˚C (at 42 DAP) and 29.3˚C to 32.7˚C (at 47 DAP), ambient temperature ranged from 32.24˚C to 33.85˚C (at 42 DAP) and 29.2˚C to 32.2˚C (at 47 DAP), reference CO2 concentration was 370 µ·mol·mol−1. Water use efficiency was calculated as CO2 assimilation rate to transpiration, and intrinsic WUE as CO2 assimilation rate to stomatal conductance.
2.5. Statistical Analysis
Statistical analysis was conducted by using analysis of variance (ANOVA) (Web Based Agricultural Statistics Software Package (WASP-2)). Effects of CO2, soil moisture levels, and interactions were tested using the least significant difference tests at P = 0.05. Correlation between fresh weight of soybean plant and various gas exchange parameters were also analyzed. Data at each observational date were analyzed separately. Results are presented in tabular and graphical representation with the standard error bars.
3. Results
3.1. Photosynthetic Rate or Net Assimilation Rate
Net assimilation rate (NAR) of soybean was highly significant due to CO2 levels and decrease in photosynthetic rate by 11.1% and 10.8% under elevated CO2 compared to ambient CO2 level at 42 and 47 DAP (P ≤ 0.01), respectively (Table 1 & Figure 1). Plants under normal and high soil moisture levels shows significantly high assimilation rate and it was high by 7% to 11.6% at 42 DAP (P ≤ 0.05), and 11.7% to 16.0% at 47 DAP (P ≤ 0.01), over plants under low soil moisture level. On the other hand, statistical analysis shows that there was significant interaction effect between CO2 and soil moisture levels on NAR both at 42 and 47 DAP (P ≤ 0.05). Significantly low NAR was under elevated CO2 with normal soil moisture level (11.6%) at 42 DAP, normal (11.7%) and high soil moisture level (16.0%) at 47 DAP compared to low soil moisture level. Results of this study also revealed that plants had high NAR at 47 DAP compared to 42 DAP.
3.2. Stomatal Conductance
Leaf stomatal conductance of soybean was extremely significant due to CO2 levels at both 42 and 47 DAP (P ≤ 0.001). Conductance was high by 40.5% and 36% under ambient CO2 in comparison to elevated CO2 level at 42 and 47 DAP, respectively (Table 1 & Figure 1). No significant effect of soil moisture levels and interaction between CO2 and soil moisture levels on leaf stomatal conductance. However, conductance was high under normal soil moisture level (14.7% and 11.2% under low and high soil moisture level, respectively) at 42 DAP and where at 47 DAP, high soil moisture level had high leaf conductance by 27% and 11.5% compared to low and normal soil moisture levels, respectively.
3.3. Intercellular CO2 Concentration
Plants grown under elevated CO2 had low intercellular CO2 concentration (16.68% & 12.28%) at 42 and 47
Table 1. Statistical results of gas exchange parameters of soybean genotypes influenced by [CO2] (elevated & ambient) and soil moisture levels (L: Low, N: Normal & H: High) and their interaction effects at 42 and 47 DAP [Assimilation rate (A), Stomatal conductance (gs), Intercellular CO2 (Ci), Transpiration rate (E), water use efficiency (WUE), relative intercellular (CO2) concentration (Ci/Ca), and intrinsic water use efficiency (A/gs)].
*,**,*** = significant at 0.05, 0.01, and 0.001 levels, respectively; NS= not-significant.
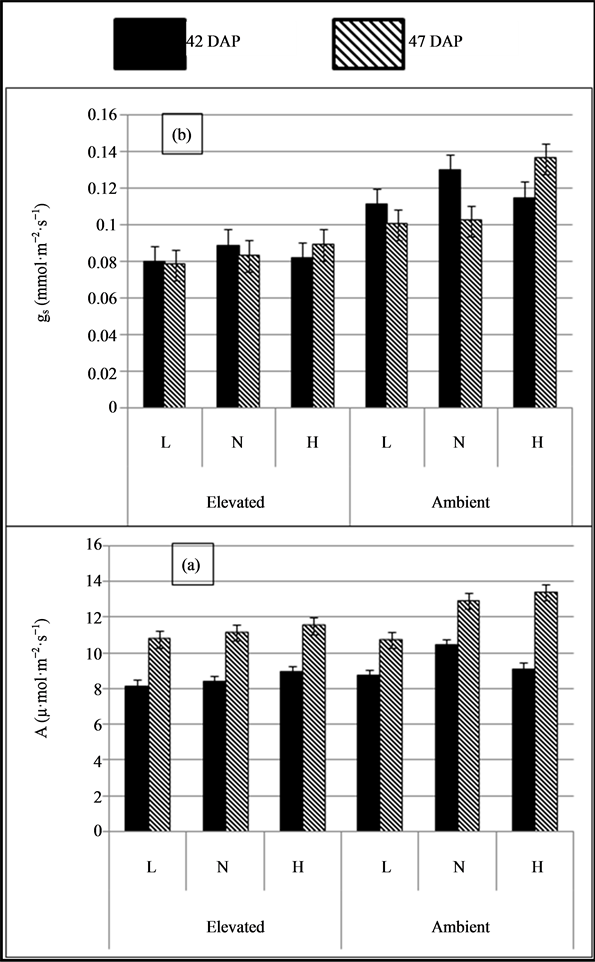
Figure 1. (a) Assimilation rate (a), and (b) stomatal conductance (gs) of soybean under (CO2) (elevated & ambient) and soil moisture levels (L: Low, N: Normal & H: High).
DAP but it was significant only at 42 DAP (P ≤ 0.01) (Table 1 & Figure 2). Soil moisture levels and interaction effect of CO2 and soil moisture were not significant at both 42 and 47 DAP. However, plants under ambient CO2 with normal soil moisture at 42 DAP and with low soil moisture at 47 DAP showed relatively high intercellular CO2 concentration compared to elevated CO2 with low to high soil moisture content. On the other hand, plants had high intercellular CO2 concentration (72%) at 42 DAP compared to 47 DAP.
3.4. The Relative Intercellular CO2 Concentration
The relative intercellular CO2 concentration (Ci:Ca intercellular CO2 concentration:ambient CO2 concentration) was extremely significant due to CO2 levels only at 42 DAP (P ≤ 0.001) and it was significantly low under elevated CO2 level by 17.4% and 11.2%, respectively at 42 and 47 DAP compared to plants under ambient CO2

Figure 2. (a) Intercellular [CO2] concentration (Ci) and (b) relative intercellular (CO2) concentration (Ci/Ca) of soybean under (CO2) (elevated & ambient) and soil moisture levels (L: Low, N: Normal & H: High).
level (Table 1 & Figure 2). Soil moisture levels and interaction between CO2 and soil moisture levels were not significant. However, results of the study shows that plants had high relative intercellular CO2 concentration under ambient CO2 level with normal soil moisture at 42 DAP and with high soil moisture at 47 DAP. Plants had high relative intercellular CO2 concentration by about 76% at 42 DAP compared to that of 47 DAP.
3.5. Transpiration and WUE
Transpiration rate was extremely significant due to CO2 levels both at 42 and 47 DAP (P ≤ 0.001). Transpiration rate was low in plants under elevated CO2 level by 43.6% and 39% over ambient CO2 at 42 and 47 DAP, respectively (Table 1 & Figure 3). Soil moisture levels significantly affected transpiration rates only at 47 DAP (P ≤ 0.01) and it was high under high soil moisture level by 26% and 18% compared to low and normal soil moisture levels. However, interaction effects of CO2 and soil moisture levels were not significant. On an average, high transpiration rate of about 76.4% at 47 DAP compared to 42 DAP.
Plants under elevated CO2 had significantly high WUE of 29% and 14% at 42 (P ≤ 0.001) and 47 DAP (P ≤ 0.01), respectively compared to ambient CO2 level. Water use efficiency not influenced significantly by soil
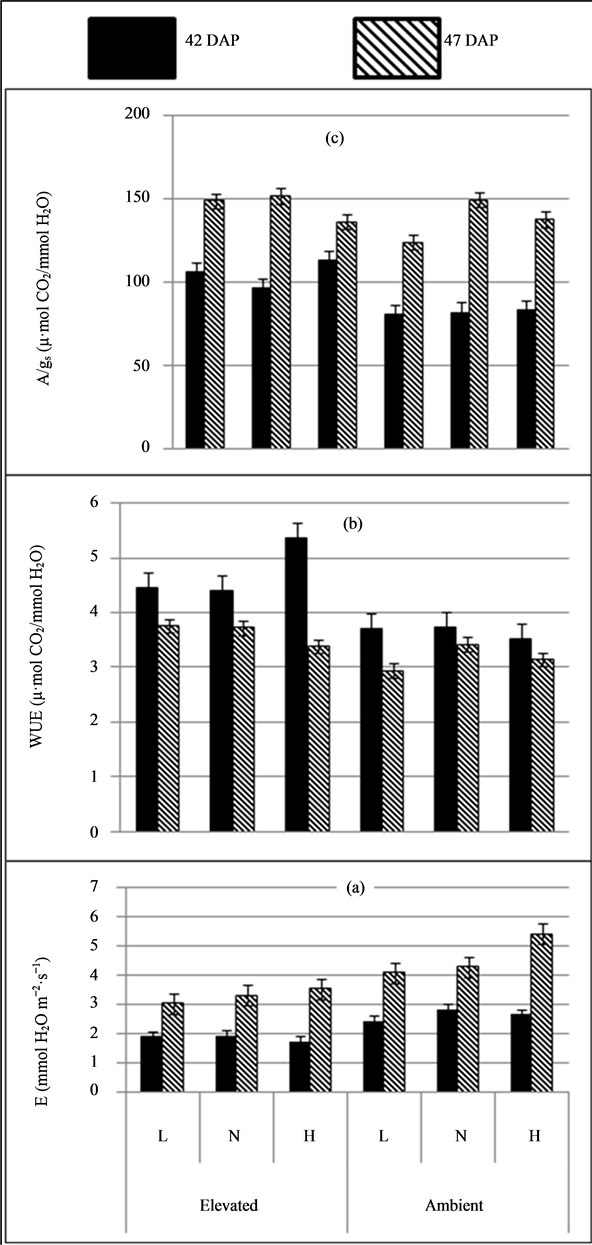
Figure 3. (a) Transpiration rate (E), (b) Water use efficiency (WUE) and (c) intrinsic water use efficiency (A:gs) of soybean under (CO2) (elevated & ambient) and soil moisture levels (L: Low, N: Normal & H: High).
moisture levels. Statistical analysis revealed that, interaction effect of CO2 and soil moisture levels were significant only at 42 DAP (P ≤ 0.05) and where plants under elevated CO2 with high soil moisture level had high WUE. Relatively, WUE was high by 27.5% at 42 DAP compared to that of 47 DAP.
3.6. Intrinsic Water Use Efficiency (A/gs)
Intrinsic water use efficiency (A/gs) was extremely significant for CO2 levels at 42 DAP (P ≤ 0.001) and it was high in plants under elevated CO2 level at 42 and 47 DAP (28.02% and 6.3%) compared to ambient CO2 level (Table 1 & Figure 3). On the other hand, no significant effect of soil moisture and interaction between CO2 and soil moisture levels on intrinsic water use efficiency of plants.
3.7. Total Dry Weight of Plants
Total dry weight of plants differed significantly due to CO2 and soil moisture level only at later stage of crop growth (58 DAP). Plants under elevated CO2 produced significantly high total dry matter (55%) compared to plants under ambient CO2 level at 58 DAP (P ≤ 0.001) (Figure 4). At 29 DAP, total dry matter production was high in plants under elevated CO2 but at 44 DAP it was high under ambient CO2 level. At all the sampling dates, plants under normal soil moisture level had maximum dry matter but it was significant only at later stage of the crop (58 DAP) (P ≤ 0.002). Plants under normal soil moisture level produced 56.6% and 50.1% high total dry matter compared to low and high soil moisture levels, respectively at 58 DAP. Interaction effect of CO2 and soil moisture levels was significant for total dry matter at early (29 DAP) and at later stage (58 DAP) of crop growth. At 29 (P ≤ 0.01) and 58 DAP (P ≤ 0.05), total dry matter production was significantly high when plants grown under elevated CO2 with low (9.4% to 34.6% high) and with normal (74.3% - 137.3% high) soil moisture levels, respectively.
Correlation coefficient (r) of total fresh weight of soybean plant was significantly positively correlated with net assimilation rate (r = 0.294*, P ≤ 0.05), leaf conductance (r = 0.286*, P ≤ 0.05) and transpiration(r = 0.0.266*, P ≤ 0.05) at 42 DAP (Table 2). Net assimilation rate showed a significant positive correlation with leaf conductance (r = 0.0.467* & r = 0.643*, P ≤ 0.05) and intercellular CO2 concentration at both 42 and 47 DAP. However, WUE was significantly negatively correlated with leaf conductance (r = −0.691* & −0.833*, P ≤ 0.05), intercellular CO2 concentration (r = −0.757* & −0.988*, P ≤ 0.05) and transpiration rate (r = −0.721* & −0.837*, P ≤ 0.05). The relative intercellular concentration CO2 significantly positive correlation with leaf conductance, intercellular CO2 concentration and transpiration rate but significantly negative correlation with WUE at both 42 and 47 DAP.
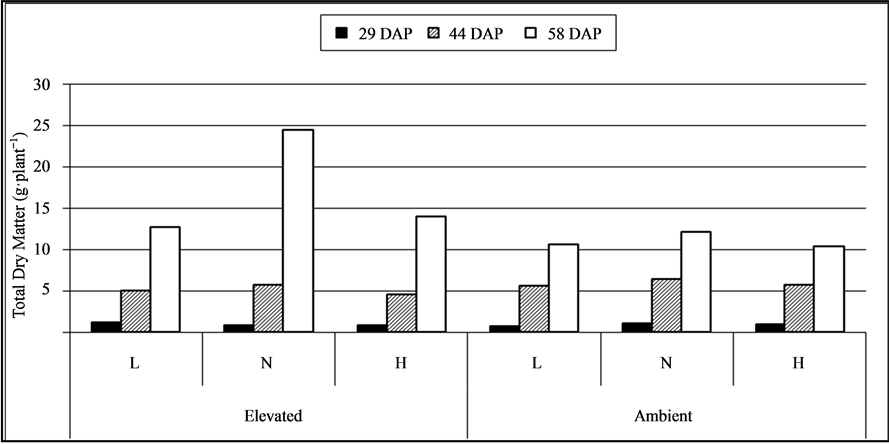
Figure 4. Total dry matter production (g·plant−1) of soybean under elevated & ambient (CO2) with three soil moisture levels (L: Low:,N: Normal & H: High) at 29, 44, and 58 DAP.
Table 2. Correlation coefficient (r) of total fresh weight of plant versus different gaseous exchange parameters at 42 (72 h of watering) and 47 DAP (24 h of watering) of soybean due to (CO2) and soil moisture levels during day.
aDegrees of freedom (n − 2) = 58; *significant at 0.05; NS= not-significant.
4. Discussion
4.1. Effect of Elevated CO2 on Gaseous Exchange Parameters
The results of this study showed down regulation of photosynthetic rate under elevated CO2. But many previous studies reported that in many C3 species and terrestrial plants, increase leaf photosynthesis at elevated CO2 due to enhancement of ribulosebiphosphate carboxylase/oxygenase (Rubisco) activity for carboxylation, but longterm exposure offsets this advantage by down-regulation of the process - and partly because the export of photosynthate from source to sink does not necessarily increase photosynthetic capacity. This reduction in photosynthetic capacity at high CO2 attributed to lower concentration of Rubisco and more pronounced at low N supply but reduction in Rubisco concentration is caused by accumulation of soluble carbohydrates is still a matter of debate . Reference argued that photosynthesis exceeds the capacity for carbohydrate export and utilization due to genetic limitations (such as determinate growth patterns) and environmental limitations (such as N deficiency or low temperature). Down-regulation of photosynthetic activity under CO2 enrichment is caused by decreasing leaf N concentration, and reduced rate of transpiration owing to decreased stomatal conductance is partially responsible for poor N translocation as reported by but photosynthesis acclimation ameliorated when N was added to growth medium adequately under elevated CO2 . This finding emphasizes the role of transpiration in acquisition of nitrogen (N) by leaves from the root environment and reveals existence of a feedback mechanism for photosynthetic acclimation at elevated CO2. The findings also supports an explanation that decreased transpiration resulting from stomatal closure possibly limit plant N uptake by causing declines in mass flow of mobile N forms to the mycorrhizophere However, the associated effects of reduced transpiration on leaf N concentration and its influence on Rubisco/photosynthetic activity are undetermined in the literature. Concentration of CO2 is not a limiting factor for photosynthesis under N deficiency but CO2 enrichment under such conditions down-regulates leaf photosynthesis . References observed a positive correlation between low leaf N content and low photosynthesis at high CO2 concentration. The results obtained in our study confirmed these findings where long-term exposure to high CO2 increase specific leaf area and decrease leaf N concentration (Figure 5) resulting in decreased leaf photosynthesis, stomatal conductance and transpiration rate significantly. Our study also demonstrated that leaf N content was

Figure 5. Specific leaf area (SLA), Leaf Nitrogen content and leaf C: N ratio of soybean under elevated & ambient (CO2) at 44 DAP.
low by 5.7% (at 29 DAP) and 8.7% (at 44 DAP) under elevated CO2 compared to ambient CO2. A reduction in leaf N concentration with elevated CO2 has been reported in different crop species . Our study suggests that the reduction in leaf N might be due to a greater increase in leaf area, as a result, the lack of proportionate gain in N by the plants (Figure 5). Increase in carbon (C) concentration and reduction in N content with increased C:N ratio due to elevated CO2 has been reported in various investigations . Uptake of N may also be reduced at high CO2 due to lower transpiration rate as reported by earlier studies .
On the contrary, many previous studies reported by increased photosynthetic rates of soybean leaves with elevated CO2 - . On the other hand, reported that crop canopy photosynthetic rates can vary throughout the growing season mainly due to different requirements of photo-assimilates. However, attributed this response to the typical characteristics of the soybean crop, which include: 1) high symbiotic N fixation capability; 2) the capacity to form an additional layer of palisade cells in the leaf tissue; 3) the capacity to shunt much of the photoassimilates into relatively inert starch rather than soluble sugars during photosynthesis; 4) a relatively strong leaf and stem sink during vegetative development; and 5) a strong seed sink during reproductive development. Plants that lack these capacities, either inherently or because of growth in limiting environments, are more likely to demonstrate some degree of down-regulation of photosynthesis . Results of the other studies reported that four biochemical mechanisms for down-regulation of photosynthesis caused by source sink imbalance at elevated CO2 are: 1) sugar repression of gene expression ; 2) insufficient N uptake ; 3) triose phosphate utilization rate limitation ; and 4) direct inhibition by saccharide content. On the other hand, the degree of down-regulation of photosynthesis was highly correlated with leaf glucose, fructose, and sucrose content and less correlated with starch content . These differential responses could be related to growth conditions and nutrient stress. Our results agree with these results wherein no external fertilizers or nutrients were supplied to the plants during the study period and probably plants were under nutritional stress. The study on dry beans by found that the net photosynthetic rate of plants grown in high nutrient levels did not show a down-regulation under elevated CO2. This clearly suggests that acclimation response varies with the growth environment, age, nutrient status of the soil, and ability of plant roots to grow in an unrestricted volume.
The mechanism by which stomatal aperture responds to elevated CO2 is mysterious but the most important phenomena responsive/sensitive to increasing CO2 levels is partial stomatal closure. In the present experiment, transpiration rate and leaf conductance was decrease under CO2 enrichment. This agrees with the previous studies . A review on soybean showed that stomatal conductance was decreased by 31% at 450 - 550 μmol·mol−1 CO2, 36% at 600 - 800 μmol·mol−1, and 51% at > 850 μmol·mol−1 of CO2 with respect to ambient 330 - 360 μmol·mol−1 CO2 and 30% decrease in stomatal conductance with a doubling of CO2 . In addition to decreased stomatal conductance, partial stomatal closure increases leaf resistance to transpiration water loss resulting in lower leaf transpiration rates. Reduction in transpiration was mainly attributed to decrease leaf conductance under elevated CO2. This result, in agreement with the results of , concluded that although stomatal conductance may be decreased by about 40% for doubled CO2, but water use by C3 crops under field conditions may probably be decreased only by up to 12%. If increases in leaf area due to doubled CO2 are small, then the transpiration reductions would be meaningful, albeit small but on the contrary, if the increase in leaf area is too large, then no reductions in transpiration would be expected and even small increases might be possible. In our result, depletion of leaf N under elevated CO2 could be attributed to reduced flow of the element in xylem owing to poor rate of transpiration due to partial closure of stomata at high CO2. Nitrogen transfer from roots to leaves occurs in the xylem and the movement is mainly regulated by water flow derived from leaf transpiration.
Our experimental result demonstrated an increased WUE and intrinsic WUE under elevated CO2 for soybean. However, WUE generally increases under high CO2 in almost all previous experiments - . In the present experiment, increased WUE under elevated CO2 resulted from by increasing growth linked to prolonged photosynthetic activity more than increasing water consumption. This effect would be highly beneficial especially in water-limited rainfed areas where water conservation is necessary . In some studies, total canopy water use of soybean was reduced by CO2 enrichment , while in others it was unaffected . Numerous experiments have demonstrated that increased WUE is due to decreases in leaf transpiration rate .
4.2. Effect of Soil Moisture on Gas Exchange Parameters
Exposure of plants to water stress leads to decreases in photosynthetic rate, stomatal conductance, transpiration rate, and concomitant increase intercellular CO2 in soybean. Stomatal closer reduces intercellular CO2 concentration in leaves which imposes limitation on CO2 assimilation, which causes an imbalance in photochemical activity at photosystem 2 (PS-2) and electron requirement for photosynthesis . Our results are also in conformity with the results of these studies. Except photosynthetic rate, all other parameters were not significant at both 42 and 47 DAP. Our experimental results indicated that plants under normal and high soil moisture levels, shows a significantly higher assimilation rate (7% to 16%) over plants under low soil moisture level. On the other hand, at all soil moisture levels photosynthetic rate was high (31.1%) at 47 DAP over 42 DAP. This high photosynthetic rate was attributed to favorable soil moisture conditions at 47 DAP compared to 42 DAP where gaseous exchange parameters measured at 24 h (47 DAP) and 72 h (42 DAP) of watering the soybean plants. Our study revealed that, high transpiration rate (18% to 26%) at high soil moisture compared to low and normal soil moisture level. On the other hand, 27.5% higher WUE at 72 h after watering (at 42 DAP) over 24 h after watering of plants. The plants showed adaptation mechanisms with progressive depletion of soil moisture by reducing stomatal conductance and transpiration rate. The favorable soil moisture content in soil increases the specific water content of soybean plants and shows an increase in trend with soil moisture availability from low to high (Figure 6) resulted in a positive impact on photosynthetic rate of plants. Our result, also in agreement with previous studies, where available soil water is necessary to maintain adequate photosynthetic rate during crop development and water deficit is known to decrease photosynthetic and transpiration rate . Reduction of net photosynthesis in soybean plants can be induced by both stomatal and non-stomatal factors (of both biochemical and photochemical origin). On the other hand, reported that decrease in photosynthetic rate under deficit soil moisture conditions may not necessarily be related to stomatal opening, but rather non-stomatal control of photosynthesis might have greater influence. In environments where there is water restriction caused by a lack of water from the soil or by a high atmospheric water demand, plants tend to close their stomata to conserve water via reducing transpiration losses and also reduces stomatal conductance (gs), which limits the entry of CO2 into the substomatal chambers reducing the diffusion of C to the site of carboxylation, resulting in significant decreases in C assimilation - . Furthermore, reports that the effects of water stress on the initial activity of Rubisco may be reproduced by induction of stomatal closure, independent of the reduction in the relative water content in the leaves of soybean plants. Thus, we can expect a lower regulation of photochemical and biochemical processes when the availability of CO2 is the most limiting component for photosynthesis in plants under severe water stress . The decrease in CO2 diffusion from the atmosphere to the carboxylation site of Rubisco is generally considered to be the main cause of reduced photosynthesis under conditions of mild and moderate water deficits - . The response of photosynthesis under water stress has been debated for decades, particularly with respect to what the most limiting factors for photosynthesis are . However, there is still some controversy regarding the importance of the main physiological parameters and the time period over which they limit photosynthesis .
Soil-plant–atmospheric interactions will be altered by future climate change scenarios and leads to changes in the water balance and the amount of water available in the soil is crucial for crop yield. In warmer climates, increased evapotranspiration favors soil dryness and predicts that potential evaporation increases by about 2% - 3% for each 1˚C rise in temperature . Thus, sites which are already at the limit with respect to water supply under current conditions are likely to be most sensitive to climate change, leading to an increase in the need for irrigation in dry areas, while more humid areas may be less affected .
4.3. Interaction Effect of CO2 and Soil Moisture on Gaseous Exchange Parameters
Our results indicated that, an interactive effect of CO2 and soil moisture levels was significant only on
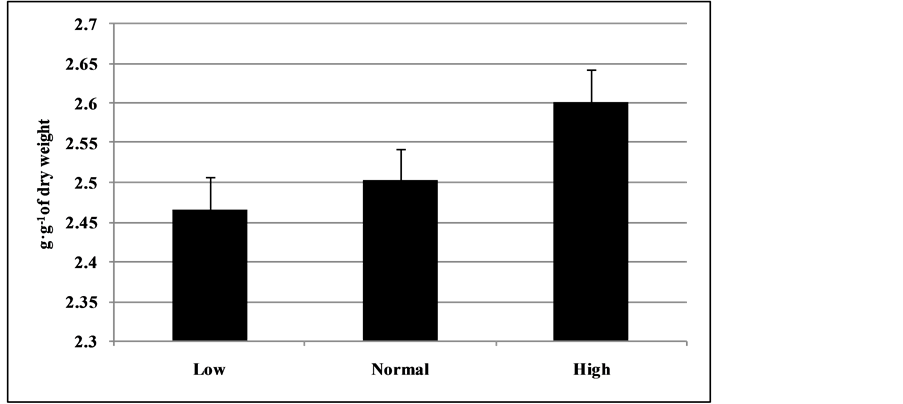
Figure 6. Specific water content of soybean (g of H2O g−1 of dry weight) at 44 DAP under different soil moisture conditions (Low; Normal; and High).
photosynthetic rate at both 42 and 47 DAP of soybean. Photosynthetic rate was low when plants exposed to elevated CO2 with favorable soil moisture conditions (normal & high) which was attributed to low leaf stomatal conductance and intercellular CO2 concentration. Previous studies reported that stomatal closer reduces intercellular CO2 concentration in leaves which imposes limitation on CO2 assimilation, which causes an imbalance in photochemical activity at photosystem 2 (PS-2) and electron requirement for photosynthesis . This current study shows down regulation of photosynthetic rate under elevated CO2 even with favorable soil moisture conditions. This down regulation of photosynthetic has been attributed to low leaf N concentration due to decline uptake of water from the N limited soil environment. Our results supports findings that leaf N content was low by 5.7% to 8.7% under elevated CO2 compared to ambient CO2. These results agree with many other studies who reported reduction in leaf N concentration with elevated CO2 in many crop species.
The effects of elevated CO2 on plants can vary depending on other interacting environmental factors including water, temperature, and available soil nutrients status. Elevated CO2 makes C more available but plants equally need other resources in optimum to harvest benefit of raising atmospheric CO2. Elevated CO2 does not directly make these resources more available particularly uptake of water and nutrients. Uptake of nutrients, particularly N, was reduced at high CO2 due to lower transpiration rate coupled with a decrease in stomatal opening resulting in a low uptake of water, which increases soil moisture content. Therefore, the ability of plants to respond to elevated CO2 with increased photosynthesis and growth may be limited under conditions of low mineral nutrients availability, particularly documented for N. In Free-Air Carbon Dioxide Exchange (FACE) experiments, there is less enhancement of photosynthesis by elevated CO2 under low than high soil N conditions . Crop yield in FACE also appears to be enhanced by elevated CO2 to a lesser extent under low-N than under high-N . We suspect that improved growth of soybean under elevated CO2 has further limited the available N in the present study where no external source of nutrients were supplied to the crop during the study period which reflected in low leaf N and high C:N ratio. The resultant inability of soybean plants to produce new sinks causes an imbalance in supply and demand which feedback on photosynthetic processes. This is supported by our leaf total N content (low by 5.7% at 29 DAP and 8.7% at 44 DAP) with more accumulation of carbohydrates resulting high C:N ratio. Our findings agree with those of , who concluded that photosynthetic acclimation results primary sink limitation in low N soils.
On the other hand, reported that higher stomatal conductance in plants is known to increase CO2 diffusion into leaves thereby favoring higher photosynthetic rates. Our result shows significant relations between photosynthetic rate and stomatal conductance (r = +0.467* at 42 DAP and +0.643* at 47 DAP) and similarly between stomatal conductance and intercellular CO2 concentration (r = +0.695* at 42 DAP and +0.816* at 47 DAP). Due to favorable soil moisture condition at 47 DAP (24 h of watering) photosynthetic rate was relatively high both at ambient and elevated CO2 over 72 h of watering (42 DAP). Further, our study revealed that low transpiration rate and C-i/CA ratio, high WUE, and A/gs ratio when plants were exposed to elevated CO2 over ambient conditions. At 24 h and 72 h of watering (47 DAP), low transpiration rate and high WUE under elevated CO2 over ambient CO2, but increased transpiration rate and a reduction in WUE with increase in soil moisture levels from low to high. Under conditions of non-limiting soil water and elevated atmospheric CO2, a reduction in stomatal conductance and decline in transpiration were observed in many herbaceous plants . Decreases in stomatal conductance and transpiration rate of crops under elevated CO2 in both irrigated and drought conditions has been reported by earlier studies - .
This physiological behavior of crop plants under elevated CO2 has greater implications, particularly in arid and semi-arid regions, where water is the most critical input . Two mechanisms are hypothesized to explain improved plant water relations under elevated CO2. First, increases in plant photosynthetic rates in elevated CO2 might lead to lower osmotic potential (OP) in leaf cells from higher concentrations of organic solutes, especially sugars of photosynthetic origin, which constitute a major fraction of osmotic substances in cells . Thus, for a given water potential, lower values of OP should result in higher turgor potential and tissue water content. Second, since decreased gs in elevated CO2 decreases leaf transpiration, it should also decrease the water potential gradient from soil to leaves. All these findings supporting the facts that for favorable crop growth under elevated CO2 needs optimal resources particularly nutrients and soil moisture for positive feedback of crop plants but interactive effective of many resources with elevated CO2 and environmental factors are inconclusive and greater dearth of literatures.
5. Conclusion
High CO2 decreases photosynthetic rate, stomatal conductance, intercellular CO2 concentration, relative intercellular CO2 concentration, and transpiration rate. The down-regulation of photosynthesis is mainly caused by low leaf N content and high C:N ratio. This is supported with many other studies reported in the literature. Carbon dioxide enrichment increases leaf area causing low leaf N content and was found to increase C:N ratio and C content per unit mass of leaves resulting to lower protein content that will deteriorate the quality of grains may have feedback on nutritional security of future world. Water use efficiency enhanced under elevated CO2 mainly through reduction in transpiration rate and water use by plants coupled with increased total dry matter production. In the present study, interactive effects of elevated CO2 and soil moisture levels are significant but interactive effects of many resources with elevated CO2 and environmental factors are inconclusive and greater dearth of literatures on this aspect. The effect of high CO2 on plants depends on the availability of other resources, mainly nutrients and soil moisture. A positive feedback of crops from CO2 enrichment will occur only when these resources are sufficient to meet crop demand.
Acknowledgements
The technical support and guidance of all staff of USDA National Laboratory for Agriculture and the Environment and Iowa State University, Ames, Iowa, USA is thankfully acknowledged. The financial support of USAID and USDA is gratefully acknowledged under USDA-GRA fellowship program.
Declaration
Trade names and company names are included for the benefit of the reader and do not imply any endorsement or preferential treatment of the product by the authors or the USDA-ARS. USDA is an equal opportunity provider and employer.
References
- Singh, G. and Shivakumar, B.G. (2010) The Role of Soybean in Agriculture. In: Singh, B., Ed., The Soybean: Botany, Production and Uses, CAB International, UK, 24-47.
- Mutei, H. (2011) Papel do Brasil no combate a fome no mundo. In: Siqueira, F., Caju, J. and Moreira, M., Eds., Boletim de Pesquisa da Soja, Fundação MT, Mato Grosso, 45-48.
- Lisar, S.Y.S., Motafakkerazad, R., Hossain, M.M. and Rahman, I.M.M. (2012) Water Stress in Plants: Causes, Effects and Responses. In: Rahman, I.M.M. and Hasegawa, H., Eds., Water Stress, In Tech, Croatia, 1-14.
- Sionit, N., Hellmers, H. and Strain, B.R. (1982) Interaction of Atmospheric CO2 Enrichment and Irradiance on Plant Growth. Agronomy Journal, 74, 721-725. http://dx.doi.org/10.2134/agronj1982.00021962007400040029x
- Kimball, B.A. (1983) Carbon Dioxide and Agricultural Yield: An Assemblage and Analysis of 430 Prior Observations. Agronomy Journal, 75, 779-788. http://dx.doi.org/10.2134/agronj1983.00021962007500050014x
- Morison, J.I.L. (1985) Sensitivity of Stomata and Water Use Efficiency to High CO2. Plant, Cell and Environment, 8, 467-474. http://dx.doi.org/10.1111/j.1365-3040.1985.tb01682.x
- Morison, J.I.L. (1998) Stomatal Response to Increased CO2 Concentration. Journal of Experimental Botany, 49, 443- 452. http://dx.doi.org/10.1093/jxb/49.Special_Issue.443
- Cure, J.D. and Acock, B. (1986) Crop Responses to Carbon Dioxide Doubling: A Literature Survey. Agriculture and Forest Meteorology, 38, 127-145. http://dx.doi.org/10.1016/0168-1923(86)90054-7
- Sharma, A. and Sengupta, U.K. (1990) Carbon Dioxide Enrichment Effect on Photosynthesis and Related Enzymes in Vigna Radiate Wilczek. Indian Journal of Plant Physiology, 33, 340-346.
- Eamus, D. (1991) The Interaction of Rising CO2 and Temperatures with Water Use Efficiency. Plant, Cell and the Environment, 14, 843-852. http://dx.doi.org/10.1111/j.1365-3040.1991.tb01447.x
- Drake, B.G. and Leadley, P.W. (1991) Canopy Photosynthesis of Crops and Native Plant Communities Exposed to Long-Term Elevated CO2. Plant, Cell and the Environment, 14, 853-860. http://dx.doi.org/10.1111/j.1365-3040.1991.tb01448.x
- Lawlor, D.W. and Mitchell, R.A.C. (1991) The Effect of Increasing CO2 on Crop Photosynthesis and Productivity: A Review of Field Studies. Plant, Cell and Environment, 14, 807-918. http://dx.doi.org/10.1111/j.1365-3040.1991.tb01444.x
- Stitt, M. (1991) Raising CO2 Level and Their Potential Significance for Carbon Flow in Photosynthetic Cell. Plant, Cell and the Environment, 14, 741-762. http://dx.doi.org/10.1111/j.1365-3040.1991.tb01440.x
- Sasek, T.W. and Strain, B.R. (1991) Effect of CO2 Enrichment on the Growth and Morphology of a Native and Introduced Honey Suckle Vine. American Journal of Botany, 78, 69-75. http://dx.doi.org/10.2307/2445229
- Bowes, G. (1993) Facing the Inevitable: Plants and Increasing Atmospheric CO2. Annual Review of Plant Physiology, 44, 309-332. http://dx.doi.org/10.1146/annurev.pp.44.060193.001521
- Idso, K.E. and Idso, S.B. (1994) Plant Responses to Atomspheric CO2 Enrichment in the Face of Environmental Constraint: A Review of the Past 10 Year’s Research. Agriculture and Forest Meteorology, 69, 153-203. http://dx.doi.org/10.1016/0168-1923(94)90025-6
- Sage, R.F. (1994) Acclimation of Photosynthesis to Increasing Atmospheric CO2: The Gas Exchange Perspective. Photosynthesis Research, 39, 351-368. http://dx.doi.org/10.1007/BF00014591
- Jiang, G.M. (1995) The Impact of Global Increasing CO2 on Plants. Chinese Bulletin of Botany, 12, 1-7.
- Allen, L.H., Kirkham, M.B., Olszyk, D.M. and Whitman, C.E. (1997) Advances in Carbon Dioxide Effects Research. ASA Special Publication N. 61, Madison, 228 p.
- Drake, B.G., Gonzalez-Meler, M.A. and Long, S.P. (1997) More Efficient Plants: A Consequence of Rising Atmospheric CO2? Annual Review of Plant Physiology and Plant Molecular Biology, 48, 609-639. http://dx.doi.org/10.1146/annurev.arplant.48.1.609
- Wang, X.L., Xu, S.H. and Liang, H. (1998) The Experimental Study of the Effects of CO2 Concentration Enrichment on Growth, Development and Yield of C3 and C4 Crops. Agricultural Sciences in China, 31, 55-61.
- Das, M., Pal, M., Zaidi, P.H., Raj, A. and Sengupta, U.K. (2000) Growth Response of Mung Bean to Elevated CO2. Indian Journal of Plant Physiology, 5, 137-140.
- Ulman, P., Catsky, J. and Pospisilova, J. (2000) Photosynthetic Traits in Wheat Grown under Decreased and Increased CO2 Concentration, and after Transfer to Natural CO2 Concentration. Biologia Plantarum, 43, 227-237. http://dx.doi.org/10.1023/A:1002752210237
- Wu, D.X. and Wang, G.X. (2000) Interaction of CO2 Enrichment and Drought on Growth, Water Use, and Yield of Broad Bean (Vicia faba). Environmental Experimental Botany, 43, 131-139. http://dx.doi.org/10.1016/S0098-8472(99)00053-2
- Hui, D F., Luo, Y.Q., Cheng, W.X., Coleman, J.S., Johnson, D.W. and Sims, D.A. (2001) Canopy Radiationand Water-Use Efficiencies as Affected by Elevated CO2. Global Change Biology, 7, 75-91. http://dx.doi.org/10.1046/j.1365-2486.2001.00391.x
- Wu, D.X., Wang, G.X., Bai, Y.F., Liao, J.X. and Ren, H.X. (2002) Responses of Growth and Water Use Efficiency of Spring Wheat to Whole Season CO2 Enrichment and Drought. Acta Botany Sinica, 44, 1477-1483.
- Kimball, B.A., Kobayahi, K. and Bindi, M. (2002) Responses of Agricultural Crops to Free-Air CO2 Enrichment. Advances in Agronomy, 77, 293-368. http://dx.doi.org/10.1016/S0065-2113(02)77017-X
- Srivastava, A.C., Khanna, Y.P., Meena, R.C., Pal, M. and Sengupta, U.K. (2002) Diurnal Changes in Photosynthesis, Sugars, and Nitrogen of Wheat and Mungbean Grown under Elevated CO2 Concentration. Photosynthetica, 40, 221- 225. http://dx.doi.org/10.1023/A:1021393607410
- IPCC (2007) Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, Cambridge University Press, Cambridge.
- Prior, S.A., Runion, G.B., Rogers, H.H. and Arriaga, F.J. (2010) Elevated Atmospheric Carbon Dioxide Effects on Soybean and Sorghum Gas Exchange in Conventional and No Tillage Systems. Journal of Environmental Quality, 39, 596-608. http://dx.doi.org/10.2134/jeq2009.0181
- IPCC (2001) Climate Change 2001: Impact, Adaptation, and Vulnerability. A Report of Working Group II of the Intergovernmental Panel on Climate Change, 18 p.
- Rosenzweig, C., Iglesias, A., Yang, X.B., Epstein, P.R. and Chivian, E. (2001) Climate Change and Extreme Weather Events; Implications for Food Production, Plant Diseases, and Pests. Global Change and Human Health, 2, 90-104. http://dx.doi.org/10.1023/A:1015086831467
- Schneider, S.H. (2001) What is “Dangerous” Climate Change. Nature, 411, 17-19. http://dx.doi.org/10.1038/35075167
- Long, S.P., Ainsworth, E.A., Rogers, A. and Ort, D.R. (2004) Rising Atmospheric Carbon Dioxide: Plants Face the Future. Annual Review of Plant Biology, 55, 591-628. http://dx.doi.org/10.1146/annurev.arplant.55.031903.141610
- Bates, B.C., Kundzewicz, Z.W., Wu, S. and Palutikof, J.P. (2008) Climate Change and Water. Technical Paper of the Intergovernmental Panel on Climate Change, IPCC Secretariat, Geneva.
- Kruijt, B., Witte, J.P.M., Jacos, C.M.J. and Kroon, T. (2008) Effects of Rising Atmospheric CO2 on Evapotranspiration and Soil Moisture: A Practical Approach for the Netherlands. Journal of Hydrology, 349, 257-267. http://dx.doi.org/10.1016/j.jhydrol.2007.10.052
- Lotze-Campen, H. and Schellnuber, H.J. (2009) Climate Impacts and Adaptation Options in Agriculture: What We Know and What We Don’t Know. Journal für Verbraucherschutz und Lebensmittelsicherheit, 4, 145-150. http://dx.doi.org/10.1007/s00003-009-0473-6
- Poorter, H., Berkel, Y.V. and Baxter, R. (1997) The Effects of Elevated CO2 on the Chemical Composition and Construction Costs of Leaves of 27 C3 Species. Plant, Cell and the Environment, 20, 472-482. http://dx.doi.org/10.1046/j.1365-3040.1997.d01-84.x
- Allen Jr., L.H. and Boote, K.J. (2000) Crop Ecosystem Responses to Climate Change: Soybean. In: Reddy, K.R. and Hodges, H.F., Eds., Climate Change and Global Crop Productivity, CABI Publishing, UK, 133-160. http://dx.doi.org/10.1079/9780851994390.0133
- Ainsworth, E.A., Davey, P.A., Bernacchi, C.J., Dermody, O.C., Heaton, E.A., Moore, D.J., Morgan, P.B., Naidu, S.L., RA, H.S.Y., Zhu, X.G., Curtis, P.S. and Long, S.P. (2002) A Meta-Analysis of Elevated CO2 Effects on Soybean (Glycine max) Physiology, Growth and Yield. Global Change Biology, 8, 695-709. http://dx.doi.org/10.1046/j.1365-2486.2002.00498.x
- Pickering, N.B., Allen Jr., L.H., Albrecht, S.L., Jones, P., Jones, J.W. and Baker, J.T. (1994) Environmental Plant Chambers: Controls and Measurements Using CR-10T Data Loggers. In: Watson, D.G., Zuzueta, F.S. and Harrison, T.V., Computers in Agriculture: Proceedings of the 5th International Conference, American Society of Agricultural Engineers, St. Joseph, 29-35.
- Reddy, K.R., Kakani, V.G., Zhao, D., Koti, S. and Gao, W. (2004) Interactive Effects of Ultraviolet-B Radiation and Temperature on Cotton Physiology, Growth, Development and Hyperspectral Reflectance. Photochemistry and Photobiology, 79, 416-427. http://dx.doi.org/10.1562/2003-11-19-RA.1
- Ewert, F., Rodriguez, D., Jamieson, P., Semenov, M.A., Mitchell, R.A.C., Goudriaan, J., Porter, J.R., Kimball, B.A., Pinter, Jr., P.J., Manderscheid, R., Weigel, H.J., Fangmeier, A., Fereres, E. and Villalobos, F. (2002) Effects of Elevated CO2 and Drought on Wheat: Testing Crop Simulation Models for Different Experimental and Climatic Conditions. Agriculture, Ecosystems and Environment, 93, 249-266. http://dx.doi.org/10.1016/S0167-8809(01)00352-8
- Amthor, J.S. (2001) Effects of Atmospheric CO2 Concentration on Wheat Yield: Review of Results from Experiments Using Various Approaches to Control CO2 Concentration. Field and Crop Research, 73, 1-34. http://dx.doi.org/10.1016/S0378-4290(01)00179-4
- Gifford, R.M. (1979) Growth and Yield of CO2-Enriched Wheat under Water-Limited Conditions. Australian Journal of Plant Physiology, 6, 367-378. http://dx.doi.org/10.1071/PP9790367
- Kramer, P.J. (1981) Carbon Dioxide Concentration, Photosynthesis, and Dry Matter Production. BioScience, 31, 29-33. http://dx.doi.org/10.2307/1308175
- Pooter, H. (1993) Interspecific Variation in the Growth Response of Plants to an Elevated Ambient CO2 Concentration. Vegetatio, 104-105, 77-97. http://dx.doi.org/10.1007/BF00048146
- Thompson, G.B. and Woodward, F.J. (1994) Some Influences of CO2 Enrichment, Nitrogen Nutrition and Competition on Grain Yield and Quality in Spring Wheat and Barley. Journal of Experimental Botany, 45, 937-942. http://dx.doi.org/10.1093/jxb/45.7.937
- Ziska, L.H., Weerakoon, W., Namuco, O.S. and Pamplona, R. (1996) The Influence of Nitrogen on the Elevated CO2 Response in Field Grown Rice. Australian Journal of Plant Physiology, 23, 45-52. http://dx.doi.org/10.1071/PP9960045
- Bunce, J.A. (1998) The Temperature Dependence of the Stimulation of Photosynthesis by Elevated Carbon Dioxide in Wheat and Barley. Journal of Experimental Botany, 49, 1555-1561. http://dx.doi.org/10.1093/jxb/49.326.1555
- Pooter, H. (1998) Do Slow-Growing Species and Nutrient-Stressed Plants Respond Relatively Strongly to Elevated CO2? Global Change Biology, 4, 693-697. http://dx.doi.org/10.1046/j.1365-2486.1998.00177.x
- Mo, X.G., Lin, Z.H. and Liu, S.X. (2007) Climate Change Impacts on the Ecohydrological Processes in the Wuding River Basin. Acta Ecologica Sinica, 27, 4999-5007.
- Thomas, A. (2008) Agriculture Irrigation Demand under Present and Future Climate Scenarios in China. Global and Planetary Change, 60, 306-326. http://dx.doi.org/10.1016/j.gloplacha.2007.03.009
- Cox, P.M., Betts, R.A., Jones, C.D., Spall, S.A. and Totterdell, I.J. (2000) Acceleration of Global Warming Due to Carbon-Cycle Feedbacks in a Coupled Climate Model. Nature, 408, 184-187. http://dx.doi.org/10.1038/35041539
- Hansen, W.J., Sato, M., Ruedy, R., Lacis, A. and Oinas, V. (2000) Global Warming in the Twenty-First Century: An Alternative Scenario. Proceedings of the National Academy of Sciences of the United States of America, 97, 9875-9880. http://dx.doi.org/10.1073/pnas.170278997
- Kaspar, T.C., Moorman, T.B. and Logsdon, S.D. (1992) The National Soil Tilth Laboratory and Rhizotron. In: Reetz, H.F., Ed., Roots of Plant Nutrition, Potash and Phosphate Institute, Atlanta, 52-60.
- Klepper, B. and Kaspar, T.C. (1994) Rhizotrons: Their Development and Use in Agricultural Research. Agronomy Journal, 86, 745-753. http://dx.doi.org/10.2134/agronj1994.00021962008600050002x
- Makino, A. (1994) Biochemistry of C3-Photosynthesis in High CO2. Journal of Plant Research, 107, 79-84. http://dx.doi.org/10.1007/BF02344533
- Peet, M.M. (1984) CO2 Enrichment of Soybeans. Effects of Leaf/Pod Ratio. Physiologia Plantarum, 60, 38-42. http://dx.doi.org/10.1111/j.1399-3054.1984.tb04246.x
- Idso, S.B. and Kimball, B.A. (1991). Downward Regulation of Photosynthesis and Growth at High CO2 Levels. No Evidence for Either Phenomenon in Three-Year Study of Sour Orange Trees. Plant Physiology, 96, 990-992. http://dx.doi.org/10.1104/pp.96.3.990
- Rogers, G.S., Milham, P.J., Thibaud, M.C. and Conroy, J.P. (1996) Interaction between Rising CO2 Concentration and Nitrogen Supply in Cotton. I. Growth and Leaf Nitrogen Concentration. Australian Journal of Plant Physiology, 23, 119-125. http://dx.doi.org/10.1071/PP9960119
- Moore, B.D., Cheng, S.H., Sims, D. and Seeman, J.R. (1999) The Biochemical and Molecular Basis for Photosynthetic Acclimation to Elevated Atmospheric CO2. Plant, Cell and Environment, 22, 567-582. http://dx.doi.org/10.1046/j.1365-3040.1999.00432.x
- Ainsworth, A.E., Rogers, A., Nelson, R. and Long, S.P. (2004) Testing the “Source-Sink” Hypothesis of Down Regulation of Photosynthesis in Elevated CO2 in the Field with Single Gene Substitution in Glycine max. Agriculture and Forest Meteorology, 122, 85-94. http://dx.doi.org/10.1016/j.agrformet.2003.09.002
- Kanemoto, K., Yamashita, Y., Ozawa, T., Imanishi, N., Nguyen, N.T., Suwa, R., Mohapatra, P.K., Kanai, S., Moghaieb, R.E., Ito, J., El-Shemy, H. and Fujita, K. (2009) Photosynthetic Acclimation to Elevated CO2 Is Dependent on N Partitioning and Transpiration in Soybean. Plant Science, 177, 398-403. http://dx.doi.org/10.1016/j.plantsci.2009.06.017
- Farage, P.K., McKee, I.F. and Long, S.P. (1998) Does a Low Nitrogen Supply Necessarily Lead to Acclimation of Photosynthesis to Elevated CO2? Plant Physiology, 118, 573-580. http://dx.doi.org/10.1104/pp.118.2.573
- Conroy, J. and Hocking, P. (1993) Nitrogen Nutrition of C3 Plants at Elevated Atmospheric CO2 Concentrations. Physiologia Plantarum, 89, 570-576. http://dx.doi.org/10.1111/j.1399-3054.1993.tb05215.x
- Pettersson, R. and McDonald, A.J.S. (1994) Effects of Nitrogen Supply on the Acclimation of Photosynthesis to Elevated CO2. Photosynthesis Research, 39, 389-400. http://dx.doi.org/10.1007/BF00014593
- Nakano, H., Makino, A. and Mae, T. (1997) The Effect of Elevated Partial Pressures of CO2 on the Relationship between Photosynthetic Capacity and N Content in Rice Leaves. Plant Physiology, 115, 191-198.
- Hocking, P.J. and Meyer, C.P. (1991) CO2 Enrichment Decreases Critical Nitrate and Nitrogen Concentration in Wheat. Journal of Plant Nutrition, 14, 571-584. http://dx.doi.org/10.1080/01904169109364225
- Conroy, J.P. (1992) Influence of Elevated Atmosphere CO2 Concentration on Plant Nutrition. Australian Journal of Botany, 40, 445-456.
- Gifford, R.M., Barrett, D.J. and Lutze, J.L. (2000) The Effect of Elevated CO2 on C:N and C:P Mass Ratio of Plant Tissues. Plant and Soil, 224, 1-14. http://dx.doi.org/10.1023/A:1004790612630
- Manderscheid, R., Bender, H.J., Jager, H.J. and Weigel, H.J. (1995) Effect of Season Long CO2 Enrichment on Cereals. II. Nutrient Concentrations and Grain Quality. Agriculture, Ecosystems and Environment, 54, 175-185. http://dx.doi.org/10.1016/0167-8809(95)00602-O
- Cotrufo, M.F., Ineson, P. and Scott, A. (1998) Elevated CO2 Reduces the Nitrogen Concentration of Plant Tissues. Global Change Biology, 4, 43-54. http://dx.doi.org/10.1046/j.1365-2486.1998.00101.x
- Vara Prasad, P.V., Allen Jr., L.H. and Boote, K.J. (2005) Crop Responses to Elevated Carbon Dioxide and Interaction with Temperature—Grain Legumes. Journal of Crop Improvement, 13, 113-155. http://dx.doi.org/10.1300/J411v13n01_07
- Huber, S.C., Rogers, H.H. and Mowry, F.L. (1984) Effects of Water Stress on Photosynthesis and Carbon Partitioning in Soybean (Glycine max [L.] Merr.) Plants Grown in the Field at Different CO2 Levels. Plant Physiology, 76, 244-249. http://dx.doi.org/10.1104/pp.76.1.244
- Hesketh, J.D., Woolley, J.T. and Peters, D.B. (1984) Leaf Photosynthetic CO2 Exchange-Rates in Light and CO2 Enriched Environments. Photosynthetica, 18, 536-540.
- Sionit, N., Rogers, H.H., Bingham, G.E. and Strain, B.R. (1984) Photosynthesis and Stomatal Conductance with CO2- Enrichment of Container and Field-Grown Soybeans. Agronomy Journal, 76, 447-451 http://dx.doi.org/10.2134/agronj1984.00021962007600030018x.
- Chen, X.M., Begonia, G.B., Alm, D.M. and Hesketh, J.D. (1995) Soybean Stomatal Acclimation to Long-Term Exposure to CO2-Enriched Atmospheres. Photosynthetica, 31, 51-57.
- Ferris, R., Wheeler, T.R., Hadley, P. and Ellis, R.H. (1998) Recovery of Photosynthesis after Environmental Stress in Soybean Grown under Elevated CO2. Crop Science, 38, 948-955. http://dx.doi.org/10.2135/cropsci1998.0011183X003800040012x
- Ziska, L.H, Bunce, J.A. and Caulfield, F.A. (2001) Rising Atmospheric Carbon Dioxide and Seed Yield of Soybean Genotypes. Crop Science, 41, 385-391. http://dx.doi.org/10.2135/cropsci2001.412385x
- Acock, B., Reddy, V.R., Hodges, H.F., Baker, D.N. and McKinion, J.M. (1985) Photosynthetic Response of Soybean Canopies to Full-Season Carbon Dioxide Enrichment. Agronomy Journal, 77, 942-947. http://dx.doi.org/10.2134/agronj1985.00021962007700060025x
- Allen Jr., L.H., Baker, J.T., and Boote, K.J. (1996) The CO2 Fertilization Effect: Higher Carbohydrate Production and Retention as Biomass and Seed Yield. In: Bazzaz, F. and Sombroek, W., Eds., Global Climate Change and Agricultural Production, FAO, Rome, 65-100.
- Allen Jr., L.H. (1994) Carbon Dioxide Increase: Direct Impacts on Crops and Indirect Effects Mediated through Anticipated Climate Changes. In: Boote, K.J., Bennett, J.M., Sinclair, T.R. and Paulsen, G.M., Eds., Physiology and Determination of Crop Yield, ASA, CSSA and SSSA, Madison, 425-459.
- Van Oosten, J.J. and Besford, R.T. (1995) Some Relationship between the Gas Exchange, Biochemistry and Molecular Biology of Photosynthesis during Leaf Development of Tomato Plants after Transfer to Different Carbon Dioxide Concentrations. Plant, Cell and the Environment, 18, 1253-1266. http://dx.doi.org/10.1111/j.1365-3040.1995.tb00185.x
- Stitt, M. and Krapp, A. (1999) The Interaction between Elevated Carbon Dioxide and Nitrogen Nutrition: The Physiological and Molecular Background. Plant, Cell and the Environment, 22,583-621. http://dx.doi.org/10.1046/j.1365-3040.1999.00386.x
- Socias, F.X., Medrano, H. and Sharkey, T.D. (1993) Feedback Limitation of Photosynthesis of Phaseolus vulgaris L Grown in Elevated CO2. Plant, Cell and the Environment, 16, 81-86. http://dx.doi.org/10.1111/j.1365-3040.1993.tb00847.x
- Prasad, P.V.V., Boote, K.J., Vu, J.C.V. and Allen Jr., L.H. (2004) The Carbohydrate Metabolism Enzymes Sucrose-P Synthase and ADG-Pyrophosphorylase in Phaseolus Bean Leaves Are Up-Regulated at Elevated Growth Carbon Dioxide and Temperature. Plant Science, 166, 1565-1573. http://dx.doi.org/10.1016/j.plantsci.2004.02.009
- Rodoglou, K.M., Aphalo, P. and Jarvis, P.G. (1992) Response of Photosynthesis, Stomata1 Conductance and Water Use Efficiency to Elevated CO2, and Nutrient Supply in Acclimated Seedlings of Phaseolus valgaris L. Annals of Botany, 70, 257-264.
- Law, B.E., Falge, E., Gu, L., Baldocchi, D.D., Bakwin, P., Berbigier, P., Davis, K., Dolman, A.J., Falk, M., Fuentes, J.D., Goldstein, A., Graanier, A., Grelle, A., Hollinger, D., Janssens, I.A., Jarvis, P., Jensen, N.O., Katul, G., Mahl, Y., Metteucci, G., Meyers, T., Monson, R., Munger, W., Oechel, W., Olson, R., Pilegaard, K., Paw, K.T., Thorgeirsson, H., Valentini, R., Verma, S., Vesala, T., Wilson, K. and Wofsy, S. (2002) Environmental Controls over Carbon Dioxide and Water Vapor Exchange of Terrestrial Vegetation. Agriculture and Forest Meteorology, 113, 97-120. http://dx.doi.org/10.1016/S0168-1923(02)00104-1
- Park, Y.-I., Chowa, W.S., Anderson, J.M. and Hurry, V.M. (1996) Differential Acclimated Susceptibility of Photosystem II to Light Stress in Light Assimilated Pea Leaves Depends on the Capacity for Photochemical and Non-Radiactive Dissipation of Light. Plant Science, 115, 137-149. http://dx.doi.org/10.1016/0168-9452(96)04339-7
- Cure, J.D. (1985) Carbon Dioxide Doubling Response: A Crop Survey. In: Strain, B.R. and Cure, J.D., Eds., Direct Effects of Increasing Carbon Dioxide on Vegetation, US Department of Energy, Carbon Dioxide Research Division, Washington DC, 99-116.
- Allen Jr., L.H. (1999) Evapotranspiration Responses of Plant and Crops to Carbon Dioxide and Temperature. Journal of Crop Production, 2, 37-70. http://dx.doi.org/10.1300/J144v02n02_02
- Clifford, S.C., Stronach, I.M., Black, C.R., Singleton-Jones, P.R., Azam-Ali, S.N. and Crout, N.M.J. (2000) Effects of Elevated CO2, Drought and Temperature on the Water Relations and Gas Exchange of Groundnut (Arachis hypogaea) Stands Grown in Controlled Environment Glasshouses. Physiologia Plantarum, 110, 78-88. http://dx.doi.org/10.1034/j.1399-3054.2000.110111.x
- Wallace, J.S. (2000) Increasing Agricultural Water Use Efficiency to Meet Future Food Production. Agriculture, Ecosystem and Environment, 82,105-119. http://dx.doi.org/10.1016/S0167-8809(00)00220-6
- Jones, P., Jones, J.W. and Allen Jr., L.H. (1985) Response of Soybean Canopy Growth, Photosynthesis, and Transpiration to Whole-Day Temperature Changes in Different CO2 Environment. Agronomy Journal, 77, 119-126.
- Rogers, H.H., Cure, J.D., Thomas, J.F. and Smith, J.M. (1984) Influence of Elevated CO2 on Growth of Soybean Plants. Crop Science, 24, 361-366. http://dx.doi.org/10.2135/cropsci1984.0011183X002400020036x
- Kimball, B.A., Mauney, J.R., Guinn, G., Nakayama, F.S., Pinter Jr., P.J., Clawson, K.L., Idso, S.B., Butler, G.D. and Radin, J R. (1984) Response of Vegetation to Carbon Dioxide, Ser 023. Effect of Increasing Atmospheric CO2 on the Yield and Water of Crops. US Department of Energy and USDA, US Water Conservation Laboratory and US Western Cotton Research Laboratory, USDA-ARS, Phoenix.
- Jones, P., Jones, J.W. and Allen Jr., L.H. (1985b) Seasonal Canopy CO2 Exchange, Water Use, and Yield Components in Soybean Grown under Different CO2 and Water Stress Conditions. Transactions in American Society of Agricultural Engineers, 28, 2021-2028.
- He, J.X., Wang, J. and Liang, H.J. (1995) Effect of Water Stress on Photochemical Function and Protein Metabolism of Photosystem II in Wheat Leaves. Plant Physiology, 93, 771-777. http://dx.doi.org/10.1111/j.1399-3054.1995.tb05130.x
- Flagella, Z., Campanile, R.G., Stoppeli, M.C., De Caro, A. and Di Fonzo, N. (1998) Drought Torlance of Photosynthetic Electron Transport under CO2-Enriched and Normal Air in Cereal Species. Plant Physiology, 104, 753-759. http://dx.doi.org/10.1034/j.1399-3054.1998.1040434.x
- Boyer, J.S. (1982) Plant Productivity and Environment. Science, 218, 443-448. http://dx.doi.org/10.1126/science.218.4571.443
- Farquhar, G.D., Wong, S.O., Evans, T.R. and Hubiok, K.T. (1989) Photosynthesis and Gas Exchange. In: Flowers, H.G. and Jones, M.B., Eds., Plant under Stress, Cambridge University Press, Cambridge, 47-67. http://dx.doi.org/10.1017/CBO9780511661587.005
- Kaiser, W.M. (1987) Effects of Water Deficit on Photosynthetic Capacity. Physiologia Plantarum, 71, 142-149. http://dx.doi.org/10.1111/j.1399-3054.1987.tb04631.x
- Yu, G.R., Wang, Q.F. and Zhuang, J. (2004) Modeling the Water Use Efficiency of Soybean and Maize Plants under Environmental Stress: Application of a Synthetic Model of Photosynthesis-Transpiration Based on Stomatal Behavior. Journal of Plant Physiology, 161, 303-318. http://dx.doi.org/10.1078/0176-1617-00972
- Flexas, J., Barón, M., Bota, J., Ducruet, J., Gallé, A., Galmeés, J., Jiménez, M., Pou, A., Ribas-Carbó, M., Sajnani, C., Tomás, M. and Medrano, H. (2009) Photosynthesis Limitations during Water Stress Acclimation and Recovery in the Drought-Adapted Vitis Hybrid Richter-110 (V. berlandieri × V. rupestri). Journal of Experimental Botany, 60, 2362- 2377. http://dx.doi.org/10.1093/jxb/erp069
- Pinheiro, C. and Chaves, M.M. (2011) Photosynthesis and Drought: Can We Make Metabolic Connections from Available Data? Journal of Experimental Botany, 62, 869-882. http://dx.doi.org/10.1093/jxb/erq340
- Flexas, J., Ribas-Carbó, M., Bota, J., Galmés, J., Henkle, M., Martínez-Cañellas, S. and Medrano, H. (2006) Decrease Rubisco Activity during Water Stress Is Not Induced by Decreased Relative Water Content but Related to Conditions of Low Stomatal Conductance and Chloroplast CO2 Concentration. New Phytology, 172, 73-82. http://dx.doi.org/10.1111/j.1469-8137.2006.01794.x
- Flexas, J., Bota, J., Galmés, J., Medrano, H. and Ribas-Carbó, M. (2006) Keeping a Positive Carbon Balance under Adverse Conditions: Responses of Photosynthesis and Respiration to Water Stress. Physiologia Plantarum, 127, 343- 352. http://dx.doi.org/10.1111/j.1399-3054.2006.00621.x
- Chaves, M.M., Maroco, J.P. and Pereira, J. (2003) Understanding Plant Responses to Drought from Genes to the Whole Plant. Functional Plant Biology, 30, 239-264. http://dx.doi.org/10.1071/FP02076
- Chaves, M.M., Flexas, J. and Pinheiro, C. (2009) Photosynthesis under Drought and Salt Stress: Regulation Mechanisms from Whole Plant to Cell. Annals of Botany, 103, 551-560. http://dx.doi.org/10.1093/aob/mcn125
- Grassi, G. and Magnani, F. (2005) Stomatal, Mesophyll Conductance and Biochemical Limitations to Photosynthesis as Affected by Drought and Leaf Ontogeny in Ash and Oak Trees. Plant, Cell and Environment, 28, 834-849. http://dx.doi.org/10.1111/j.1365-3040.2005.01333.x
- Lawlor, D.W. and Cornic, G. (2002) Photosynthetic Carbon Assimilation and Associated Metabolism in Relation to Water Deficits in Higher Plants. Plant, Cell and Environment, 25, 275-294. http://dx.doi.org/10.1046/j.0016-8025.2001.00814.x
- Lawlor, D.W. and Tezara, W. (2009) Causes of Decreased Photosynthetic Rate and Metabolic Capacity in Water-Deficient Leaf Cells: A Critical Evaluation of Mechanisms and Integration of Processes. Annals of Botany, 103, 561-579. http://dx.doi.org/10.1093/aob/mcn244
- Lockwood, J.G. (1999) Is Potential Evapotranspiration and Its Relationship with Actual Evapotranspiration Sensitive to Elevated Atmospheric CO2 Levels? Climatic Change, 41, 193-212. http://dx.doi.org/10.1023/A:1005469416067
- Brumbelow, K. and Georgakakos, A. (2001) An Assessment of Irrigation Needs and Crop Yield for the United States under Potential Climate Changes. Journal of Geophysical Research: Atmospheres, 106, 27383-27405. http://dx.doi.org/10.1029/2001JD900034
- Ainsworth, E.A. and Long, S.P. (2005) What Have We Learned from 15 Years of Free-Air CO2 Enrichment (FACE)? A Meta-Analytic Review of the Responses of Photosynthesis, Canopy Properties and Plant Production to Rising CO2. New Phytology, 165, 351-372. http://dx.doi.org/10.1111/j.1469-8137.2004.01224.x
- Ainsworth, E.A. and Rogers, A. (2007) The Response of Photosynthesis and Stomatal Conductance to Rising CO2: Mechanisms and Environmental Interactions. Plant, Cell and the Environment, 30, 258-270. http://dx.doi.org/10.1111/j.1365-3040.2007.01641.x
- Long, S.P., Ainsworth, E.A., Leakey, A.D.B., Nosberger, J. and Ort, D.R. (2006) Food for Thought: Lower-Than-Expected Crop Yield Stimulation with Rising CO2 Concentrations. Science, 312, 1918-1921. http://dx.doi.org/10.1126/science.1114722
- Ainsworth, E.A. (2008) Rice Production in a Changing Climate: A Meta-Analysis of Responses to Elevated Carbon Dioxide and Elevated Ozone Concentration. Global Change Biology, 14, 1642-1650. http://dx.doi.org/10.1111/j.1365-2486.2008.01594.x
- Rogers, A., Fischer, B.U., Bryant, J., Frehner, M., Blum, H., Raines, C.A. and Long, S.P. (1998) Acclimation of Photosynthesis to Elevated CO2 under Low-Nitrogen Nutrition Is Affected by the Capacity for Assimilate Utilization. Perennial Ryegrass under Free Air CO2 Enrichment. Plant Physiology, 118, 683-689. http://dx.doi.org/10.1104/pp.118.2.683
- Taiz, L. and Zeiger, E. (1998) Plant Physiology. 2nd Edition, Sinauer Associates Publishers, Sunderland, Massachusetts. http://dx.doi.org/10.1071/PP9840361
- Morison, J.I.L. and Gifford, R.M. (1984) Plant Growth and Water Use with Limited Water Supply in High CO2 Concentrations. I. Leaf Area, Water Use and Transpiration. Australian Journal of Plant Physiology, 11, 361-374. http://dx.doi.org/10.1071/PP9840361
- Chen, J.J. and Sung, J.M. (1990) Gas Exchange Rate and Yield Responses of Virginia-Type Peanut to Carbon Dioxide Enrichment. Crop Science, 30, 1085-1089. http://dx.doi.org/10.2135/cropsci1990.0011183X003000050025x
- Stronach, I.M., Clifford, S.C., Mohamed, A.D., Singleton-Jones, P.R., Azamali, S.N. and Crout, N.M.J. (1994) The Effects of Elevated Carbon Dioxide, Temperature and Soil Moisture on the Water-Use of Stands of Groundnut (Arachis hypogaea L.). Journal of Experimental Botany, 45, 1633-1638. http://dx.doi.org/10.1093/jxb/45.11.1633
- Clifford, S.C., Black, C.R., Roberts, J.A., Stronach, I.M., Singleton-Jones, P.R., Mohamed, A.D. and Azamali, S.N. (1995) The Effect of Elevated Atmospheric CO2 and Drought Onstomatal Frequency in Groundnut (Arachis hypogaea L.). Journal of Experimental Botany, 46, 847-852. http://dx.doi.org/10.1093/jxb/46.7.847
- Prasad, P.V.V., Boote, K.J., Allen Jr., L.H. and Thomas, J.M.G. (2002) Effects of Elevated Temperature and Carbon Dioxide on Seed-Set and Yield of Kidney Bean (Phaseolus vulgaris L.). Global Change Biology, 8, 710-721. http://dx.doi.org/10.1046/j.1365-2486.2002.00508.x
NOTES

*Corresponding author.


