Low Carbon Economy
Vol.3 No.1(2012), Article ID:17876,13 pages DOI:10.4236/lce.2012.31004
Technico-Economical Evaluation of CO2 Transport in an Adsorbed Phase
![]()
1CIIA, Department of Civil and Environmental Engineering, Universidad de Los Andes, Bogotá, Colombia; 2S3D, Nantes, France; 3Ecole Supérieure des Sciences et Technologies de l’Ingénieur de Nancy, Université Henri Poincaré, Nancy, France.
Email: *adelebar@esstin.uhp-nancy.fr
Received September 16th, 2011; revised October 20th, 2011; accepted November 8th, 2011
Keywords: CO2; transport; adsorption; activated carbon; economical analysis
ABSTRACT
This work considers the possibility to transport CO2 in an adsorbed phase and analyzes its cost as a function of transported quantities, transport conditions and transportation means. CO2 adsorption capacities of 6 different adsorbents, comprising 4 activated carbons and 2 zeolites, were empirically evaluated in a given range of pressure and temperature. The adsorbent with the highest mass adsorption capacity (AC1), as well as another sorbent described in the literature (AC5) were selected to be used for CO2 transportation by ships, trains or trucks. Their characteristics and performances were then used to develop an economic analysis of transportation costs and CO2 emissions generated by the transport with or without storage. Economic evaluation of CO2 batch transport shows that CO2 transported in an adsorbed phase by train was seen to be almost competitive on distances between 250 and 500 km, in comparison to liquefied CO2. One of the activated carbon appeared to be competitive on short distances by truck when transport was not followed by storage. Ship transport of adsorbed CO2 on distances around 1500 km was competitive, when CO2 was used as delivered; there was an over cost of only 16%, when there was storage after the transport. The CO2 emissions generated by CO2 transport and storage when transport is carried out in an adsorbed phase were smaller than the ones generated by liquid phase transport below 1200 km, 500 km and 300 km by ship, train and truck respectively, as a function of the adsorbent used. Adsorbed CO2 transported on 1500 km by ship generated 27% less CO2 emissions than liquid phase and 17% by train for a distance of 250 km and 16% by truck on 150 km, although these differences were decreasing with the distance of transport.
1. Introduction
Control of greenhouse gases emission, especially carbon dioxide (CO2), is the principal goal of World Climate Change Protocols. CO2 capture and storage (CCS) looks like an option to achieve this objective, avoiding CO2 emissions at the atmosphere by holding them in geological formations or by injection in ocean. CCS is principally composed of three steps: capture, transport and storage. Capture includes CO2 separation and concentration to being transported and stored in a disposal place where the CO2 release will be reduced. The CO2 transport is normally processed in liquid or supercritical phase which generate additional costs and CO2 emissions. The supercritical state is used with pipeline in continuous flux, and the liquid one for batch transportation (e.g. train, truck, ship). CO2 transport via pipeline has been used for several years in the oil industry for the extraction system. Conditions of pressure and temperature handled in the supercritical phase need temperatures below –60˚C and pressures higher than 7.38 MPa [1]. In addition, due to problems caused by static pressure drops, and friction within pipes, CO2 must be compressed at pressure between 10 and 80 MPa [1,2]. The batch CO2 transport condition is generally liquid state because solidification requires twice of energy than liquefaction [3] doing it energetic and economic inefficient [4,5]. Besides CCS, CO2 transport via truck or train is used for delivery in industry that uses CO2 such as food industry which demands 100,000 tons/year [1]. CO2 transport by ship, still in development, is similar to the liquefied gas petroleum (LPG) using high pressure, low temperature or both conditions [5].
CO2 transportation costs are not known in detail because it has not been yet scaled up to large amounts of CO2 or for CCS projects [5]. The cost assessment has been made according to different scenarios in which quantity, distance and media are relevant. Svensson et al., [1] made a cost comparison between on shore pipeline, on shore pipeline-water carrier, offshore pipeline and railway-water carrier for 1.0 Mt/y of CO2 and 10 Mt/y of CO2 transported. The on shore for 110 km and 10 Mt CO2/y scenario, showed the best price per ton of CO2 transported: 0.95 EUR/ton. In the same way, for longer distances (600 km) offshore pipeline-water carrier was the cheapest option (5.61 EUR/ton) in comparison with railway-water carrier scenario (8.58 EUR/ton) and on shore off shore transportation (17.75 EUR/ton) (pressure and temperature conditioning are not included). Some other studies have been reported about CO2 ship transportation [4-6]. They concluded that CO2 shipping could be a promised route for CO2 transportation on long distances with a cost comprised between 13 and 55 US$/t.
The economy of scale plays an important role reducing transportation cost in 70% and 80% by pipeline and ship, if raising the amount of CO2 transported per year by 10 times [1]; 64% if it is increased 7 times [6]; 30% by tanker if the amount is increased 3 times [4] or 33% by 2.5 increments [2-7].
Also, conditioning process (i.e. liquefaction) impacts the total transportation cost. Kaarstad and Hustad [8] estimated the cost of transport by ship of 5.5 Mt/y of CO2 with or without liquefaction. Their results showed a difference of 57% when conditioning is included, 34 US$/t- 55 US$/t. Decarre et al., [6] also compared investment cost of two conditioning systems: –50˚C, 0.7MPa and –35˚C, 0.15MPa. The second scenario was the more economically efficient, minimizing the energy requirements for liquefaction from 37kWh/t CO2 to 12 kWh/t CO2.
The different economic studies have shown that the cost effective CO2 transportation are pipeline or ship [1,4], the last one being the cheapest one for distances longer than 1000 Km compared to offshore pipeline [5,9]. It was also demonstrated that the cost per ton of CO2 decrease when the whole transported quantity increase [2,4-7].
The CO2 transportation could represent 20% of total CCS cost [10] that ranged between 30 - 70 US$/t in the case of a pulverized coal power plant (PC) and for a capture of 1.6 Mt CO2/y. The cost of capture is the largest component cost (23 - 35US$/t) that includes the compression until required condition for transport by pipeline: 14MPa [5]. The total cost of CCS will change with the quantity of CO2, the distance of transport and the storage system. The capturing of 0.19 Mt CO2/y from a 24 MW biomass IGCC plant is estimated to be about 82 US$/t CO2 while for a 400MW of 2.1 Mt CO2/y could be 11 US$/t CO2 [5].
In the case of CCS, CO2 storage is the ending process after transportation. It could be made in geological formations (i.e. oil-gas depleted reservoirs, saline water-saturated reservoir rocks, coal seams, basalts), or inside ocean by injection, searching CO2 dilution, or keeping on a hollow of sea floor. Storage cost ranges between 0.6 and 8.3 US$/t CO2 [5]. This value depends of zone location, depth, number of injection wells as conditioning for CO2 disposition (i.e. pressure). Shafeen et al., [11] estimated CO2 sequestration cost from a power plant in Ontario, Canada, inside sandstone formation including pipeline transportation and storage 1000 km of depth by injection in 10 wells. Their calculations gave US$ 7.5/t of CO2, 20% of total capture and sequestration cost. Overpressure is required to avoid changing phase problems inside pipelines. Injection in 1000 km well could require 13 - 18 MPa of pressure [5]. Ocean storage cost estimation at 3000 m depth is US$ 5.3/t CO2 including injection platform, pipe and nozzles.
The CO2 capture technology could modify the transporttation cost considering its selectivity (i.e. impurity) and operation conditions (i.e. pressure, temperature). In CO2 post combustion capture, options are solvents absorption, membranes, solid sorbents and liquefaction. Solid sorbents like activated carbon and zeolites have a high sorption capacity in operating low pressures and recovery sorbent material option [12]. In the present work, as a new method of transportation which could link capture and transport, batch CO2 transport in an adsorbed phase was studied and compared with liquefied one, in scenarios with or without geological or ocean disposal. The following scenarios studied thereafter, include geological or ocean storage which correspond to CCS application evaluation, as well as absence of storage step corresponding to the use of CO2 for food industry, greenhouse agricultural applications… The proposed principle of transport in an adsorbed phase takes advantage of recent industrial developments of activated carbon media and filters, based on optimized disposition of fixed activated carbon fibres and cloths [13], or of compartments of fixed bed of immobilized granular activated carbon particles. It is thus possible to conceive and construct reservoirs with an adapted and structured adsorbent filling content that allow a homogenous conditioning and an easy filling, emptying and thus reuse as a batch transport system. In contrast, the adsorbed CO2 transport was not compared in detail to pipeline transport because the extrapolation of activated particles supporting adsorbed CO2 and being continuously transported in pipes was not straightforward. Nevertheless, the comparison of batch adsorbed and liquefied CO2 transport with continuous transport in pipeline is given through some cost evaluation available in literature.
In the present study, adsorption capacity of activated carbon and zeolites were first evaluated at temperature of 0˚C, 25˚C, 75˚C at pressure below 0.8 MPa to define CO2 transportation sorbent. Another activated carbon described in literature was also selected. The economic analysis and environmental evaluation of the CO2 transport by ship, truck and train were carried out for adsorbed CO2 and liquefied CO2 to compare them. The different parameters that influence the transport cost (i.e. distance, tanker size, operating conditions, means of transport) were studied for CO2 adsorbed or liquid scenarios. Finally, adsorbed-liquefied transport comparison was made. Operating conditions for adsorbed CO2 transport were fixed at 0˚C and 1.5 MPa by extrapolation of experimenttal results. Geological storage was chosen for CO2 transported by truck or train, and ocean storage by shipping. Moreover, the CO2 emissions resulting from transportation and storage were also evaluated as a function of the same parameters.
2. Experimental Materials and Methods
2.1. Experimental Assembly
The equipment used for adsorption tests consists in a bottle of CO2 linked through a pipe to the system. A cylindrical cell stainless steel reactor (254 mL) was equipped with an internal thermocouple that allowed measuring the temperature within the bed of the adsorbents; the cell was immerged in a liquid bath (i.e. water, glycol) having a controlled temperature in order to fix the chosen adsorption temperature. Equipment is described in detail in [14].
A pressure monitoring system, which gives the pressure within the system and a mass flow-meter Brooks 5850S, allowed knowing the input mass of the gas. Experiments were carried out at 0˚C, 25˚C, 75˚C and the pressure was increased step by step up (0.04, 0.1, 0.2, 0.4, 0.8 MPa) to 0.8 MPa. Prior to adsorption measurements, the moisture of the solid sorbent sample was eliminated in oven, in air atmosphere, for 12 h at 100˚C and after it was inserted in the cell. Before loading the system with gas, the circuit was purged using a vacuum pump until reaching a pressure around 2 kPa. The loading process was started by opening main input valve and regulated with a manual fine valve in order to respect the max flow imposed by the flow meter. The gas flow rate and the gas mass input were continuously recorded as a function of time. The data of temperature, pressure and CO2 mass flow were acquired at the acquisition frequency of 1Hz. The assembly was previously tested to prevent leakage and tests were repeated several times with an empty cell assessing the reproducibility and accuracy of the experiments.
2.2. Characterisation of Adsorbents
Four activated carbons (AC) and two zeolites were tested in this study. The samples were named AC1, AC2, AC3 and AC4 for activated carbons characterized in the present study and AC5 for another one from literature that will be considered later, and Z1 and Z2 for zeolites. Sample AC4 was used with two different conditioning: one as a woven material, which was a delivered form of an activated carbon tissue (a); the second made of bulk fibres of the tissue not woven (b) (Table 1).
Surface areas and information about micropores were determined using a Micrometrics model ASAP 2010 micropore-volume reactor. Before the nitrogen analysis, samples were conditioned at 350˚C for 24 h. Nitrogen adsorption measurements were conducted at liquid nitrogen temperature (77˚K). Different methods were used for porosity characterization: t-plot (size pore distribution), Horvath-Kawazoe (micropore volume), BJH (mesoporosity) and BET (surface area). The porosity type classification was based on International Union of Pure and Applied Chemistry (IUPAC).
2.3. Expressions of Adsorption Capacities
Effective adsorbed quantity of CO2 (Qeff) on the adsorbents materials is deduced from two measured quantities: Qtot that is the CO2 mass contained in the total circuit, composed of the pipes and the cell containing the adsorbents, and Qdead the CO2 mass contained only in the pipes. Qeff was then calculated making the difference Qtot-Qdead.

Table 1. Physical properties of adsorbent.
Once Qeff was determined, adsorption isotherms were represented by the amount of CO2 on the adsorbent as a function of pressure. Quantity of CO2 adsorbed can be expressed in terms of sorbent mass or volume. Thus, isotherms were represented as adsorbed moles of CO2 per kilogram of sorbent (Qm) or as adsorbed moles of CO2 per litre of sorbent (Qv). Qm and Qv are related one to each other through material bulk density. These two adsorption efficiency criteria are useful depending on the constraints imposed to the adsorption application. In case of transportation and its cost evaluation, the weight was considered to be the limiting factor and thus, comparison of adsorption performances between adsorbents was done by the means of Qm. In contrast, Qv would be useful if the constraint comes from the size (volume) of a separation reactor. Qm and Qv are appreciated within an accuracy range of ±5% regarding repetition of tests presented in [14].
3. Technico-Economical Evaluation Methodology
3.1. Cost Evaluation Methodology Generalities
Considered specifications to cost evaluation were the following: total costs were expressed in US$/ton of CO2 (annual total cost basis). The assessment of the CO2 transportation cost includes different stages: conditioning (liquefaction-adsorption), storage, transport and load and unloading [15]. Storage cost evaluation includes: requirements of booster compressors, injection wells and off shore platforms [11]. To calculate annual investment costs, the life utility (15 years), the effective annual interest rate (7%) and the maintenance rate (45%) were considered constant for each step of the total transport and storage costs [15]. To analyse transportation costs by ship, train and truck, the proposed methodologies by Ozaki et al. [15], Öztürk [16], and Edwards [17] were considered respectively. The storage cost methodology used is reported by Shafeen et al. [11] and Aka et al. [18].
The boundary conditions of cost and environmental impact calculations for transport in liquid phase and in an adsorbed phase were:
—Initial conditions 15˚C and 0.11 MPa with a gas purity of 100% of CO2 (after Aspelund et al. [19])—Final conditions for transport in case of applications using CO2, other than storage, are “as delivered”: –50˚C and 0.7 MPa in case of liquid and 0˚C and 0.02 MPa for adsorbed phase transport (i.e. desorption conditions)—Final conditions for transport + storage, i.e. sequestration, were for both liquid and adsorbed CO2: 0˚C, 33 MPa (geological), 34 MPa (ocean).
Both liquid and adsorbed transport processes may be economically advantaged or disadvantaged in their comparison by the choice of some of the initial or final conditions, knowing these conditions might be relevant of the CO2 capture or of the CO2 storage processes, particularly when optimizing the global capture—transport— storage costs and environmental impacts. Energy necessary to the adsorption process to cool and to compress CO2 from initial conditions to the transport operational conditions were calculated on the basis of three sets of transport settings: 0˚C - 0.8 MPa, 0˚C - 1 MPa, 0˚C - 1.5 MPa.
3.2. Transport Scenario
CO2 is commonly transported by pipelines or in containers (tank or cistern). Only the last ones were considered in this scenario. Different states of CO2 can be transported in tanks according to the operating conditions: solid state, at atmospheric pressure and –80˚C, compressed liquid CO2, at 8 MPa and room temperature, but the most used condition to transport CO2 is liquefied, at –50˚C and 0.7 MPa or –25˚C and 1.5 MPa [2]. The set of conditions of –50˚C and 0.7 to 1.5 MPa were used in this work for the evaluation of the transport costs and their comparison with the adsorbed phase transport.
Table 2 shows the transport scenario with different amount of produced gas, as a function of power plants capacity, tanker size and the distances which were considered for this study. Coal based thermal power stations (PC) of 1000 and 300 MW with an amount of produced CO2 of 20,000 and 5500 t CO2/day and ship transport were considered. For plants of 100 MW, an amount of produced CO2 of 2000 t CO2/day and transport by train was assessed. And for plants of 30 MW with a generation of 500 ton of CO2 per day, truck transport was selected.
The different sizes for the ship evaluated in this study were: 50,000 deadweight tonnage (DTW), 80,000 DTW and 170,000 DTW [15] (respectively 46,000 m3, 101,000 m3 and 157,600 m3) used for transportation distances of 1500 km, 3000 km, 6000 km and 12,000 km. The transportation distances assessed by train were: 250 km, 300 km and 500 km and by truck: 150 km, 250 km and 300 km. Table 3 shows detailed specifications about ship transport.
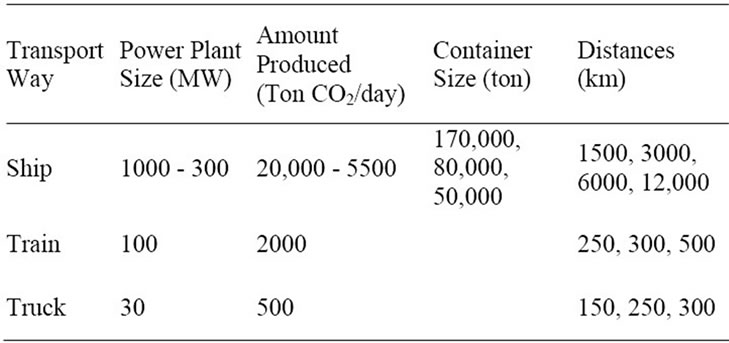
Table 2. Transport scenario.
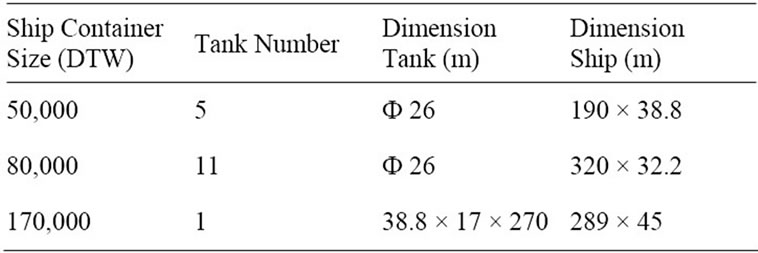
Table 3. Ship reservoir dimension and quantity.
3.3. Storage Scenario
Transported CO2 could be held in geological or ocean reservoirs for long periods. To calculate cost, storage scenarios were linked with means of transport: by ship in ocean reservoir, by train and truck in geological formations. Both liquefied and adsorbed CO2 must be compressed to reach a higher pressure than injection point (9% - 18% above) [11]. Either supposed geological or ocean injection depth was 3000m. The number of injecttion wells was calculated according to flow rate of CO2 of each scenario based on Shafeen et al., methodology [11]. Off shore platforms for ocean storage were included supposing a depth of 25 m. Standards for maximum injection pressures ranged from 13 to 18 kPa/m for oil and gas wells [5]. A pressure of 11 kPa/m was chosen, so 33 MPa is required to geological injection. Ocean storage requires a pressure at the bottom of 34 MPa. The cost calculation included thus: platform (offshore), injection wells, booster, energy cost, operation and maintenance. The CO2 retention both in geological and ocean storage has been modelled for injection wells of 3000 m, and around 100% should be holding in a period of 100 years [5]. For these reasons, the leakage of CO2 accounted for 0% of total transported.
3.4. Emissions
The CO2 emissions for the different transport ways were assessed in order to measure their impact to the environment. The CO2 quantity emitted during the transport and storage was evaluated with the help of literature data as the sum of three principal steps. The first one is the CO2 emitted during the conditioning step; the second one is the CO2 emitted during the transportation step; and the third one is CO2 emitted during booster compression for injection in geological or ocean storage. The emissions were calculated considering electric energy and fuel used respectively during the two transport steps and diesel engine in platform for CO2 injection. For CO2 conditioning 0.833 kg∙CO2/kW [15] is the environmental impact related to the electric energy consumed during this step. For the transport step emissions are depending on the way of transport: by ship transport 3.19 ton CO2/t fuel [15] and [20] was considered; by truck transport 2.772 kg∙CO2/l diesel fuel [20] and by train transport 10.78 kg∙CO2/km [21], based on emission data from electric energy consumption per km. In storing process, 2.772 kg∙CO2/L diesel fuel and 0% leaking of transported CO2 was assumed [5].
4. Results and Discussion
4.1. Adsorption Results and Selection of the Best Adsorbents
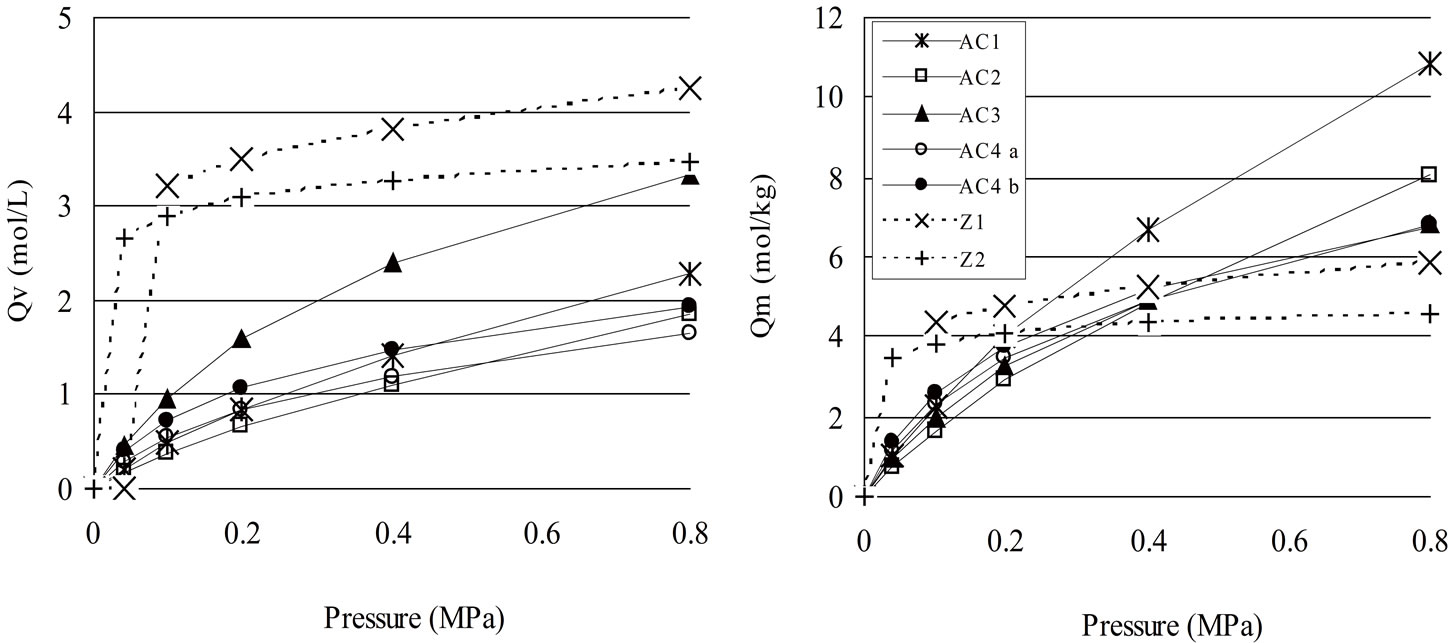
One topic to selecting sorbents to be used in pressure swing process is the adsorption capacity of the material for the considered gas. The adsorption isotherms of pure CO2 on the samples mentioned above are shown in Figures 1(a) and (b), which show that the best sorbent in
 (a) (b)
(a) (b)
Figure 1. Adsorption isotherms moles CO2: (a) based on adsorbent volume Qv at 25˚C and (b) based on adsorbent mass Qm.
terms of capacity depends on the parameter of selection (i.e. (Qm), mass capacity or (Qv) volumetric capacity), confirming that the bulk density of the material could make a difference. Figure 1 shows that for the zeolites Z1 and Z2 the volumetric and mass adsorption capacities increased almost at a plateau value when the pressure was risen up to 0.1 MPa. After 0.1 MPa till up to 0.8 MPa, the adsorption capacity remained almost constant. For activated carbon the adsorption increased moderately up to 0.8 MPa and may grow up again after that, as confirmed by results from [12], who indicated that some AC adsorption capacity increases up to 3 MPa and becomes steady after 3 - 3.5 MPa. It can be noted that at low pressure the best sorbent resulted from zeolites (Z1 and Z2) despite their reduced surface area. Thus for zeolites it can be supposed that the surface area not the sole determining factor. The homogeneous structure and chemical affinity of CO2 in zeolites can be important factors for enhancing adsorption capacity. At higher pressure, activated carbons AC1 and AC3 proved comparably or more efficient than zeolites regarding respectively mass and volumetric adsorption capacity. For AC, Table 1 together with Figure 1 shows that the bigger the surface, higher the adsorption capacity. In fact at 0.8 MPa the adsorbent are ranked between themselves by their BET surface value. The BET surface area of the sample AC1 was measured as being the highest of the considered samples. Zeolites Z1 and Z2 are characterized by having a high microporosity despite having a low surface area. When the adsorption isotherms are denoted in moles of gas per unit of volume of adsorbent (Qv) (Figure 1(a)), the best adsorbents are zeolites (i.e. sample Z1). The adsorption capacity of activated carbon AC3 is almost the same than the one of zeolite Z1 at 0.8 MPa and even seems to increase after this pressure. This affirmation is reported and confirmed in results by Siriwardane et al. [22], for activated carbons in general. Although those zeolites have a small BET surface, they demonstrated to be better sorbents than AC at low pressure. Chue et al. [23] affirmed that zeolites are the best sorbent for pressure swing adsorption (PSA) process when adsorption pressure is close to the atmospheric pressure. For adsorbent selection, operating conditions are thus an important element to consider.
Accounting for these results, it was decided to choose AC1 as the selected material for transportation cost evaluation of adsorbed CO2 because of its high sorption capacity per mass of active carbon. Another sorbent from literature, named as AC5, was taken into account thanks to its large Qv [24].
4.2. Ship Transportation and Sequestration Costs
Costs have been evaluated for different means of transport. Figure 2 shows the price per ton of delivered CO2 linked to distance, material (i.e. A1 - A5), and ship size, for an operational condition of 0˚C and 1 MPa. It should be noted that the price increases rapidly with the distance, at rates depending on sorbent material and system capacity.
As it is displayed in Figure 2(a) lot of parameters influence the cost such: the transported amount, the ship size and the distance. The cost variation between the two sorbent materials used in different ship size for transporttation of 5500 t/d of CO2 along 1500 km is weak. At higher distance, as costs increases, the ship size and sorbent used influence the cost more sensitively. In general, the transportation cost for higher delivered amounts (e.g. 20,000 t CO2/d), in the 80,000 DTW ship with AC5, appeared to be more profitable. A reduction of 16% is shifted rising 3.6 times the quantity transported (i.e. from 5500 to 20,000 t CO2/d) if AC5 and 80,000 DTW ship are selected (12,000 km, 0˚C, 1 MPa) (Figures 2(a) & (b)). The amount of transported CO2 in adsorbed via influences the cost but in different range than liquefied CO2 by ship. The saving cost is 30% by ship transport of liquefied CO2 increased by 3 times [4] or 33% in 2.5 increments [2,7]. If the operating pressure is changed to 1.5 MPa the cost is reduced as it is shown in Figure 3.
The ship size has a significant importance for large distances for both adsorbent, in accordance with observations from [8] and [15] for liquefied CO2. Nevertheless, as Ozaki et al. affirmed, when the greatest size ship is used an opposite effect is exerted and the cost increases [15].
The quantity of transported CO2 is associated directly with the sorbent material and its capacity of adsorption, which grows when the pressure rises. The sorbent cost and its capacity play a major role on the total transport expenses linked with the number of required ships and the fuel intake which influence investment, operational and maintenance costs, representing between 78% - 93% of total transportation expenses. The cost of the sorbent material ranked from 1.30 to 13.81 US$/t CO2 (Figure 3). Otherwise storage cost in geological or ocean reservoir could represent between 12% to 33% of total sequestration (transport + storage) cost, being 10.97 US$/t by adsorbed phase and 6.12 US$/t by liquefied CO2. The distance raises both numbers of required ships and of fuel intake, increasing the CO2 transportation cost linearly, as the Figure 4 shows. The rate of increase is higher in adsorbed CO2 by AC1 than by AC5 and by adsorbed phase than by liquefied CO2. Moreover, the liquefied CO2 transport is generally the most attractive condition on economic viewpoint, attributed to its bigger capacity per trip (i.e. investment, operational and maintenance) shown in the transport cost participation of 51% - 78% of total expenses. Nevertheless, the adsorbed CO2 transport shows competitive costs in a distance of 1500 km, with sequestration cost of 32 US$/t for an annual quantity of 7.3 Mt of CO2 (Figure 3).
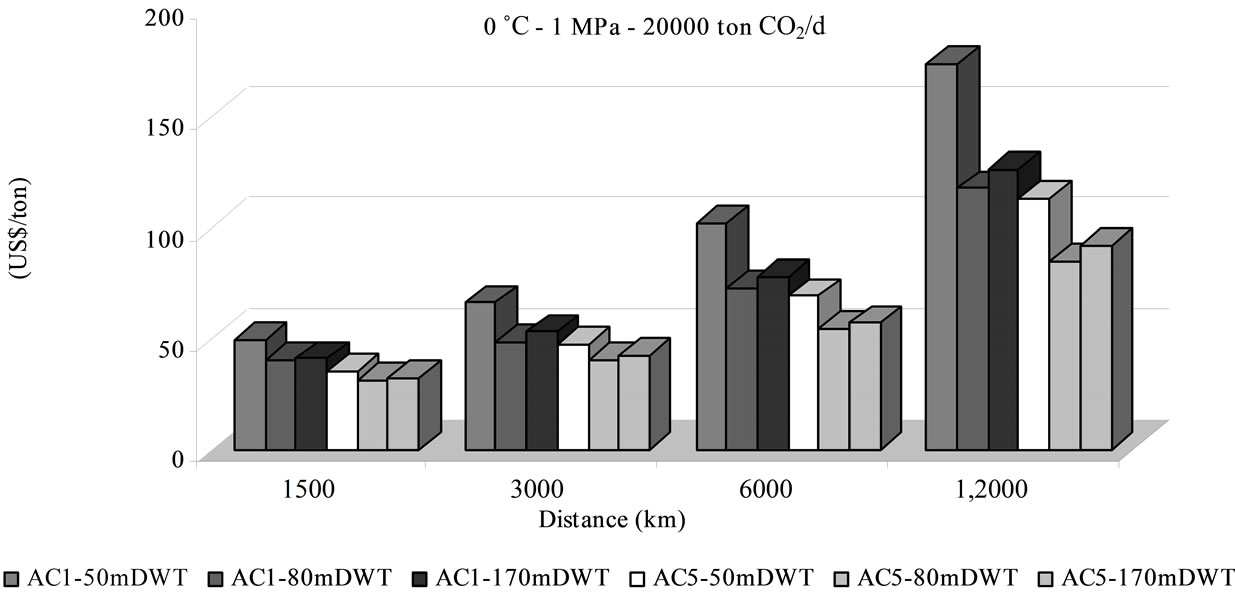
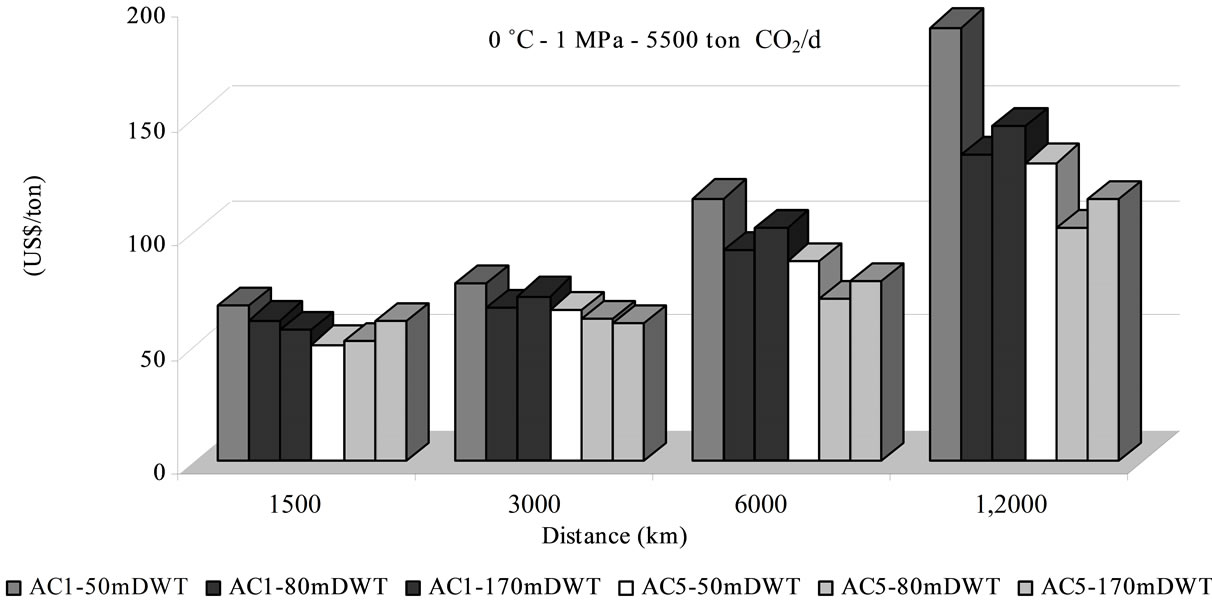
Figure 2. Total transport cost at operating conditions (0˚C, 1 MPa) in adsorbed phase for the considered adsorbent AC1 and AC2 and reservoir and ships from Tables 2 and 3: (a) amount of CO2 produced of 20,000 ton CO2/d and (b) 5500 ton CO2/d.
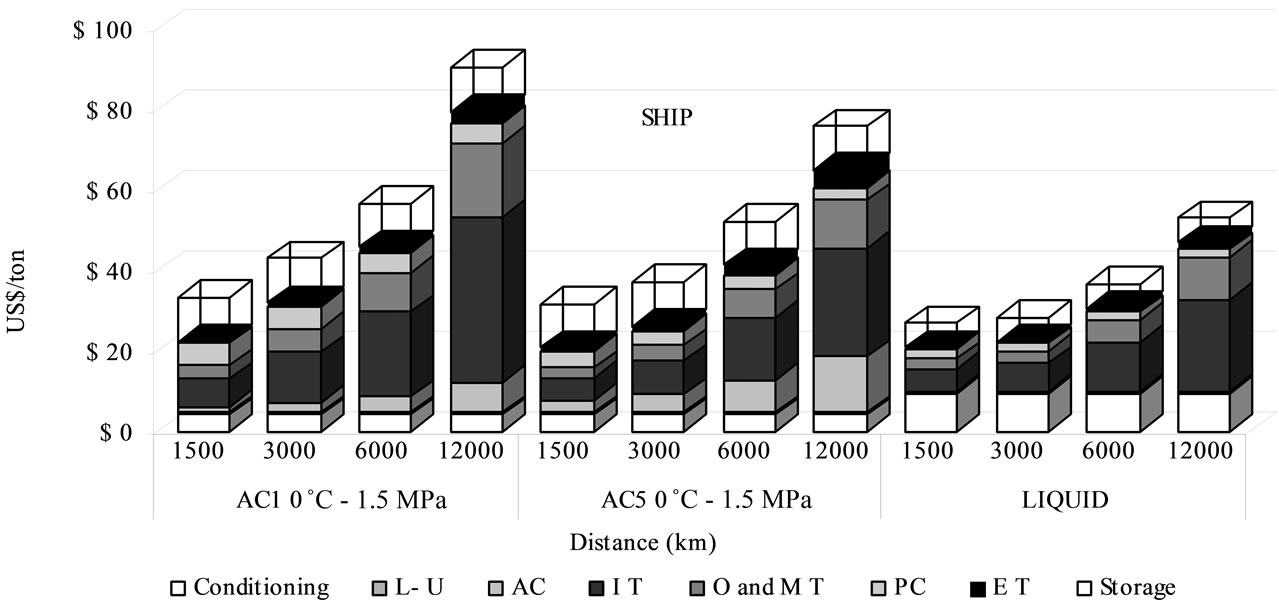
Figure 3. Cost comparison between adsorbed and liquid routes in ship transport for 20,000 ton/d. L-U: Load-download; AC: Activated carbon; IT: Investments of transport; O and MT: Operation and maintenance of transport; PC: Port cost; ET: Energy cost in transport.

Figure 4. Relationship between distance and cost in ship transport.
The CO2 transport in the adsorbed phase brings a smaller cost contribution during the conditioning process, 0˚C for adsorbed phase instead of –50˚C for liquid phase (Figure 3), but the storage cost increases more for adsorbed CO2 than for liquefied transport, due to higher energy requirements, giving a more expensive global cost. Although there is a saving cost in transport fees for conditioning energy requirements by adsorbed phase, it decreases with the distance for the other input costs (i.e. fuel, investment, maintenance, operative cost) that increase the total transportation price. For liquid CO2 transport several authors [2,7,15] attribute to the liquefaction step, the biggest part of the total cost, although its influence in total cost decreases as the distance becomes longer.
The difference cost between the two considered adsorbents is obviously due to the different adsorption capacities of the two sorbents.
4.3. Train Transportation-Sequestration Costship Transportation and Sequestration Costs
In the case of train transport, the considered operating conditions were of 1.5 MPa at 0˚C. Figure 5 shows that, for train, the adsorbed phase is often more economical than liquid one in transportation, especially for a distance of 300 km and adsorbent AC5, with a cost of 14.4 US$/t and a difference of 21%. The total cost of the absorbent material ranked from 0.12 to 0.37 US$/t of transported CO2 (Figure 5).
Components of total transportation cost show that the conditioning price for liquefied CO2 represents 62% of the total (11.3 US$/t CO2), whilst for adsorbed CO2 transportation by AC5 is only 32% (5.4 US$/t CO2). This fact explains why the adsorbed phase transportation is cheaper than the liquid one (Figure 5).
The total transport cost by AC1 material is higher than AC5 because of the lower AC1 volume adsorption capacity (Qv). As a result, by AC5, it is possible to store a larger quantity of CO2 in smaller number of containers and lower maintenance and investment costs are required. The lower cost is also resulting from the reduced energy consumption due to the lower numbers of transport travels.
If the storage step is considered, the total sequestration cost is less for liquefied CO2 than for adsorbed, as the storing cost by adsorbed phase is 10.95 US$/t and by liquefied is 5.81 US$/t. The total sequestration cost by train ranked between 28 and 34 US$/t by AC1, 25 - 28 US$/t by AC5 and 23 - 26 US$/t by liquefied CO2. Adsorbed CO2 transport appears thus to be a more competitive route than liquid route, if transported CO2 is to be used for applications which demand conditioning conditions near the desorption conditions.
4.4. Truck Transportation-Sequestration Costs
The truck system is the worst economical option. Nevertheless, at very short distances and for reduced amounts, it might be a possible alternative compared to pipeline transport because of its lower infrastructure investment cost [25].
For transport by truck, the liquefied CO2 is the most viable condition (Figure 6). For adsorbed CO2 transportation the cost using AC1 material is lower than AC5 material due to the larger fuel consumption, because with AC5, it might be possible to save money regarding the number of necessary trucks, but the fuel cost becomes larger. The fuel cost contribution in the total transport fee of the truck system is the biggest of the three via, with a 25% - 33% for A1, 38% - 48% for A5 and 17% - 24%
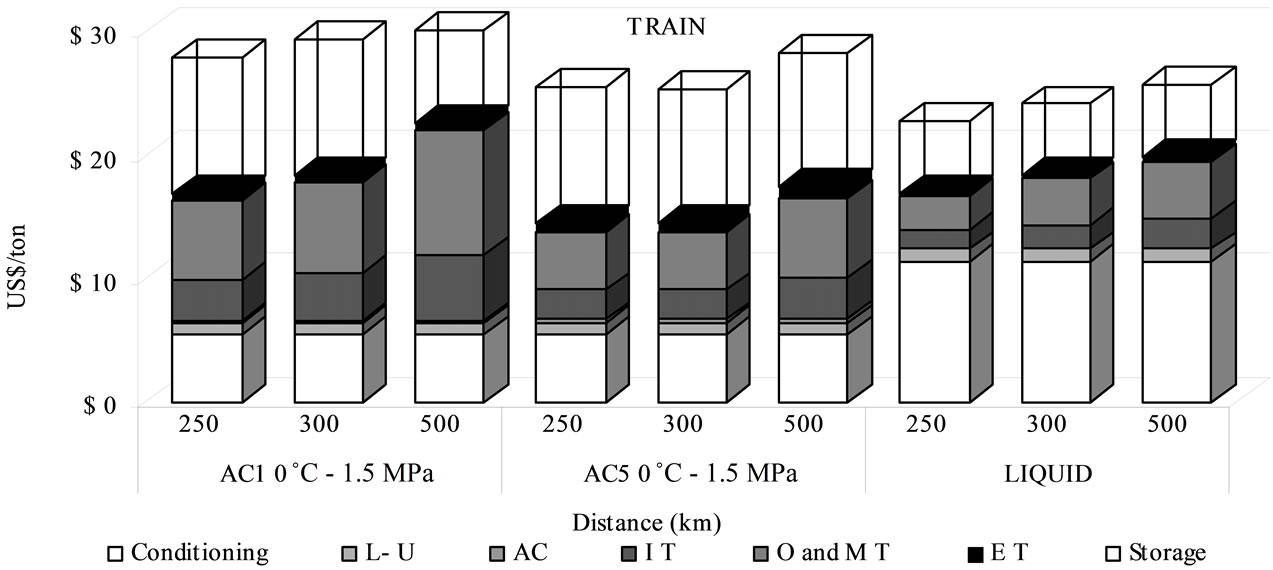
Figure 5. Cost comparison between adsorbed and liquid routes in train transport.
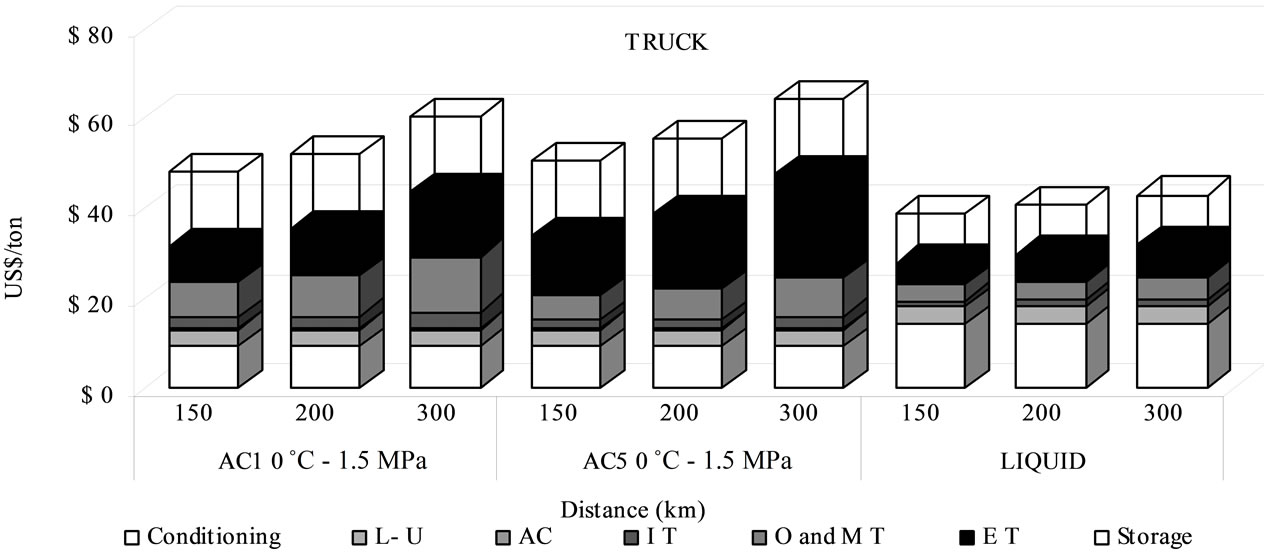
Figure 6. Cost comparison between adsorbed and liquid routes for truck transport.
for the liquid phase (Figure 6) while for ship and train, it is below to 7% (Figures 3-5). The cost of the absorbent material (AC1 - AC5) represents only 0.7% of the total transport fee, between 0.12 and 0.37 US$/t CO2 transported compared to the transportation cost of 32 - 44 US$/t by AC1, 34 - 48 US$/t by AC5.
The storage of CO2 transported in adsorbed phase increases the total sequestration cost in 16 US$/t CO2 whilst for liquefied CO2 it rises in 11 US$/t CO2 (Figure 6).
4.5. CO2 Emissions by CO2 Transport and Storage
As it is shown in Figures 7, 8 and 9, CO2 transport by adsorbed phase exhibits lower CO2 emissions compared to those caused by liquid transport in distances below of 6000 km by ship, 300 km by train and 150 km by truck, although these differences are not above 3% of total CO2 transported. Transport by ship in adsorbed phase with active carbon AC1 is the most effective environmentally with low CO2 emissions compared with liquefied CO2 or adsorbed in AC5. This advantage becomes less signifycant as the distance becomes larger for the bigger fuel use and the storage energy requirements as these last represent between 23% - 49% of emissions whilst by liquefied is only 2%. The fuel ship emissions for adsorbed transport is more significant than for liquid one with a contribution of 9% - 50% in comparison with 3% - 20% (Figure 7).
CO2 emissions of train transportation by adsorbed phase are lower than liquid one when AC5 is used as sorbent. In this case, as mentioned above for train costs, the limiting factor is the volume and as lower energy intake is required, lower CO2 quantities are emitted. Similar to ship via, the fuel contribution is bigger in the adsorbed phase, ranging between 24% - 49%, while for liquid phase conditioning emission are the most important with 79% - 89% of total CO2 (Figure 8). The storage emission is still being most important in adsorbed CO2 than liquefied: it represents 34% - 41% of the total and only 1% for liquid one.
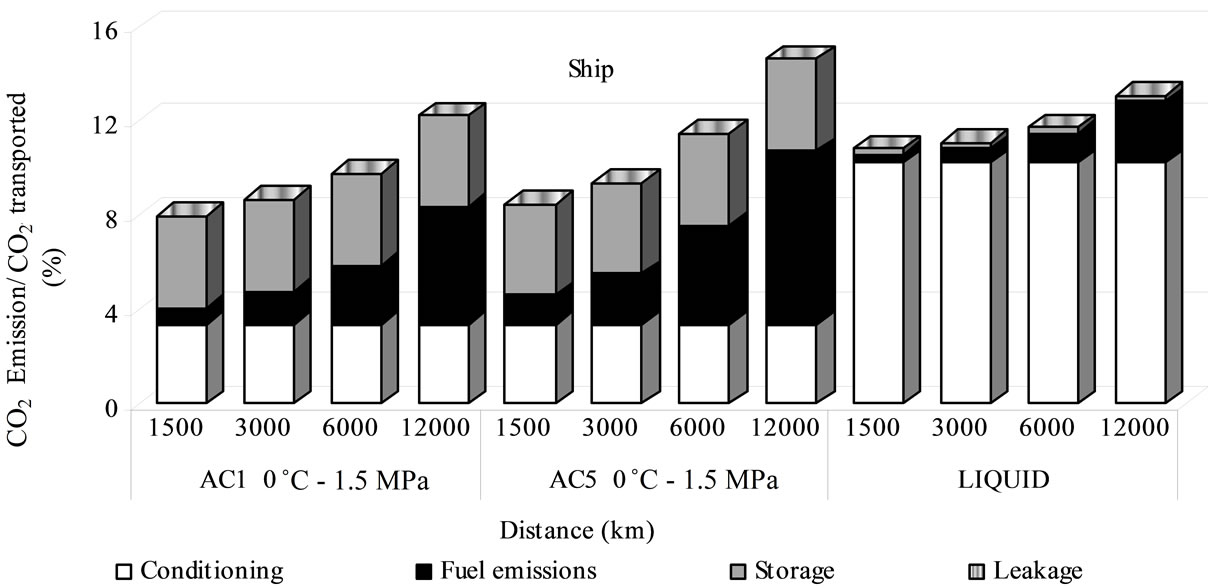
Figure 7. Emission percentage for sequestration by ship transport.
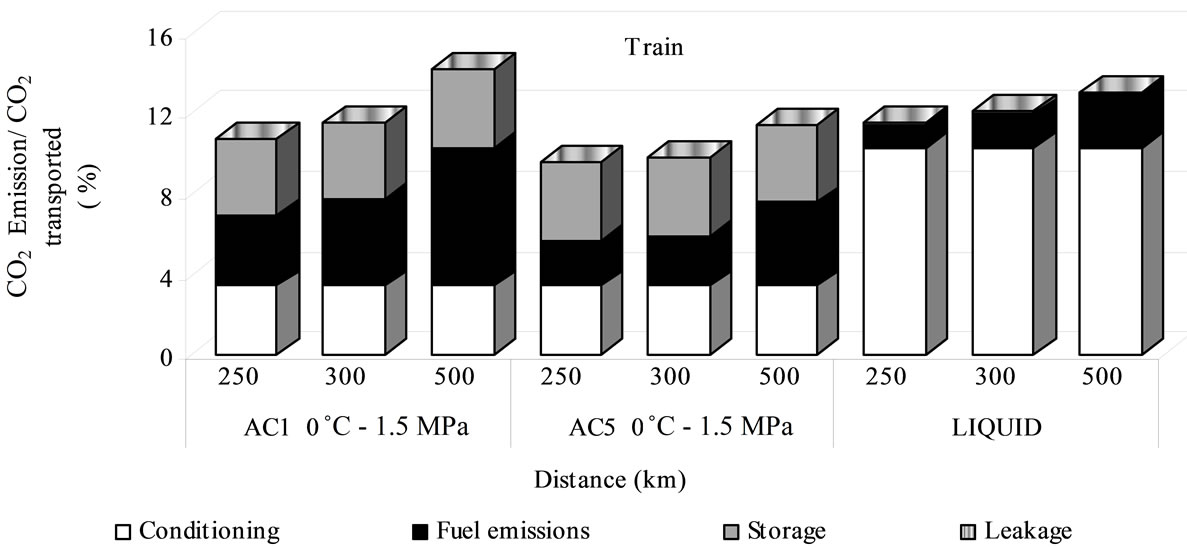
Figure 8. Emission percentage for sequestration by train transport.
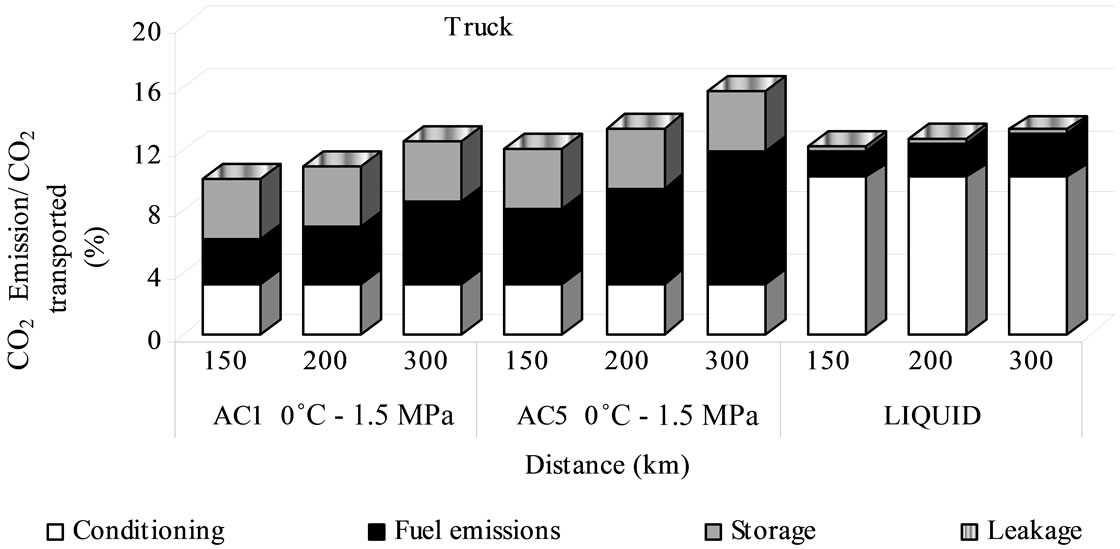
Figure 9. Emission percentage for sequestration by truck transport.
Although, there is a difference between the truck emissions by adsorbed phase and liquid one below 150 km at great distances, these variation becomes weak, mainly when AC5 is used changing the system more environmentally valuable of adsorbed to liquefied. The fuel emissions become the main factor for larger distance (i.e. 300 km) with a contribution of 54% followed for the storing emissions with 25% and a relationship between the CO2 emitted and CO2 transported of 16%; 3% more than the liquid phase (Figure 9).
As it is observed in Figures 7 to 9, CO2 emissions increase with distance, as much for AC5 as for AC1 by ship and by truck, showing that volume capacity of adsorption of active carbon has higher influence on the emission due to fuel consumption of these means, while for train transport the mass capacity of adsorption is the factor that reduces the emissions linked to energy requirements; in contrast with transport in liquid phase which remains almost constant because the larger part of CO2 emissions is produced during the liquefaction not during transportation.
Others parameters as transported amount, change in operating conditions, do not bring important changes to items contributions to emissions. Adsorbent with a larger adsorption capacity could reduce numbers of vehicles necessary to transportation in order to decrease the fuel consumption and to allow emission reductions.
The net cost of CO2 transport and storage is the cost of transport and storage of CO2 accounting for the difference between the CO2 stored and the CO2 emitted during the whole process. It is shown in Figure 10(a), accounting for storage, and Figure 10(b) without storage. The
 (a)
(a)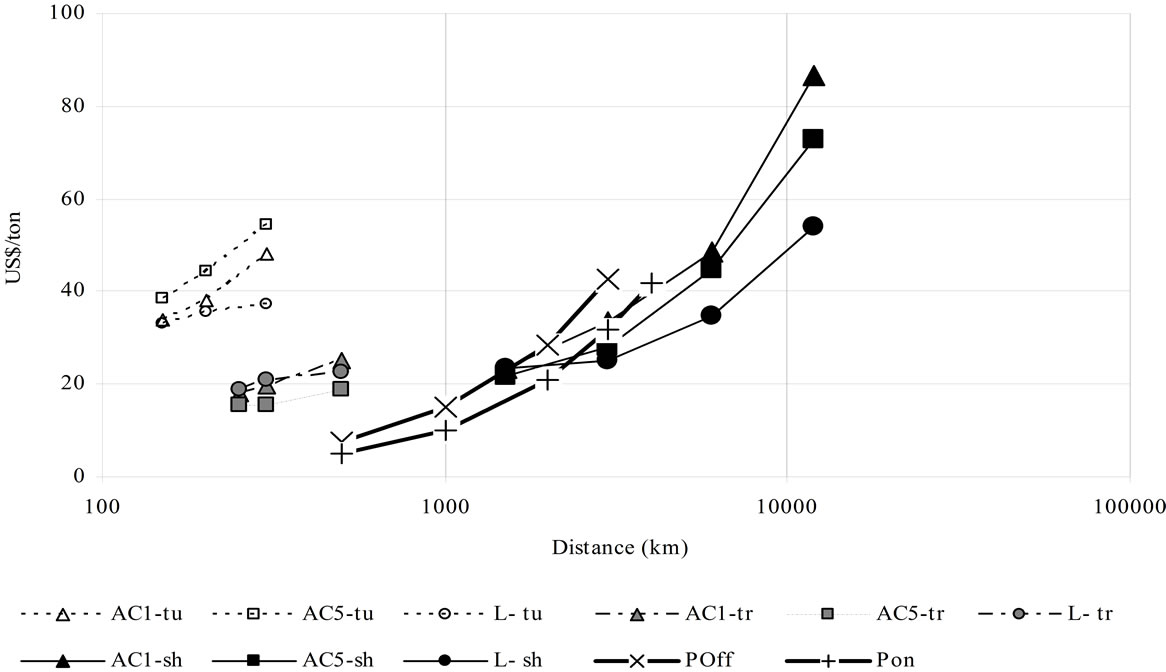 (b)
(b)
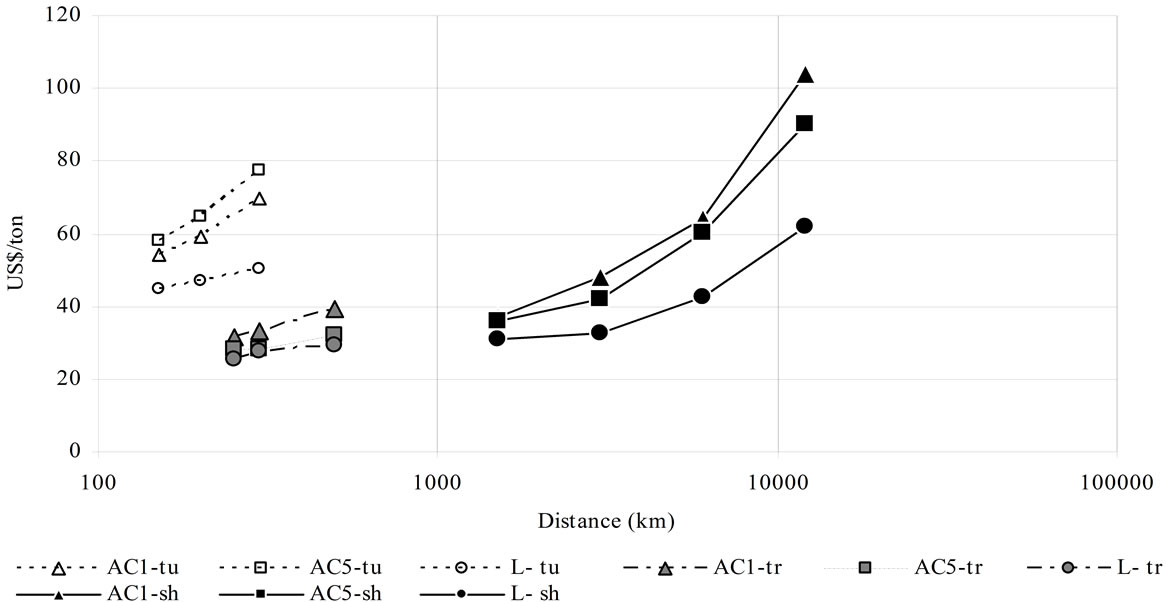
Figure 10. Transport cost for the different means and CO2 quantities. AC1 and AC5 (adsorbed phase on AC1 and AC5) L = liquid phase, tu = truck, tr = train, sh = ship, POff = pipeline offshore without storage [6 Mt CO2/y] [5], POn = pipeline onshore without storage [6 Mt CO2/y] [5]. Colour refers to: Black: 7.3 Mt/y, gray: 0.73 Mt/y, white: 0.18 Mt/y. Above with CO2 storage (a). Below without CO2 storage (b).
CO2 transported by the scenarios of the present study are: 0.18 Mt/y by truck, 0.73 Mt/y by train, 7.3 Mt/y by ship. Notice that direct comparison is not possible because of the cost per ton decreasing with quantity per year. Ranges of net costs are: 0.007 - 0.025 US$/t-km by ship; 0.01 - 0.015 US$/t-km by pipeline; 0.06 - 0.12 US$/t-km by train and 0.17 - 0.4 US$/t-km by truck; with an almost linear relationship between distance and price per ton. Figure 10 also shows a competitive net cost of adsorbed CO2 transport by ship for distances around 1500 km. It is also the case for train transport, especially when transport is not followed by storage.
5. Conclusions
CO2 adsorption capacities of 6 different adsorbents, comprising 4 activated carbons and 2 zeolites, were empirically evaluated in a given range of pressure and temperature. One of the activated carbons named AC1 was able to adsorb nearly 10 CO2 mol/kg when pressure was as high as 0.8 MPa. It was also shown that, in contrast with zeolite, activated carbon capacities were increasing with gas pressure. It was thus selected to being a good candidate for CO2 transport, as well as one other activated carbon, named AC5, selected after literature.
Economic evaluation of CO2 batch transport shows that CO2 adsorbed by AC5 and transported by train is quite competitive on distances between 250 - 500 km in comparison to liquefied CO2, generating a slight over cost of 2% - 10% considering the total cost of transport + storage. AC1 and AC5 were evaluated as being cheaper, if the transported CO2 is used after transport as delivered without storage. Activated carbon AC1 appears to be competitive on short distances by truck when transport is not followed by storage. Ship transport of adsorbed CO2 for small distances (i.e. around 1500 km) appear to be competitive when CO2 is used as delivered and there is no storage; there is an overcost of only 16%, when there is storage after the transport.
The CO2 emissions generated by CO2 sequestration (transport + storage) when transport is carried out in an adsorbed phase were smaller than the ones generated by liquid phase transport below 1200 km with AC1, 500 km with AC5 and 300 km with AC1, by ship, train and truck respectively. Sequestration of adsorbed CO2 transported on 1500 km by ship, generated 27% less CO2 emissions than liquid phase; 17% by train for a distance of 250 km; 16% by truck on 150 km, although all these differences were decreasing with the distance of transport. CO2 emissions of liquid phase transportation were due principally to the liquefaction step, which is not present in the adsorbed phase transportation, whilst for adsorbed mean the fuel intake, booster and energy requirements were the most important factors that increased the emissions.
The CO2 transport in an adsorbed phase thus appears to be an environmentally attractive and rather competitive route compared to liquid one in certain conditions (i.e. short distances). The growing requirements in the active carbon industry for CCS as well as optimization of use may increase interest for CO2 transport in an adsorbed phase. CO2 capture conditions as well as delivering conditions for its use or storage after transport, are determining factors to demonstrate the economic suitability of CO2 transport in an adsorbed phase.
REFERENCES
- R. Svensson, M. Odenberger, F. Johnsson and L. Strömberg, “Transportation systems for CO2—application to carbon capture and storage,” Energy Conversion and Management, Vol. 45, No. 15-16, 2004, pp. 2343-2353. doi:10.1016/j.enconman.2003.11.022
- A. Aspelund, M. J. Molnvik and G. De Koeijer, “Ship transport of CO2: Technical Solutions and analysis of Costs, Energy Utilization, Exergy Efficiency and CO2 emissions,” Chemical Engineering Research and Design, Vol. 84, No. 9, 2006, pp. 847-855. doi:10.1205/cherd.5147
- D. Golomb, “Transport systems for Ocean Disposal of CO2 and Their Environmental Effects,” Energy Conversion and Management, Vol. 38, 1997, pp. 279-286. doi:10.1016/S0196-8904(96)00282-8
- Y. Fujioka, M. Ozaki, K. Takeuchi, Y. Shindo and H. J. Herzog, “Cost comparison in various CO2 Ocean Disposal Options,” Energy Conversion and Management, Vol. 38, 1997, pp. 273-277. doi:10.1016/S0196-8904(96)00281-6
- B. Metz, O.Davidson, H. De Coninck, M. Loos and L. Meyer, “Carbon Dioxide Capture and Storage: IPCC Special Report,” Cambridge University Press, Cambridge, 2005.
- S. Decarre, J. Berthiaud, N. Butin and J.-L. GuillaumeCombecave, “CO2 Maritime Transportation,” International Journal of Greenhouse Gas Control, Vol. 4, No. 5, 2010, pp. 857-864.
- M. Barrio, A. Aspelund, T. Weydahl, T. E. Sandvik, L. R. Wongraven, H. Krogstad, R. Henningsen, M. Molnvik, and S. I. Eide, “Ship-Based Transport of CO2,” Proceedings of the 7th International Conference on Greenhouse Gas Control Technologie, Vancouver, 5-9 September 2004, pp. 1655-1660.
- O. Kaarstad and C. W. Hustad, “Delivering CO2 to Gullfaks and the Tampen Area,” 2003. http:// www.co2.no
- G. Hegerland, T. Jorgensen and J. O. Pande, “Liquefaction and handling of Large Amounts of CO2 for EOR,” Proceedings of the 7th International Conference on Greenhouse Gas Control Technologies, Vancouver, 5-9 September 2004, pp. 2541-2544.
- F. Neele, C. Hendriks and R. Brandsma, “Geocapacity: Economic Feasibility of CCS in Networked Systems,” Energy Procedia, Vol. 1, No. 1, 2009, pp. 4217-4224. doi:10.1016/j.egypro.2009.02.232
- A. Shafeen, E. Croiset, P. L. Douglas and I. Chatzis, “CO2 sequestration in Ontario, Canada. Part I: Storage Evaluation of Potential Reservoirs,” Energy Conversion and Management, Vol. 45, No. 17, 2004, pp. 2645-2659. doi:10.1016/j.enconman.2003.12.003
- S. Sircar, T. C. Golden and M. B. Rao, “Activated carbon for Gas Separation and storage,” Carbon, Vol. 34, No. 1, 1996, pp. 1-12. doi:10.1016/0008-6223(95)00128-X
- A. Subrenat and P. Le Cloirec, “Volatile Organic Compound (VOC) removal by adsorption onto Activated Carbon Fiber Cloth and Electrothermal Desorption: An Industrial Application,” Chemical Engineering Communications, Vol. 193, No. 4, 2006, pp. 478-486. doi:10.1080/00986440500191768
- M. Pellerano, P. Pré, M. Kacem and A. Delebarre, “CO2 capture by adsorption on Activated Carbons Using Pressure Modulation,” Energy Procedia, Vol. 1, No. 1, 2009, pp. 647-653. doi:10.1016/j.egypro.2009.01.085
- M. Ozaki, J. Davison, J. Minamiura, E. S. Rubin, D. W. Keith, C. F. Gilboy, M.Wilson, T. Morris, J. Gale and K. Thambimuthu, “Marine transportation of CO2,” Proceedings of the 7th International Conference on Greenhouse Gas Control Technologies, Vancouver, 5-9 September 2004, pp. 2535-2539.
- Z. Oztürk, “Investigating Optimum Speed in High-Speed Railway: Istanbul-Ankara Corridor,” ARI The Bulletin of the Istanbul Technical University, Vol. 54, No. 3, 2004, pp. 78-89.
- W. Edwards, “Estimating Farm Machinery Costs,” 2009. http://www.extension.iastate.edu
- M. Aka, M. Nishio, M. Iijima, M. Ozaki, J. Minamiura, T. Tanaka, E. S. Rubin, D. W. Keith, C. F. Gilboy, M. Wilson, T. Morris, J. Gale and K. Thambimuthu, “Performance and Economic Evaluation of CO2 capture and sequestration technologies,” Proceedings of the 7th International Conference on Greenhouse Gas Control Technologies, Vancouver, 5-9 September 2004, pp. 1213-1219.
- A. Aspelund, T. E. Sandvik, H. Krogstad and G. Koeijer, “Liquefaction of captured CO2 for Ship-Based Transport,” Proceedings of the 7th International Conference on Greenhouse Gas Control Technologies, Vancouver, 5-9 September 2004, pp. 2545-2549.
- J. A. Kent, “Riegel’s Hand Book of Industrial Chemistry,” 10th Edition, Kluwer Academic Press, New York, 2003.
- Y. Haseli, G. F. Naterer and I. Dincer, “Comparative assessment of Greenhouse Gas Mitigation of Hydrogen Passenger Trains,” International Journal of Hydrogen Energy, Vol. 33, No. 7, 2008, pp. 1788-1796. doi:10.1016/j.ijhydene.2008.02.005
- R. V. Siriwardane, M. S. Shen and E. P. Fisher, “Adsorption of CO2, N2, and O2 on Natural Zeolites,” Energy Fuels, Vol. 17, No. 3, 2003, pp. 571-576. doi:10.1021/ef020135l
- K. T. Chue, J. N. Kim, Y. J. Yoo, S. H. Cho and R. T. Yang, “Comparison of Activated Carbon and zeolite 13X for CO2 recovery from Flue Gas by Pressure Swing Adsorption,” Industrial & Engineering Chemistry Research, Vol. 34, No. 2, 1995, pp. 591-598. doi:10.1021/ie00041a020
- X. Py, V. Goetz and G. Plantard, “Activated Carbons Textural Optimization for Gas Storage Processes,” Chemical Engineering and Processing: Process Intensification, Vol. 47, No. 3, 2008, pp. 308-315. doi:10.1016/j.cep.2007.01.007
- Centre de la Technologie de l’Energie du CANMET (CTEC), “Carbon Capture & Storage CO2 Capture and Storage Roadmap,” 2006. http://www.co2trm.gc.ca
NOTES
*Corresponding author.

