Journal of Agricultural Chemistry and Environment
Vol.2 No.3(2013), Article ID:35678,6 pages DOI:10.4236/jacen.2013.23009
Effects of metal ions on the activity and stability of peroxidase in Tartary buckwheat shoots
![]()
1Faculty of Fisheries Sciences, Hokkaido University, Hakodate, Japan
2National Agriculture and Food Research Organization, Hokkaido Agricultural Research Center, Sapporo, Japan; *Corresponding Author: tsuzu@affrc.go.jp
3National Agriculture and Food Research Organization, Hokkaido Agricultural Research Center, Memuro Upland Farming Research Station, Memuro, Japan
Received 1 May 2013; revised 17 June 2013; accepted 2 July 2013
Copyright © 2013 Daisuke Mikami et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
This is the first report to purify and characterize POX in shoots of buckwheat species. POX was partially purified from Tartary buckwheat shoots by 48.0 fold with a final yield of 9.07%. During ion-exchange and gel filtration chromatography, only one peak corresponding to POX activity was found. The molecular weight of POX was determined to be 37.5 kDa using gel filtration chromatography. The optimal pH of POX activity was 5.5 (guaiacol, quercetin) and 5.0 (ABTS). The Km of POX activity was 22.3 mM (guaiacol), 6.3 mM (ABTS) and 0.92 mM (quercetin). In contrast, the Km for quercetin in the presence of Fe3+ ions was two orders of magnitude less (0.018 mM) than that in its absence. The stability of POX activity was increased in the presence of trivalent metal ions, even after 186 h in solution. POX activity was retained by 83.6% and 56.1% in the presence of 1 mM Fe3+ and Al3+ ions, respectively, whereas it was completely inactivated in their absence. To the best of our knowledge, this is the first study to detail the activation and stabilization of POX activity in relation to trivalent metal ions.
Keywords: Tartary Buckwheat; Characterization; Peroxidase; Metal Ion; Quercetin; Km
1. INTRODUCTION
Buckwheat is a rich source of functional polyphenols such as rutin and quercetin [1-4]. Tartary buckwheat leaves contain rutin and anthocyanins in high concentrations (rutin: ca 100 mg/g DW, anthocyanin: ca 0.8 mg/g DW) [5,6]. For plants, the physiological roles of polyphenols are reported to provide UV-B protection and have antimicrobial properties [7]. Although some physiological roles of polyphenols, including UV screening, anti-desiccation and anti-cold stresses [5], related to stress resistance have been studied in buckwheat, the roles of polyphenols still remain unclear.
On the other hand, plants have polyphenol-oxidizing enzymes such as peroxidase, which is related to lignification [8] and resistance against infection by pathogens. Tartary buckwheat (Fagopyrum tataricum) shoots contain a substantial amount of peroxidase (POX) activity, the characteristics and physiological roles of which have not been studied. Peroxidases are classified by various categories on the basis of substrate specificity and comparison of amino acid sequences. For example, ascorbate peroxidase (APX, EC 1.11.1.11) and plant peroxidase (POX, EC 1.11.1.7) are heme-containing proteins and glutathione peroxidase (GPX, EC 1.11.1.9) is a protein that forms oxidized glutathione, which is related to antioxidants and detoxification. POXs are considered to have different properties in different tissues [8]. Therefore, characterization of POX is important in order to understand the physiological roles of polyphenols in buckwheat leaves.
POX has been partially purified and characterized in both common (F. esculentum) and Tartary buckwheat seeds [9,10]. These reports revealed that POX consists of at least two isozymes and has a lower Km for phenolic substrates such as quercetin and guaiacol than other substrates. However, to the best of our knowledge, the purification and characterization of POX in buckwheat shoots has not yet been reported in buckwheat species. Some papers have described POX as being activated by Ca2+, Fe3+ or Al3+ ions [21,22]. Some kinds of POX protein have been reported to contain metal ions, but Ca2+ sometimes inhibited POX activities [23]. These reports revealed the importance concerning the effects of metal ions on POX activity in studying the physiological roles of POX. Therefore, we investigated the effects of metal ions on purified POX activities as well.
2. MATERIALS AND METHODS
2.1. Plant Material
Tartary buckwheat (F. tataricum var. Hokkai T10) was grown in the experimental field at the National Agricultural Research Center for the Hokkaido Region in Memuro, Hokkaido, Japan. On June 7th, 2012, shoots grown to 5 days after germination were collected and stored at −80˚C until they were used.
2.2. Extraction and Purification of POX
Buckwheat shoots (20 g fresh weight) were homogenized with 200 ml of extraction buffer containing 50 mM acetate-LiOH buffer (pH 5.5) and 0.3% TritonX-100 (v/v) for 30 min according to the method in previous report [11]. A crude enzyme solution was obtained by centrifugation and then precipitated with 0% - 80% saturation of solid (NH4)2SO4. The precipitate was dissolved in buffer A, which contained 50 mM acetate-LiOH buffer (pH 5.5), and dialyzed. The dialyzed enzyme solution was applied to a 2.4 × 7.0 cm SP-Sepharose column (GE healthcare Japan cooperation; Asahigaoka, 191-0065 Hino Tokyo Japan) and equilibrated with buffer A. POX was eluted with a linear gradient of 50 - 500 mM LiCl in the same buffer. Active fractions were collected and further purified by gel filtration chromatography on a 1.5 × 55 cm Sephacryl S-200 column (GE healthcare Japan cooperation) with equilibrated buffer A, which contained 150 mM LiCl, and eluted with the same buffer. All of the above steps were carried out as soon as possible at 4˚C. In each purification step, POX activity was measured using guaiacol as the substrate. After gel filtration, POX activity became unstable, and did not improve, compared with cation exchange chromatography, following a subsequent purification fold. Therefore, we tried to stabilize POX by adding both FeCl3 and AlCl3. However, addition of these ions caused a decrease in performance after gel filtration (data not shown). We then decided to add FeCl3 or measure enzymatic properties as soon as possible after active fractions were obtained by gel filtration. Using guaiacol as substrate, one unit of POX activity was defined as the amount of enzyme which increased absorbance by 1.0 min−1 at 490 nm. Total protein content was measured using bovine serum albumin (BSA) as the standard protein.
2.3. Assay of POX Activity
POX was assayed according to the method of previous report [11]. POX activities were determined at 25˚C by measuring the initial rate of the increase in absorbance at 490 nm (guaiacol) and 415 nm (ABTS), and the decrease in absorbance at 370 nm (quercetin). The assay mixture contained 50 mM buffer at optimal pH, 9.3 mM H2O2, 85 mM guaiacol 87 mM ABTS and 0.3 mM quercetin as the substrate.
2.4. Estimation of Molecular Weight of POX
Gel filtration chromatography was used to estimate the molecular weight of POX according to the method in previous report [11]. A POX solution was loaded on a 1.5 × 55 cm Sephacryl S-200 column with equilibrated buffer A, which contained 150 mM LiCl. The molecular weight was determined using a calibration curve derived from the following standard proteins: β-amylase (200 kDa), BSA (66 kDa), carbonic anhydrase (29 kDa), and cytochrome C (12.4 kDa).
2.5. Determination of Optimal pH and Km
Using guaiacol, ABTS and quercetin as substrates, POX activity was determined under the following pH levels at 25˚C: 2.0 (50 mM glycine-HCl buffer); 3.0 - 4.0 (50 mM citrate-LiOH buffer); 5.0 - 6.0 (50 mM acetate-LiOH buffer); 7.0 (50 mM phosphate-LiOH buffer); 8.0 (50 mM Tris-HCl buffer); and 9.0 - 10.0 (50 mM borate-LiOH buffer). Km was determined by LineweaverBurk plots at different concentrations of each substrate. The substrate mixture contained 1 mM FeCl3.
2.6. Effect of Various Metal Ions on Purified POX Activity
After the gel filtration step, purified POX was incubated for 0 h, 96 h and 186 h at 4˚C with 1 mM of various metal chlorides (FeCl3, AlCl3, CaCl2, MnCl2, MgCl2, KCl and NaCl), and POX activity was measured using guaiacol as a substrate. Assay was performed according to the method of previous report [11].
3. RESULTS AND DISCUSSION
3.1. Purification of Peroxidase from Tartary Buckwheat Shoots
POX in Tartary buckwheat shoot was purified by ammonium sulfate precipitation, cation-exchange chromatography and gel filtration (Table 1). We found one large peak corresponding to POX activity on cation-exchange chromatography (Figure 1). In comparison, buckwheat seeds contain at least two peaks corresponding to POX

Table 1. Purification of the POX in Tartary buckwheat shoots.
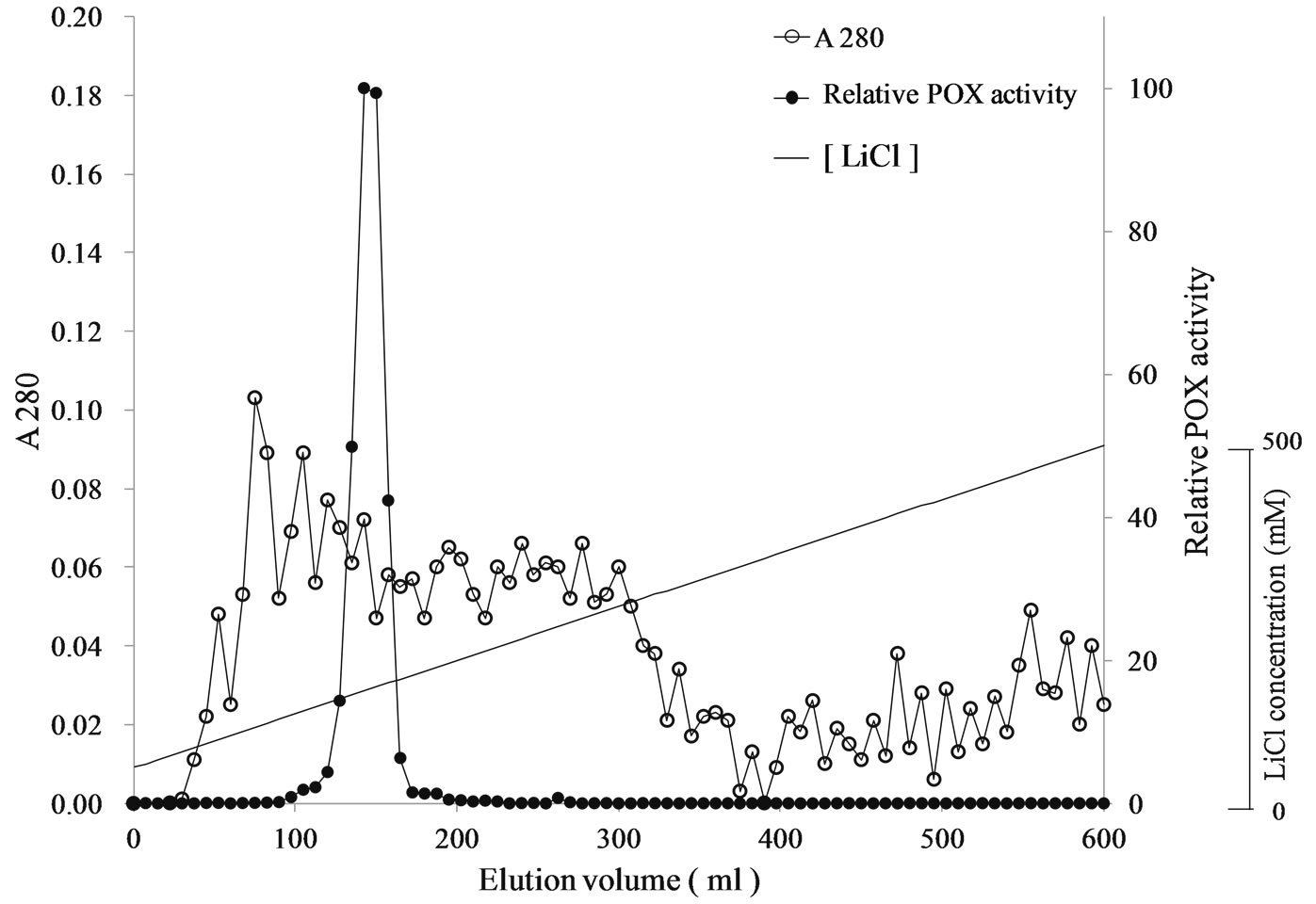
Figure 1. Elution profile of POX activity on the SP-sepharose column
activity [3,10]. Therefore, the composition of cationic POX isozymes in Tartary buckwheat shoots must be different from that found in common and Tartary buckwheat seeds. Slight POX activity was also found on anion-exchange chromatography (data not shown), even though anionic POX was too weak to purify. It reported that POX isozymes induced by UV-B radiation in buckwheat seeds differed from those induced in leaves [9]. These results suggest that POX may serve different roles in seeds compared with shoots. Based on gel filtration chromatography, the molecular weight of POX from buckwheat shoots was estimated to be 37.5 kDa. This value was smaller than that of POX from both common (46.1 kDa) [10] and Tartary buckwheat seeds (46.8 kDa) [11], but similar to that of both Allium sativa L. (37.8 kDa) [12] and Withania somnifera (34 kDa) [13].
3.2. Optimal pH of Purified POX
The optimal pH of purified POX was investigated using guaiacol, ABTS and quercetin as substrates, and found to be similar to each substrate used in this study (Table 2). The optimal pH for guaiacol was 5.5. This value was similar to that of peroxidase from both Spinacia oleracea leaves (pH 5.2) [14] and Brassica napus L. (pH 5.5) [15]. POX activity decreased with either an increase or a decrease of pH. The optimal pH for guaiacol in common buckwheat seed was 9.0, which was different from that in POX from Tartary buckwheat shoots [10]. On the other hand, the optimal pH for guaiacol in Tartary buckwheat seed was 6.0, which was similar to that of POX in Tartary buckwheat shoots for guaiacol [11]. The optimal pH of POX in Tartary buckwheat shoots for ABTS was 5.0. This value was similar to those of peroxidase from Cucumis melo L. (pH 5.5) [16] and Capsicum annuum L. (pH 4.5) [17]. The optimal pH for quercetin was 5.5, which was higher than that of peroxidase from both Vitis vinifera and Allium sativum L. [12,18]. When quercetin was used as a substrate, POX could retain activity in a wider pH range compared to guaiacol and ABTS. Buckwheat shoots, and especially leaves and cotyledon, contain a large amount of rutin [3,19] and exhibit rutinosidase activity [19]. When these structures are damaged by UV and cold stress, quercetin is generated from rutin via rutinosidase activity [3]. Therefore, a wider pH range of POX activity against quercetin may relate to quercetin metabolism in vivo.

Table 2. Optimum pH of purified POX.
This reinforces the idea that the anti-fungal agent 3,4- dihydroxybenzoic acid is formed by peroxidase-dependent oxidation of quercetin on browning onion scales [20].
3.3. Effect of Metal Ions on Purified POX Activity
After gel-filtration chromatography, the effects of various metal ions on purified POX activity were investigated at 0 h, 96 h and 186 h in storage (Table 3). At 0 h, POX activity ranged between 93.8% (CaCl2) to 127% (FeCl3) compared to the activity without metal ions (= defined as 100%). After storage at 96 h and 186 h, POX did not retain activity in either the absence or presence of divalent and monovalent metal ions. On the other hand, when FeCl3 or AlCl3, where added POX activity was retained even after storage at 186 h at 83.6% (FeCl3) and 56.1% (AlCl3). These results suggest that POX in Tartary buckwheat shoots can be stabilized by trivalent metal ions. In addition, it suggests that FeCl3 stabilizes POX more effectively than AlCl3. Numerous reports have shown that POX is activated by divalent ions such as Ca2+ ions [21,22]. On the other hand, only a few studies have reported that POX is activated by both Fe3+ [23] and Al3+ [24]. In addition, in these reports, POX activities were activated only about 2.1 to 3.3 folds. However, no studies have found that the stability of POX is enhanced in the presence of trivalent metal ions. Therefore, our finding reveals novel characteristics concerning POX and suggests the importance of studying its physiological roles in plants.
3.4. Kinetics of Purified POX
We measured the Km of POX both in the presence and absence of FeCl3 and AlCl3 using guaiacol, ABTS and quercetin as substrates. The results are summarized in Table 4. In the absence of the trivalent ions, Km values

Table 3. Effect of metal ions on POX activity.
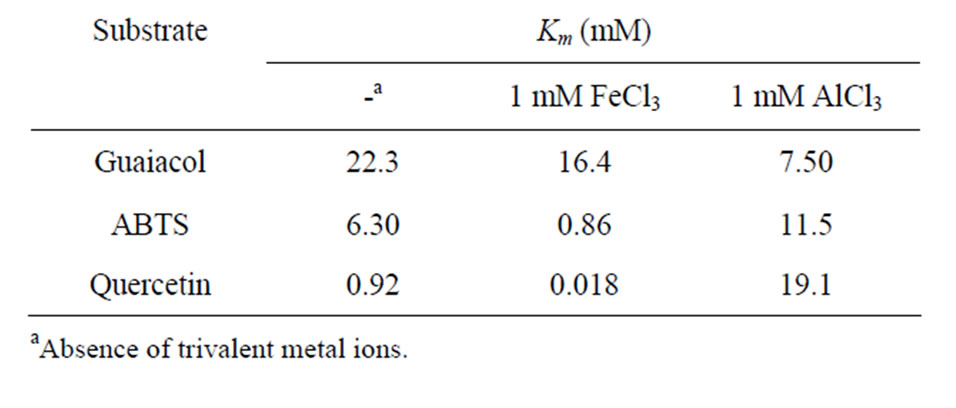
Table 4. Km of purified POX.
were 22.3 mM, 6.3 mM and 0.92 mM for guaiacol, ABTS and quercetin, respectively. Compared to POX in other plants, the Km for guaiacol was higher than that from Jastropha curcas leaves (0.17 mM) [25] and Brassica napus roots (3.7 mM) [15]. The Km for ABTS was also higher than that from Eupatrium odoratutum (0.12 mM) [26] and Brassica napus roots (0.7 mM) [15]. In addition, the Km for quercetine was much higher than that from both Allium sativum L. (0.033 mM) [12] and Vicia faba leaves (0.023 mM) [27]. Furthermore, the presence of FeCl3 enhanced the affinity of POX, resulting in a decreased Km of 16.4 mM, 0.86 mM and 0.018 mM for guaiacol, ABTS and quercetin, respectively. The presence of FeCl3 led to a dramatic decrease of Km, about 51.1 fold, for quercetin compared to its absence, which, when compared to the Km for quercetin in other plants, is one of the lowest values so far reported in: both common buckwheat seed (0.028 mM) [10] and Vicia faba leaves (0.023 mM) [27]. On the other hand, the presence of AlCl3 increased the Km for quercetin about 20 fold compared to Km in its absence. These results suggest that POX has different mechanisms for controlling its activity in relation to both FeCl3 and AlCl3. Based on these results, we suggest that POX in Tartary buckwheat shoots have unique characteristics, and novel aspects related to the physiological roles of POX that warrant for further investigation.
4. ACKNOWLEDGEMENT
We thank to Mr. S. Nakamura and Mr. T. Fukaya for their assistance in the field. We also thank to Ms. K. Fujii, Ms. M. Hayashida and Ms. T. Ando for their technical assistance.
REFERENCES
- Kitabayashi, H., Ujihara, A., Hirose T. and Minami, M. (1995) Varietal differences and heritability for rutin content in common buckwheat, Fagopyrum esculentum Moench. Breeding Science, 45, 75-79.
- Kitabayashi, H., Ujihara, A., Hirose, T. and Minami, M. (1995b) On the genotypic differences for rutin content in Tartary buckwheat, Fagopyrum tataricum Gaerth. Breeding Science, 45, 189-194
- Suzuki, T., Honda, Y. and Mukasa, Y. (2005) Effect of UV-B radiation, cold and desiccation stress on rutin concentration and rutin glucosidase activity in tartary buckwheat (Fagopyrum tataricum) leaves. Plant Science, 168, 1303-1307. doi:10.1016/j.plantsci.2005.01.007
- Dadakova, E. and Kalinova, J. (2010) Determination of quercetin glycosides and free quercetin in buckwheat by caplillary micellar electrokinetic chromatography. Journal of Separation Science, 33, 1633-1638. doi:10.1002/jssc.200900809
- Fabjan N, Rode J, Kosir IJ, Wang Z, Zhang Z and Kreft I. (2003) Tartary buckwheat (Fagopyrum tataricum Gaertn.) as a source of dietary rutin and quercitrin. Journal of Agricultural and Food Chemistry, 51, 6452-6455. doi:10.1021/jf034543e
- Suzuki, T., Watanabe, M., Iki, M., Aoyagi, Y., Kim, S.J., Mukasa, Y., Yokota, S., Takigawa, S., Hashimoto, N., Noda, T., Yamauchi, H. and Matsuura, C. (2009) Timecourse study and effects of drying method on concentrations of γ-aminobutyric acid, flavonoids, anthocyanin, and 2’-hydroxynicotianamine in leaves of buckwheats. Journal of Agricultural and Food Chemistry, 57, 259-264. doi:10.1021/jf802731d
- Harborne, J.B. and Williams, A.C. (2000) Advances in flavonoid research since 1992. Phytochemistry, 55, 481- 504. doi:10.1016/S0031-9422(00)00235-1
- Logrimini, L.M. and Rothstein, S. (1987) Tissue specificity of tobacco peroxidase isozymes and their induction by wounding and tobacco mosaic virus infection. Plant physiology. 84, 438-442. doi:10.1104/pp.84.2.438
- Jovanovic, Z.S., Milosevic, J.D. and Radovic, S.R. (2006) Antioxidative enzymes in the response of buckwheat (Fagopyrum esculentum Moench) to ultraviolet B radiation. Journal of Agricultural and Food Chemistry, 54, 9472-9478. doi:10.1021/jf061324v
- Suzuki, T., Honda, Y., Mukasa, Y. and Kim, S.J. (2006) Characterization of peroxidase in buckwheat seed. Phytochemistry, 67, 219-224. doi:10.1016/j.phytochem.2005.11.014
- Suzuki, T., Shin, D.H., Woo, S.H., Mukasa, Y., Morishita, T., Noda, T., Takigawa, S., Hashimoto, N., Yamauchi, H. and Matsuura-Endo, C. (2012) Caracterization of peroxidase in Tartary buckwheat seed. Food Science and Technology Research, 18, 571-575. doi:10.3136/fstr.18.571
- Marzouki, M.S., Almagro, L., Sabater-Jara, B.A., Barcelo, R. and Pedreno, A. (2010). Kinetic characterization of a basic peroxidase from Garlic (Allium sativum L.) cloves. Journal of Food Science, 75, 740-746. doi:10.1111/j.1750-3841.2010.01848.x
- Johri, S., Jamwal, U., Rasool, S., Kumar, A., Verma, V. and Qazi, G.N. (2005) Purification and characterization of peroxidases from Withania somnifera (AGB 002) and their ability to oxidize IAA. Plant science, 169, 1014- 1021. doi:10.1016/j.plantsci.2005.05.015
- Koksal, E. (2011) Peroxidase from leaves of spinach (Spinacia oleracea): Partial purification and some biochemical properties. International Journal of Pharmacology, 7, 135-139.
- Duarte-Vazquez, M.A., Garacia-Almendarez, B.E., Regalado, C. and Whitaker, J.R. (2001) Purification and properties of a neutral peroxidase isozyme from turnip (Brassica napus L. var. purple Top White Globe) root. Journal of Agricultural and Food Chemistry, 49, 4450- 4456. doi:10.1021/jf010043e
- Rodriguez-Lopez, N., Espin, J.C., Amor, F.D., Tudela, J., Martines, V., Cerda, A. and Garcia-Canovas, F. (2000) Purification and kinetic characterization of an anionic peroxidase from melon (Cucumis melo L.) cultivated under different salinity conditions. Journal of Agricultural and Food Chemistry, 48, 1537-1541. doi:10.1021/jf9905774
- Serrano-Martinez, A., Fortea, I.M., Amor, M.F. and Nunez-Delicado, E. (2008) Kinetic characterization and thermal inactivation study of partially purified red pepper (Capsicum annuum L.) peroxidase. Food Chemistry, 107, 193-199. doi:10.1016/j.foodchem.2007.08.028
- Morales, M. and Barcelo, R.A. (1997) A basic peroxidase isozyme from vacuoles and cell walls of Vitis vinifera. Phytochemistry, 45, 229-232. doi:10.1016/S0031-9422(96)00825-4
- Suzuki, T., Kim, S.J., Takigawa, S., Mukasa, Y., Hashimoto, N., Saito, K., Noda, T., Matsuura-Endo, C., Zaidul, I.S.M. and Yamauchi, H. (2007) Changes in rutin concentration and flavonol-3-glucosidase activity during seedling growth in Tartary buckwheat (Fagopyrum tataricum Gaertn). Canadian Journal of Plant Science, 87, 83-87. doi:10.4141/P05-151
- Takahama, U. and Hirota, S. (2000). Deglucosidase of quercetin glucosides to the aglycone and formation of antifungal agents by peroxidase dependent oxidation of quercetin on browing of onion scales. Plant and Cell Physiolpgy, 41, 1021-1029. doi:10.1093/pcp/pcd025
- Mura, A., Lougu, S., Padiglia, A., Rinaldi, C.A., Floris, G. and Medda, R. (2005) Reversible thermal inactivation and conformational states in denaturant guanidium of a calcium-dependent peroxidase from Euphorbia characias. International Journal of Biological Macromolecules, 37, 205-211. doi:10.1016/j.ijbiomac.2005.10.010
- Mohamed, A.S., Abulnaja, O.K., Ads, S.A., Khan, A.J. and Kumosani, A.T. (2011) Characterisation of an anionic peroxidase from horseradish cv. Balady. Food Chemistry, 128, 725-730. doi:10.1016/j.foodchem.2011.03.096
- Manu, B.T. and Rao, U.S.J.P. (2009) Calcium modulated activity enhancement and thermal stability study of a cationic peroxidase purified from wheat bran. Food Chemistry, 114, 66-71. doi:10.1016/j.foodchem.2008.09.028
- Onsa, H.G., Saari, B.N., Selamat, J. and Bakar, J. (2004) Purification and characterization of membrane-bound peroxidases from Metroxylon sagu. Food Chemistry, 85, 365-376. doi:10.1016/j.foodchem.2003.07.013
- Cai, F., Yang, C., Duan, P., Gao, S., Xu, Y. and Chen, F. (2012) Purification and characterization of a novel thermal stable peroxidase from Jastropha curcas leaves. Journal of Molecular Catalysis B: Enzymatic, 77, 59-66. doi:10.1016/j.molcatb.2011.12.002
- Rani, D.N. and Abraham, T.E. (2006) Kinetic study of a purified anionic peroxidase isolated from Eupatorium odoratum and its novel application as time temperature indicator for materials. Journal of Food Engineering, 77, 594-600.
- Takahara, U. and Egashira, T. (1991) Peroxidases in vacuoles of Vicia faba leaves. Phytochemistry, 30, 73-77. doi:10.1016/0031-9422(91)84101-W
ABBREVIATIONS
ABTS: 2,2’-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid);
DW: dry weight.

