Food and Nutrition Sciences
Vol.05 No.20(2014), Article ID:50617,12 pages
10.4236/fns.2014.520209
The Role of Copper and Vitamin A Deficiencies Leading to Neurological Signs in Captive Cheetahs (Acinonyx jubatus) and Lions (Panthera leo) in the United Arab Emirates
Claudia Kaiser1,2, Ulrich Wernery1, Jörg Kinne1, Laurie Marker3, Annette Liesegang2,4
1Central Veterinary Research Laboratory, Dubai, United Arab Emirates
2Institute of Animal Nutrition, Vetsuisse Faculty Zurich, Zürich, Switzerland
3Cheetah Conservation Fund, Farm Elandsvreugde #367, Otjiwarongo, Namibia
4Center for Applied Biotechnology and Molecular Medicine, University of Zurich, Zürich, Switzerland
Email: claudiakaiser-vet@gmx.de
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 24 August 2014; revised 20 September 2014; accepted 25 September 2014
ABSTRACT
Neurological signs like ataxia and hind limb paresis have often been reported in cheetahs (Acinonyx jubatus), lions (Panthera leo) and snow leopards (Panthera unica). As a cause, copper and Vitamin A deficiencies have been discussed. Many cases were seen in cheetahs and lions in the United Arab Emirates (UAE) within the last years. The aim of this study was to find correlations between nutrition, serum, and tissue levels, focusing on copper and Vitamin A. Blood and tissue samples of affected and unaffected animals were analyzed at the Central Veterinary Research Laboratory in Dubai, UAE. Animals were split into three different groups (A, B and C) according to their diets. Minerals were determined in serum, tissue, food and water samples, and serum was additionally analyzed for Vitamin A and E. Liver, kidney and spinal cord samples were taken for histopathological investigations. Mean serum copper and liver copper levels of animals fed pure chicken muscle meat without supplements were significantly lower (0.41 ± 0.71 µM/L; 2.16 ± 0.95 ppm wet weight) than in animals fed a whole carcass prey diet (12.16 ± 3.42 µM/L; 16.01 ± 17.51 ppm wet weight) (p < 0.05). Serum Vitamin A and E levels were highest in animals fed whole carcass prey diets (1.85 ± 0.68; 27.31 ± 5.69 µM/L). Liver zinc concentrations were highest in animals fed pure chicken meat only (43.75 ± 16.48 ppm wet weight). In histopathology, demyelination of the spinal cord was found in all of the affected animals and most commonly when fed a diet based on poultry without supplements.
Keywords:
Ataxia, Cheetah (Acinonyx jubatus), Copper, Lion (Panthera leo), Vitamin A

1. Introduction
Copper (Cu) is an essential trace element and is required for a number of body functions like hemoglobin synthesis, myelination of the nerve sheaths, mineralization of the skeleton, cross linking of elastin and collagen, as well as a constituent of several enzymes [1] -[3] . It also plays a synergistic and antagonistic role in other minerals and vitamins. Vitamin B1, B10, B12, D and minerals like calcium (Ca), cobalt (Co), selenium (Se), sodium (Na) and iron (Fe) enhance Cu retention whereas molybdenum (Mo), cadmium (Cd), mercury (Hg), zinc (Zn), lead (Pb), potassium (K), phosphorous (P), Vitamin A (VA), B3, B5, B6 and C as well as iron (Fe) act antagonistically to Cu. An increased corticoid production, which can arise in stress situations, decreases Cu retention as well [3] . Cu deficiency is assumed to affect the central nervous system causing demyelination of nerves in the spinal cord and therefore ataxia and other neurological signs [4] [5] .
Dietary factors have been found as possible contributors to a number of health issues in cheetahs (Acinonyx jubatus) [6] - [13] and other captive bred felids like lions (Panthera leo) [14] - [18] and snow leopards (Panthera unica) [13] [19] . In cheetahs, ataxia, as well as hind limb paralysis and paresis due to degenerative lesions in the spinal cord, has been reported in zoos, private collections and wildlife parks and is known as “cheetah myelopathy” [13] [20] - [29] . Deficiencies of the trace element Cu [20] [21] [29] [30] and VA [14] [23] have been discussed.
A similar phenomenon has been described in lions where skull malformations have been found as a cause for neurological signs, possibly caused by hypovitaminosis A [14] [15] [31] .
Felids require preformed VA in their diet due to the lack of ability to convert β-carotene into retinol [32] .
In captivity, a meat diet which is not supplemented and does not contain bones, viscera, fur or feathers may lead to these deficiencies. It is essential to offer a carnivorous diet to captive felids which is balanced and includes all necessary nutrients, including vitamins and minerals. Since there is only limited information published about requirements of most non-domestic felid species, the domestic cat is often used as a reference [33] [34] .
In the United Arab Emirates (UAE), many different felids are kept in zoos, wildlife centers and private collections. Some of the most commonly reported clinical signs in these felids are ataxia, swaying gait and hind limb paresis.
This study is a retrospective study, based on pathology findings, tissue and serum results of cheetahs and lions, tested at the Central Veterinary Research Laboratory (CVRL), Dubai, UAE between 2002 and 2013. Some records were incomplete and not every sample of each animal was available for each parameter tested. The study was designed to determine vitamin and trace element levels of captive cheetahs and lions in the UAE which were fed different diets and to find a correlation between neurological signs and the food these animals received, with particular focus on Cu and VA.
2. Material and Methods
2.1. Animals
Animals in this study originated from various private collections within the UAE. The age of these animals varied from 11 month to 12 years. Individual’s history was taken from the medical history form or from pathology records when available.
In total, 76 animals were included in this study consisting of: 61 cheetahs (49 adults and 12 sub-adults) and 15 lions (8 adults and 7 sub-adults). They were divided into three groups, according to their base diet (Table 1 and Table 2).
Thirty-nine blood samples of adult and juvenile felids including 30 cheetahs (Acinonyx jubatus sp.) and 9 lions (Panthera leo) were tested for Cu, Se, Zn, VA and Vitamin E (VE) serum concentrations.
Necropsies were conducted on 43 animals, including 36 cheetahs and 7 lions. Liver samples were analyzed for Cu, Se and Zn. Histological examination was conducted on kidney, liver samples as well as spinal cord samples from the lumbar region.
Table 1. Number of cheetahs (Acinonyx jubatus) by age group (number of affected animals)a and diet composition grouped by base diet.
aAnimals were declared as affected when they showed neurological signs like ataxia and/or swaying gait and/or hind limb paresis. bDepending on age and size of the animal. cMazuri® Carnivore Supplement 58QC for Slab Meat (Dietex International Ltd., P.O. Box 705, Witham, Essex, CM8 3TH, United Kingdom). dCopper-Trition (Equine Products [U.K.] Ltd, 22 Riversdale Court, Newburn Haugh Industrial State, Newcastle upon Tyne, NE15 8SG United Kingdom). eNutrobal (VETARK Professional, P.O. Box 60, Winchester, SO23 9XN, United Kingdom). fPredator supplement (HEALTHtech laboratories (Pty) Ltd, Cnr Douglas and Old Pretoria Rd, Midrand, South Africa, P.O. Box. 12285, Vorna Valley 1686, South Africa).
Table 2. Number of lions (Panthera leo) by age group (number of affected animals)a and diet composition grouped by base diet.
aAnimals were declared as affected when they showed neurological signs like ataxia and/or swaying gait and/or hind limb paresis. bDepending on age and size of the animal. cPredator supplement (HEALTHtech laboratories (Pty) Ltd, Cnr Douglas and Old Pretoria Rd, Midrand, South Africa, P.O. Box. 12,285, Vorna Valley 1686, South Africa).
2.2. Diet
Analysis of the food and supplements the three groups were fed is presented in Table 3.
Group A consisted of cheetahs only and included 17 individuals (15 adults and 2 sub-adults). Adult cheetahs of Group A were fed six days a week (Table 1). Their daily diet consisted of whole quails (1200 - 1500 g, depending on the size and weight of the animal), including viscera and feathers. Birds were cut open and coated with 3.5 g Mazuri® Carnivore Supplement 58QC for Slab Meat (Dietex International Ltd., P.O. Box 705, Witham, Essex, CM8 3TH, United Kingdom) and 1.5 g Copper-Trition (Equine Products [U.K.] Ltd, 22 Riversdale Court, Newburn Haugh Industrial Estate, Newcastle upon Tyne, NE15 8SG, United Kingdom) per day, and 2 - 4 times per month pieces of a whole animal carcass like a rabbit or a gazelle was fed. The sub-adults (up to 1.5 years) in Group A received 1000 - 1200 g of whole quails, sprinkled with 1 g of Nutrobal (VETARK Professional, P.O. Box 60, Winchester, SO23 9XN, United Kingdom) each day.
Group B included 24 animals (18 cheetahs: 13 adults and 5 sub-adults; 6 lions: 2 adults and 4 sub-adults), which were fed six days a week, primarily defrosted chicken with bones but without viscera and feathers (Table 1 and Table 2). Cheetahs were fed a daily amount of 1000 - 1500 g and lions 3000 - 5000 g (depending on the age and size of the animal). Birds were defrosted in a warm water bowl for about 30 minutes. Additionally this
Table 3. Food, supplement and water analysis from captive cheetahs (Acinonyx jubatus) and lions (Panthera leo) in the United Arab Emirates which were divided into three groups according to their base diet.
aCa = calcium, P = phosphorus, Cu = copper, Se = selenium, Zn = zinc, VA = Vitamin A, VE = Vitamin E. bF. E. D. I. A. F. Nutritional Guidelines for complete and complementary pet food for dogs and cats. cna = not analyzedd Mazuri® Carnivore Supplement # 58QC for Slab meat (Dietex International Ltd., P.O. Box 705, Witham, Essex, CM8 3TH, United Kingdom). eCopper-Trition (Equine Products [U.K.] Ltd, 22 Riversdale Court, Newburn Haugh Industrial Estate, Newcastle upon Tyne, NE15 8SG, United Kingdom). fNutrobal (VETARK Professional, P.O. Box 60, Winchester, SO23 9XN, United Kingdom). gPredator supplement (HEALTHtech laboratories (Pty) Ltd, Cnr Douglas and Old Pretoria Rd, Midrand, South Africa, P.O. Box 12285, Vorna Valley 1686, South Africa). hVitamin E from mg/100g, 1.49. i[47] Kölle W. (2007) Wasseranalysen-richtig beurteilt. Wiley- VCH GmbH & Co. KGaA Hannover, Germany.
group received Predator supplement (HEALTHtech laboratories (Pty) Ltd, Cnr Douglas and Old Pretoria Rd, Midrand, South Africa, P.O. Box 12285, Vorna Valley 1686, South Africa), 10 g per 2 kg chicken every other day. Felids of this group were fed pieces of a whole carcass (without viscera and fur) of a gazelle, sheep or a goat 2 - 4 times a month.
Group C consisted of 35 animals (26 cheetahs: 21 adults and 5 sub-adults; 9 lions: 6 adults and 3 sub-adults).
Animals were fed six days a week (Table 1 and Table 2). Three days a week they received chicken with bones, and three days pure chicken muscle meat. Cheetahs got fed 1000 - 1500 g and lions 3000 - 10,000 g (depending on the age and size of the animal). No additional supplements were given.
All animals had access to water ad libitum.
2.3. Analytical Chemistry of Food and Water Samples
One hundred mL of water samples and 200 g of food samples from three facilities were tested for Ca, Cu, P, Se, and Zn concentrations at CVRL (Table 3).
The food and water samples were analyzed by emission spectrometry in a Thermo Scientific iCAP 6000 Series ICP Spectrometer (Thermo Fisher Scientific Inc., 81 Wyman Street, Waltham, Massachusetts, 02454 USA).
2.4. Analytical Chemistry of Serum
Blood samples of 39 individual felids were collected and analyzed for Cu, Se, Zn, VA and VE concentrations (Table 4).
Table 4. Blood vitamin and trace element analysis (mean ± standard deviation [SD] (range); µM/L) [n cheetahs: lions] of captive cheetahs (Acinonyx jubatus) and lions (Panthera leo) in the United Arab Emirates compared to published serum parameters for cheetahs; liver and kidney analysis (mean ± standard deviation [SD] (range); ppm wet weight) [n] of captive felids in the United Arab Emirates and published reference values for domestic cats. Conversion factors were used to change reference values into µM/L.
a,bBechert et al., 2002, supplemented meata and commercial diet. b(Retinol, from µg/mL, 3.49; α-tocopherol, from µg/mL, 2.32; copper, from µg/mL, 15.7; selenium, from ng/mL, 0.0127; zinc, from µg/mL, 15.3; serum). cDierenfeld, 1993 (copper, from mEq/L, 502; zinc, from mEq/L, 505;serum). dBeckmann et al., 2013. ePuls, R. 1994. Mineral Levels in Animal Health, Diagnostic Data, 2nd Edition, Sherpa International, Clearbrook, BC, Canada.
For serum analysis, 5 - 6 mL of blood were collected either during an annual routine health check or before euthanasia by venopuncture of the V. jugularis or V. cephalica, using a 19-gauge needle and a 10-mL disposable syringe. The blood was collected into serum vacutainers (BD Vacutainer® Blood Collection tubes, BD Vacutainer Systems, Plymouth, Devon, PL6 7BP, United Kingdom) and transported in cooled containers to CVRL for analysis.
At CVRL the sera were diluted 10-times with de-ionized water. VA (retinol) and VE (α-tocopherol) concentrations were determined in serum, using an Alliance Waters 2695 Separations Module High-performance liquid chromatography (HPLC), and a Waters 2487 Dual Absorbance Detector (Waters Corporation, 34 Maple Street, Milford, Massachusetts, 01757, USA).
Serum Cu and Zn concentrations were determined using a Thermo Elemental iCE 3000 Series Atomic Absorption Spectrometer (AAS) (Thermo Fisher Scientific Inc., 101 Constitution Blvd, Franklin, Massachusetts, 02038, USA). For Cu, a wavelength of 324.8 nm was set and 213.9 nm for Zn. Three standards were performed for both elements for plotting a standard curve. For deuterium background correction a STAT (slotted tube atom trap) was used to increase the sensitivity of the analysis.
Se concentrations in serum were measured using a Graphite Furnace Atomic Absorption Spectrometer (Fisher Scientific UK Holding Company Limited, Solaar House, 19 Mercers Row, Cambridge, CB5 8BZ, United Kingdom). The serum was diluted 10-times with 0.05% Triton. The element was measured at a wavelength of 196.0 nm and deuterium was taken for background correction. Three standards were performed for plotting a standard curve. A matrix modifier solution containing 2% hydroxyl ammonium hydrochloride and 500 mg/L palladium was used to reduce the interferences.
2.5. Pathology and Histopathology
Necropsies were performed on 43 carcasses immediately after arriving at CVRL (Table 5). Fresh tissue samples were taken from kidney, liver and spinal cord of the lumbar region. Tissue samples were fixed in a 10% formalin solution. After using routine methods for the sample preparation, the samples were then stained with Hematoxylin and Eosin (H & E) and histological examined.
2.6. Analytical Chemistry of Liver Tissue Samples
Forty-three liver samples of 43 dead felids were processed and analyzed at CVRL after necropsy (Table 4). For the analysis of Cu, Se and Zn concentrations in liver tissue samples were digested with 5 mL 65% nitric acid and 1 mL 31% H2O2 in Ethos Milestone Microwave Labstation (200˚C, 800 W for 10 minutes) (Milestone Inc., 25 Controls Drive, Shelton, Connecticut, 06484, USA). After the digestion the samples were made up to 10 mL or 100 mL with deionised water, depending on the concentration of minerals present. After sample preparation solutions were measured with the same method as performed with serum samples.
2.7. Statistics
For each blood and tissue parameter the mean concentrations (µM/L for serum and ppm wet weight for tissue samples), standard deviation (SD) and range were determined. All data are expressed as mean ± SD. The three groups (A, B and C) were compared to each other for serum Cu, Se, VA, VE and Zn as well as for liver Cu and Zn concentrations by one-way ANOVA using Tukey Post hoc tests in R (R Core development team [2013]: a language and environment for statistical computing and graphics. www.r-project.org). Values of p < 0.05 were considered as statistically significant.
3. Results
3.1. Clinical Signs
Out of the 76 individuals tested, 30.3% (23 animals) were affected with clinical neurological signs including ataxia, lack of coordination, swaying gait and hind-limb weakness from moderate to severe (Table 5). These animals all showed normal appetite. In the final stages they were unable to stand, developed hind limb paresis and finally died or were euthanized due to poor prognosis.
None of the animals in Group A were affected with neurological signs whereas 4.0% (3 animals) in Group B were affected. In Group C 26.3% (20 animals) of the animals were affected including three animals (one sub- adult cheetah, one sub-adult lion and one adult lion) which also showed signs of ataxia but were still alive until the date tested (Table 5).
3.2. Diet
Table 3 shows the results of the five parameters tested (Ca, P, Cu, Se, and Zn) in five different meat products and of the four different dietary supplements (including VA and VE levels of the supplements, received from the manufacturers) feed to Group A and B. Reference values for domestic cats are also presented for comparison [33] .
Table 5. Gross pathology and histopathology results of cheetahs (Acinonyx jubatus): lions (Panthera leo) in the United Arab Emirates which were split into three different groups (A, B and C)a.
aGroups were categorized by their base diet. bAnimals were declared as affected when they showed neurological signs like ataxia and/or swaying gait and/or hind limb paresis. cIn total 23 animals showed neurological signs. Out of these, three (1:2) were still alive. Hence only blood was analyzed. dnf = nothing found.
Meat on bone (beef and chicken) including quails, had a Ca:P level of 1:1 whereas pure muscle meat had a Ca:P ratio of 1:55 (beef) and 1:28 (chicken). All five meat products were low in Cu, Se and Zn. Quail, beef muscle meat and beef on bone possessed the highest Cu values (0.0003 g/100g dry matter; 0.0002 g/100g dry matter respectively). When compared with the supplements, most of the elements were above the reference values for cats, especially Ca, Cu, and Zn [33] . However, Predator supplement (HEALTHtech laboratories (Pty) Ltd, 79 Landmarks Ave., Centurion 0157, South Africa) had a very low Cu content (0.0001 g/100g dry matter) and Copper-Trition (Equine Products [U.K.] Ltd, 22 Riversdale Court, Newcastle upon Tyne, NE I5 8SG, United Kingdom) a very low Ca content (0.049 g/100g dry matter).
Tab water samples from three different private collection breeding places showed similar ranges (Table 3). Ranges fell within the WHO values for standard water [35] .
3.3. Analytical Analysis of Serum
Reference values for serum and hepatic VA, VE and trace elements are limited for lions and other big felids, hence the results were compared to recently published data from cheetahs [8] [36] [37] . The values are presented in Table 4 and Figure 1.
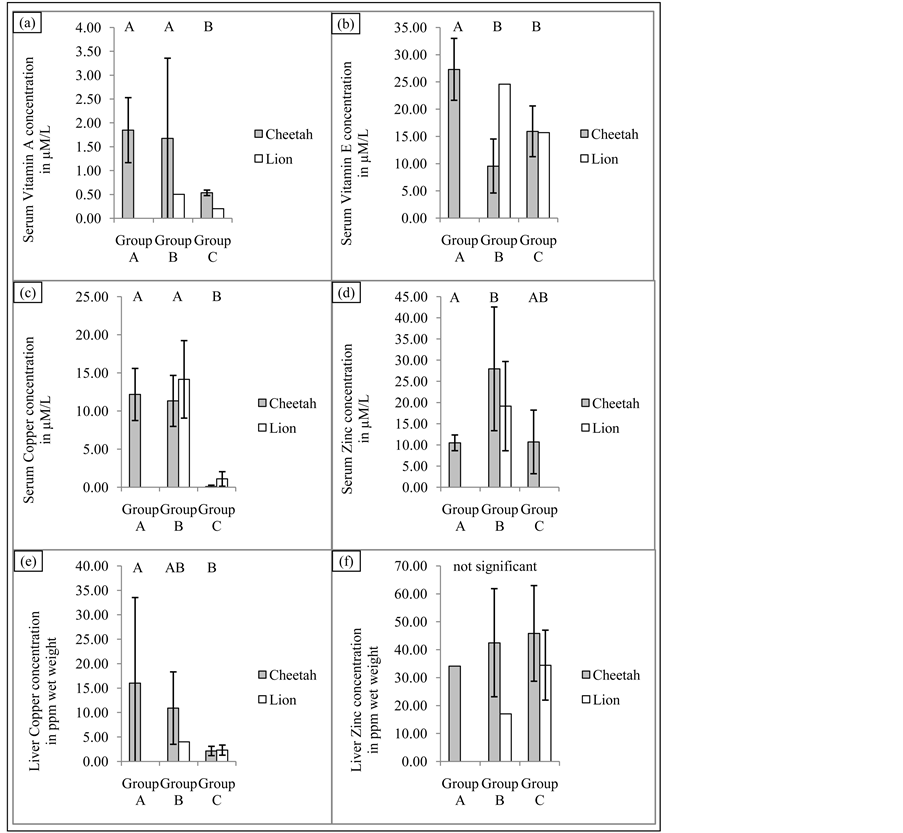
Figure 1. Comparison of serum vitamin and mineral results (mean ± SD; µM/L) as well as liver values (mean ± SD; ppm wet weight) of captured cheetahs (Acinonyx jubatus) and lions (Panthera leo) in the United Arab Emirates. Different letters ((A), (B)) denote significant differences.
Overall there were significant differences between the three groups for VA (F2,20 = 3.84, p = 0.039), VE (F2,20 = 14.06, p < 0.001), Cu (F2,28 = 45.29, p < 0.001) and Zn (F2,18= 3.97, p = 0.037). Group A had the highest VA (1.85 ± 0.68 µM/L) and VE (27.31 ± 5.69 µM/L) concentrations whereas Group C had the lowest VA (0.45 ± 0.17 µM/L) and Cu (0.41 ± 0.71 µM/L) levels of all groups. There was no significant difference for serum Se between the three groups (F2,21 = 2.909, p = 0.077). Group A was significantly higher than Group C for serum VA, VE (27.31 ± 5.69 µM/L; 15.88 ± 4.04 µM/L) and Cu (12.16 ± 3.42 µM/L; 0.41 ± 0.71 µM/L) values but not for serum Zn (10.48 ± 1.86 µM/L; 10.7 ± 7.51 µM/L). There were no significant differences between Group A and B for serum VA and Cu. Group A had significantly higher serum VE levels than Group B (27.31 ± 5.69 µM/L; 12.56 ± 7.98 µM/L) whereas Group A had significantly lower levels for serum Zn than Group B (10.48 ± 1.86 µM/L; 18.84 ± 6.26 µM/L). Group B was significantly higher than Group C for serum Cu (12.74 ± 4.26 µM/L; 0.41 ± 0.71 µM/L) but not for serum VA, VE and Zn (Figure 1).
3.4. Pathology and Histopathology
The results of the gross pathology and histopathology of necropsies are presented in Table 5.
Forty-three percent (n = 10) of the affected animals (n = 23) showed slight hind limb muscle atrophy.
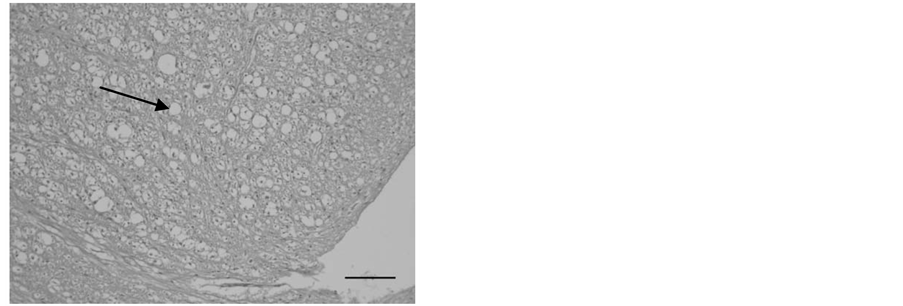
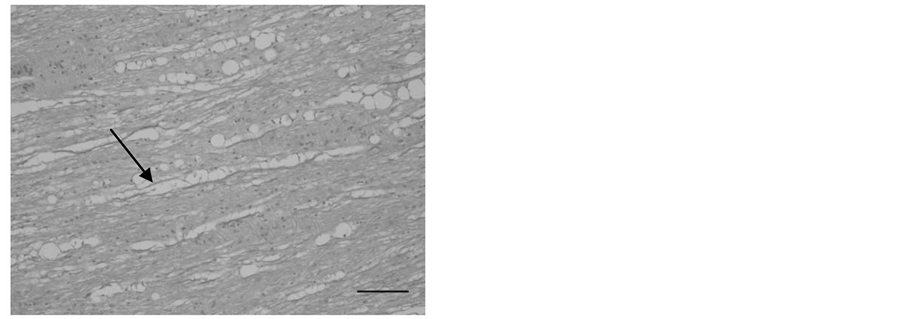
In histopathology, significant pathological changes were found in the central nervous system. Demyelination of the spinal cord, characterized by bilateral dilated myelin sheath of the white matter and vacuolization of the tissue was seen in all 20 felids that showed neurologic signs before death. Diffuse vacuolated myelin sheaths were seen in the spinal cord (Figure 2 and Figure 3).
Skull malformation was seen in one lion of Group C. No amyloid was found in the liver or the kidney and no inflammation was seen in any of the cases.
Figure 2. Transverse section of the lumbar region of the spinal cord from a cheetah (Acinonyx jubatus) showing severe vacuolation due to demyelination (H & E, bar = 100 µm).
Figure 3. Longitudinal section of the lumbar part of the spinal cord from a cheetah (Acinonyx jubatus) with marked myelin loss (H & E, bar = 100 µm).
3.5. Analysis of Liver Tissue Samples
The results of liver tissue sample analysis are shown in Table 4.
Overall there were significant differences between the three groups for liver Cu (F2,41 = 14.40, p < 0.001) but not for liver Zn (F2,19 = 0.204, p = 0.817) concentrations. Group A was significantly higher for liver Cu concentrations than Group C (16.01 ± 17.51 ppm wet weight; 2.16 ± 0.95 ppm wet weight) but not significantly higher than Group B (9.41 ± 6.41 ppm wet weight).
4. Discussion
Neurological disorders like ataxia, paresis and hind limb weakness have been described numerous times in cheetahs. Some authors associated their findings with Cu deficiency [21] [29] [30] , while others with viral infections in combination with high stress levels [27] [28] , where animals started showing ataxia and respiratory signs like nasal discharge and sneezing after stressful events like hand capture, transport or relocation, but no virus was found. However, an increased corticoid production in stress situations can occur which may decrease Cu retention [3] and could trigger Cu deficiency.
Cheetahs with ataxia and hind limb paresis in the above mentioned studies, showed atrophy of the hind limb muscles and severe demyelination with vacuolation of the spinal cord, which validates our findings [12] [21] [26] [29] [38] . The neurological disease in cheetahs, characterized by degenerative lesions of the spinal cord, causing ataxia and paresis is called “cheetah encephalomyelopathy” [39] . This disease is responsible for 25% of all deaths in the European Endangered Species Programme (EEP) cheetah population [39] however the exact etiology is still uncertain [13] .
Drug therapy had no real influence on the progress of this disease in previous cases [25] [26] except in one animal where progression of ataxia was inhibited by an anti-herpes drug [27] . Copper sulfate supplementation had no impact on the improvement of the clinical signs in cheetah cubs with ataxic signs at Nuremberg Zoo, Germany [27] but in another case in the USA, where cubs received oral and injectable Cu supplements, signs of ataxia improved within four weeks [20] .
Liver Cu values, which are only available through liver biopsy, are a better indicator for the Cu status than serum values and are the most responsive to changes in dietary Cu intake [40] . Serum Cu levels may remain within normal ranges whereas liver levels may fluctuate according to the needs of the animals. Therefore, liver values provide the best analysis to detect Cu deficiency [41] , though, with continued deficient Cu intake, serum Cu concentration will decline over time, which was shown to occur in animals of Group C. Since this study is a retrospective study, liver vitamin levels were unfortunately not available.
This study shows that felids which were only fed pure poultry diet developed ataxia and hind limb paresis more often than animals that were fed an appropriately balanced diet with regular supplements. Since muscle meat does not contain a high amount of trace elements and vitamins, [15] not only Cu but other minerals like Ca and P should be monitored closely. The Ca: P ratio, which is ideally between 1:1 - 2:1 [34] , is too low if only pure muscle meat is fed (Table 3) and can result in metabolic bone disease [42] as well as Cu deficiency since high P intake depletes Cu retention [3] . As cheetahs are not able to consume large bones [43] , Ca needs to be supplemented when only pure muscle meat is fed.
A primary cause of Cu deficiency, which can be assumed for animals of Group C, can occur when not enough Cu is fed [44] . A secondary cause can be due to an excessive intake of antagonistic acting mineral such as Zn [1] [2] not only when fed in large amounts but also when animals do extensive licking at cages and therefore absorb a lot of Zn [44] . This behavior has been seen in some of the animals in this study.
Chicken muscle meat does not only contain an inappropriate amount of Cu for a balanced diet for captive felids, but it also has a high level of Zn (Table 3) and therefore, a diet based on chicken muscle meat without supplements may be a cause for the etiology of ataxia [19] [20] . Although serum Cu levels are not the best indicator for Cu deficiency, they should be analyzed when common signs like ataxia or paresis are visible. An early start on Cu supplements is recommended because felids will more likely respond to therapy [30] , therefore prognosis seems better for a reversal in signs of ataxia.
One unaffected cheetah was analyzed twice for serum values four weeks apart. This cat used to receive pure chicken muscle meat without any supplements before it was put on a more appropriate diet. The new diet included beef on bone with Predator supplement (HEALTHtech laboratories (Pty) Ltd, Cnr Douglas and Old Pretoria Rd, Midrand, South Africa, P.O. Box 12,285, Vorna Valley 1686, South Africa). The serum Cu level was 8.00 µM/L before the diet was changed. After feeding beef on bone with supplements for four weeks, serum Cu concentration reached 17.80 µM/L. The same was seen with serum Se levels with 2.80 µM/L before and 4.79 µM/L after the diet change. VE levels were 5.40 µM/L and 16.20 µM/L respectively. There was not a big change in Zn (14.7 µM/L before and 13.6 µM/L after the diet change) and VA levels (0.20 µM/L before and 0.30 µM/L respectively). It is assumed that VA serum levels may react slower to diet changes than Cu serum levels. If the animal has a VA deficiency, serum VA levels may still be adequate for an uncertain amount of time [31] .
VA deficiency has also been associated with cases of ataxia in cheetahs [12] [13] and lions [14] - [18] [45] , particularly when fed a lean red meat diet. Hypovitaminosis A has especially been reported in lions under one year of age. The rapid growth in that age seems a causal factor for that deficiency [15] . Skull malformations have been found in affected lions and was also seen in one of the tested lions in this study which showed neurological disorders before death [14] [15] [17] [31] .
A balanced nutrition for these obligate carnivores is important because hyper VA intake has shown to antagonize ingestion of VE and Cu [3] . Some supplements contain a high level of VA, therefore the level of VE and Cu needs to be monitored and it needs to be considered that VA can be toxic when fed >10,000 IU/kg in a diet [34] . Since the liver is the major storage organ for VA, liver VA concentrations are more significant than serum values. Therefore, a liver biopsy would be recommended as a diagnostic tool. Since this study is a retrospective study, liver VA values were unfortunately not available. Only an extreme depletion or overconsumption of VA is reflected in serum VA concentrations [31] .
It is very important that animals, especially cubs and infants, get a balanced nutrition with an adequate level of VA to avoid deficiencies. Blood mineral and vitamin concentrations may vary according to gender, age, environmental factors and nutritional intake (interactions with other minerals and vitamins) [8] .
In the wild, carnivores consume a wide variety of whole vertebrate prey (e.g. antelopes, birds) including skin, fur/feathers, viscera and bones. These prey animals eat a different diet in the wild than captive animals that are then fed to captive carnivores. For instance, a wild bird eats a more insectivorous diet in the wild than what a captive chicken is fed [46] . Hence a wild predator like a cheetah or a lion gets a more balanced diet in the wild than in captivity. Therefore, feeding whole carcass diets is more natural and enhances the animal’s physical and psychological well-being.
5. Conclusions
In summary, the results in this study show that multiple nutritional deficiencies appear to be the cause for neurological disorders in these species. Not all animals in this study were tested for all analysis conducted in this paper due to the opportunistic collection of the samples.
Further investigations and correlations to minerals and vitamins influencing Cu and VA absorption need to be conducted. Because this study is a retrospective one, not all Cu and VA influencing factors are analyzed.
This paper provides an overview of the correlation between mineral and vitamin serum and tissue levels and further investigations are important and to be conducted.
Acknowledgements
The authors would like to thank H.H. Sheikh Mohammed bin Rashid Al Maktoum for his support at the Central Veterinary Research Laboratory, the staff of CVRL for their help with the sample-preparation and analysis and for their help to create this study. They also thank Alan Stephenson, Christina Hebel, Corina Berner-Schultheiss, Birger Kruse, Claudia Szentiks, Eli Walker, Kathrine Forsythe and Fabiano Ezekiel from the Cheetah Conservation Fund for their help during the preparation of this publication, and are thankful to the reviewers for their helpful comments on the development for this manuscript.
References
- Jubb, K.V.F. and Huxtable, C.R. (1993) The Nervous System. In: Jubb, K.V.F., Kennedy, P.C. and Palmer, N., Eds., Pathology of Domestic Animals, 5th Edition, Vol. 1, Academic Press, New York, 360-362. http://dx.doi.org/10.1016/B978-0-08-057133-1.50011-4
- Turnlund, J.R. (1994) Copper. In: Shils, M.E., Olson, J.A. and Shike, M., Eds., Modern Nutrition in Health and Disease, Vol. 8, Lea and Febiger, Philadelphia, 231-241.
- Watts, D.L. (1989) The Nutritional Relationships of Copper. Journal of Orthomolecular Medicine, 4, 99-108.
- Goodman, B.P., Chong, B.W., Patel, A.C., Fletcher, G.P. and Smith, B.E. (2006) Copper Deficiency Myeloneuropathy Resembling B12 Deficiency: Partial Resolution of MR Imaging Findings with Copper Supplementation. American Journal of Neuroradiology, 27, 2112-2114.
- Prodan, C.I., Holland, N.R., Wisdom, P.J., Burstein, S.A. and Bottomley, S.S. (2002) CNS Demyelination Associated with Copper Deficiency and Hyperzincemia. Neurology, 59, 1453-1456. http://dx.doi.org/10.1212/01.WNL.0000032497.30439.F6
- Allan, G., Portas, T., Bryant, B., Howlett, R. and Blyde, D. (2008) Ulnar Metaphyseal Osteochondrosis in Seven Captive Bred Cheetahs (Acinonyx jubatus). Veterinary Radiology Ultrasound, 49, 551-556. http://dx.doi.org/10.1111/j.1740-8261.2008.00430.x
- Allen, M.E. and Ullrey, D.E. (2004) Relationships among Nutrition and Reproduction and Relevance for Wild Animals. Zoo Biology, 23, 475-487. http://dx.doi.org/10.1002/zoo.20029
- Bechert, U., Mortenson, J., Dierenfeld, E.S., Cheeke, P., Keller, M., Holick, M., Chen, T.C. and Rogers, Q. (2002) Diet Composition and Blood Values of Captive Cheetahs (Acinonyx jubatus) Fed Either Supplemented Meat or Commercial Food Preparations. Journal of Zoo and Wildlife Medicine, 33, 16-28.
- Bolton, L.A. and Munson, L. (1999) Glomerulosclerosis in Captive Cheetahs (Acinonyx jubatus). Veterinary Pathology, 36, 14-22. http://dx.doi.org/10.1354/vp.36-1-14
- Depauw, S., Hesta, M., Whitehouse-Tedd, K., Stagegaard, J., Buyse, J. and Janssens, G.P.J. (2012) Blood Values of Adult Captive Cheetahs (Acinonyx jubatus) Fed Either Supplemented Beef or Whole Rabbit Carcasses. Zoo Biology, 31, 629-641. http://dx.doi.org/10.1002/zoo.20427
- Gosselin, S.J., Setchell, K.D.R., Harrington, G.W., Welsh, M.B.B., Pylypiw, H., Kozeniauskas, R., Dollard, D., Tarr, M.J. and Dresser, B.L. (1989) Nutritional Considerations in the Pathogenesis of Hepatic Veno-Occlusive Disease in Captive Cheetahs. Zoo Biology, 8, 339-347. http://dx.doi.org/10.1002/zoo.1430080404
- Palmer, A.C. and Cavanagh, J.B. (1995) Encephalo Myelopathy in Young Cats. Journal of Small Animal Practice, 36, 57-64. http://dx.doi.org/10.1111/j.1748-5827.1995.tb02822.x
- Robert, N. (2008) Neurologic Disorders in Cheetahs and Snow Leopards. In: Fowler, M.E. and Miller, R.E., Eds., Zoo and Wild Animal Medicine: Current Therapy 6. Saunders Elsevier, St. Louis, 265-271. http://dx.doi.org/10.1016/B978-141604047-7.50035-X
- De Risio, L., Beltran, E., Matiasek, K., de Stefani, A. and Holloway, A. (2010) Neurological Dysfunction and Caudal Fossa Overcrowding in a Young Cheetah with Hypovitaminosis A. Veterinary Record, 167, 534-536. http://dx.doi.org/10.1136/vr.c4802
- Hartley, M.P., Kirberger, R.M., Haagenson, M. and Sweers, L. (2005) Diagnosis of Suspected Hypovitaminosis A Using Magnetic Resonance Imaging in African Lions (Panthera leo). Journal of the South African Veterinary Association, 76, 132-137. http://dx.doi.org/10.4102/jsava.v76i3.414
- Maratea, K.A., Hooser, S.B. and Ramos-Vara, J.A. (2006) Degenerative Myelopathy and Vitamin A Deficiency in a Young Black-Maned Lion (Panthera leo). Journal of Veterinary Diagnostic Investigation, 18, 608-611. http://dx.doi.org/10.1177/104063870601800617
- O’Sullivan, B.M., Mayo, F.D. and Hartley, W.J. (1977) Neurologic Lesions in Young Captive Lions Associated with Vitamin A Deficiency. Australian Veterinary Journal, 53, 187-189. http://dx.doi.org/10.1111/j.1751-0813.1977.tb00168.x
- Wenker, C.J. and Robert, N. (2012) Stargazing in Lions. In: Fowler, M.E. and Miller, R.E., Eds., Zoo and Wild Animal Medicine, 7th Edition, W.B. Saunders Co., Philadelphia, 470-476.
- Stidworthy, M.F., Lewis, J.C.M., Penderis, J. and Palmer, A.C. (2008) Progressive Encephalo Myelopathy and Cerebellar Degeneration in a Captive-Bred Snow Leopard (Uncia uncia). Veterinary Record, 162, 522-524. http://dx.doi.org/10.1136/vr.162.16.522
- Eldridge, R. (1997) LIOC Endangered Species Conservation Federation: Maintaining Mineral Balance. Newsletter, 41, 8-10.
- Hafner, A., Schmidt, P., Hänichen, T. and Schmahl, W. (1996) Degenerative Myelopathie bei Geparden (Acinonyx jubatus) [Degenerative Myelopathy in Cheetahs (Acinonyx jubatus)]. Berliner und Münchener tierärztliche Wochenschrift, 109, 403.
- Kotsch, V., Walzer, C. and Kübber-Heiss, A. (2001) Evaluation of a Questionnaire Concerning the Cheetah Cub Ataxia within the EEP. Proceedings of 40th International Symposium on Diseases of Zoo- and Wild Animals, Rotterdam, 23-26 May 2001, 333-334.
- Palmer, A.C. and Franklin, R.J.M. (1999) Review of Neuropathological Material from Ataxic Cheetah Cubs at Fota Wildlife Park. Workshop on Ataxia in Cheetah Cubs, Dublin, 13-15.
- Palmer, A.C., Callanan, J.J., Guerin, L.A., Sheahan, B.J., Stronach, N. and Franklin, R.J. (2001) Progressive Encephalomyelopathy and Cerebellar Degeneration in 10 Captive-Bred Cheetahs. Veterinary Record, 149, 49-54. http://dx.doi.org/10.1136/vr.149.2.49
- Robert, N., Walzer, C., Petit, T. and Vitaud, C. (2006) Neurological Diseases in Cheetah. Proceedings of EAZWV 6th Scientific Meeting, Budapest, 24-28 May 2006.
- Walzer, C. and Kübber-Heiss, A. (1995) Progressive Hind Limb Paralysis in Adult Cheetahs (Acinonyx jubatus). Journal of Zoo and Wildlife Medicine, 26, 430-435.
- Walzer, C., Kuebber-Heiss, A., Gelbmann, W., Suchy, A., Bauder, B. and Weissenboeck, H. (1998) Acute Hind Limb Paresis in Cheetah (Acinonyx jubatus) Cubs. Proceedings of the 2nd Scientific Meeting of the European Association of Zoo- and Wildlife Veterinarians (EAZWV), Chester, 21-24 May 1998, 267-274.
- Walzer, C., Url, A., Robert, N., Kübber-Heiss, A., Nowotny, N. and Schmidt, P. (2003) Idiopathic Acute Onset Myelopathy in Cheetah (Acinonyx jubatus) Cubs. Journal of Zoo and Wildlife Medicine, 34, 36-46.
- Zwart, P., von der Hage, M., Schotman, A.J.H., Dorrestein, G.M. and Rens, J. (1985) Copper Deficiency in Cheetah (Acinonyx jubatus). Verhandlungsbericht des 27. Internationalen Symposiums über die Erkrankungen der Zootiere, St Vincent/Torino, 9-13 June 1985, 253-257.
- Brand, D.J. (1980) Captive Propagation at the National Zoological Gardens of South Africa, Pretoria. International Zoo Yearbook, 20, 107-112.
- Shamir, M.H., Rubin, G., Aizenberg, Z., Berkovich, Z., Reifen, R., Horowitz, I., Bdolah-Abram, T. and Aroch, I. (2012) Needle Biopsy for Hepatic Vitamin A Levels in Lions (Panthera leo). Journal of Zoo and Wildlife Medicine, 43, 207- 213. http://dx.doi.org/10.1638/2010-0038.1
- Schweigert, F.J., Raila, J., Wichert, B. and Kienzle, E. (2002) Cats Absorb ß-Carotene, but It Is Not Converted to Vitamin A. Journal of Nutrition, 132, 1620-1612.
- FEDIAF (2011) Nutritional Guidelines for Complete and Complementary Pet Food for Cats and Dogs. European Pet Food Industry Federation, 17.
- National Research Council of the National Academies (2006) Nutrient Requirements of Dogs and Cats. The National Academic Press, Washington DC.
- Kölle, W. (2007) Wasseranalysen-Richtig Beurteilt [Evaluation of Water Analysis]. Wiley-VCH GmbH & Co. KGaA, Hannover.
- Beckmann, K.M., O’Donovan, D., McKeown, S., Wernery, U., Basu, P. and Bailey, T.A. (2013) Blood Vitamins and Trace Elements in Northern-East African Cheetahs (Acinonyx jubatus soemmeringii) in Captivity in the Middle East. Journal of Zoo and Wildlife Medicine, 44, 613-626. http://dx.doi.org/10.1638/2011-0156R2.1
- Dierenfeld, E.S. (1993) Nutrition of Captive Cheetahs (Acinonyx jubatus): Food Composition and Blood Parameters. Zoo Biology, 12, 143-150. http://dx.doi.org/10.1002/zoo.1430120113
- Shibly, S., Schmidt, P., Robert, N., Walzer, C. and Url, A. (2006) Immunohistochemical Screening for Viral Agents in Cheetah (Acinonyx jubatus) Myelopathy. Veterinary Record, 159, 557-561. http://dx.doi.org/10.1136/vr.159.17.557
- Robert, N. and Walzer, C. (2009) Pathological Disorders in Captive Cheetahs. In: Vargas, A., Breitenmoser-Würsten, C. and Breitenmoser, U., Eds., Iberian Lynx ex Situ Conservation: An Interdisciplinary Approach, Fundación Biodiversidad, Madrid, 265-272.
- Doong, G., Keen, C.L., Rogers, Q., Morris, J. and Rucker, R.B. (1983) Selected Features of Copper Metabolism in the Cat. Journal of Nutrition, 113, 1963-1971.
- Fascetti, A.J., Rogers, Q.R. and Morris, J.G. (2000) Dietary Copper Influences Reproduction in Cats. Journal of Nutrition, 130, 1287-1290.
- Fowler, M.E. (1978) Metabolic Bone Disease. In: Fowler, M.E., Ed., Zoo and Wild Animal Medicine, Saunders, Philadelphia, 53-76.
- Phillips, J.A. (1993) Bone Consumptions by Cheetahs at Undisturbed Kills: Evidence of Lack of Focal-Palatine Erosion. Journal of Mammalogy, 74, 487-492. http://dx.doi.org/10.2307/1382408
- Hendriks, W.H., Allan, F.J., Tarttelin, M.F., Collett, M.G. and Jones, B.R. (2001) Suspected Zinc-Induced Copper Deficiency in Growing Kittens Exposed to Galvanized Iron. New Zealand Veterinary Journal, 49, 68-72. http://dx.doi.org/10.1080/00480169.2001.36205
- Cassidy, J.P., Caulfield, C., Jones, B.R., Worrall, S., Conlon, L., Palmer, A.C. and Kelly, J. (2007) Leukoencephalomyelopathy in Specific Pathogen-Free Cats. Veterinary Pathology, 44, 912-916. http://dx.doi.org/10.1354/vp.44-6-912
- Bell, K.M. (2010) Nutrient Requirements, Utilization and Metabolism. In: Tucker, L.A. and Thomas, D.G., Eds., Spot the Difference: Are Cheetahs Really Just Big Cats? Nottingham University Press, Nottingham, 67-91.
- Puls, R. (1994) Mineral Levels in Animal Health. Diagnostic Data. 2nd Edition, Sherpa International, Clearbrook, 82.