American Journal of Plant Sciences
Vol.5 No.8(2014), Article ID:44203,9 pages DOI:10.4236/ajps.2014.58120
Bioremediation of Diuron in Soil Environment: Influence of Cyanobacterial Mat
Jamal Safi1,2, Yousef Awad2, Yasser El-Nahhal1,3
1Environmental Protection and Research Institute, Gaza, Palestine
2Al-Azhar University of Gaza, Gaza, Palestine
3The Islamic University of Gaza, Gaza, Palestine
Email: y_el_nahhal@hotmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 27 January 2014; revised 28 February 2014; accepted 10 March 2014
ABSTRACT
Diuron is a herbicide widely used in the Middle East; its field application creates many environmental problems. This study investigated the bioremediation of Diuron in soil environment by Cyanobacterial mats collected from Gaza Wadi. Various concentrations of Diuron were injected in water saturated soil samples pre-treated with Cyanobacterial mat for several periods. Percent growth of Jews mallow as a test plant was taken as indicator of biodegradation of Diuron. Results showed that Diuron was degraded in soil and degradation was more pronounced when Diuron was incubated with Cyanobacterial in the irrigation water. Larger applied rate of Cyanobacterial mat did not affect the biodegradation of Diuron. These encouraging results suggest that application of Cyanobacterial mat could be a suitable method to remediate soil pollution.
Keywords:Cyanobacterial Mat; Diuron; Soil

1. Introduction
Diuron (N-(3,4-dichlorophenyl)-N,N-dimethyl urea) is one of the phenyl urea herbicide derivatives. It is applied for weed control either pre or post-emergence treatment of cotton, fruit, cereal and other agricultural crops [1] . It was also used in urban and industrial areas. It is also used as an active ingredient in antifouling boat paints and in algaecide formulations used in fountains and aquaculture [2] . The frequent application of Diuron has created environmental problems to soil and ground water systems due to their residues and possible leaching and/or runoff to the ground or surface water. For instance it has been detected in ground water [3] and surface water [4] in California and in Canada [5] . Recent report [6] demonstrated that Diuron is very mobile in sandy soil and has the potential for ground water contamination. Furthermore, the remaining concentration of Diuron in the top soil can cause damage to the crops in the next crop cycle. Diuron has relatively low soil adsorption coefficient, while its hydrolysis and aqueous photolysis half-lives are relatively long. Its average field dissipation half-life is 90 days [1] . Consequently Diuron is both mobile and relatively persistent in soil [7] .
Enhanced biodegradation of diuron in agricultural soils was previously studied [8] -[11] . Several Diuron-degrading bacteria have been isolated from different agricultural soils [12] -[14] and river waters [15] .
In addition Diuron residues in water samples can be harmful to animal and human being. These residues must be removed and/or remediate from soil and/or water systems to have clean and environmentally sound systems. Our approach for studying biodegradation of Diuron in soil based on the idea that Cyanobacterial mats were able to degrade organic pollution from water. Cyanobacterial mats are composed of physiologically different groups of microorganisms such as cyanobacteria and diatoms as oxygenic phototrophs, chemolithottrophic colorless sulfur bacteria, anaerobic phototrophic purple sulfur bacteria, anaerobic sulfate-reducing bacteria as well as aerobic heterotrophs. Cyanobacteria constitute a group of prokaryotic microorganisms able to carry out oxygen photosynthesis [16] . The idea behind using bioassay technique to evaluate biodegradation of herbicide in soil based on the fact that bioassay technique is more sensitive tool than chemical technique as previously reported [17] . The bioassay technique allows us to determine a very low remaining concentration of herbicide in soil [18] [19] . The objectives of this research are to: Evaluate the biodegradation behavior of Diuron in soil and liquid media by Cyanobacterial mats and to provide answers to the following research questions: 1) Dose the application rate of Diuron affects the degradation process? 2) Dose the number of Cyanobacterial mat cells affects the degradation process? 3) Dose the degradation process is time controlled?
2. Materials and Methods
Analytical grade Diuron was purchased from Fluka (Germany). It is a hydrophobic herbicide with an octanol/ water partitioning coefficient (Kow = 2.85) and weakly soluble in water (36.4 mg/L) 25˚C [20] .
2.1. Soil Collection and Analysis
Soil samples were collected from top 0 - 30 cm of agricultural land located in Al Zawaida village in the Middle Governorate of Gaza Strip, which has no history of Diuron application in the past 10 years. Soil samples were analyzed to determine the physicochemical properties.
2.2. Collection of Cyanobacterial Mat Samples
Following the procedure recently published [21] the samples of Cyanobacterial mats were collected on 15.04— 2012 at 9.00 am from three locations in Wadi Gaza have the following GPS data: Location 1: 31˚27′46.74″N, 34˚22′38.95″E; Location 2: 31˚27′47.81″N, 34˚22′46.02″E and Location 3: 31˚27′53.12″N, 34˚22′57.74″E.
The collected Cyanobacterial mats were transferred to a 20 liter plastic pot. The samples are physically filtered through 2 mm mesh sieved and then diluted by tab water free from chlorine or any disinfecting agents. The dilution was 1:1 ratio. Due to long period of laboratory experiments, the collection of Cyanobacterial mats was repeated several times as required. This dilution assumed to produce 106 cells/ml.
2.3. Soil Sterilization
About 6 kg of soil samples were sieved using 2 mm mesh size and transferred to polyethylene packets, tolerant to high temperature, and saturated with water for 48 hours. The soil samples were transferred to the autoclave (SS-325-Automatic High-Pressure-Steam Sterilizer Class I-B) and kept inside the autoclave at 120˚C for 1.5 hours. Then the soil samples were left for cooling. The cooled soil was transferred to black plastic pots having 4 holes in the bottom, for the execution of the experiment.
2.4. Diuron Solutions
Technical amounts of Diuron (31.3 mg/L) was dissolved in 1 L distilled water and used as a stock solution throughout the experiments and used to prepare the standard solutions. Further dilutions were made to obtain the needed concentration as planned in the experiments. The following concentrations of Diuron were prepared and tested 0.055, 0.11, 0.22, 0.44, and 0.88 mg/kg soil.
2.5. Bioassay Technique
The herbicidal activity of Diuron against the test plants was determined following the procedure described previously [22] . In this procedure gradient concentration below the solubility limit was prepared and added to several plastic pots containing 10 seeds of the test plant (Jew’s mallow Corchorus olitorius). The pots were then transferred to the growth chamber for 2 weeks and irrigated as necessary. After 2 weeks % growth inhibition of the tested plant was calculated according to growth inhibition equation.
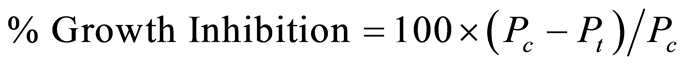 (1)
(1)
where Pc and Pt are the shoot heights of the control and the treated samples at any soil depth.
2.6. Biodegradation of Diuron
Twenty ml of Cyanobacterial mats suspension assumed to contain 106 cell/ml was mixed with 340 gm of wet soil in plastic pots and left for 2 weeks for Cyanobacterial to adapt to soil environment prior to cultivation of plant seeds and application of Diuron. Diuron was applied in the cultivation day. Five different concentrations (0.0, 0.055, 0.11, 0.22, 0.44, 0.88 mg/kg soil) were used. Each concentration had 5 replicates. The same concentrations were used in all the next experiments.
2.7. Incubation of Diuron with Cyanobacterial Mats Directly in Pots
In this experiment, soil samples (340 g/pot) were sprayed with the required Diuron concentration after pre-incubation of Cyanobacterial mats in soil and keeping the soil wet at saturation for 2 weeks. The pots were transferred to the growth chamber for 2 weeks, and 10 seeds of the test plant were sown in each pot. The pots were irrigated with 20 ml water free from chlorine after germination of seeds. The plant was irrigated as required. The growth of the test plants were taken as indicator of herbicide degradation according to Equation (1).
2.8. Incubation of Diuron with Cyanobacterial Mats in the Irrigation Water
In this procedure 5 L of natural irrigation water free from chlorine was transferred to 5 glass bottles, capacity 2 L. About 100 ml of cyanobacterial mats were transferred to each bottle 2 days before gradient Diuron concentrations were injected in the appropriate glass bottle. The bottles were transferred to the growth chambers for 20 days of incubation. Then the sterilized soil samples were transferred to plastic pots and ten seeds of Jow’s mallow were sown in each pot. The pots were organized as groups according to the tested concentrations of each herbicide. Then each group members were irrigated with 20 ml of the irrigation water pre-incubated with the tested concentration along with Cyanobacterial mats as mentioned above. This irrigation process continued everyday by adding 20 ml from the same water to the appropriate pot. The pots were then kept wet by addition of few drops of irrigation. The pots were left in the growth chamber for 2 weeks and then plant highest length was taken as indicator of Diuron degradation according to Equation (1).
2.9. Influence of Incubation Period
In the procedure series of glass bottles containing gradient concentrations of Diuron and the required amount of irrigation water and Cyanobacterial mats were incubated in the laboratory for 5, 10, 15, 25 and 30 days with 12 light and 12 dark period to match the living conditions of the Cyanobacterial mats in the natural eco-system (Wadi Gaza). Then at the end of incubation period the irrigation water was collected and used to irrigate pots pre-planted with 10 seeds of the test plant. This experiment includes positive control (Diuron without Cyanobacterial mat), and negative control (Cyanobacterial mat without Diuron). These bottles were incubated for the same period mentioned above. These bottles were used to irrigate the pot of the test plant at the end of the incubation period. Growth inhibition was taken as indicator of herbicide concentrations, and calculated according to Equation (1).
2.10. Statistical Analysis
The experiment design was randomized block. The growth inhibition data were subjected to analysis of variance, and main effects and interactions were tested for significance using repeated measures ANOVA. T-test was used to test the significant difference among concentrations. Average and standard deviation were calculated for each tested concentration.
3. Results and Discussion
The physical analysis of used soil showed that sand, silt and clay contents were 92%, 2% and 6% respectively. Chemical analysis showed that N% and P% were 1.5 and 1.2 respectively, whereas Na+, K+ and Ca2+ were 30.5, 0.4 and 29 meq/L respectively. pH was 7.5 and EC 5.3 Mmohs/cm more details are presented in El-Nahhal et al. [21] .
3.1. Cyanobacterial Mats
The diversity of Cyanobacterial mats used in this research was previously studied and characterized [23] , their ability to degrade diesel oil and hydrocarbons have been previously demonstrated [24] [25] and chloroacetanilides have recently been investigated [21] .
3.2. Dose-Response of Diuron
Dose-response relationship of Diuron (Figure 1) indicates a linear relationship at low concentrations. Storage positive association, R2 = 0.927 is shown from the results in the Figure. Accordingly, the linear regression equation can be used to estimate the remaining Diuron concentrations in soil sample after treatment with Cyanobacterial mats.
Effect of Diuron on Jew’s mallow growth as a test plant is shown in Figure 2. It is obvious that there is a decrease of Jew’s mallow growth as Diuron concentration increased in the soil. Furthermore, Diuron concentration higher that 0.055 mg/kg soil gave similar growth reduction of Jew’s mallow plants, indicating the sensitivity of the test plant to Diuron. Statistical analyses of Jew’s mallow growth as a response to Diuron concentration indicate significant differences. Differences at level 0.05 are given by different letters in the columns in Figure 2.
Influence of Cyanobacterial mats on Diuron degradation after 20 days incubation in soil media are shown in Figure 3. It can be seen that Jew’s mallow plants grow normally similar to the control sample at the lowest Diuron concentration (0.055 mg/kg soil). Regardless to the statistical variations, Jew’s mallow plants grow quite
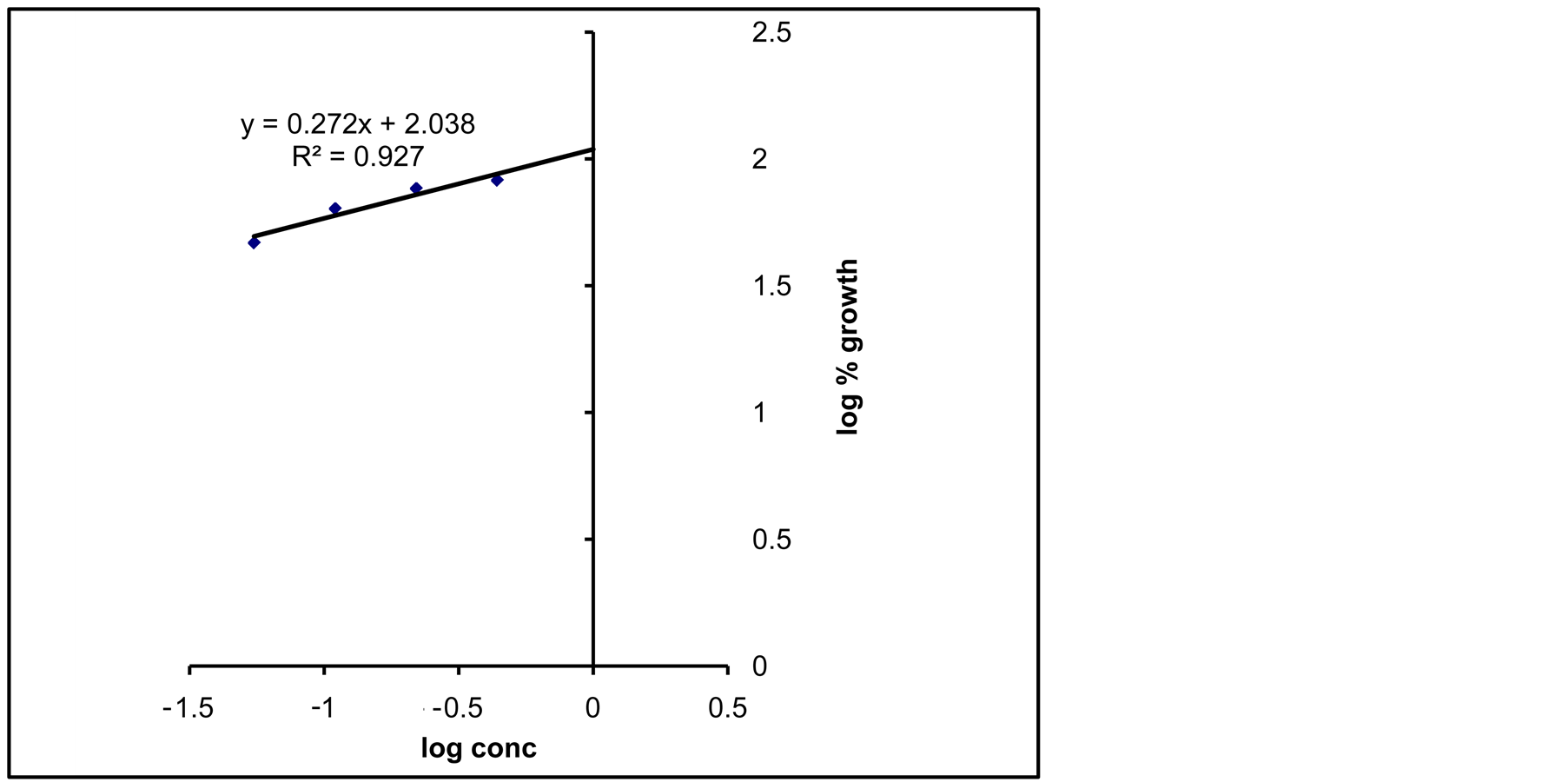
Figure 1. Dose response relationships between Diuron concentration and Jew’s mallow growth as test plant.
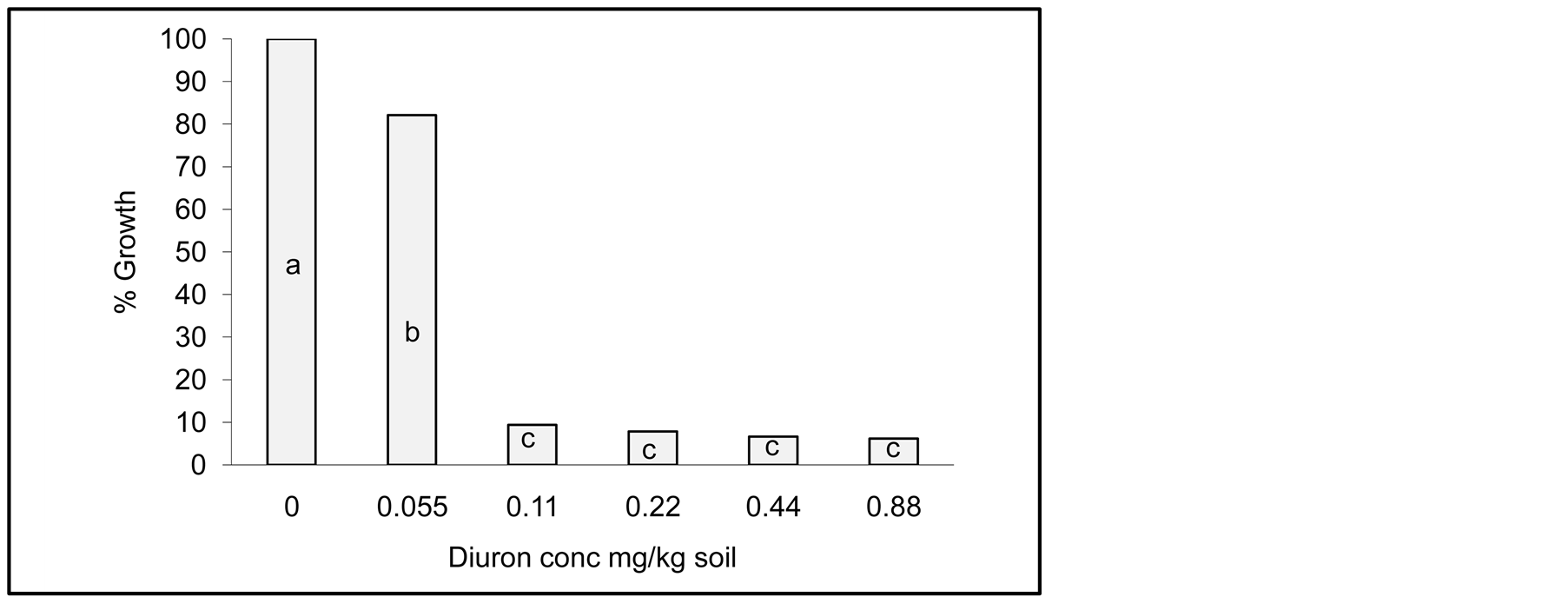
Figure 2. Influence of various concentration of Diuron on Jew’s mallow growth without incubation with Cyanobacterial mats. Effect measured was plant height. Error bars represent standard deviation. Columns which have the common letter are not significantly different at P = 0.05.
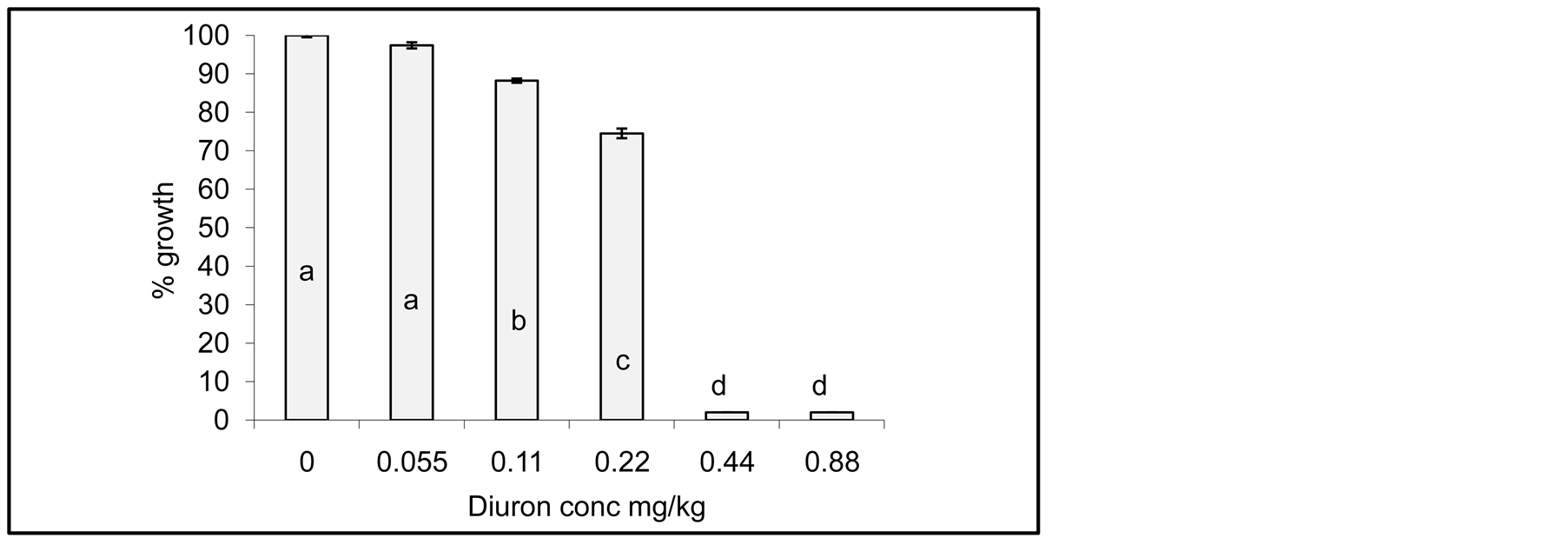
Figure 3. Influence of Cyanobacterial mats on Diuron degradation in soil media after 20 days of incubation time. Effect measured was plant height. Error bars represent standard deviation. Columns which have the common letter are not significantly different at P = 0.05.
normal at 0.11 and 0.22 mg/kg. These results indicate that Cyanobacterial mats degrade tremendous amounts of Diuron. However, growth reduction is still obvious at Diuron concentration of 0.44 and 0.88 mg/kg, indicating no degradation of Diuron. The explanation of these results is that Diuron is nearly toxic to Cyanobacterial mats at high concentration as demonstrated by Ma el al. [26] , or the degradation metabolites has similar toxicity to the parent compounds.
In addition we estimated the remaining concentrations of Diuron (mg/kg soil) and presented them in Table 1.
It is obvious that the remaining concentrations of Diuron are high at high initial concentration whereas at low initial concentration, the remaining concentration is low due to high fraction of degradation, these results agree with the data in Figure 3.
3.3. Kinetics of Diuron Degradation
Effect of incubation time on Diuron degradation by Cyanobacterial mat was shown in Figure 4.
Degradation of Diuron due to incubation with cyanobacterial mats indicated that the process did not go so fast and no complete degradation was observed as seen by growth reduction of Jew’s mallow plant. However, after one day of incubation little growth of Jew’s mallow plant was observed indicating that Diuron was still active in soil. After 15 days of incubation little Diuron was degraded as seen by increasing the growth of Jew’s mallow. Further degradation was also observed after 20 and 25 days but not complete. In contrast degradation of acetochlor
Table 1. Remaining concentrations of Diuron (mg/kg soil) after different periods of incubation (1-25 d).
D = day.
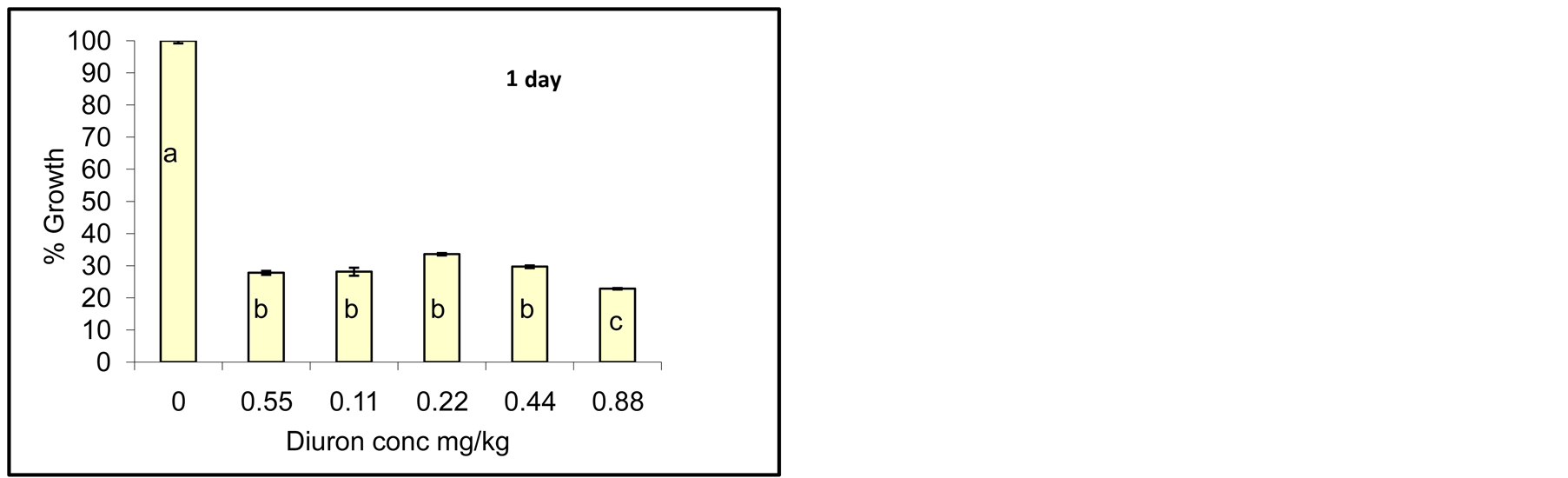
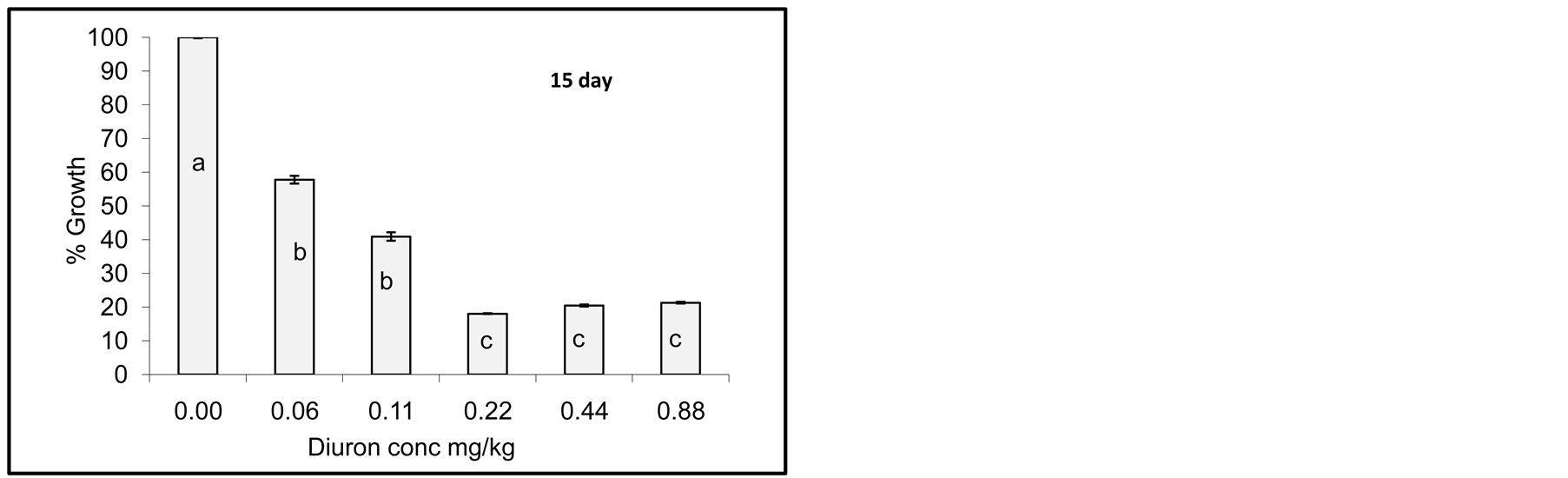
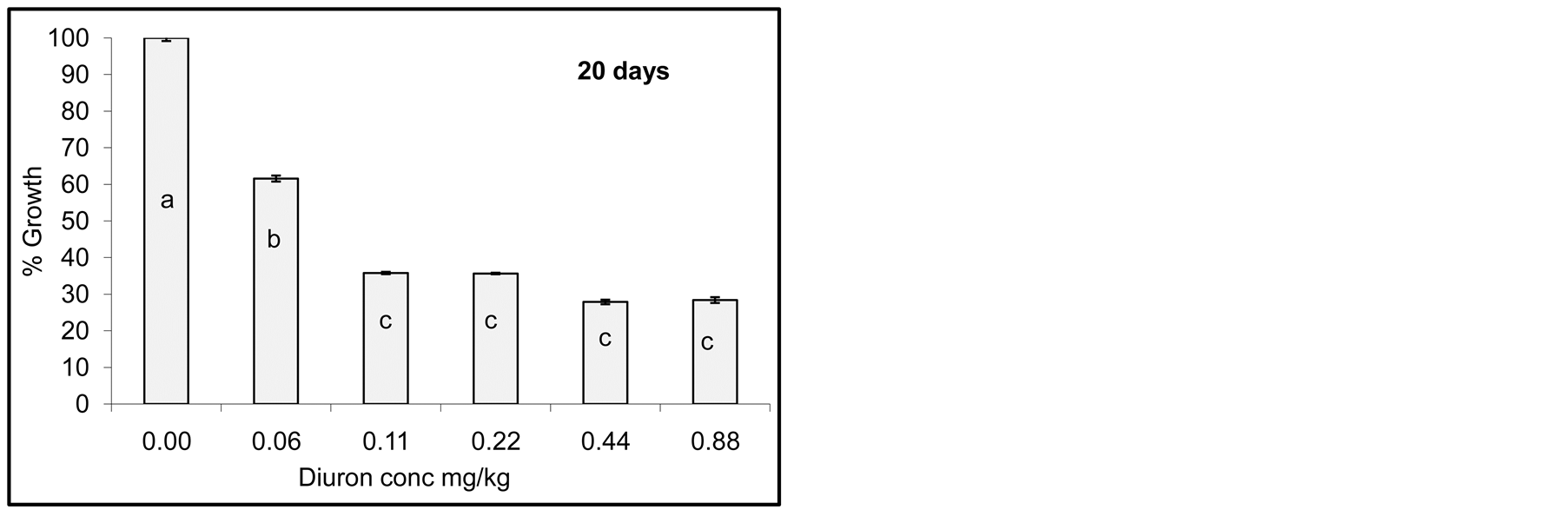
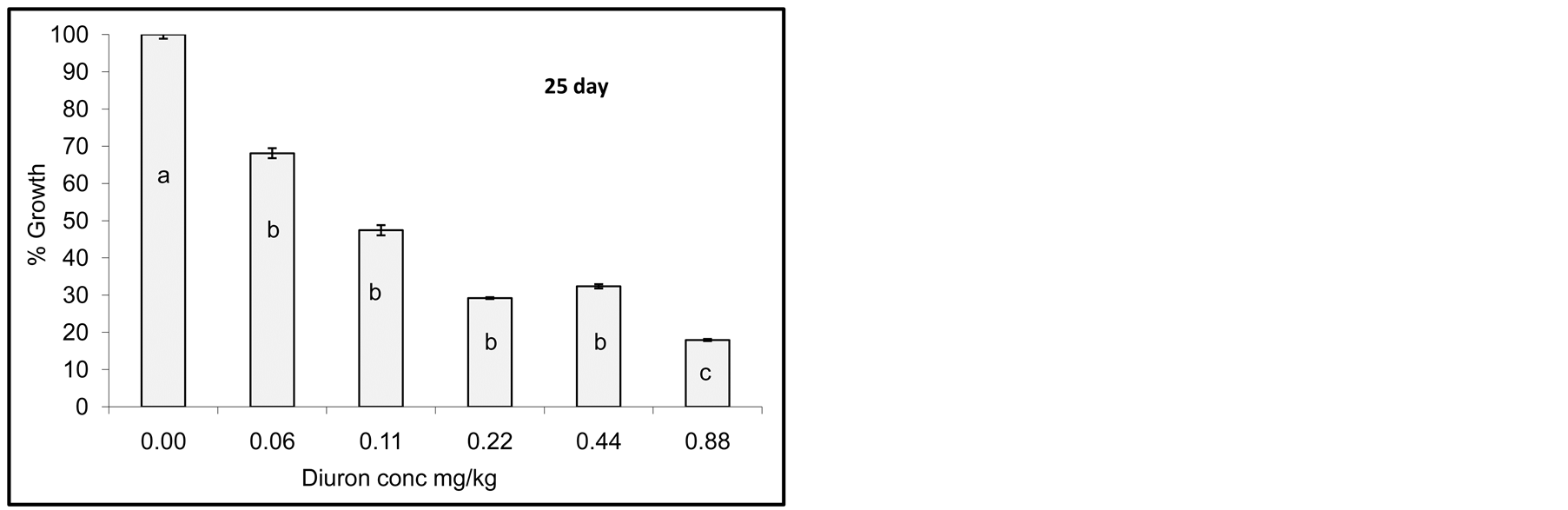
Figure 4. Effect of incubation times (1-25 day) on Diuron degradation by Cyanobacterial mat on soil media. Effect measured was plant height. Error bars represent standard deviation. Columns which have the same letter in an incubation time are not significantly different at P = 0.05.
was nearly completed after 25 days of incubation [21] . These results suggest that Diuron is nearly a toxic agent to Cyanobacterial mats at high concentration. This suggestion is supported by previous results [26] [27] who found that the acute toxicity of Diuron “the effective concentration that required to kill 50% of green algae (EC50)” 1.3 × 10−3 mg/L, indicating high toxicity. The low value for Diuron indicates that Diuron is several times more toxic to green algae than acetochlor, which agrees with the presented results. These results are in agreement with recent report [28] who found high toxicity of Diuron to Cyanobacterial mats collected from Waddy Gaza.
Due to this high toxicity of Diuron to green algae which is a component of Cyanobacterial mats a complete degradation was not observed. Furthermore the tested Diuron concentrations were several times higher than the acute toxicity of green algae. This indicated that Cyanobacterial mat can adopt themselves to different concentrations of Diuron. In addition, we estimated the remaining concentrations of Diuron (Table 1). It is obvious that there considerable reduction of Diuron concentrations over time up to 25 days. However, these results suggest that the degradation is a slow process or the biodegradation metabolites have similar toxicity to those of the
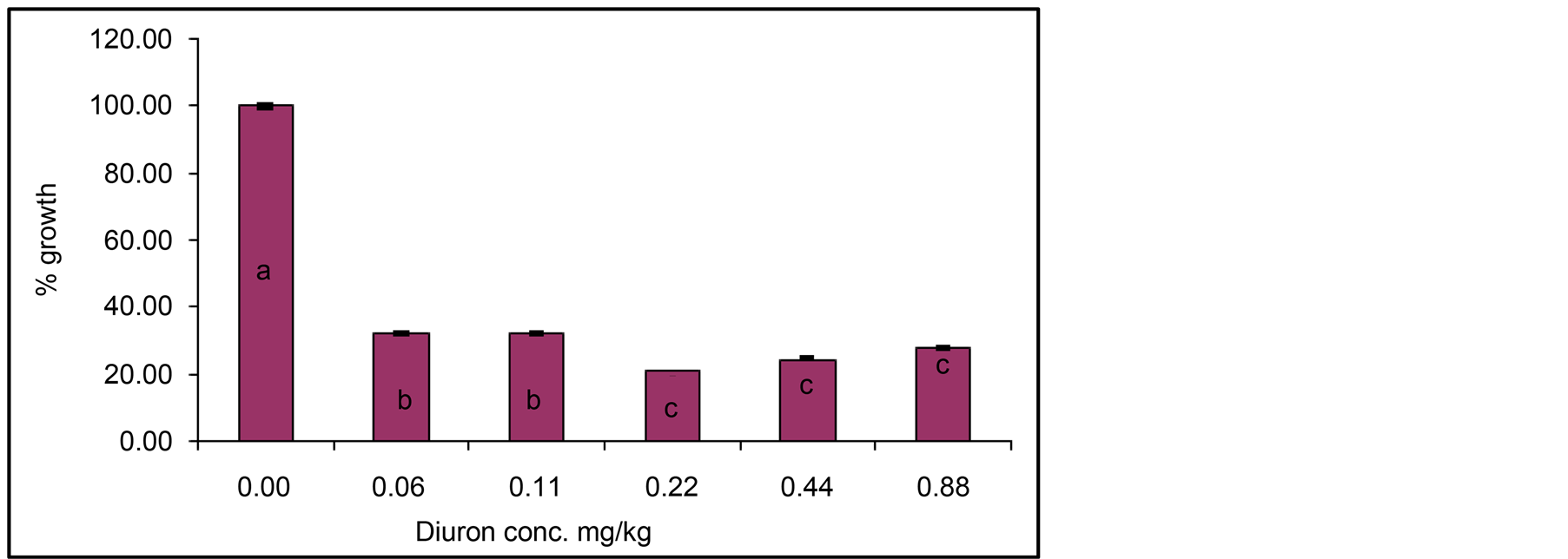
Figure 5. Influence of various concentrations of Diuron on Jew’s mallow height after 20 day of exposure to light without Cyanobacterial mat. Effect measured was plant height. Error bars represent standard deviation. Columns have the common letter are not significantly different at P = 0.05.
parent compounds. This explanation is supported by the results of Ma et al. [26] , who found similar toxicity of Diuron and its metabolites.
Statistical analysis of the data in Figure 4 indicates significant difference among concentrations for any incubation time. Columns which have the same letter at any incubation time are not significantly different at P = 0.05.
Despite the discrepancy of some results in Table 1 it is obvious that there was a gradient of degradation regardless of the fact that it was not completed. However, the tested concentrations 0.11 - 0.88 mg/kg soil are several times higher than the effective concentration that required for killing 50% green algae [26] [28] .
4. Effect of Light or Hydrolysis on Degradation of Diuron
Effect of Light or hydrolysis on degradation of Diuron is presented in Figure 5. It is clear that responses of Jew’s mallow plants to various concentrations of Diuron incubated with water without Cyanobacterial mats indicate high percent of growth reduction. These results indicate that Diuron is stable against hydrolysis or photochemical degradation. An interesting outcome of the data in Figure 5 is that stability of Diuron occurs under light or aquatic hydrolysis.
Comparison of the data in Figures 2 and 5 indicates that growth reduction in Figure 2 is higher than that in Figure 5. This indicates that Diuron either underwent photochemical degradation or slight hydrolysis as show by increased growth percent. Nevertheless, the data in Figure 4 and the estimated remaining concentration of Diuron (Table 1) indicate the stability of Diuron against photochemical degradation or hydrolysis.
5. Conclusion
Degradation of Diuron was successfully accomplished by Cyanobacterial mats at low Diuron concentrations. Kinetic studies indicated that biodegradation of Diuron was not reached due to possible acute toxicity at high concentrations. Kinetic studies for Diuron indicate considerable degradation levels. Degradation of Diuron was very fast at low concentrations and more pronounced at liquid media than at soil media. Kinetic studies indicate that Diuron or its metabolite is still active even after 30 days of incubation. Diuron can be toxic to Cyanobacterial mats at concentrations above 0.22 mg/kg soil. It can be concluded that application of Cyanobacterial mats to soil containing low concentrations of Diuron could be degraded by Cyanobacterial mats within a period of time equivalent to 30 days. These encouraging and promising results illustrate that Cyanobacterial mats can be used for bioreduction of soil and/or water pollution by herbicides.
References
- Kidd, H. and James, D.R. (1991) The Agrochemicals Handbook. 3rd Edition, Royal Society of Chemistry Information Services, Cambridge.
- Sørensen, S., Albers, C. and Aamand, J. (2008) Rapid Mineralization of the Phenylurea Herbicide Diuron by Variovorax sp. SRS16 in Pure Culture and within a Two-Member Consortium. Applied and Environmental Microbiology, 74, 2332-2340. http://dx.doi.org/10.1128/AEM.02687-07
- Braun, A.L. and Hawkins, L.S. (1991) Presence of Bromacil, Diuron, and Simazine in Surface Water Runoff from Agricultural Fields and Non-Crop Sites in Tulare County, California. Department of Pesticide Regulation, Pest Management and Analysis Program, Sacramento.
- Powell, S., Neal, R. and Leyva, J. (1996) Runoff and Leaching of Simazine and Diuron Used on Highway Rights of Way. Department of Pesticide Regulation, Environmental Monitoring and Pest Management Branch, Sacramento.
- Struger, J, Grabuski, J, Cagampan, S., Rondeau, M., Sverko, E. and Marvin, C. (2011) Occurrence and Distribution of Sulfonylurea and Related Herbicides in Central Canadian Surface Waters 2006-2008. Bulletin of Environmental Contamination and Toxicology, 87, 420-425. http://dx.doi.org/10.1007/s00128-011-0361-5
- El-Nahhal, Y., Abadsa, M. and Affifi, S. (2013) Adsorption of Diuron and Linuron in Gaza Soils. American Journal of Analytical Chemistry, 4, 94-99. http://dx.doi.org/10.4236/ajac.2013.47A013
- Moncada, A. (2004) Environmental Fate of Diuron, Environmental Monitoring Branch, Department of Pesticide Regulation, Sacramento.
- Bulcke, R., Cools, K., Dekkers, T., Eelen, H., Neus, O. and Rouchaud, J. (2000) Soil Dissipation of Diuron, Chlorotoluron, Simazine, Propyzamide, and Diflufenican Herbicides after Repeated Application in Fruit Tree Orchards. Archives of Environmental Contamination and Toxicology, 29, 60-65.
- Piutti, S., Marchand, A.L., Lagacherie, B., Martin-Laurent, F. and Soulas, G. (2002) Effect of Cropping Cycles and Repeated Herbicide Applications on the Degradation of Diclofopmethyl, Bentazone, Diuron Isoproturon and Pendimethalin in Soil. Pest Management Science, 58, 303-312. http://dx.doi.org/10.1002/ps.459
- Rouchard, J., Neus, O., Bulcke, R., Cools, K., Eelen, H. and Dekker, T. (2000) Soil Dissipation of Diuron, Chlorotoluron, Simazine, Propyzamide, and Diflufenican Herbicides after Repeated Applications in Fruit Tree Orchards. Archives of Environmental Contamination and Toxicology, 39, 60-65. http://dx.doi.org/10.1007/s002440010080
- Dellamatrice, P.M. and Monteiro, R.T.R. (2004) Isolation of Diuron-Degrading Bacteria from Treated Soil. Brazilian Archives of Biology and Technology, 47, 999-1003. http://dx.doi.org/10.1590/S1516-89132004000600020
- El-Deeb, B.A., Sollan, S.M., Ali, A.M. and Ali, K.A. (2000) Detoxification of the Herbicide Diuron by Pseudomonoas sp. Folia Microbiologica 45, 211-216. http://dx.doi.org/10.1007/BF02908946
- Sørensen, S.R., Ronen, Z. and Aamand, J. (2001) Isolation from Agricultural and Characterization of a Sphingomonas sp. Able to Mineralize the Phenylurea Herbicide Isoproturon. Applied and Environmental Microbiology, 67, 5403- 5409. http://dx.doi.org/10.1128/AEM.67.12.5403-5409.2001
- Widehem, P., Aït-Aïssa, S., Tixier, C., Sancelme, M., Veschambre, H. and Truffaut, N. (2002) Isolation, Characterization and Diuron Transformation Capacities of a Bacterial Strain Arthrobacter sp. N2. Chemosphere, 46, 527-534. http://dx.doi.org/10.1016/S0045-6535(01)00192-8
- Batisson, I., Pesce, S., Besse-Hoggan, P., Sancelme, M. and Bohatier, J. (2007) Isolation and Characterization of Diuron-Degrading Bacteria from Lotic Surface Water. Microbial Ecology, 54, 761-770. http://dx.doi.org/10.1007/s00248-007-9241-2
- Garcia-Pichel, F. (2000) Cyanobacteria. Encylopedia of Microbiology. Academic Press, San Diego, 907-927.
- El-Nahhal, Y. (2004) Leaching Behavior of Metolachlor in Soil. Journal of Environmental Engineering and Science, 3, 187-194. http://dx.doi.org/10.1139/s03-075
- El-Nahhal, Y. (2003) Persistence, Mobility, Efficacy and Safety of Chloroacetanilide Herbicide Formulation under Field Conditions. Environmental Pollution, 124, 33-38. http://dx.doi.org/10.1016/S0269-7491(02)00431-1
- El-Nahhal, Y., Lagaly, G. and Rabinovitz, O. (2005) Organo-Clay Formulations of Acetochlor: Effect of High Salt. Journal of Agricultural and Food Chemistry, 53, 1620-1624. http://dx.doi.org/10.1021/jf040383a
- Tomlin, C.D.S. (2000) The Pesticide Manual. British Crop Protection Council (BCPC), Farnham.
- El-Nahhal, Y., Awad, Y. and Safi, J. (2013) Bioremediation of Acetochlor in Soil and Water Systems by Cyanobacterial Mat. International Journal of Geosciences, 4, 880-890. http://dx.doi.org/10.4236/ijg.2013.45082
- El-Nahhal, Y., Nir, S., Polubesova, T., Margulies, L. and Rubin, B. (1998) Leaching, Phytotoxicity and Weed Control of New Formulations of Alachlor. Journal of Agricultural. Food Chemistry, 46, 3305-3313. http://dx.doi.org/10.1021/jf971062k
- Abed, R.M.M., Safi, N.M.D., Koster, J., de Beer, D., El-Nahhal, Y., Rullkotter, J. and Garcia-Pichel, F. (2002) Microbial Diversity of a Heavily Polluted Microbial Mat and Its Community Changes Following Degradation of Petroleum Compounds. Applied and Environmental Microbiology, 68, 1674-1683. http://dx.doi.org/10.1128/AEM.68.4.1674-1683.2002
- Safi, N.M.D., Koster, J. and Rulkotter, J. (2001) Fossil Fuel Pollution in Wadi Gaza and Biodegradation of Petroleum Model Compounds by Cyanobacterial Mats. The 36th Congress of the International Commission for the Scientific Exploration of the Mediterranean Sea, Monaco, 2001, 209.
- Safi, N. (2004) Environment Organic Geochemistry of Sediments from Wadi Gaza and Investigation of Bioremediation of Petroleum Derivatives and Herbicides by Cyanobacterial Mats under Different Experimental Conditions. Doktors der Naturwissenschaften (Ph.D. Thesis). Universitat Oldenburg, Oldenburg.
- Ma, J., Liang, W., Xu, L., Wang, S., Wei, Y. and Lu, J. (2001) Acute Toxicity of 33 Herbicides to the Green Alga Chlorella pyrenoidosa. Bulletin of Environmental Contamination and Toxicology, 66, 536-541. http://dx.doi.org/10.1128/AEM.68.4.1674-1683.2002
- Ma, J. (2002) Differential Sensitivity to 30 Herbicides among Populations of Two Green Algae Scenedesmus obliquus and Chlorella pyrenoidosa. Bulletin of Environmental Contamination and Toxicology, 68, 275-281.
- Kerkez, S. (2013) Potential Toxicity of the Herbicides; Diuron, Diquat, Terbutryn and their Mixtures to Cyanobacterial Mats, Wadi Gaza, Palestine. Master Thesis, The Islamic University, Gaza.


