Pharmacology & Pharmacy
Vol.06 No.02(2015), Article ID:53910,9 pages
10.4236/pp.2015.62009
The Adverse Event Profile in Patients Treated with TransferonTM (Dialyzable Leukocyte Extracts): A Preliminary Report
Toni Homberg1, Violeta Sáenz2, Jorge Galicia-Carreón2, Iván Lara2, Edgar Cervantes-Trujano1,2, Maria C. Andaluz1,2, Erika Vera1, Oscar Pineda1, Julio Ayala-Balboa1, Alejandro Estrada-García3, Sergio Estrada-Parra3, Mayra Pérez-Tapia4, Maria C. Jiménez-Martínez5*
1Clinical Trials Branch and Clinical Immunology Service, Unit of External Services and Clinical Research (USEIC), National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico
2Unit of Pharmacovigilance, Unit of External Services and Clinical Research (USEIC), National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico
3Department of Immunology, National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico
4Unit of R&D in Bioprocesses (UDIBI), National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico
5Department of Biochemistry, Faculty of Medicine, National Autonomous University of Mexico, Mexico City, Mexico
Email: *mcjimenezm@bq.unam.mx
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 22 December 2014; accepted 7 February 2015; published 10 February 2015
ABSTRACT
Background: Dialyzable leukocyte extracts (DLE) are heterogeneous mixtures of peptides less than 10 kDa in size that are used as immunomodulatory adjuvants in immune-mediated diseases. TransferonTM is DLE manufactured by National Polytechnic Institute (IPN), and is registered by Mexican health-regulatory authorities as an immunomodulatory drug and commercialized nationally. The proposed mechanism of action of TransferonTM is induction of a Th1 immunoregulatory response. Despite that it is widely used, to date there are no reports of adverse events related to the clinical safety of human DLE or TransferonTM. Objective: To assess the safety of TransferonTM in a large group of patients exposed to DLE as adjuvant treatment. Methods: We included in this study 3844 patients from our Clinical Immunology Service at the Unit of External Services and Clinical Research (USEIC), IPN. Analysis was performed from January 2014 to November 2014, searching for clinical adverse events in patients with immune-mediated diseases and treated with TransferonTM as an adjuvant. Results: In this work we observed clinical nonserious adverse events (AE) in 1.9% of patients treated with TransferonTM (MD 1.9, IQR 1.7 - 2.0). AE were 2.8 times more frequently observed in female than in male patients. The most common AE were headache in 15.7%, followed by rash in 11.4%, increased disease-related symptomatology in 10%, rhinorrhea in 7.1%, cough in 5.7%, and fatigue in 5.7% of patients with AE. 63% of adverse event presentation occurred from day 1 to day 4 of treatment with TransferonTM, and mean time resolution of adverse events was 14 days. In 23 cases, the therapy was stopped because of adverse events and no serious adverse events were observed in this study. Conclusion: TransferonTM induced low frequency of nonserious adverse events during adjuvant treatment. Further monitoring is advisable for different age and disease groups of patients.
Keywords:
Dialyzable Leukocyte Extracts, Adverse Events, Monitoring, Drug Safety, Adjuvant Therapy, Immunoregulation, Guidelines, Transfer Factor, Pharmacovigilance

1. Introduction
Dialyzable leukocyte extracts (DLE) are heterogeneous mixtures of peptides under 10 kDa, released after disruption of peripheral blood leukocytes from healthy donors [1] . It has been reported that administration of DLE improves clinical responses in allergies [2] , in infections and immunodeficiency syndromes [3] - [5] , and in some other immune-mediated diseases (reviewed in [6] ). The therapeutic adjuvant effect of DLE is associated with their ability to modulate immune responses changing innate signaling pathways, such as TLRs [7] , NF-kB, and cyclic adenosine monophosphate (cAMP) in cultured cells [8] [9] ; DLE could modulate production of cytokines, including TNF-a, IL-6 [10] [11] , and induce IFN-g secretion, driving immune response to a Th1 immune-regu- latory response [12] [13] .
TransferonTM is a human dialyzable leukocyte extract manufactured by National Polytechnic Institute (IPN), Mexico, at Good Manufacturing Practice (GMP) facilities. TransferonTM is registered by Mexican health authorities as a drug and is commercialized nationally. Although TransferonTM is a mixture of peptides, it has been demonstrated that there is a high batch-to-batch reproducibility in the chromatographic profile and also in the biological efficacy, demonstrated in vitro by up-regulation of IFN-g in a lymphocytic cell line (Jurkat clone E6-1) [1] .
DLEs have been widely used since the 1970s due to their immune-regulatory functions for various clinical purposes [6] [14] [15] ; to date, there are no current reports about the safety of TransferonTM; thus, it was the aim of our study.
2. Methods
2.1. Patients
We included a total of 3844 patients in this study. The medical staff from the Clinical Immunology Service, Unit of External Services and Clinical Research (USEIC) at IPN, was responsible for clinical evaluation. Analysis was performed from January 2014 to November 2014, searching for clinical adverse events in patients who received oral formulation of TransferonTM (Pharma-ft, UDIMEB formerly Laboratorio de Investigación Científica, IPN, MEX) as adjuvant therapy in immune-mediated diseases.
Patients selected for the use of DLE as an adjuvant treatment were those with immune-related disease, and whose symptoms remained in spite of standard treatment for that specific disease. DLE dosing was indicated based on the guidelines suggested by Berrón-Pérez et al. [6] . Adverse events were evaluated in the population with the following selection criteria: all patients who were treated with DLE during the period of January 2014- November 2014 who took at least one dose of DLE, and followed dosing instructions as indicated. Pediatric patients were considered those younger than 11years. Patients signed an informed consent as part of running clinical protocols IC 12-001, IC 12-002, IC-12-003 designed to determine the safety of TransferonTM. Patients aged between 8 and 17 years also gave their verbal assent to participate in these protocols. Adverse events were defined in this study according to local law regulation in drug safety [16] , and to the international regulation [17] . Serious adverse events were considered as any drug effect that results in death, life-threatening events, hospitalization, and disability; while nonserious adverse events were defined as drug related signs or symptoms that are tolerable, not life-threatening, sometimes needing additional treatment and/or require stopping the drug. The clinical terms used to describe each adverse event were based on terminology of the Medline Plus medical dictionary, from the National Institutes of Health [18] . As part of the national pharmacovigilance program, both serious and nonserious adverse events need to be reported to the federal authorities as stated by the Mexican Official Standard [16] . Adverse event surveillance was performed by the medical staff, and the pharmacovigilance unit helped to classify and report each event according to [16] , defined in numeral 6.1.3. “Clinical Investigation Notification Method” of the Mexican Official Standard. All involved personnel were properly trained for reporting adverse events and knowledgeable in all relevant Mexican regulation.
2.2. Statistical Analysis
Statistical analyses were performed using the GraphPad Prism software, version 6.0f (San Diego, CA). Demographic variables were analyzed with descriptive statistics, and results are presented in tables and plots. In order to determine differences between groups, T test and X2 were used, and a p < 0.05 was considered as statistically significant.
3. Results
3.1. Characteristics of Patients
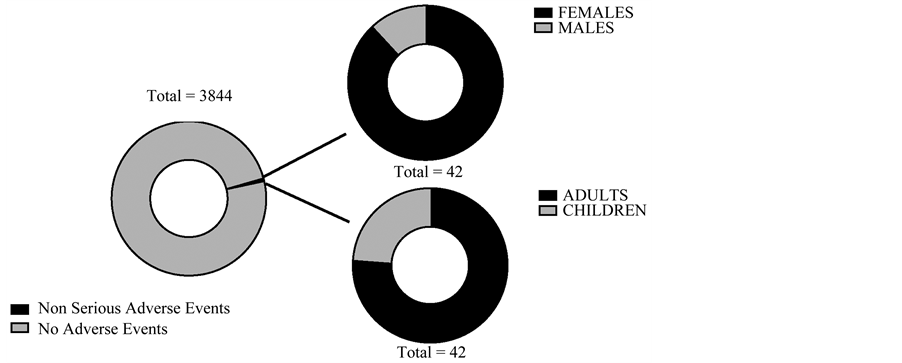
From a total of 3844 patients that were included in this study, 42 patients of them developed 70 nonserious adverse events (AE). Incidence of nonserious adverse events was observed in 1.9% of patients treated with TransferonTM (MD 1.9%, IQR 1.7 - 2.0). AE were 2.8-times more frequently observed in female than in male patients (p < 0.0001); the mean age of adverse event presentation in adult patients was 47 ± 15 years old; while in children it was 5 ± 2.8 years old. AE were 3.2-times more frequent in adults than in children (p = 0.0002) (Figure 1). No serious AE were reported in patients treated with TransferonTM.
3.2. Immune-Mediated Diseases and Adverse Events
Immune-mediated diseases in which adjuvant treatment with TransferonTM is indicated were classified into one of three types: Allergic Diseases, Infectious Diseases and Autoimmune Diseases. From a total of 42 patients that developed an AE, 41.4% were observed in patients with diagnosis of allergic diseases, 24.3% were observed in patients with diagnosis of autoimmune diseases, and 34.3% were observed in patients with diagnosis of infectious disease. Frequency of immune-mediated diseases and frequency of adverse events are depicted in Table 1.
Figure 1. Distribution of adverse events and demographic data of patients treated with TransferonTM.
Table 1. Immune-mediated diseases in which TransferonTM is indicated and adverse events observed.
3.3. Adverse Events
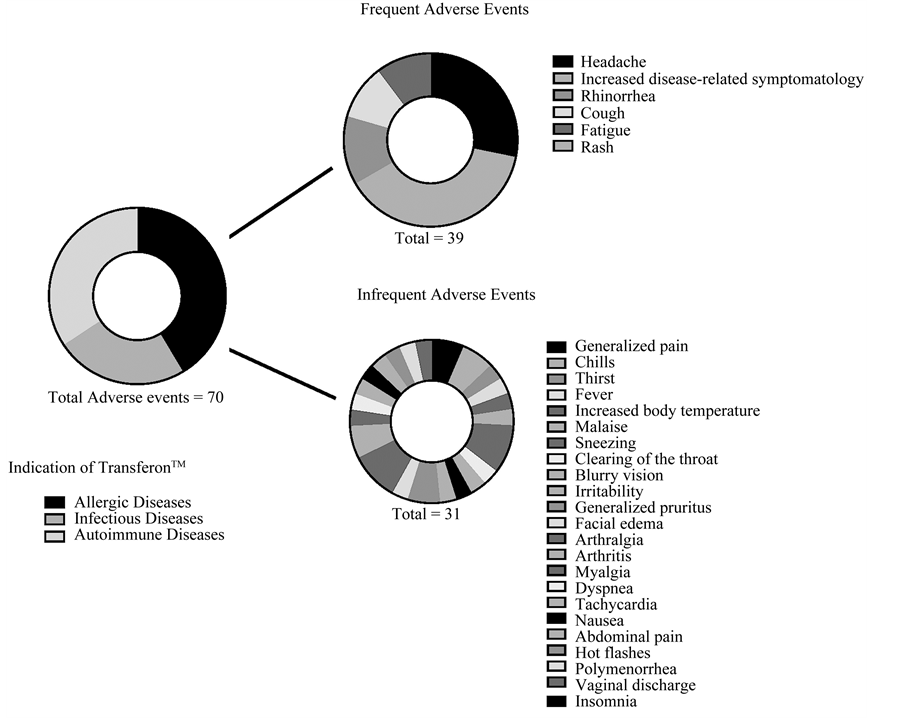
The most common AE observed with DLE was headache in 15.7% of patients, followed by rash in 11.4%, increased disease-related symptomatology in 10%, rhinorrhea in 7.1%, cough in 5.7%, and fatigue in 5.7% of patients (Figure 2). The remaining adverse events in patients treated with DLE are described in Table 2.
3.4. Patterns of Onset and Resolution of Adverse Events
TransferonTM was administered in units, in a dose-reduction scheme, as suggested by Berrón-Pérez et al. [6] . One unit of TransferonTM is a standardized vial containing 2 mg of dialyzed peptides/5mL [1] . Total units received per patient were dependent of the diagnosis, and patients who developed AE were at different stages of treatment: 27.14% of patients were taking 1 U every day for the first week, 20% of patients were taking 2 U/week, 28.6% of patients were taking 1 U once a week, 2.3% of patients were taking 1 U every 10 days, and 21.3% of patients were taking 1 U every 2 weeks. We did not find a significant correlation between higher dosage and higher frequency of AE.
The majority of AE (63%) appeared between 1 - 4 days of treatment with TransferonTM. AE lasted from 15 min to 14 days, and 77.1% of AE were resolved within the next 72 h after onset (Figure 3). In 32.9% of patients, TransferonTM was stopped because of adverse events; in 14.3% of patients, TransferonTM dosage was decreased after AE onset; and 2.86% of patients continued treatment with DLE without changes.
3.5. Drug Interactions
Although AE were more frequently observed in patients taking thyroid hormone or estrogen substitution, and/or
Figure 2. Distribution of type of adverse events in patients treated with TransferonTM.
glucocorticoid therapy, no significant interactions between TransferonTM and other concomitant medication were identified.
4. Discussion
Dialyzable leukocyte extracts are heterogeneous mixtures of polar and hydrophilic peptides under 10 kDa, released after disruption of peripheral blood leukocytes from healthy donors [1] . DLE have been used to modulate immune response [1] [10] - [13] in patients suffering immune-mediated diseases [6] [14] [15] . TransferonTM is a human dialyzable leukocyte extract manufactured by National Polytechnic Institute (IPN), Mexico, at GMP facilities. The use of TransferonTM is widely extended in our country [6] ; but despite their broad clinical use, to our knowledge no adverse events have been studied before.
In this work, we observed a low frequency of AE with the use of DLE. A significant adverse effect is usually considered as a reaction present in at least 2% of the studied population [19] . Of all included patients, only 1.9% of them developed adverse events. These results suggest that DLE is a safe and tolerable substance, but the unexpected low incidence of adverse events also opens the possibility of TransferonTM-related adverse events may remain under-diagnosed.
Comparing our adverse event profile with adverse effects of glatiramer acetate (GA), a four peptide-based product with 5 - 9 kDa in size [19] - [21] , we did not find similarities. GA is an injectable synthetic amino acid- based product with immune regulatory properties, reported incidence of adverse effects is at least 10%, mostly related to injection-site reaction. DLE is an oral peptide mixture lower than 10 kDa in size, and even though oral and injectable formulations cannot be fully compared, we expected to find some similar adverse effects between TransferonTM and GA. Nevertheless, some interesting patterns in adverse events presentation with the use of
Table 2. Adverse events identified in patients using TransferonTM.
Figure 3. Time of onset and resolution of adverse events in patients treated with TransferonTM.
TransferonTM are worth analyzing, i.e. AE were more frequent in females, and we detected 2 vulnerable age groups: ages 1 to 10, and 40 - 60 years old; this suggests that dosing should be reevaluated in order to reduce adverse events in both groups, and physicians need to increase clinical supervision to opportunely detect AE in children. It is unknown if hormonal factors may be involved in the female predominant adverse event profile showed in our study, but it is recognized that hormones may influence drug metabolism. Gender differences in adverse drug events have been observed before [22] [23] , and factors other than hormonal effects such as body weight and differences in pharmacokinetics may be involved. In order to determine if hormonal factors are related in this pattern, it would be desirable to further study adverse event incidence in different female groups, such as before menarche and after menopause.
In this study we observed that most common AE were headache, followed by rash, increased disease-related symptomatology, rhinorrhea, cough and fatigue. In line with our results, the most common side effects of IFN- gamma include fever, chills, fatigue, myalgia, and headache [24] - [26] , and it is well known that TransferonTM induces IFN-g in vivo [12] [13] , and in vitro [1] . On the other hand, some DLE have been associated to an in vitro transient elevation of cAMP [8] [9] , and increased concentrations of cAMP are related with headache [27] - [29] , rhinorrhea [30] [31] , and fatigue [32] - [34] . If TransferonTM is able to increase cAMP it is unknown, and needs further investigation. Importantly, no serious AE were observed in this study.
Limitations
The study population was heterogeneous, involving an extensive age group and patients presenting various different diseases as well as varied concomitant treatments for these diseases. Additional studies should be conducted in a controlled population. The low incidence of adverse events elevates the possibility that adverse events may be under-reported, and strengthening our pharmacovigilance protocols will aide in this matter. Finally, the lack of other adverse reaction reports associated to human DLE limits our possibility of comparison.
5. Conclusion
To our knowledge, no data on the adverse event profile of other DLEs have been published, and this study is an adequate first step to describe adverse events associated with DLE and TransferonTM; TransferonTM induced low frequency of nonserious adverse events during adjuvant treatment; however, further monitoring is advisable for different age and disease groups of patients.
Conflicts of Interest
S. E-P and M. P-T have been compensated for their work by UDIMEB, the producer of TransferonTM. All other authors declare no conflict of interest.
Author Contributions
T.H.―performed clinical evaluation, analyzed and interpreted clinical immunological data, and wrote the paper; V.S.―trained medical staff in pharmacovigilance and developed pharmacovigilance procedures according to Mexican regulation. V.S., J.G.-C.; I.L.―collected clinical information, and analyzed data; E.C.-T., M.C.A., E.V., O.P., J.A.-B.―performed clinical-immunological evaluation of patients; A.E-G.―analyzed data, wrote paper; S.E.-P.―contributed with critical criticism of paper and wrote paper; M.P.-T. and M.C.J.-M.―designed the study, analyzed data, wrote paper and conducted research. A.E.-G. received a PhD scholarship from CONACyT number 164000.
References
- Medina-Rivero, E., Merchand-Reyes, G., Pavón, L., Vázquez-Leyva, S., Pérez-Sánchez, G., Salinas-Jazmín, N., Estrada- Parra, S., Velasco-Velázquez, M. and Pérez-Tapia, S.M. (2014) Batch-to-Batch Reproducibility of Transferon™. Journal of Pharmaceutical and Biomedical Analysis, 88, 289-294. http://dx.doi.org/10.1016/j.jpba.2013.09.004
- Navarro Cruz, D., Serrano Miranda, E., Orea, M., Estrada Parra, S., Teran-Ortiz, L., Gomez-Vera, J. and Flores- Sandoval, G. (1996) Transfer Factor in Moderate and Severe Atopic Dermatitis. Revista Alergia México, 43, 116-123.
- Byston, J., Cech, K., Pekarek, J. and Jilkova, J. (1996) Effect of Anti-Herpes Specific Transfer Factor. Biotherapy, 9, 73-75. http://dx.doi.org/10.1007/BF02628660
- Masi, M., De Vinci, C. and Baricordi, O.R. (1996) Transfer Factor in Chronic Mucocutaneous Candidiasis. Biotherapy, 9, 97-103. http://dx.doi.org/10.1007/BF02628665
- Viza, D., Fudenberg, H.H., Palareti, A., Ablashi, D., De Vinci, C. and Pizza, G. (2013) Transfer Factor: An Overlooked Potential for the Prevention And Treatment of Infectious Diseases. Folia Biologica (Prague), 59, 53?67.
- Berron-Perez, R., Chavez-Sanchez, R., Estrada-Garcia, I., Espinosa-Padilla, S., Cortez-Gomez, R., Serrano-Miranda, E., Ondarza-Aguilera, R., Perez-Tapia, M., Pineda Olvera, B., Jimenez-Martinez, M.C., Portugues, A., Rodriguez, A., Cano, L., Pacheco, P.U., Barrientos, J., Chacon, R., Serafin, J., Mendez, P., Monges, A., Cervantes, E. and Estrada- Parra, S. (2007) Indications Usage, and Dosage of the Transfer Factor. Revista Alergia México, 54, 134?139.
- García-Hernández, U., Robledo-Avila, F.H., Alvarez-Jiménez, V.D., Rodríguez-Cortés, O., Wong-Baeza, I., Serafín- López, J., Aguilar-Anguiano, L.M., Estrada-Parra, S., Estrada-García, I., Pérez-Tapia, S.M. and Chacón-Salinas, R. (2014) Dialyzable Leukocyte Extracts Activate TLR-2 on Monocytes. Natural Product Communications, 9, 853-856.
- Herlin, T., Jensen, J.R., Thestrup-Pedersen, K. and Zachariae, H. (1981) Dialyzable Leukocyte Extract Stimulates cAMP in T Gamma Lymphocytes. Allergy, 36, 337-343. http://dx.doi.org/10.1111/j.1398-9995.1981.tb01585.x
- Tsuneta, H. (1984) The Role of Cultured Thymic Epithelium and Dialyzable Leukocyte Extracts on the Maturation Process of T Cell. Study of Their Effects on Cyclic Nucleotides Levels in Thymocytes. Hokkaido Igaku Zasshi, 59, 128-139.
- Franco-Molina, M.A., Mendoza-Gamboa, E., Castillo-Leon, L., Tamez-Guerra, R.S. and Rodriguez-Padilla, C. (2005) Bovine Dialyzable Leukocyte Extract Modulates the Nitric Oxide and Pro-Inflammatory Cytokine Production in Lipopolysaccharide-Stimulated Murine Peritoneal Macrophages in Vitro. Journal of Medicinal Food, 8, 20-26. http://dx.doi.org/10.1089/jmf.2005.8.20
- Hernandez, M.E., Mendieta, D., Pérez-Tapia, M., Bojalil, R., Estrada-Garcia, I., Estrada-Parra, S. and Pavon, L. (2013) Effect of Selective Serotonin Reuptake Inhibitors and Immunomodulator on Cytokines Levels: An Alternative Therapy for Patients with Major Depressive Disorder. Clinical and Developmental Immunology, Article ID: 267871. http://dx.doi.org/10.1155/2013/267871
- Estrada-Parra, S., Nagaya, A., Serrano, E., Rodriguez, O., Santamaria, V., Ondarza, R., Chavez, R., Correa, B., Monges, A., Cabeza, R., Calva, C. and Estrada-Garcia, I. (1998) Comparative Study of Transfer Factor and Acyclovir in the Treatment of Herpes Zoster. International Journal of Immunopharmacology, 20, 521-535. http://dx.doi.org/10.1016/S0192-0561(98)00031-9
- Luna-Baca, G.A., Linares, M., Santacruz-Valdes, C., Aguilar-Velazquez, G., Chavez, R., Perez-Tapia, M., Estrada- Garcia, I., Estrada-Parra, S. and Jimenez-Martinez, M.C. (2007) Immunological Study of Patients with Herpetic Stro- mal Keratitis Treated with Dialyzable Leukocyte Extracts. 13th International Congress of Immunology-ICI, Proceed- ings Immunology, Rio de Janeiro, 21-25 August 2007, 67-70.
- Kirkpatrick, C.H. and Smith, T.K. (1976) The Nature of Transfer Factor and Its Clinical Efficacy in the Management of Cutaneous Disorders. Journal of Investigative Dermatology, 67, 425-430. http://dx.doi.org/10.1111/1523-1747.ep12514723
- Nekam, K., Kalmar, L., Gergely, P., Kelemen, G., Fekete, B., Lang, I., Levai, J. and Petranyi, G.Y. (1977) In Vitro Effect of Transfer Factor on Active Rosettes and Leucocyte Migration of Patients with Cancer. Clinical & Experi- mental Immunology, 27, 416-420.
- Mexican Official Standard NOM-220-SSA1-2012, Installation and Operation of Pharmacovigilance.
- US Department of Health and Human Services Food and Drug Administration, Office of the Commissioner, Center for Drug Evaluation and Research, Center for Biologics Evaluation and Research, Center for Devices and Radiological Health, Office of Good Clinical Practice (2009) Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs―Improving Human Subject Protection.
- Reviewed on Line (2014). www.nlm.nih.gov/medlineplus/mplusdictionary.html
- Copaxone Product Monograph (2011).
- Johnson, K.P., Brooks, B.R., Cohen, J.A., Ford, C.C., Goldstein, J., Lisak, R.P., Myers, L.W., Panitch, H.S., Rose, J.W., Schiffer, R.B., Vollmer, T., Weiner, L.P. and Wolinsky, J.S. (1998) Extended Use of Glatiramer Acetate (Copaxone) Is Well Tolerated and Maintains Its Clinical Effect on Multiple Sclerosis Relapse Rate and Degree of Disability. Copolymer 1 Multiple Sclerosis Study Group. Neurology, 50, 701-708. http://dx.doi.org/10.1212/WNL.50.3.701
- Bornstein, M.B., Miller, A., Slagle, S., Weitzman, M., Crystal, H., Drexler, E., Keilson, M., Merriam, A., Wassertheil- Smoller, S., Spada, V., et al. (1987) A Pilot Trial of COP 1 in Exacerbating-Remitting Multiple Sclerosis. The New England Journal of Medicine, 317, 408-414. http://dx.doi.org/10.1056/NEJM198708133170703
- Tran, C.L., Knowles, S.R., Liu, B.A. and Shear, N.H. (1998) Gender Differences in Adverse Drug Reactions. Journal of Clinical Pharmacology, 38, 1003-1009. http://dx.doi.org/10.1177/009127009803801103
- Pistone, G., Gurreri, R., Alaimo, R., Curiale, S. and Bongiorno, M.R. (2014) Gender Differences in Adverse Drug Reactions in Dermatological Patients in West Sicily: An Epidemiological Study. Journal of Dermatolog Treat, 25, 510-512. http://dx.doi.org/10.3109/09546634.2013.814755
- Murray, H.W. (1994) Interferon-Gamma and Host Antimicrobial Defense: Current and Future Clinical Applications. The American Journal of Medicine, 97, 459-467. http://dx.doi.org/10.1016/0002-9343(94)90326-3
- Gallin, J.L., Farber, J.M., Holland, S.M. and Nutman, T.B. (1995) Interferon-γ in the Management of Infectious Dis- eases. Annals of Internal Medicine, 123, 216-224. http://dx.doi.org/10.7326/0003-4819-123-3-199508010-00009
- Hübel, K.K., Dale, D.C. and Liles, W.C. (2002) Therapeutic Use of Cytokines to Modulate Phagocyte Function for the Treatment of Infectious Diseases: Current Status of Granulocyte Colony-Stimulating Factor, Granulocyte-Macrophage Colony-Stimulating factor, macrophage Colony-Stimulating Factor, and Interferon-γ. The Joural of Infectious Diseases, 185, 1490-1501. http://dx.doi.org/10.1086/340221
- Birk, S., Kruuse, C., Petersen, K.A.T., Felt-Hansen, P. and Olesen, J. (2006) The Headache-Inducing Effect of Cilos- tazol in Human Volunteers. Cephalalgia, 26, 1304-1309. http://dx.doi.org/10.1111/j.1468-2982.2006.01218.x
- DeSantana, J.M. and Sluka, K.A. (2008) Central Mechanisms in the Maintenance of Chronic Widespread Noninflam- matory Muscle Pain. Current Pain and Headache Reports, 12, 338-343. http://dx.doi.org/10.1007/s11916-008-0057-7
- Guo, S., Olesen, J. and Ashina, M. (2014) Phosphodiesterase 3 Inhibitor Cilostazol Induces Migraine-Like Attacks via Cyclic AMP Increase. Brain, 137, 2951-2959. http://dx.doi.org/10.1093/brain/awu244
- Schmidt, B.M., Kusma, M., Feuring, M., Timmer, W.E., Neuhäuser, M., Bethke, T., Stuck, B.A., Hörmann, K. and Wehling, M. (2001) The Phosphodiesterase 4 Inhibitor Roflumilast Is Effective in the Treatment of Allergic Rhinitis. Journal of Allergy and Clinical Immunology, 108, 530-536. http://dx.doi.org/10.1067/mai.2001.118596
- Lee, R.J., Chen, B., Doghramji, L., Adappa, N.D., Palmer, J.N., Kennedy, D.W. and Cohen, N.A. (2013) Vasoactive Intestinal Peptide Regulates Sinonasal Mucociliary Clearance and Synergizes with Histamine in Stimulating Sinonasal Fluid Secretion. The FASEB Joural, 27, 5094-103. http://dx.doi.org/10.1096/fj.13-234476
- Kolbeck, R.C. and Speir, W.A. (1991) Theophylline, Fatigue, and Diaphragm Contractility: Cellular Levels of 45Ca and cAMP. Journal of Applied Physiology, 70, 1933-1937.
- Staines, D.R. (2006) Phosphodiesterase Inhibitors May Be Indicated in the Treatment of Postulated Vasoactive Neuro- peptide Autoimmune Fatigue-Related Disorders. Medical Hypotheses, 66, 203-204. http://dx.doi.org/10.1016/j.mehy.2005.08.006
- Staines, D.R., Brenu, E.W. and Marshall-Gradisnik, S. (2009) Postulated Vasoactive Neuropeptide Immunopathology Affecting the Blood-Brain/Blood-Spinal Barrier in Certain Neuropsychiatric Fatigue-Related Conditions: A Role for Phosphodiesterase Inhibitors in Treatment? Neuropsychiatric Disease and Treatment, 5, 81-89.
NOTES

*Corresponding author.