Neuroscience & Medicine
Vol.5 No.4(2014), Article ID:49702,13 pages
DOI:10.4236/nm.2014.54021
The Biological Actions and Mechanisms of Brain-Derived Neurotrophic Factor in Healthy and Disordered Brains
Toshiyuki Mizui1,2, Yuichiro Tanima1,2, Hiroko Komatsu3, Haruko Kumanogoh1,2*, Masami Kojima1,2*
1Bio-Interface Research Group, Health Research Institute, National Institute of Advanced Industrial Science and Technology (AIST), Ikeda, Japan
2Core Research for Evolutional Science and Technology (CREST), Japan Science and Technology Agency (JST), Kawaguchi, Japan
3Global Discovery Research, Neuroscience and General Medicine Product Creation Unit, Eisai Product Creation Systems, Eisai Co., Ltd., Tsukuba, Japan
Email: *h-kumanogou@aist.go.jp, *m-kojima@aist.go.jp
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 13 July 2014; revised 10 August 2014; accepted 2 September 2014
Abstract
Brain-derived neurotrophic factor (BDNF) is a neurotrophin that elicits neuronal survival and differentiation, synaptic transmission, and the modulation of synaptic plasticity. The biological actions of BDNF are mediated via two distinct receptors: the high-affinity tropomyosin-related kinase B (TrkB) receptor and the low-affinity p75 neurotrophin receptor (p75NTR). Recent findings regarding the actions and mechanisms of BDNF are reviewed here. Activity-dependent synaptic plasticity, as exemplified by long-term potentiation (LTP) and long-term depression (LTD), underlies the cellular mechanism of learning and memory. An accumulating body of evidence shows that BDNF modulates synaptic plasticity. This function requires extracellular neurotrophin release, synaptic activity-dependent local protein synthesis. In addition, a precursor of BDNF, proBDNF, is emerging as a new ligand with biological activities that are distinct from those of BDNF. The proteolytic cleavage of proBDNF is also proposed as a mechanism that determines the direction of BDNF actions. This review discusses the post-translational processing of proBDNF, the modulatory roles of the human BDNF polymorphism Val66Met, recent reports of the novel mechanisms of BDNF expression, and clinical reports showing the roles of BDNF in the blood. Taken together, these data provide new insights into the biological roles of BDNF and its related molecules in the central nervous system.
Keywords: Component, Brain-Derived Neurotrophic Factor (BDNF), Tropomyosin-Related Kinase B (TrkB), Synaptic Plasticity, Polymorphism, Proteolytic Cleavage

1. Introduction
Levi-Montalcini and Hamburger first discovered that a mouse sarcoma tumor implanted near the spinal cord of the developing chicken released a soluble protein that increased neurite outgrowth from sympathetic neurons [1] . This factor is named nerve growth factor (NGF) [2] -[4] . Over 20 years after the discovery of NGF, Barde and colleagues isolated a neuronal survival-eliciting factor from the pig brain, named brain-derived neurotrophic factor (BDNF) [5] ; the successful molecular cloning of BDNF cDNA came not long after [6] . BDNF is highly homologous to NGF at the amino acid sequence level. This finding led to the identification of two additional neurotrophins: neurotrophin-3 (NT-3) and neurotrophin-4/5 (NT-4/5) [7] -[9] .
There are two forms of BDNF: proBDNF and mature BDNF (mBDNF), which are synthesized by intracellular and/or extracellular proteolytic enzymes (e.g., furin, pro-hormone convertase, and plasmin) [10] . The prodomain of neurotrophin is implicated in the protein folding of BDNF [11] . In the BDNF gene, a single nucleotide polymorphism (SNP), which substitutes methionine (Met) to valine (Val) at amino acid position 66 in the pro-region of human BDNF protein, was identified, and it was demonstrated that the Val66Met mutation (Val66Met or Rs6265) influences the activity-dependent secretion of BDNF [12] . Since this report, accumulating evidence suggests that the Val66Met genetic variant increases the susceptibility of an individual to a variety of brain disorders [13] .
2. Neurotrophin Receptors
The NTs interact with two distinct classes of receptors. The first receptor, the p75 neurotrophin receptor (p75NTR), is a member of the tumor necrosis factor (TNF) receptor family [8] . p75NTR was originally identified as a receptor for NGF, but was subsequently shown to bind to BDNF, NT-3, and NT-4/5 with a similar low affinity [8] [9] . The extracellular domain of p75NTR has cysteine-rich motifs. The cytoplasmic domain includes a “death” domain, the cytoplasmic domain found in other members of the TNF receptor family [14] [15] . Although p75NTR does not contain a catalytic kinase motif, it interacts with several proteins that play important roles in regulating neuronal survival and differentiation [16] .
The second class is the tropomyosin-related kinase (Trk) family of receptor tyrosine kinases (TrkA, TrkB, and TrkC) [9] . NGF specifically binds TrkA; BDNF and NT4 activates TrkB; NT3 activates TrkC. In some cellular contexts, NT3 is able to activate TrkA and TrkB [9] . The extracellular domain of each Trk receptor consists of a cysteine-rich cluster followed by a series of leucine-rich repeats and two immunoglobulin-like domains. The Trk receptors have a transmembrane domain and a cytoplasmic domain that has tyrosine kinase activity. The activation of NTs, through Trk receptors, leads to the activation of Ras, phosphatidylinositol 3-kinase (PI3K), and phospholipase C-γ1. These signaling pathways control the activity of downstream molecules such as mitogen-activated protein (MAP) kinases [9] . The NT receptors and their signaling mechanisms have been extensively reviewed by others [9] [10] [16] and so will not be discussed in this review article.
3. ProBDNF, Pro-Peptide of BDNF, and BDNF
Lee and colleagues reported that the proteolytic cleavage of NTs (from the precursor form to the mature form) is an important process in their post-transcriptional mechanism, and determines their specific action [17] . A precursor of NGF (proNGF) promotes neuronal death, while mature NGF elicits neuronal survival [17] . Also, proBDNF induces the death of sympathetic neurons, cerebellar neurons, and motor neurons in vitro [18] -[20] . Furthermore, proNGF and proBDNF bind to the p75NTR and the sortilin receptor complex with high affinity [18] -[21] . Thus, the cellular responses to proand mature neurotrophins can be engaged by different combinations of Trk receptors, p75NTR, or sortilin family members. The roles of the precursor and the mature forms of BDNF in neuronal morphology and synaptic transmission will now be discussed.
The intracellular cleavage of the pro-domain of BDNF is performed by the pro-protein convertases (PC1/3, PC2, PC4, PC5/6, PC7) and furin [22] . The activity of these serine proteinases is dependent on Ca2+ concentrations (10 μM) and pH is in the range of 5 - 7, typically present in the trans-Golgi network (TGN) and secretory granules [23] . Pro-protein convertases are dominantly expressed in neurons, while furin is expressed in all tissues [24] . These convertases cleave the pro-peptide behind a pair of or single basic residues (Arg-Arg or Lys-Arg) and at the C-terminus of the pro-domain [23] [24] .
In the case of BDNF, 120 amino acid N-terminal fragment, namely the BDNF pro-peptide, is produced from proBDNF by proteinase cleavage (Figure 1). Dieni and her colleagues demonstrated that the BDNF pro-peptide is detectable in hippocampal tissues [25] . They investigated the distribution of both BDNF and its pro-peptide in the adult hippocampus using light and electron microscopy and by using genetically engineered mouse strains and antibodies generated by Koshimizu et al. [19] . BDNF and its pro-peptide are present in hippocampal extracts at similar concentrations [25] , suggesting that both peptides are stored after cleavage. Both BDNF and its pro-peptide are detected in the same sub-population of large secretory vesicles in presynaptic terminals, showing that neuronal stimulation leads to the exocytosis of secretory vesicles and may promote their delivery to the postsynaptic neurons. Another possibility raised from this report is that, as in the case of neuropeptides such as beta-endorphin [26] , the BDNF pro-peptide may exert biological functions beyond assisting the folding of BDNF. This notion is indicated by several reports. Second, as described earlier, Val66Met polymorphism also affects the activity-dependent secretion of BDNF and memory function in human [12] . This report provided two interpretations. First, the BDNF pro-domain plays an important role in activity-dependent-secretion of BDNF. Second, this regulated-secretion mechanism might be crucial for human memory function.
Interestingly, Anastasia et al. (2012) showed that Val66Met polymorphism alters prodomain structure, resulting in a change in the biological activity of the BDNF pro-peptide to induce neuronal growth cone retraction [27] . This group also investigated whether the Val66 and Met66 BDNF pro-peptides adopted different structures to elicit distinct biological activities, and showed that the structural changes induced by the Val66Met substitution confer bioactivity to the BDNF pro-peptide (as only the Met66 BDNF pro-peptide acutely alters neuronal
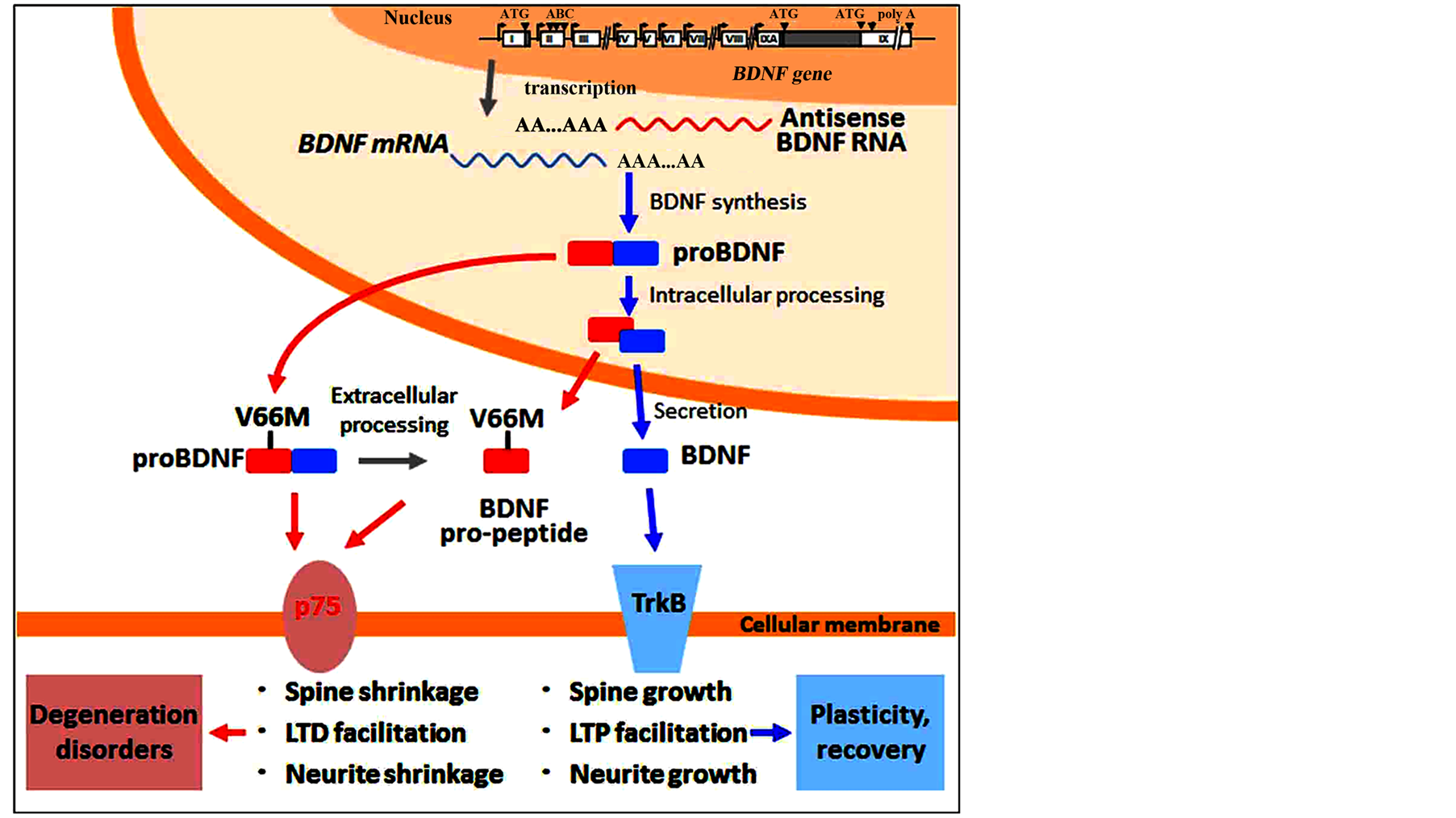
Figure 1. Synthesis and processing of BDNF. The BDNF gene is transcribed to BDNF mRNA, which is then targeted by antisense RNA molecules [76] [86] . BDNF mRNA is translated to proBDNF, which is either released or cleaved to yield mature BDNF (mBDNF) and the pro-peptide. mBDNF activates the TrkB receptor to promote plasticity and recovery from neuronal trauma or disorders. proBDNF interacts with the p75 receptor to induce degeneration, which leads to neurological and neuropsychiatric disorders.
morphology) and exerts this effect through differential interaction with SorCS2, the sortilin-related Vps10p domain sorting receptor [27] .
4. Molecular Mechanisms Underlying BDNF and ProBDNF Actions on Neurite Growth
BDNF promotes alternatively the growth or pruning of the axons intracellular mechanisms, which depend on the extent of the activity of TrkB or p75NTR receptors. These mechanisms must feed into the cytoskeletal regulation required for axonal and dendritic growth. BDNF induces branch formation via a combination of local and long-range signals, and the formation and maintenance of axonal branches is tightly coupled with activity-dependent events [28] . BDNF-mediated TrkB activation at the distal axon induces a retrograde Erk1/2 signal, which in turn activates immediate early gene transcription in the nucleus [29] . In addition to the axon growth, BDNF mediates the degeneration of sympathetic axons that express TrkA and p75NTR. The sympathetic neurons do not express the BDNF receptor TrkB; however, BDNF is secreted from active axons to stimulate p75NTR activity. The two receptors in the sympathetic neurons, p75NTR and TrkA, form high affinity binding sites for NGF, a process that depends upon the ratio of the two receptors [30] [31] . Another mechanism underlying the negative control of axonal growth involves components of myelin, which are largely responsible for inhibitory signals during regeneration [32] . BDNF-p75NTR signaling plays a role in the myelin-induced axonal degeneration of sympathetic axons. The myelin/BDNF/p75NTR signaling pathway activates the RhoA ROCKcofilin pathway [33] , which mediates axonal retraction, suggesting that BDNF-p75NTR signaling can potentially lead to axonal degeneration. Indeed, BDNF is a potential regulator of dendritic growth in CNS neurons [34] . We previously reported that BDNF led to increases in the number of primary dendrites [35] .
Proneurotrophins inhibit the outgrowth of neurites in basal forebrain cholinergic neurons, dorsal root ganglia neurons, and cortical and hippocampal neurons [19] [36] [37] . The growth-inhibiting activities assigned to mBDNF signaling via p75NTR may also be a consequence of proBDNF rather than mBDNF. Recently, Sun and colleagues showed that proBDNF causes a collapse in neurite outgrowth of primary neurons. An alternative mechanism of proBDNF action on the actin cytoskeleton is the activation of the RhoA/ROCK/cofilin pathway downstream of p75NTR [37] .
5. Morphological Alterations within Synapses
Long-term potentiation (LTP) and long-term depression (LTD) are synaptic phenomena expressed at excitatory synapse in the mammalian brain. The synaptic activity-induced LTP/LTD may lead to the morphological changes within synaptic structures. The neuronal growth activity of BDNF [7] raises the possibility that the bidirectional structural changes within synapses could be mediated by BDNF. One hypothesis is opened with a body of observations [7] ; “the neuronal growth activity of BDNF” and “the killing effect of precursor neurotrophins”. Indeed, exposure to BDNF causes the refinement of synapses in an activity-dependent manner [38] . Long-term treatment of hippocampal slices with BDNF increases the synapse number and the spine density of the apical dendrites of pyramidal neurons in the hippocampus [39] . BDNF acts on different types of spines depending on the presence of spontaneous synaptic transmission. Moreover, BDNF-mediated regulation of spine formation in the dendrites of hippocampal neurons is controlled by cyclic AMP (cAMP), a signaling molecule involved in late-LTP [40] . Taken together, the spontaneous neuronal activity and the consequent rise in the intracellular concentration of cAMP might be involved in BDNF-dependent spine formation.
The killing effect of precursor neurotrophins may correlate with pathological conditions of the nervous system, and may extend to other aspects of neurotrophin function. A recent study showed that proBDNF levels were decreased in the parietal cortex of subjects with Alzheimer’s disease [41] . Also, proNGF induces neuronal growth cone retraction through the inactivation of Rac and the interaction between p75NTR and SorCS2 [42] . Using cultured hippocampal neurons, we demonstrated that proBDNF reduces the density of dendritic spines, and that the amplitude of spontaneous activity was significantly reduced in proBDNF-treated cultures [19] . These findings show that BDNF could mediate bidirectional structural changes in the spines at the excitatory synapses.
6. The Roles of BDNF and ProBDNF in Synaptic Plasticity
Synaptic plasticity is a crucial mechanism for the processing and encoding of information in the central nervous system, and impairments in synaptic plasticity are associated with aging and brain diseases. LTP and LTD are forms of activity-dependent synaptic plasticity, which increase or decrease the strength of synaptic transmission, respectively. LTP and LTD are divided into at least two temporally distinct phases: early-LTP/LTD (E-LTP/ LTD) and late-LTP/LTD (L-LTP/LTD). E-LTP and L-LTP are parallel to the two forms of memory, short-term memory (STM) and long-term memory (LTM), respectively. In acute slices, E-LTP occurs during the first hour or two of LTP. It is also induced by high frequent tetanus stimulation (100 pulses at 100 Hz), leading to an increase in the strength of synaptic transmission and the activation of signaling molecules. L-LTP persists for longer and involves de novo synthesis of synaptic plasticity-related proteins (PRPs).
Given that BDNF expression is predominantly observed in the hippocampal pyramidal cell layer, it raises questions regarding the roles that BDNF and proBDNF play in synaptic plasticity. A substantial body of evidence shows that mBDNF is necessary and sufficient for the regulation of E-LTP and the maintenance of L-LTP through the TrkB receptor, whereas proBDNF promotes LTD by activating p75NTR [43] . High-frequency stimulation favors BDNF release and TrkB-dependent LTP in the CA1 zone of the hippocampus, while low-frequency stimulation activates proBDNF release [44] .
The relationship between BDNF action and the balance between excitatory and inhibitory synaptic activity is an interesting issue in the field of synaptic plasticity [45] . Glutamate receptors, including NMDA (N-methyl-Daspartate) and non-NMDA receptors, play distinct roles in synaptic plasticity. BDNF phosphorylates the NR1 and NR2B subunits of the NMDA receptor and up-regulates the GluR1 subunit of the a-amino-3-hydroxy-5- methyl-4-isoxazolepropionic acid (AMPA) receptor (in addition to the GluR2/3 subunits). Moreover, mature form of BDNF reduces the excitability of GABAergic inter-neurons by activating TrkB, whereas proBDNF does not affect GABAergic activity [46] . GABAergic function, at least under certain conditions, plays a crucial role in regulating the induction of LTP and LTD [47] .
The regulation of hippocampal E-LTP by BDNF was demonstrated using BDNF gene knockout mice and BDNF-scavenging proteins. For example, expression of E-LTP is markedly impaired in BDNF knockout mice [48] [49] . Furthermore, the in vitro scavenging of endogenous BDNF by a TrkB-IgG fusion protein [50] reduces the magnitude of E-LTP. Numerous studies indicate that BDNF acutely enhances E-LTP expression by increasing synaptic responses to tetanic stimulation and by enhancing the docking of synaptic vesicles, possibly through changes in the extent of phosphorylation of several synaptic proteins [51] -[53] . To this end, the application of BDNF-scavenging proteins and BDNF blocking antibodies abolish E-LTP after LTP-inducing tetanic stimulation [54] [55] . Moreover, BDNF elicits E-LTP in the dentate gyrus of the hippocampus, thereby increasing the function of NMDA receptors [56] [57] .
L-LTP is modulated by BDNF/TrkB signaling. L-LTP expression was inhibited in both BDNF knockout mice and in the presence of a TrkB blocking antibody [58] . The conversion of proBDNF to BDNF by tissue plasminogen activator (tPA)/plasmin is essential for L-LTP [59] , and L-LTP is impaired in mice harboring a targeted mutation in the phospholipase C-γ (PLC-γ) docking site (not Shc docking site) on TrkB [60] . Initial reports suggested that BDNF and proBDNF have opposite effects on synaptic plasticity [10] . Woo and colleagues showed that proBDNF facilitates hippocampal LTD by activating p75NTR, and that this facilitation requires NR2B activation [43] . Thus, BDNF and proBDNF might modulate synaptic plasticity bidirectionally through TrkB and p75NTR, respectively. The BDNF pro-peptide is cleaved out of proBDNF to produce mBDNF. The pro-peptide was detectable endogenously. However, the biological role of the BDNF pro-peptide is not fully understood. We found that the BDNF pro-peptide itself facilitates hippocampal LTD and elicits endocytosis of the AMPA-type receptor (Mizui et al., in submission), suggesting a novel role for the BDNF pro-peptide in synaptic plasticity.
7. Transcriptional Regulation of BDNF Expression by Neuronal Activity and a Newly Emerging Role in the Regulation of BDNF by Non-Coding BDNF Transcripts
Learning, training, and memory formation can alter the amount of bdnf mRNA and protein in the hippocampus. For example, the induction of LTP in the hippocampus rapidly and selectively increases the level of bdnf mRNA [61] [62] , and learning increases hippocampal BDNF, proBDNF, and TrkB protein content [63] . Tongiorgi and colleagues reported that bdnf mRNA is transported into the dendrites of hippocampal neurons by neuronal depolarization in vitro [64] , and by epileptogenic stimulation in vivo [65] [66] . These findings suggest that the dendritic movement of bdnf mRNA and the local translation of BDNF may promote activity-dependent synaptic plasticity.
BDNF-induced L-LTP depends on local protein synthesis in hippocampal dendrites [67] [68] . A body of biochemical data showed that BDNF stimulated the dendritic synthesis of different proteins by trafficking mRNA into the dendrites and by increasing the local translation of existing dendritic mRNA. In cultured neurons, BDNF induces the dendritic synthesis of synaptic proteins, which include activity-controlled cytoskeleton-associated proteins (Arc/Arg3.1), Ca2+/calmodulin-dependent protein kinase IIα (CaMKIIα), Homer2, and Lim kinase 1 (Limk1) [69] -[73] .
There are important questions regarding how and where this factor is produced by transcriptional and translational mechanisms in neuronal cells. The first report identified four promoters upstream of the 5' untranslated region (UTR) on rat BDNF gene (termed promoter I, II, III, and IV) [74] and, until recently, additional six (human) and five (rodent) promoters in human and rodent BDNF have been identified, respectively (leading to the new nomenclature of BDNF 5' UTR promoters termed I-V, Vh, VI-IXa in human and I-IXa in rodent) [75] [76] . Notably, in both rodent and human, BDNF gene expression is controlled by multiple activity-dependent (see below) and tissue-specific promoters [72] [73] . In rodent, promoter IV (previously known as promoter III) is activated by the stimulation with neuronal activity. Promoter IV also contains multiple Ca2+-responsive sequences and elements to which CaRF and CREB bind, respectively; therefore, the activation of this promoter is positively controlled by various stimuli encoded by Ca2+ and/or cAMP signals [77] -[80] . Human promoter I is induced by neuronal activity and the activation was controlled by bHLH-PAS-type transcription factor ARNT2-NPAS4 heterodimer. In promoter IV, however, CREB, ARNT2-NPAS4, and USFs are the key factors required for activation of the promoter by neuronal activity, and PasREand CRE-like elements regulate human BDNF promoter IX [81] . Furthermore, bdnf transcripts differ in their subcellular localization. An et al. (2008) recently reported that mouse bdnf mRNA with a short 3' UTR accumulates in the neuronal soma whereas bdnf mRNA with a long 3' UTR is trafficked to dendrites [82] . It was also reported that spatial segregation of bdnf transcripts allowed BDNF to differentially shape distinct dendritic compartments [83] .
Several reports suggest that non-coding natural antisense RNA controls the expression of protein-coding trascription in human and mouse (e.g. Wrap53 and BACE1-AS) [84] [85] . The human BDNF gene comprises 11 exons and nine functional promoters that are used in specific tissue and brain regions, and the non-coding natural antisense RNAs are transcribed from the BDNF-AS (anti-sense) gene within the BDNF gene locus. At least four BDNF-AS transcripts are co-expressed in the brain, and form RNA: anti-sense RNA duplexes [76] . Furthermore, a recent study found that inhibition of mouse bdnf-AS results in the up-regulation of bdnf mRNA expression [86] . Bdnf-AS represses the transcription of Bdnf-SENSE (sense) by altering the chromatin landscape at the gene locus, causing a decrease in the levels of BDNF protein. Oligonucleotides targeting bdnf-AS were intra-cerebroventricularly delivered to mice; consequently, bdnf mRNA levels in the forebrain regions of these were higher than those in control oligonucleotide-treated mice. Several neurodegenerative and psychiatric disorders are associated with impaired levels of neurotrophins, suggesting that the up-regulation of BDNF has beneficial effects on several disorders. Therefore, pharmacological approaches that target growth factor-related antisense transcripts warrant further study in preclinical model systems of neurological and psychiatric disease, and could complement new drug treatments aimed at shifting the balance between active and repressive chromatin markers in the brain [87] .
8. The Production and Secretion of ProBDNF
As previously mentioned, BDNF is initially synthesized as proBDNF, which is post-translationally cleaved by intracellular proteases to yield the mBDNF protein [88] . Recently, however, it was mentioned that this processing mechanism plays a key role in determining the cellular functions of the neurotrophin [88] [89] .
Two leading groups of Professors Barde and Hempstead recently carried out extensive studies into the effect of the presence and absence of endogenous proBDNF [90] [91] . However, their findings appeared to be inconsistent. Matsumoto and colleagues demonstrated that proBDNF produced by neurons is converted into mBDNF very rapidly, and proposed that proBDNF is the intermediate product. However, Yang and colleagues showed that neurons could produce and secrete proBDNF, and that high levels of proBDNF were present in the postnatal central nervous system. To demonstrate this, they treated primary cultures that were almost free of glial cells with a plasmin inhibitor to block proBDNF proteolysis. They were not able to detect mBDNF, probably because the TrkB-Fc protein added to the culture medium sequestered the mBDNF produced by the neurons. Several proteolytic enzymes must be present within distinct subcellular and extracellular locations to cleave proBDNF. Thus, when and where proBDNF is processed remains a key question that must be answered to understand the biological actions of mBDNF and proBDNF.
Although BDNF, its pro-peptide and proBDNF is now detectable [25] , the intracellular trafficking and processing of BDNF is not fully understood [10] . After synthesis in the endoplasmic reticulum (ER), BDNF needs to be folded correctly and conveyed into the constitutive or regulated secretory pathway for secretions. It has been shown that BDNF-containing large dense core vesicles (LDCVs) are present and undergoes anterograde transport to axonal terminals [25] [92] . On the other hand, in the dendritic side, the activity-dependent translocation of BDNF mRNA into dendrites was significantly found [64] .
The pro-domain of BDNF is conserved among vertebrate species, indicating that they have an important role in intracellular processing [93] [94] . Previously, it was demonstrated that Val66Met mutation in the BDNF prodomain (Figure 1) affected the trafficking and secretion of BDNF [12] [95] . However, this result does not reveal the mechanisms by which the pro-domain controls the intracellular sorting of neurotrophins. Interestingly, a putative sorting motif in the mature domain of BDNF was found through homology modeling of BDNF to pro-opiomelanocortin (POMC). Indeed, replacing the two acidic residues with alanine resulted in decrease in the regulated secretion of BDNF as well as an increase in constitutive secretion of BDNF [96] . Additionally, a crucial interaction between the acidic residues in BDNF and basic residues in a sorting receptor carboxypeptidase E (CPE) was evaluated [96] , and the activity-dependent secretion of BDNF was significantly decreased in cultured hippocampal neurons prepared from CPE-knockout mice. These reports suggest that the mature domain of BDNF contains a motif essential for sorting from the endoplasmic reticulum and/or Golgi network to the regulated secretory pathway.
What is the role of the BDNF pro-domain in secretion mechanism? There is a report suggesting that the interaction of the BDNF pro-domain with sortilin, a receptor that is localized mainly intracellularly, control the mode of BDNF secretion [97] . Sortilin is co-localized with BDNF in secretory granules in neurons, and interacts with two sub-regions in the BDNF pro-domain. Deletion of these sub-regions significantly reduced regulated secretion of BDNF. Therefore, these results indicate that the BDNF prodomain, in particular, the two sub-regions in pro-domain, is crucial for the regulated secretion of BDNF.
Thus, there are several intracellular mechanisms for controlling the intracellular maturation, trafficking and secretion of BDNF: 1) the interaction of CPE with the mature domain of BDNF for controlling the activity-dependent secretion of BDNF, 2) for facilitating proper folding of the mature domain, the interaction of intracellular sortilin with proBDNF in Golgi and 3) for sorting BDNF to the regulated secretory pathway, the interaction of sortilin with sub-regions in the BDNF pro-domain. Incorrect folding stimulates an accumulation of the protein in the endoplasmic reticulum and/or Golgi network, leading to an inability to target BDNF-containing vesicles to neuronal processes and/or synapses. To prevent this intracellular failure, three intracellular mechanisms might be crucial and promote an appropriate configuration, intracellular trafficking and secretion of BDNF. However, the Val66Met mutation selectively impaired the regulated secretion of BDNF without significant change in constitutive secretion of BDNF [12] [95] , showing that at least the prodomain surrounding Val66 does not be important for the binary control of sorting neurotrophins to the constitutive or regulated pathway. Finally, there are reports showing the pro-domain facilitates the proper folding of NGF [94] [98] . In the case of NGF, while interactions of the pro-domain with sortilin might allow correct folding, the absence of a sorting motif on the mature domain of NGF prevents it from entering the regulated secretory pathway.
9. Serum BDNF and Its Role in Brain Disorders
BDNF is thought to be a biomarker and potential drug target in patients with mental disorders. Many reports show a relationship between serum levels of BDNF and the onset of mental disorders, in particular depression. Patients with major depressed disorder (MDD) and schizophrenia have low levels of serum BDNF [99] [100] . Eight weeks of antidepressant treatment raises serum BDNF levels in MDD patients [101] . Sen and colleagues independently tested these possibilities and replicated those findings [102] . Patients with post-traumatic stress disorder (PTSD) have significantly higher serum BDNF levels than controls [103] . By contrast, PTSD patients have low plasma BDNF levels [104] .
Furthermore, Karege and colleagues measured the concentration of BDNF in the serum, plasma, and whole blood of MDD patients and controls and showed that although the BDNF levels in the serum and plasma of MDD patients were significantly lower than those of the control group, there was no difference in the whole blood BDNF levels. However, the plasma concentrations of platelet factor 4 (PF4), an important indicator of platelet activity, were higher in MDD patients. There were no significant differences in the PF4 serum levels between MDD patients and the control group. Thus, this report suggests that platelet activity, as indicated by PF4 activity, does not regulate the BDNF concentration in serum [100] .
BDNF is abundant in platelets [105] , and MDD patients have significantly lower levels of platelet BDNF than controls [106] . However, there are issues that remain unaddressed with regard to BDNF levels in the blood. These include differences in the concentrations of blood BDNF, the correlation between BDNF levels in the brain and blood, the source of blood BDNF, and the mechanisms controlling blood BDNF concentrations. Molendijk and colleagues report that human serum BDNF levels vary according to the season [107] . Epidemiological studies of blood BDNF levels would provide biological insights into clinical psychiatry, and facilitate the development of a more convenient method of diagnosis.
10. Conclusion
Here, we reviewed the biological actions and functional mechanisms of BDNF, and, through those descriptions, sought to extend the discussion for the understanding of the pathological mechanisms underlying brain disorders and physiological dynamics within the brain. In spite of the extension of BDNF biology, this knowledge would be expected to help to develop new therapies and aid the diagnosis of brain diseases in the future. We previously demonstrated that a human BDNF polymorphism Val66Met causing amino acid substitution in the BDNF prodomain impaired activity-dependent secretion of BDNF and human episodic memory. Since then, the intracellular mechanisms for controlling the intracellular maturation, trafficking and secretion of BDNF were extensively elucidated: 1) the interaction of CPE with the mature domain of BDNF for controlling the activity-dependent secretion of BDNF, 2) the interaction of intracellular sortilin with proBDNF in Golgi for facilitating proper folding of the mature domain and 3) the interaction of sortilin with sub-regions in the BDNF pro-domain for sorting BDNF to the regulated secretory pathway. Furthermore, by the development of the specific antibodies, the endogenous presence of BDNF, its pro-peptide and proBDNF has been ensured and their biological activities have been clarified. We expect that these new findings would develop the biology and provide new insights into the mechanical understanding of disordered brains.
References
- Levi-Montalcini, R. and Hamburge, V. (1953) A Diffusible Agent of Mouse Sarcoma, Producing Hyperplasia of Sympathetic-Ganglia and Hyperneurotization of Viscera in the Chick Embryo. Journal of Experimental Zoology, 123, 233-287. http://dx.doi.org/10.1002/jez.1401230203
- Cohen, S. and Levi-Montalcini, R. (1956) A Nerve Growth-Stimulating Factor Isolated from Snake Venom. Proceedings of the National Academy of Sciences of the United States of America, 42, 571-574.http://dx.doi.org/10.1073/pnas.42.9.571
- Cohen, S. (1960) Purification of a Nerve-Growth Promoting Protein from the Mouse Salivery Gland and Its Neuro-Cytotoxic Antiserum. Proceedings of the National Academy of Sciences of the United States of America, 46, 302-311. http://dx.doi.org/10.1073/pnas.46.3.302
- Bocchini, V. and Angeletti, P.U. (1969) The Nerve Growth Factor: Purification as a 30,000-Molecular-Weight Protein. Proceedings of the National Academy of Sciences of the United States of America, 64, 787-794.http://dx.doi.org/10.1073/pnas.64.2.787
- Barde, Y.A., Edgar, D. and Thoenen, H. (1982) Purification of a New Neurotrophic Factor from Mammalian Brain. The EMBO Journal, 1, 549-553.
- Leibrock, J., Lottspeich, F., Hohn, A., Hofer, M., Hengerer, B., Masiakowski, P., Thoenen, H. and Barde, Y.A. (1989) Molecular Cloning and Expression of Brain-Derived Neurotrophic Factor. Nature, 341, 149-152.http://dx.doi.org/10.1038/341149a0
- Bibel, M. and Barde, Y.A. (2000) Neurotrophins: Key Regulators of Cell Fate and Cell Shape in the Vertebrate Nervous System. Genes & Development, 14, 2919-2937. http://dx.doi.org/10.1101/gad.841400
- Chao, M.V. (2003) Neurotrophins and Their Receptors: A Convergence Point for Many Signalling Pathways. Nature Reviews Neuroscience, 4, 299-309. http://dx.doi.org/10.1038/nrn1078
- Reichardt, L.F. (2006) Neurotrophin-Regulated Signalling Pathways. Philosophical Transactions of the Royal Society B: Biological Sciences, 361, 1545-1564. http://dx.doi.org/10.1098/rstb.2006.1894
- Lu, B., Pang, P.T. and Woo, N.H. (2005) The Yin and Yang of Neurotrophin Action. Nature Reviews Neuroscience, 6, 603-614. http://dx.doi.org/10.1038/nrn1726
- Kolbeck, R., Jungbluth, S. and Barde, Y.A. (1994) Characterisation of Neurotrophin Dimers and Monomers. European Journal of Biochemistry, 225, 995-1003. http://dx.doi.org/10.1111/j.1432-1033.1994.0995b.x
- Egan, M.F., Kojima, M., Callicott, J.H., Goldberg, T.E., Kolachana, B.S., Bertolino, A., Zaitsev, E., Gold, B., Goldman, D., Dean, M., Lu, B. and Weinberger, D.R. (2003) The BDNF Val66Met Polymorphism Affects Activity-Dependent Secretion of BDNF and Human Memory and Hippocampal Function. Cell, 112, 257-269.http://dx.doi.org/10.1016/S0092-8674(03)00035-7
- Bath, K.G. and Lee, F.S. (2006) Variant BDNF (Val66Met) Impact on Brain Structure and Function. Cognitive, Affective, & Behavioral Neuroscience, 6, 79-85. http://dx.doi.org/10.3758/CABN.6.1.79
- Liepinsh, E., Ilag, L.L., Otting, G. and Ibáñez, C.F. (1997) NMR Structure of the Death Domain of the p75 Neurotrophin Receptor. The EMBO Journal, 16, 4999-5005. http://dx.doi.org/10.1093/emboj/16.16.4999
- He, X.L. and Garcia, K.C. (2004) Structure of Nerve Growth Factor Complexed with the Shared Neurotrophin Receptor p75. Science, 304, 870-875. http://dx.doi.org/10.1126/science.1095190
- Hempstead, B.L. (2002) The Many Faces of p75NTR. Current Opinion in Neurobiology, 12, 260-267.http://dx.doi.org/10.1016/S0959-4388(02)00321-5
- Lee, R., Kermani, P., Teng, K.K. and Hempstead, B.L. (2001) Regulation of Cell Survival by Secreted Proneurotrophins. Science, 294, 1945-1948. http://dx.doi.org/10.1126/science.1065057
- Teng, H.K., Teng, K.K., Lee, R., Wright, S., Tevar, S., Almeida, R.D., Kermani, P., Torkin, R., Chen, Z.Y., Lee, F.S., Kraemer, R.T., Nykjaer, A. and Hempstead, B.L. (2005) ProBDNF Induces Neuronal Apoptosis via Activation of a Receptor Complex of p75NTR and Sortilin. The Journal of Neuroscience, 25, 5455-5463.http://dx.doi.org/10.1523/JNEUROSCI.5123-04.2005
- Koshimizu, H., Kiyosue, K., Hara, T., Hazama, S., Suzuki, S., Uegaki, K., Nagappan, G., Zaitsev, E., Hirokawa, T., Tatsu, Y., Ogura, A., Lu, B. and Kojima, M. (2009) Multiple Functions of Precursor BDNF to CNS Neurons: Negative Regulation of Neurite Growth, Spine Formation and Cell Survival. Molecular Brain, 2, 27.http://dx.doi.org/10.1186/1756-6606-2-27
- Taylor, A.R., Gifondorwa, D.J., Robinson, M.B., Strupe, J.L., Prevette, D., Johnson, J.E., Hempstead, B., Oppenheim, R.W. and Milligan, C.E. (2012) Motoneuron Programmed Cell Death in Response to ProBDNF. Developmental Neurobiology, 72, 699-712. http://dx.doi.org/10.1002/dneu.20964
- Nykjaer, A., Lee, R., Teng, K.K., Jansen, P., Madsen, P., Nielsen, M.S., Jacobsen, C., Kliemannel, M., Schwarz, E., Willnow, T.E., Hempstead, B.L. and Petersen, C.M. (2004) Sortilin Is Essential for ProNGF-Induced Neuronal Cell Death. Nature, 427, 843-848. http://dx.doi.org/10.1038/nature02319
- Khatib, A.M., Siegfried, G., Chrétien, M., Metrakos, P. and Seidah, N.G. (2002) Proprotein Convertases in Tumor Progression and Malignancy: Novel Targets in Cancer Therapy. The American Journal of Pathology, 160, 1921-1935.http://dx.doi.org/10.1016/S0002-9440(10)61140-6
- Seidah, N.G. and Chrétien, M. (1999) Proprotein and Prohormone Convertases: A Family of Subtilases Generating Diverse Bioactive Polypeptides. Brain Research, 848, 45-62. http://dx.doi.org/10.1016/S0006-8993(99)01909-5
- Zhou, A., Webb, G., Zhu, X. and Steiner, D.F. (1999) Proteolytic Processing in the Secretory Pathway. Journal of Biological Chemistry, 274, 20745-20748. http://dx.doi.org/10.1074/jbc.274.30.20745
- Dieni, S., Matsumoto, T., Dekkers, M., Rauskolb, S., Ionescu, M.S., Deogracias, R., Gundelfinger, E.D., Kojima, M., Nestel, S., Frotscher, M. and Barde, Y.A. (2012) BDNF and Its Pro-Peptide Are Stored in Presynaptic Dense Core Vesicles in Brain Neurons. The Journal of Cell Biology, 196, 775-788. http://dx.doi.org/10.1083/jcb.201201038
- Jessop, D.S. (1999) Review: Central Non-Glucocorticoid Inhibitors of the Hypothalamo-Pituitary-Adrenal Axis. Journal of Endocrinology, 160, 169-180. http://dx.doi.org/10.1677/joe.0.1600169
- Anastasia, A., Deinhardt, K., Chao, M.V., Will, N.E., Irmady, K., Lee, F.S., Hempstead, B.L. and Bracken, C. (2013) Val66Met Polymorphism of BDNF Alters Prodomain Structure to Induce Neuronal Growth Cone Retraction. Nature Communications, 4, 2490. http://dx.doi.org/10.1038/ncomms3490
- Jeanneteau, F., Deinhardt, K., Miyoshi, G., Bennett, A.M. and Chao, M.V. (2010) The MAP Kinase Phosphatase MKP-1 Regulates BDNF-Induced Axon Branching. Nature Neuroscience, 13, 1373-1379.http://dx.doi.org/10.1038/nn.2655
- Brondello, J.M., Pouysségur, J. and McKenzie, F.R. (1999) Reduced MAP Kinase Phosphatase-1 Degradation after p42/p44MAPK-Dependent Phosphorylation. Science, 286, 2514-2517.http://dx.doi.org/10.1126/science.286.5449.2514
- Chao, M.V. and Hempstead, B.L. (1995) p75 and Trk: A Two-Receptor System. Trends in Neurosciences, 18, 321-326.http://dx.doi.org/10.1016/0166-2236(95)93922-K
- Hempstead, B.L., Martin-Zanca, D., Kaplan, D.R., Parada, L.F. and Chao, M.V. (1991) High-Affinity NGF Binding Requires Coexpression of the trk Proto-Oncogene and the Low-Affinity NGF Receptor. Nature, 350, 678-683.http://dx.doi.org/10.1038/350678a0
- Filbin, M.T. (2003) Myelin-Associated Inhibitors of Axonal Regeneration in the Adult Mammalian CNS. Nature Reviews Neuroscience, 4, 703-713. http://dx.doi.org/10.1038/nrn1195
- Park, K.J., Grosso, C.A., Aubert, I., Kaplan, D.R. and Miller, F.D. (2010) p75NTR-Dependent, Myelin-Mediated Axonal Degeneration Regulates Neural Connectivity in the Adult Brain. Nature Neuroscience, 13, 559-566.http://dx.doi.org/10.1038/nn.2513
- McAllister, A.K., Lo, D.C. and Katz, L.C. (1995) Neurotrophins Regulate Dendritic Growth in Developing Visual Cortex. Neuron, 15, 791-803. http://dx.doi.org/10.1016/0896-6273(95)90171-X
- Suzuki, S., Numakawa, T., Shimazu, K., Koshimizu, H., Hara, T., Hatanaka, H., Mei, L., Lu, B. and Kojima, M. (2004) BDNF-Induced Recruitment of TrkB Receptor into Neuronal Lipid Rafts: Roles in Synaptic Modulation. The Journal of Cell Biology, 167, 1205-1215. http://dx.doi.org/10.1083/jcb.200404106
- Deinhardt, K., Kim, T., Spellman, D.S., Mains, R.E., Eipper, B.A., Neubert, T.A., Chao, M.V. and Hempstead, B.L. (2011) Neuronal Growth Cone Retraction Relies on Proneurotrophin Receptor Signaling through Rac. Science Signaling, 4, ra82. http://dx.doi.org/10.1126/scisignal.2002060
- Sun, Y., Lim, Y., Li, F., Liu, S., Lu, J.J., Haberberger, R., Zhong, J.H. and Zhou, X.F. (2012) ProBDNF Collapses Neurite Outgrowth of Primary Neurons by Activating RhoA. PLoS One, 7, 35883.http://dx.doi.org/10.1371/journal.pone.0035883
- Cabelli, R.J., Hohn, A. and Shatz, C.J. (1995) Inhibition of Ocular Dominance Column Formation by Infusion of NT-4/5 or BDNF. Science, 267, 1662-1666. http://dx.doi.org/10.1126/science.7886458
- Tyler, W.J. and Pozzo-Miller, L.D. (2001) BDNF Enhances Quantal Neurotransmitter Release and Increases the Number of Docked Vesicles at the Active Zones of Hippocampal Excitatory Synapses. The Journal of Neuroscience, 21, 4249-4958.
- Ji, Y., Pang, P.T., Feng, L. and Lu, B. (2005) Cyclic AMP Controls BDNF-Induced TrkB Phosphorylation and Dendritic Spine Formation in Mature Hippocampal Neurons. Nature Neuroscience, 8, 164-172.http://dx.doi.org/10.1038/nn1381
- Michalski, B. and Fahnestock, M. (2003) Pro-Brain-Derived Neurotrophic Factor Is Decreased in Parietal Cortex in Alzheimer’s Disease. Molecular Brain Research, 111, 148-154.http://dx.doi.org/10.1016/S0169-328X(03)00003-2
- Skeldal, S., Matusica, D., Nykjaer, A. and Coulson, E.J. (2011) Proteolytic Processing of the p75 Neurotrophin Receptor: A Prerequisite for Signalling? Neuronal Life, Growth and Death Signalling Are Crucially Regulated by Intra-Membrane Proteolysis and Trafficking of p75NTR. BioEssays, 33, 614-625.http://dx.doi.org/10.1002/bies.201100036
- Woo, N.H., Teng, H.K., Siao, C.J., Chiaruttini, C., Pang, P.T., Milner, T.A., Hempstead, B.L. and Lu, B. (2005) Activation of p75NTR by ProBDNF Facilitates Hippocampal Long-Term Depression. Nature Neuroscience, 8, 1069-1077.http://dx.doi.org/10.1038/nn1510
- Nagappan, G., Zaitsev, E., Senatorov, V.V., Yang, J., Hempstead, B.L. and Lu, B. (2009) Control of Extracellular Cleavage of ProBDNF by High Frequency Neuronal Activity. Proceedings of the National Academy of Sciences of the United States of America, 106, 1267-1272. http://dx.doi.org/10.1073/pnas.0807322106
- Lessmann, V. and Brigadski, T. (2009) Mechanisms, Locations, and Kinetics of Synaptic BDNF Secretion: An Update. Neuroscience Research, 65, 11-22. http://dx.doi.org/10.1016/j.neures.2009.06.004
- Holm, M.M., Nieto-Gonzalez, J.L., Vardya, I., Vaegter, C.B., Nykjaer, A. and Jensen, K. (2009) Mature BDNF, but Not ProBDNF, Reduces Excitability of Fast-Spiking Interneurons in Mouse Dentate Gyrus. The Journal of Neuroscience, 29, 12412-12418. http://dx.doi.org/10.1523/JNEUROSCI.2978-09.2009
- Chevaleyre, V. and Castillo, P.E. (2003) Heterosy-naptic LTD of Hippocampal GABAergic Synapses: A Novel Role of Endocannabinoids in Regulating Excitability. Neuron, 38, 461-472. http://dx.doi.org/10.1016/S0896-6273(03)00235-6
- Patterson, S.L., Abel, T., Deuel, T.A., Martin, K.C., Rose, J.C. and Kandel, E.R. (1996) Recombinant BDNF Rescues Deficits in Basal Synaptic Transmission and Hippocampal LTP in BDNF Knockout Mice. Neuron, 16, 1137-1145.http://dx.doi.org/10.1016/S0896-6273(00)80140-3
- Korte, M., Carroll, P., Wolf, E., Brem, G., Thoenen, H. and Bonhoeffer, T. (1995) Hippocampal Long-Term Potentiation Is Impaired in Mice Lacking Brain-Derived Neurotrophic Factor. Proceedings of the National Academy of Sciences of the United States of America, 92, 8856-8860. http://dx.doi.org/10.1073/pnas.92.19.8856
- Figurov, A., Pozzo-Miller, L.D., Olafsson, P., Wang, T. and Lu, B. (1996) Regulation of Synaptic Responses to High-Frequency Stimulation and LTP by Neurotrophins in the Hippocampus. Nature, 381, 706-709.http://dx.doi.org/10.1038/381706a0
- Gottschalk, W.A., Jiang, H., Tartaglia, N., Feng, L., Figurov, A. and Lu, B. (1999) Signaling Mechanisms Mediating BDNF Modulation of Synaptic Plasticity in the Hippocampus. Learning & Memory, 6, 243-256.
- Jovanovic, J.N., Czernik, A.J., Fienberg, A.A., Greengard, P. and Sihra, T.S. (2000) Synapsins as Mediators of BDNF-Enhanced Neurotransmitter Release. Nature Neuroscience, 3, 323-329. http://dx.doi.org/10.1038/73888
- Pozzo-Miller, L.D., Gottschalk, W., Zhang, L., McDermott, K., Du, J., Gopalakrishnan, R., Oho, C., Sheng, Z.H. and Lu, B. (1999) Impairments in High-Frequency Transmission, Synaptic Vesicle Docking, and Synaptic Protein Distribution in the Hippocampus of BDNF Knockout Mice. The Journal of Neuroscience, 19, 4972-4983.
- Chen, G., Kolbeck, R., Barde, Y.A., Bonhoeffer, T. and Kossel, A. (1999) Relative Contribution of Endogenous Neurotrophins in Hippocampal Long-Term Potentiation. The Journal of Neuroscience, 19, 7983-7990.
- Kang, H., Welcher, A.A., Shelton, D. and Schuman, E.M. (1997) Neurotrophins and Time: Different Roles for TrkB Signaling in Hippocampal Long-Term Potentiation. Neuron, 19, 653-664.http://dx.doi.org/10.1016/S0896-6273(00)80378-5
- Kovalchuk, Y., Hanse, E., Kafitz, K.W. and Konnerth, A. (2002) Postsynaptic Induction of BDNF-Mediated Long-Term Potentiation. Science, 295, 1729-1734. http://dx.doi.org/10.1126/science.1067766
- Levine, E.S., Crozier, R.A., Black, I.B. and Plummer, M.R. (1998) Brain-Derived Neurotrophic Factor Modulates Hippocampal Synaptic Transmission by Increasing N-Methyl-D-Aspartic Acid Receptor Activity. Proceedings of the National Academy of Sciences of the United States of America, 95, 10235-10239.http://dx.doi.org/10.1073/pnas.95.17.10235
- Korte, M., Kang, H., Bonhoeffer, T. and Schuman, E. (1998) A Role for BDNF in the Late-Phase of Hippocampal Long-Term Potentiation. Neuropharmacology, 37, 553-559. http://dx.doi.org/10.1016/S0028-3908(98)00035-5
- Pang, P.T., Teng, H.K., Zaitsev, E., Woo, N.T., Sakata, K., Zhen, S., Teng, K.K., Yung, W.H., Hempstead, B.L. and Lu, B. (2004) Cleavage of ProBDNF by tPA/Plasmin Is Essential for Long-Term Hippocampal Plasticity. Science, 306, 487-491. http://dx.doi.org/10.1126/science.1100135
- Minichiello, L., Calella, A.M., Medina, D.L., Bonhoeffer, T., Klein, R. and Korte, M. (2002) Mechanism of TrkB-Mediated Hippocampal Long-Term Potentiation. Neuron, 36, 121-137.http://dx.doi.org/10.1016/S0896-6273(02)00942-X
- Patterson, S.L., Grover, L.M., Schwartzkroin, P.A. and Bothwell, M. (1992) Neurotrophin Expression in Rat Hippocampal Slices: A Stimulus Paradigm Inducing LTP in CA1 Evokes Increases in BDNF and NT-3 mRNAs. Neuron, 9, 1081-1088. http://dx.doi.org/10.1016/0896-6273(92)90067-N
- Castrén, E., Pitkänen, M., Sirviö, J., Parsadanian, A., Lindholm, D., Thoenen, H. and Riekkinen, P.J. (1993) The Induction of LTP Increases BDNF and NGF mRNA but Decreases NT-3 mRNA in the Dentate Gyrus. NeuroReport, 4, 895-898. http://dx.doi.org/10.1097/00001756-199307000-00014
- Silhol, M., Arancibia, S., Maurice, T. and Tapia-Arancibia, L. (2007) Spatial Memory Training Modifies the Expression of Brain-Derived Neurotrophic Factor Tyrosine Kinase Receptors in Young and Aged Rats. Neuroscience, 146, 962-973. http://dx.doi.org/10.1016/j.neuroscience.2007.02.013
- Tongiorgi, E., Righi, M. and Cattaneo, A. (1997) Activity-Dependent Dendritic Targeting of BDNF and TrkB mRNAs in Hippocampal Neurons. Journal of Neuroscience, 17, 9492-9505.
- Simonato, M., Bregola, G., Armellin, M., Del Piccolo, P., Rodi, D., Zucchini, S. and Tongiorgi, E. (2002) Dendritic Targeting of mRNAs for Plasticity Genes in Experimental Models of Temporal Lobe Epilepsy. Epilepsia, 43, 153-158. http://dx.doi.org/10.1046/j.1528-1157.43.s.5.32.x
- Chiaruttini, C., Sonego, M., Baj, G., Simonato, M. and Tongiorgi, E. (2008) BDNF mRNA Splice Variants Display Activity-Dependent Targeting to Distinct Hippocampal Laminae. Molecular and Cellular Neuroscience, 37, 11-19.http://dx.doi.org/10.1016/j.mcn.2007.08.011
- Kang, H. and Schuman, E.M. (1995) Long-Lasting Neurotrophin-Induced Enhancement of Synaptic Transmission in the Adult Hippocampus. Science, 267, 1658-1662. http://dx.doi.org/10.1126/science.7886457
- Kang, H. and Schuman, E.M. (1996) A Requirement for Local Protein Synthesis in Neurotrophin-Induced Hippocampal Synaptic Plasticity. Science, 273, 1402-1406. http://dx.doi.org/10.1126/science.273.5280.1402
- Aakalu, G., Smith, W.B., Nguyen, N., Jiang, C. and Schuman, E.M. (2001) Dynamic Visualization of Local Protein Synthesis in Hippocampal Neurons. Neuron, 30, 489-502. http://dx.doi.org/10.1016/S0896-6273(01)00295-1
- Schratt, G.M., Nigh, E.A., Chen, W.G., Hu, L. and Greenberg, M.E. (2004) BDNF Regulates the Translation of a Select Group of mRNAs by a Mammalian Target of Rapamycin-Phosphatidylinositol 3-Kinase-Dependent Pathway during Neuronal Development. The Journal of Neuroscience, 24, 7366-7377.http://dx.doi.org/10.1523/JNEUROSCI.1739-04.2004
- Schratt, G.M., Tuebing, F., Nigh, E.A., Kane, C.G., Sabatini, M.E., Kiebler, M. and Greenberg, M.E. (2006) A Brain-Specific MicroRNA Regulates Dendritic Spine Development. Nature, 439, 283-289.http://dx.doi.org/10.1038/nature04367
- Takei, N., Inamura, N., Kawamura, M., Namba, H., Hara, K., Yonezawa, K. and Nawa, H. (2004) Brain-Derived Neurotrophic Factor induces Mammalian Target of Rapamycin-Dependent Local Activation of Translation Machinery and Protein Synthesis in Neuronal Dendrites. The Journal of Neuroscience, 24, 9760-9769.http://dx.doi.org/10.1523/JNEUROSCI.1427-04.2004
- Yin, Y., Edelman, G.M. and Vanderklish, P.W. (2002) The Brain-Derived Neurotrophic Factor Enhances Synthesis of Arc in Synaptoneurosomes. Proceedings of the National Academy of Sciences of the United States of America, 99, 2368-2373. http://dx.doi.org/10.1073/pnas.042693699
- Timmusk, T., Palm, K., Metsis, M., Reintam, T., Paalme, V., Saarma, M. and Persson, H. (1993) Multiple Promoters Direct Tissue-Specific Expression of the Rat BDNF Gene. Neuron, 10, 475-489.http://dx.doi.org/10.1016/0896-6273(93)90335-O
- Aid, T., Kazantseva, A., Piirsoo, M., Palm, K. and Timmusk, T. (2007) Mouse and Rat BDNF Gene Structure and Expression Revisited. The Journal of Neuroscience Research, 85, 525-535. http://dx.doi.org/10.1002/jnr.21139
- Pruunsild, P., Kazantseva, A., Aid, T., Palm, K. and Timmusk, T. (2007) Dissecting the Human BDNF Locus: Bidirectional Transcription, Complex Splicing, and Multiple Promoters. Genomics, 90, 397-406.http://dx.doi.org/10.1016/j.ygeno.2007.05.004
- Tao, X., West, A.E., Chen, W.G., Corfas, G. and Greenberg, M.E. (2002) A Calcium-Responsive Transcription Factor, CaRF, That Regulates Neuronal Activity-Dependent Expression of BDNF. Neuron, 33, 383-395.http://dx.doi.org/10.1016/S0896-6273(01)00561-X
- Metsis, M., Timmusk, T., Arenas, E. and Persson, H. (1993) Differential Usage of Multiple Brain-Derived Neurotrophic Factor Promoters in the Rat Brain Following Neuronal Activation. Proceedings of the National Academy of Sciences of the United States of America, 90, 8802-8806. http://dx.doi.org/10.1073/pnas.90.19.8802
- Hong, E.J., McCord, A.E. and Greenberg, M.E. (2008) A Biological Function for the Neuronal Activity-Dependent Component of Bdnf Transcription in the Development of Cortical Inhibition. Neuron, 60, 610-624.http://dx.doi.org/10.1016/j.neuron.2008.09.024
- Sakata, K., Woo, N.H., Martinowich, K., Greene, J.S., Schloesser, R.J., Shen, L. and Lu, B. (2009) Critical Role of Promoter IV-Driven BDNF Transcription in GABAergic Transmission and Synaptic Plasticity in the Prefrontal Cortex. Proceedings of the National Academy of Sciences of the United States of America, 106, 5942-5947.http://dx.doi.org/10.1073/pnas.0811431106
- Pruunsild, P., Sepp, M., Orav, E., Koppel, I. and Timmusk, T. (2011) Identification of Cis-Elements and Transcription Factors Regulating Neuronal Activity-Dependent Transcription of Human BDNF Gene. The Journal of Neuroscience, 31, 3295-3308. http://dx.doi.org/10.1523/JNEUROSCI.4540-10.2011
- An, J.J., Gharami, K., Liao, G.Y., Woo, N.H., Lau, A.G., Vanevski, F., Torre, E.R., Jones, K.R., Feng, Y., Lu, B. and Xu, B. (2008) Distinct Role of Long 3’UTR BDNF mRNA in Spine Morphology and Synaptic Plasticity in Hippocampal Neurons. Cell, 134, 175-187. http://dx.doi.org/10.1016/j.cell.2008.05.045
- Baj, G., Leone, E., Chao, M.V. and Tongiorgi, E. (2011) Spatial Segregation of BDNF Transcripts Enables BDNF to Differentially Shape Distinct Dendritic Compartments. Proceedings of the National Academy of Sciences of the United States of America, 108, 16813-16818.
- Mahmoudi, S., Henriksson, S., Corcoran, M., Méndez-Vidal, C., Wiman, K.G. and Farnebo, M. (2009) Wrap53, A Natural p53 Antisense Transcript Required for p53 Induction upon DNA Damage. Molecular Cell, 33, 462-471.
- Faghihi, M.A., Modarresi, F., Khalil, A.M., Wood, D.E., Sahagan, B.G., Morgan, T.E., Finch, C.E., St Laurent 3rd., G., Kenny, P.J. and Wahlestedt, C. (2008) Expression of a Noncoding RNA Is Elevated in Alzheimer’s Disease and Drives Rapid Feed-Forward Regulation of Beta-Secretase. Nature Medicine, 14, 723-730.
- Modarresi, F., Faghihi, M.A., Lopez-Toledano, M.A., Fatemi, R.P., Magistri, M., Brothers, S.P., Van der Brug, M.P. and Wahlestedt, C. (2012) Inhibition of Natural Antisense Transcripts in Vivo Results in Gene-Specific Transcriptional Upregulation. Nature Biotechnology, 30, 453-459. http://dx.doi.org/10.1038/nbt.2158
- Peter, C.J. and Akbarian, S. (2011) Balancing Histone Methylation Activities in Psychiatric Disorders. Trends in Molecular Medicine, 17, 372-379. http://dx.doi.org/10.1016/j.molmed.2011.02.003
- Barker, P.A. (2009) Whither ProBDNF? Nature Neuroscience, 12, 105-106. http://dx.doi.org/10.1038/nn0209-105
- Greenberg, M.E., Xu, B., Lu, B. and Hempstead, B.L. (2009) New Insights in the Biology of BDNF Synthesis and Release: Implications in CNS Function. The Journal of Neuroscience, 29, 12764-12767.http://dx.doi.org/10.1523/JNEUROSCI.3566-09.2009
- Matsumoto, T., Rauskolb, S., Polack, M., Klose, J., Kolbeck, R., Korte, M. and Barde, Y.A. (2008) Biosynthesis and Processing of Endogenous BDNF: CNS Neurons Store and Secrete BDNF, Not Pro-BDNF. Nature Neuroscience, 11, 131-133. http://dx.doi.org/10.1038/nn2038
- Yang, F., Je, H.S., Ji, Y., Nagappan, G., Hempstead, B. and Lu, B. (2009) Pro-BDNF-Induced Synaptic Depression and Retraction at Developing Neuromuscular Synapses. The Journal of Cell Biology, 185, 727-741.http://dx.doi.org/10.1083/jcb.200811147
- Kohara, K., Kitamura, A., Morishima, M. and Tsumoto, T. (2001) Activity-Dependent Transfer of Brain-Derived Neurotrophic Factor to Postsynaptic Neurons. Science, 291, 2419-2423. http://dx.doi.org/10.1126/science.1057415
- Suter, U., Heymach, J.V. and Shooter, E.M. (1991) Two Conserved Domains in the NGF Propeptide Are Necessary and Sufficient for the Biosynthesis of Correctly Processed and Biologically Active NGF. The EMBO Journal, 10, 2395-2400.
- Rattenholl, A., Ruoppolo, M., Flagiello, A., Monti, M., Vinci, F., Marino, G., Lilie, H., Schwarz, E. and Rudolph, R. (2001) Pro-Sequence Assisted Folding and Disulfide Bond Formation of Human Nerve Growth Factor. Journal of Molecular Biology, 305, 523-533. http://dx.doi.org/10.1006/jmbi.2000.4295
- Chen, Z.Y., Patel, P.D., Sant, G., Meng, C.X., Teng, K.K., Hempstead, B.L. and Lee, F.S. (2004) Variant Brain-Derived Neurotrophic Factor (BDNF) (Met66) Alters the Intracellular Trafficking and Activity-Dependent Secretion of Wild-Type BDNF in Neurosecretory Cells and Cortical Neurons. The Journal of Neuroscience, 24, 4401-4411.http://dx.doi.org/10.1523/JNEUROSCI.0348-04.2004
- Lou, H., Kim, S.K., Zaitsev, E., Snell, C.R., Lu, B. and Loh, Y.P. (2005) Sorting and Activity-Dependent Secretion of BDNF Require Interaction of a Specific Motif with the Sorting Receptor Carboxypeptidase. Neuron, 45, 245-255.http://dx.doi.org/10.1016/j.neuron.2004.12.037
- Chen, Z.Y., Ieraci, A., Teng, H., Dall, H., Meng, C.X., Herrera, D.G., Nykjaer, A., Hempstead, B.L. and Lee, F.S. (2005) Sortilin Controls Intracellular Sorting of Brain-Derived Neurotrophic Factor to the Regulated Secretory Pathway. The Journal of Neuroscience, 25, 6156-6166. http://dx.doi.org/10.1523/JNEUROSCI.1017-05.2005
- Rattenholl, A., Lilie, H., Grossmann, A., Stern, A., Schwarz, E. and Rudolph, R. (2001) The Pro-Sequence Facilitates Folding of Human Nerve Growth Factor from Escherichia Coli Inclusion Bodies. European Journal of Biochemistry, 268, 3296-3303. http://dx.doi.org/10.1046/j.1432-1327.2001.02232.x
- Toyooka, K., Asama, K., Watanabe, Y., Muratake, T., Takahashi, M., Someya, T. and Nawa, H. (2002) Decreased Levels of Brain-Derived Neurotrophic Factor in Serum of Chronic Schizophrenic Patients. Psychiatry Research, 110, 249-257. http://dx.doi.org/10.1016/S0165-1781(02)00127-0
- Karege, F., Perret, G., Bondolfi, G., Schwald, M., Bertschy, G. and Aubry, J.M. (2002) Decreased Serum Brain-Derived Neurotrophic Factor Levels in Major Depressed Patients. Psychiatry Research, 109, 143-148.http://dx.doi.org/10.1016/S0165-1781(02)00005-7
- Shimizu, E., Hashimoto, K., Okamura, N., Koike, K., Komatsu, N., Kumakiri, C., Nakazato, M., Watanabe, H., Shinoda, N., Okada, S. and Iyo, M. (2003) Alterations of Serum Levels of Brain-Derived Neurotrophic Factor (BDNF) in Depressed Patients with or without Antidepressants. Biological Psychiatry, 54, 70-75.http://dx.doi.org/10.1016/S0006-3223(03)00181-1
- Sen, S., Duman, R. and Sanacora, G. (2008) Serum Brain-Derived Neurotrophic Factor, Depression, and Antidepressant Medications: Meta-Analyses and Implications. Biological Psychiatry, 64, 527-532.http://dx.doi.org/10.1016/j.biopsych.2008.05.005
- Matsuoka, Y., Nishi, D., Noguchi, H., Kim, Y. and Hashimoto, K. (2013) Longitudinal Changes in Serum Brain-Derived Neurotrophic Factor in Accident Survivors with Posttraumatic Stress Disorder. Neuropsychobiology, 68, 44-50. http://dx.doi.org/10.1159/000350950
- Dell’Osso, L., Carmassi, C., Del Debbio, A., Catena Dell’Osso, M., Bianchi, C., Da Pozzo, E., Origlia, N., Domenici, L., Massimetti, G., Marazziti, D. and Piccinni, A. (2009) Brain-Derived Neurotrophic Factor Plasma Levels in Patients Suffering from Post-Traumatic Stress Disorder. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 33, 899-902. http://dx.doi.org/10.1016/j.pnpbp.2009.04.018
- Fujimura, H., Altar, C.A., Chen, R., Nakamura, T., Nakahashi, T., Kambayashi, J., Sun, B. and Tandon, N.N. (2002) Brain-Derived Neurotrophic Factor Is Stored in Human Platelets and Released by Agonist Stimulation. Thrombosis and Haemostasis, 87, 728-734.
- Lee, B.H. and Kim, Y.K. (2009) Reduced Platelet BDNF Level in Patients with Major Depression. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 33, 849-853.
- Molendijk, M.L., Haffmans, J.P., Bus, B.A., Spinhoven, P., Penninx, B.W., Prickaerts, J., Oude Voshaar, R.C. and Elzinga, B.M. (2012) Serum BDNF Concentrations Show Strong Seasonal Variation and Correlations with the Amount of Ambient Sunlight. PLoS One, 7, 48046. http://dx.doi.org/10.1371/journal.pone.0048046
NOTES

*Corresponding authors.

