American Journal of Analytical Chemistry
Vol.06 No.02(2015), Article ID:53393,8 pages
10.4236/ajac.2015.62011
Kinetics and Vapor Pressure Studies of bis(N-alkyl-2-hydroxonapthaldimine)nickel (II) (N-R = methyl to pentyl) Complexes
Maria Francis George Johnson1, Thevabakthi Siluvai Muthu Arul Jeevan2*, Sebastian Arockiasamy3, Karachalacheruvu Seetharamaiah Nagaraja1
1Department of Chemistry, Loyola Institute of Frontier Energy (LIFE), Loyola College, Chennai, India
2Department of Chemistry, College of Natural and Computational Sciences, University of Gondar, Gondar, Ethiopia
3Department of Chemistry, School of Advanced Sciences (SAS), VIT University Chennai Campus, Chennai, India
Email: *jejeevan@gmail.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 29 December 2014; accepted 16 January 2015; published 20 January 2015
ABSTRACT
The complexes of bis[N-alkyl-2-hydroxonapthaldimine]nickel(II) (N-alkyl = methyl, ethyl, propyl, butyl or pentyl) were synthesized and their volatilization in N2 atmosphere was demonstrated by the TG-based transpiration technique. The equilibrium vapor pressure of the complexes over a temperature span of 470 - 590 K was determined by adapting a horizontal dual arm single furnace thermoanalyser as a transpiration apparatus. It yielded 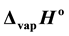 as 153.1 (±1.9), 122.9 (±0.3), 147.6 (±10.7), 151.8 (±10.9) and 114.7 (±5.3) k∙Jmol−1 respectively. The entropies of vaporization
as 153.1 (±1.9), 122.9 (±0.3), 147.6 (±10.7), 151.8 (±10.9) and 114.7 (±5.3) k∙Jmol−1 respectively. The entropies of vaporization 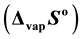 for these complexes as calculated from the intercept of the linear fit expressions were found to be 319.7 (±3.9), 229.9 (±5.8), 317.8 (±17.2), 319.7 (±19.1) and 254.6 (±9.6) Jmol−1∙K−1 respectively. The non-isothermal vaporization activation energy was determined from Arrhenius and Coats-Redfern methods.
for these complexes as calculated from the intercept of the linear fit expressions were found to be 319.7 (±3.9), 229.9 (±5.8), 317.8 (±17.2), 319.7 (±19.1) and 254.6 (±9.6) Jmol−1∙K−1 respectively. The non-isothermal vaporization activation energy was determined from Arrhenius and Coats-Redfern methods.
Keywords:
Volatile Nickel Complexes, Thermal Properties, Vapor Pressure, Transpiration Technique, Enthalpy and Entropy of Vaporization, Vaporization Kinetics

1. Introduction
Success of the metallo-organic chemical vapor deposition (MOCVD) technique for preparing thin films rests mainly on the availability of completely volatile precursors. Therefore, development of stable, non-toxic and completely volatile solid metallo-organic precursors for use in CVD of metallic nickel has attracted intensive research [1] -[7] . Many publications warned the use of highly toxic Ni(CO)4 as a precursor [8] [9] , which was first used by Mond in 1885. Other precursors used in MOCVD of Ni films are Ni(acac)2en [1] , Ni(tfacim)2 (tfacim = triflouroacetylacetone-imine) [3] [4] , Ni(L)2 [L= dimethylglyoxime [10] , diethylglyoxime, dipropylglyoxime [6] ], Ni(η5-C5H5)2 [7] , Ni(L)2 [L = acetylacetone (acac) [11] , hexafluoroacetylacetone (hfac) [12] and tetramethylheptanedione (tmhd) [13] ]. Ni(tmhd)2 [14] and Ni[(acac)2en] [1] met the requirements of an ideal precursors in CVD applications. It is essential to synthesize new precursors with mixed O and N environment around Ni employing aromatic ligands for better volatility. This paper describes the synthesis and characterization of nickel complexes by using Schiff base ligands. These complexes are characterized by elemental analyses, FT-IR, FABMS, TG/DTA, vaporization kinetics and vapor pressure measurement by the TG-based transpiration technique.
2. Experimental
NiCl2∙6H2O, 2-hydroxynaphthal (Aldrich), methylamine, ethylamine, propylamine, butylamine, pentylamine (Loba Chemie, India), liquor ammonia and methanol (S. D. Fine, India) were used as procured.
2.1. Synthesis of the Complexes
2.1.1. Synthesis of bis(2-hydroxonapthaldehydato)nickel(II) [Ni(2-hydroxynapthal)2]
NiCl2∙6H2O (9.48 g) was dissolved in water (20 cm3) to which 2-hydroxynaphthal (10 cm3) was added drop- wise under constant stirring. To this reaction mixture, liquor ammonia (12 cm3) was added drop-wise by continuously monitoring the formation of a greenish yellow precipitate. This precipitate was digested at 333 K over a water bath for 0.5 h, which was filtered, washed with ethanol and dried under vacuum [15] . Yield: 76%.
2.1.2. Syntheses of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) complexes [N-alkyl = methyl to pentyl]
The syntheses of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II), where N-alkyl = methyl to pentyl complexes were carried out by treating the appropriate primary amines with the hot alcoholic suspension of Ni(2-hydrox- onapthal)2. The resulting olive green solution was refluxed for about 0.5 h and the crystals formed were filtered, washed with ethanol and dried under vacuum. The compound was recrystallized from methanol. Yield: 56% - 82%.
2.2. Characterization
The C, H and N analyses were performed on a CARBO-ERBA-11008 rapid elemental analyzer. IR spectra were recorded as KBr pellets on a Perkin-Elmer FT-IR spectrometer (RX1, FT-IR) in the region of 4000 - 450 cm−1. Fast atom bombardment mass spectra (FABMS) of the nickel complexes were recorded by employing a JEOL SX 102/DA-6000 spectrometer using argon as the FAB gas with an accelerating voltage of 10 kV and the spectra were recorded at room temperature. M-nitrobenzyl alcohol (NBA) was used as the matrix unless specified otherwise. The thermal analyses were carried out using Perkin-Elmer, Pyris Diamond TG/DTA at a linear heating rate of 0.17 Ks−1. High purity nitrogen (purity > 99.99%) dried by passing through refrigerated molecular sieves (Linde 4A) was used as the purge gas at a flow rate of 12 dm3∙h−1.
2.3. Vapor Pressure Measurement Studies
A horizontal thermal analyzer was adapted as a transpiration setup for vapor pressure measurements. The configuration of the horizontal dual arm with a single narrow furnace chamber minimizes errors arising from convection, buoyancy, thermo molecular and electrostatic charge effects. The arms of the thermo balance served as the temperature-cum-DTA sensors.
The block diagram of the thermal analyzer, modification for its functioning in the transpiration mode, including precise flow calibration for the carrier gas using a capillary glass flow meter and corrections for apparent weight losses in isothermal mode were the same as reported [15] [16] . The choice of 6 dm3∙h−1 for N2 gas was made for the isothermal equilibrium vaporization on the basis of the existence of a plateau in the plot of the equilibrium vapor pressure 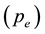 against the flow rate. The vapor pressure measurements were carried out by rapid heating 0.17 Ks−1 and after allowing for temperature stabilization, subsequent changes in isothermal steps were done at a heating rate of 0.03 Ks−1. It turned out that the vapor pressure
against the flow rate. The vapor pressure measurements were carried out by rapid heating 0.17 Ks−1 and after allowing for temperature stabilization, subsequent changes in isothermal steps were done at a heating rate of 0.03 Ks−1. It turned out that the vapor pressure 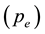 derived from the TG-based transpiration method was reliable to 10% accuracy [15] [16] .
derived from the TG-based transpiration method was reliable to 10% accuracy [15] [16] .
2.4. Vaporization Kinetics
Experiments were performed under non-isothermal conditions at a programmed linear heating rate of 10˚C∙min−1 for methyl to pentyl homologues. Among the various methods for the kinetics evaluation of TG weight loss data, Arrhenius method was followed in the present investigation to study the vaporization kinetics.
3. Results and Discussion
3.1. Thermal Analyses
The elemental analyses and their composition are presented in Table 1. The melting point endotherms (Figure 1) were calculated accurately using the built-in Pyris software and the values 280˚C, 271˚C, 205˚C, 176˚C and 175˚C for methyl to pentyl homologues respectively which exhibited a decreasing trend with increasing length of the alkyl chain. The non-isothermal TG curves (Figure 2) of bis(2-hydroxonapthaldehydato)nickel(II) indicated a 37.8% residue at 700 K making it unsuitable for CVD applications. The TG curves of bis(N-alkyl-2-hy- droxonapthaldimine)nickel(II) homologues (where N-alkyl = methyl to pentyl) showed a single step weight loss commencing from 700 K, leading to a negligible amount of residue qualifying themselves as precursors for CVD.
3.2. Spectral Characterization
The relevant frequencies such as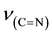 ,
, 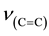 ,
, 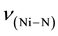
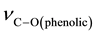 and
and 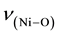 (Table 2) support the struc-
(Table 2) support the struc-
ture. The FABMS patterns (Table 3) of bis[N-alkyl-2-hydroxonapthaldimine)]nickel(II) (where N-alkyl = methyl to pentyl) complexes revealed the molecular masses (M) of the complexes and their fragmentation pattern. The values of molecular ion peaks were observed at 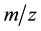 (Da) = 430, 454, 483, 510 and 539 respectively, for methyl to pentyl homologues as clusters due to the isotopic abundances of natural nickel (58Ni: 67.76%, 60Ni: 26.16%, 61Ni: 1.25%, 62Ni: 3.66%, 64Ni: 1.16%).
(Da) = 430, 454, 483, 510 and 539 respectively, for methyl to pentyl homologues as clusters due to the isotopic abundances of natural nickel (58Ni: 67.76%, 60Ni: 26.16%, 61Ni: 1.25%, 62Ni: 3.66%, 64Ni: 1.16%).
3.3. Vapor Pressure Measurements
Effective control of the process of any type of CVD and to monitor the composition, thickness and microstructure of thin films, relevant information on precursor chemistry, possible fragmentation pattern by the decomposition of precursors and vapor pressures are needed. Therefore vapor pressure measurement was deemed as essential for these complexes to be used for MOCVD.
If W is the mass loss of the sample (Table 4) at the isothermal temperature caused by the flow of 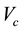 dm3 of the carrier gas (measured at
dm3 of the carrier gas (measured at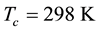 ), the vapor pressure
), the vapor pressure 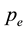 could be calculated using Dalton’s law of partial pressure for ideal gas mixtures as
could be calculated using Dalton’s law of partial pressure for ideal gas mixtures as
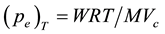 (1)
(1)
where 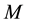 is the molar mass of the complexes,
is the molar mass of the complexes, 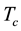 and
and 







Table 1. Elemental analyses and melting points of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) complexes.
Table 2. Relevant IR frequencies of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II).
Table 3. Assignment of peaks in FAB mass spectra of bis(N-alkyl-(2-hydroxonapthal)nickel(II) complexes; N-alkyl = methyl (a) to pentyl (e) respectively.
Table 4. Mass loss (for 60 min) and for equilibrium solid vaporization of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) homologues by TG-based transpiration method.
Table 5. Clausius-Clapeyron parameters for bis(N-alkyl-2-hydroxonapthaldimine)nickel(II).
Figure 1. DTA traces of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II).
Figure 2. Non-isothermal TG curves of nickel complexes.
Figure 3. Clausius-Clapeyron plots for bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) homologues.
Enthalpy of vaporization is obtained by multiplying the slope 
respectively. The entropies of vaporization 
linear fit expression were found to be 319.7 (±3.9), 229.9 (±5.8), 317.8 (±17.2), 319.7 (±19.1) and 254.6 (±9.6) Jmol−1∙K−1. The vapor pressure of the complexes will be helpful for fixing the metal organic chemical vapor deposition (MOCVD) process parameters for getting the desired phase and rate of deposition of nickel and composite materials.
3.4. Determination of Activation Energy
The rate constant 


where 



where 




And the plot of 

The kinetics of the complexes was followed by employing the Coats-Redfern approximation which gives the expression (6).

A plot of 

tion is used in the equation. The 
Figure 4. Arrhenius plots of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II).
Figure 5. Plots of 



Table 6. Activation energies obtained using the Coats-Redfern method for several solid state processes at heating rate of 10˚C∙min−1 for methyl to pentyl.
4. Conclusion
The thermodynamic and kinetic decomposition of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) (N-alkyl = me- thyl, ethyl, propyl, butyl or pentyl) complexes were carried out. The molecular masses of the homologous series of the nickel complexes were obtained from FABMS. The TG-based transpiration technique was used to eva- luate the vapor pressure data of bis(N-alkyl-2-hydroxonapthaldimine)nickel(II) homologues. The standard en- thalpies of vaporization and entropies of vaporization of the complexes have been evaluated. The non-isothermal vaporization activation energy values were determined by Arrhenius and Coats-Redfern methods.
Acknowledgements
This work received financial support from the Department of Science and Technology (DST), India, through Grant No. SR/S3/ME/03/2005-SERC-Engg. The authors thank the Central Drug Research Institute, Lucknow, for recording the mass spectra and C, H and N analyses.
References
- Premkumar, P.A., Dasgupta, A., Kuppusami, P., Parameswaran, P., Mallika, C., Nagaraja, K.S. and Raghunathan, V.S. (2006) Synthesis and Characterization of Ni and Ni/CrN Nanocomposite Coatings by Plasma Assisted Metal-Organic CVD. Chemical Vapor Deposition, 12, 39-45. http://dx.doi.org/10.1002/cvde.200506415
- Hunde, E.T. and Watkins, J.J. (2004) Reactive Deposition of Cobalt and Nickel Films from Their Metallocenes in Supercritical Carbon Dioxide Solution. Chemistry of Materials, 16, 498-503. http://dx.doi.org/10.1021/cm034433n
- Bakovets, V.V., Mitkin, V.N. and Gelfond, N.V. (2005) Mechanism of Ni Film CVD with a Ni(Ktfaa)2 Precursor on a Silicon Substrate. Chemical Vapor Deposition, 11, 368-374. http://dx.doi.org/10.1002/cvde.200506376
- Bakovets, V.V., Mitkin, V.N. and Gelfond, N.V. (2005) Mechanism of Ni Film CVD with a Ni(ktfaa)2 Precursor on a Copper Substrate. Chemical Vapor Deposition, 11, 112-117. http://dx.doi.org/10.1002/cvde.200406317
- Zhou, M., Lin, W.Y., de Tacconi, N.R. and Rajeshwar, K. (1996) Metal/Semiconductor Electrocomposite Photoelectrodes: Behavior of Ni/TiO2 Photoanodes and Comparison of Photoactivity of Anatase and Rutile Modifications. Journal of Electroanalytical Chemistry, 402, 221-224. http://dx.doi.org/10.1016/0022-0728(95)04368-3
- Brissonneau, L. and Vahlas, C. (1999) MOCVD-Processed Ni Films from Nickelocene. Part I: Growth Rate and Morpho- logy. Chemical Vapor Deposition, 5, 135-142. http://dx.doi.org/10.1002/(SICI)1521-3862(199908)5:4<135::AID-CVDE135>3.0.CO;2-1
- Stauf, G., Driscoll, D., Dowben, P., Barfuss, S. and Grade, M. (1987) Iron and Nickel Thin Film Deposition via Metallocene Decomposition. Thin Solid Films, 153, 421-430. http://dx.doi.org/10.1016/0040-6090(87)90202-1
- Kang, J.K. and Ree, S.W. (2000) Metalorganic Chemical Vapor Deposition of Nickel Films from Ni(C5H5)2/H2. Journal of Materials Research, 15, 1828-1833. http://dx.doi.org/10.1557/JMR.2000.0264
- Maruyama, T. and Tago, T. (1993) Nickel Thin Films Prepared by Chemical Vapor Deposition from Nickel Acetylacetonate. Journal of Materials Science, 28, 5345-5348. http://dx.doi.org/10.1007/BF00570088
- Becht, M., Gallus, J., Hunziker, M., Atamny, F. and Dahmen, K.H. (1995) Nickel Thin Films Grown by MOCVD Using Ni(dmg)2 as Precursor. Journal de Physique IV, C5, 465-472.
- Hemert, R.V., Spenlove, L. and Sievers, S. (1965) Vapor Deposition of Metals by Hydrogen Reduction of Metal Chelates. Journal of the Electrochemical Society, 112, 1123-1126. http://dx.doi.org/10.1149/1.2423376
- Lane, P.A., Crosbie, M.J., Wright, P.J., Donohue, P.P., Hirst, P.J., Reeves, C.L., Anthony, C.J., Jones, A.C., Todd, M.A. and Williams, D.J. (2003) The Metal-Organic Chemical Vapor Deposition of Lanthanum Nickelate Electrodes for Use in Ferroelectric Devices. Chemical Vapor Deposition, 9, 87-92. http://dx.doi.org/10.1002/cvde.200390007
- Anthony, C.J. (1998) MOCVD of Electroceramic Oxides: A Precursor Manufacturer’s Perspective. Chemical Vapor Deposition, 4, 169-179. http://dx.doi.org/10.1002/(SICI)1521-3862(199810)04:05<169::AID-CVDE169>3.3.CO;2-Y
- Wood, J.L. and Jones, M. (1963) Heats of Formation and Coordinate Bond Energies of Some Nickel(II) Chelates. Jour- nal of Physical Chemistry, 67, 1049-1051. http://dx.doi.org/10.1021/j100799a024
- Pankajavalli, R., Mallika, C., Sreedharan, O.M., Premila, M. and Gopalan, P. (1998) Vapor Pressure of C60 by a Transpiration Method Using a Horizontal Thermobalance. Thermochimica Acta, 316, 101-108. http://dx.doi.org/10.1016/S0040-6031(98)00304-9
- Arockiasamy, S., Sreetharan, O.M., Mallika, C., Raghunathan, V.S. and Nagaraja, K.S. (2007) Development, Characterisation and Rapid Evaluation of Standard Enthalpies of Vaporisation and Fusion of Volatile Bis(N-R-Salicylaldimine) Nickel(II) (n-R = Methyl to Pentyl) Complexes for Its MOCVD Applications. Chemical Engineering Science, 62, 1703- 1711. http://dx.doi.org/10.1016/j.ces.2006.12.001
- Burnham, L., Dollimore, D. and Alexander, K. (2001) Calculation of the Vapor Pressure-Temperature Relationship Using Thermogravimetry for the Drug Allopurinol. Thermochimica Acta, 367, 15-22. http://dx.doi.org/10.1016/S0040-6031(00)00652-3
- Joseph, K., Sridharan, R. and Gnanasekaran, T. (2000) Kinetics of Thermal Decomposition of Th(C2O4)2・6H2O. Journal of Nuclear Materials, 281, 129-139. http://dx.doi.org/10.1016/S0022-3115(00)00241-5
NOTES
*Corresponding author.









