Journal of Biosciences and Medicines
Vol.04 No.11(2016), Article ID:72162,12 pages
10.4236/jbm.2016.411004
The Preventative Effect of Dietary Apostichopus japonicus on Intestinal Microflora Dysregulation in Immunosuppressive Mice Induced by Cyclophosphamide
Hongjie Shi*, Rong Zheng*, Juan Wu, Tao Zuo, Changhu Xue, Qingjuan Tang#
College of Food Science and Engineering, Ocean University of China, Qingdao, China

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: October 26, 2016; Accepted: November 19, 2016; Published: November 22, 2016
ABSTRACT
Sea cucumbers are recognized as food and drug resources with many nutritional benefits, and Apostichopus japonicus is a kind of sea cucumber with good quality. Processing methods have some effect on its quality. This study aimed to explore the effects of Apostichopus japonicus with three different processing methods (dried, instant, and enzymatic sea cucummbers) on intestinal microflora dysregulation using a cyclophosphamide (cy) induced immunosuppressive mouse model. The expression of lysozyme, immunoglobulin A (IgA), and polymeric immunoglobulin receptor (pIgR) in the intestine and gut microbiota were investigated. The results showed that three types of A. japonicus could improve mucosal immunity and regulate gut microbiota. Dietary A. japonicus could reverse microbial imbalance, including increasing the bacterial diversity, enhancing the number of Bifidobacterium, and changing the bacterial composition. The most effect was observed with dried A. japonicus. Expression of lysozyme and IgA in the intestine was significantly increased. This study identified positive effects of dietary A. japonicus on mucosal immunity, particularly on gut microbiota, suggesting that dietary A. japonicus may aid in improving mucosal immunity and preventing exogenous infection. Additionally, the processing method has some effect on immunomodulatory function.
Keywords:
Gut Microbiota, Mucosal Immunity, Apostichopus japonicus, Cyclophosphamide

1. Introduction
The mammalian intestine is covered by a large area (400 m2) of mucosal tissue. It is one site in the body, which communicates with the external environment [1] . The mucosa is constantly in contact with a lot of material, including foods, probiotics and other harmless substances, and viruses, parasites, and other harmful organisms or substances which can invade the body through the mucosa [2] . In order to protect the body, the mucosal tissue forms part of the immune system with a tight defense function. It plays an important role in preventing the invasion of pathogens and also incorporates humoral and cellular immune functions [3] . Lysozyme and secretory IgA (SIgA), which is composed of two IgA molecules and is transported by pIgR, are two important mucosal immune system effectors. They help prevent infection and remove antigens crossing the mucosal barrier [4] [5] . Our previous study had indicated that cyclophosphamide could induce the expression of lysozyme and sIgA [6] . There are a large number of bacteria inhabiting the mammalian gut, constituting the body’s normal microbes [7] . The gut microbiota, as part of the intestinal mucosal barrier, plays an important role in maintaining intestinal homeostasis [8] [9] . Disturbances in gut microbiota homeostasis are thought to contribute to severe gastrointestinal disorders [10] [11] . It has been reported that cyclophosphamide (cy) causes intestinal microflora imbalance, resulting in bacterial transfer to mesenteric lymph nodes and spleen [12] . Infection is common during chemotherapy, resulting in diarrhea, stomach ache, emesis, etc [13] . It is painful for patients, making a search for suitable treatments imperative.
Apostichopus japonicus is a species of sea cucumber with good quality, which is popular in Asia as a foodstuff. It contains many bioactive materials such as polysaccharide, collagen, and saponin, which contribute to its numerous physiological effects. It was reported that different processing method could change the ingredient of sea cucummber [14] . Some believe that Sea cucumbers are not suitable for the cancer patients treated with chemotherapeutic drugs and this set a limitation to the application aspect of A. japonicus. However, this study aimed to investigate the effects of A. japonicus, prepared using different processing methods, on mucosal immunity and gut microbiota to clarify whether A. japonicus could reduce tumor-related pain as a dietotherapy method. Additionally, we explored the effects of processing methods on nutritional benefits of A. japonicus. Our results might provide a basic knowledge for cancer patients to choose right sea cucumber products for the sake of their health.
2. Materials and Methods
2.1. Preparation of Dietary A. japonicus
Fresh A. japonicus were purchased from Nanshan markets in Qingdao. The sea cucumbers were boiled for 10 min and processed in three different ways. To produce dried A. japonicus, the boiled sea cucumbers were put in a cold-air oven at 20˚C for 6 days. They were soaked in distilled water for 2 days and boiled for 30 - 40 minutes. After cooling, the sea cucumbers were soaked in distilled water for 2 - 3 days at 4˚C. To prepare instant A. japonicus, the boiled sea cucumbers were kept in the condiments for 1 hour and sterilized at 121˚C for 15 minutes). Finally, A. japonicus was treated with papain at 50˚C for 12 hours and the papain inactivated at 100˚C for 10 min. The resulting mixture was centrifuged and the supernatant retained. The processed A. japonicus were stored at −40˚C until use. The sea cucumbers were crushed and dissolved in saline before being administered to the mice.
All reagents used in the animal experiments were of analytical purity.
2.2. Animal Maintenance and Treatment
Male balb/c mice (19 ± 1 g, 4 weeks old) were obtained from Vital River (Beijing, China). During the experimental period, the mice were housed in a room maintained under a 12 h light/dark cycle at 24˚C. The mice had free access to standard laboratory pellet chow (Kangda, Jinan, China) and fresh water.
The mice were randomly assigned to 5 groups (10 mice/group): normal control group, model group, and A. japonicus groups with three different processing methods. The A. japonicus groups were given one of three sea cucumber regimens followed by 512 mg∙kg−1 by gavage for 28 days, while the normal and model groups were given normal saline as control. Except for the normal group, the others were treated with cyclophosphamide (50 mg∙kg−1) by intraperitoneal injection on the 26th and 27th feeding day. The normal mice were injected with saline as control. The mice body weights were recorded every two days. The feces were collected on the 28th day. Mice were sacrificed by cervical dislocation on the 29th day and the small intestine was collected for analysis. All experimental procedures were conducted according to the guidelines provided by the ethical committee of experimental animal care at Ocean University of China (OUC, Qingdao, China) and the standards for laboratory animals of China (GB 14922-94, GB 14923-94 and GB/T 14925-94).
2.3. DNA Extraction
Bacterial genomic DNA was extracted from fecal samples with the QIAamp DNA stool kit (Qiagen, Germantown, MD), according to the manufacturer’s instruction.
2.4. PCR-DGGE Analysis
Bacterial universal primers GC-338F (5’-CGCCCGGGGCGCGCCCCGGGGCGGGGC GGGGGCGC-GGGGGGCCTACGGGAGGCAGCAG-3’) and 518R (5’-ATTACCGC GGCTGCTGG-3’) were used to amplify the 16S rDNA gene V3 region by PCR. PCR amplification was carried out in a MJ Mini Personal Thermal Cycler (BIO-RAD, America). Each reaction was performed in a total volume of 50 μL containing DNA template 100 ng, 10 × PCR buffer 5 μL, dNTP mix (10 mM) 3.2 μL, MgCl2 (25 mM) 3 μL, each primer (20 mM) 0.5 μL, and 5 U/μL of Taq DNA polymerase (Takara, Japan) 1 μL. The thermal cycler profile consisted of an initial denaturation step at 94˚C for 5 min, followed by 30 cycles at 94˚C for 30 s, 55˚C for 30 s, 72˚C for 30 s, and a final extension at 72˚C for 10 min. DGGE was carried out with a Dcode System apparatus (Bio-Rad, Herts, UK) and a gradient from 35% - 55% (7 M urea and 40% deionized formamide were considered to be the 100% denaturant). The gel images were converted into digital data using Canoco. Principal component analysis (PCA) and cluster analysis were employed to compare the gut microbiota composition among groups. Important bands were retrieved, sequenced (Sangon Biotech Co. Ltd., Shanghai, China), and identified in Genbank, as described [15] .
2.5. Real-Time Quantitative PCR
Bifidobacteria and Lactobacillus were assayed by quantitative real-time PCR. 25 μL of the reaction volume was used for the quantitative real-time PCR assay that consisted of 12.5 μL of Maxima SYBR Green qPCR Master Mix (Roche, Switzerland), 10 μM of primers (0.75 μL each of forward and reverse primers), 8.5 μL ddH2O, and 2.5 μL of template. The thermal conditions consisted of an initial denaturation at 95˚C for 10 min followed by 45 cycles of denaturation at 95˚C for 15 s, annealing at 60˚C for 20 s, and extension at 72˚C for 30 s. The sequences of the primers used in this study are described in supplementary Table S.
2.6. Western Blot Analysis
The expression levels of lysozyme, immunoglobulin A (IgA) and polymeric immunoglobulin receptor (pIgR) in the small intestine were investigated by western blot analysis. Briefly, the tissues were homogenized in RIPA buffer (50 mM Tris, 150 mM NaCl, 1 mM PMSF, 1 mM EDTA, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS, pH 7.4) and put on ice for 20 minutes and then centrifuged at 12,000 × g for 5 min at 4˚C. The protein concentration of the supernatant was measured using the BCA protein assay kit (Beyotime Institute of Biotechnology, Shanghai, China) with bovine serum albumin as the standard sample. Proteins were separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinyl difluoride (PVDF) membranes (GE, Fairfield, CT, USA). The membrane was blocked with TBST (10 mM Tris, 150 mM NaCl, and 0.1% Tween 20, pH 7.6) containing 5% skim milk (BD, Franklin Lakes, NJ, USA) and incubated with the indicated primary antibody in TBST overnight at 4˚C. Subsequently, the membrane was incubated with the secondary antibody and visualized by enhanced chemiluminescence (ECL) kit (GE, Fairfield, CT, USA). Equal protein loading was assessed by blotting for GAPDH.
Table S. The 16S rRNA gene-targeted group-specific primers of bacteria.
Bacteria-specific primers were designed by Primer Premier 5.0 software.
2.7. Statistical Analysis
A one-way analysis of variance between different groups was carried out using SPSS V 18.0 software. Data were presented as mean ± standard error and statistical significance was defined to be p < 0.05.
3. Results
3.1. Structural Changes in Gut Microbiota Revealed by Polymerase Chain Reaction and Denaturing Gradient Gel Electrophoresis (PCR-DGGE)
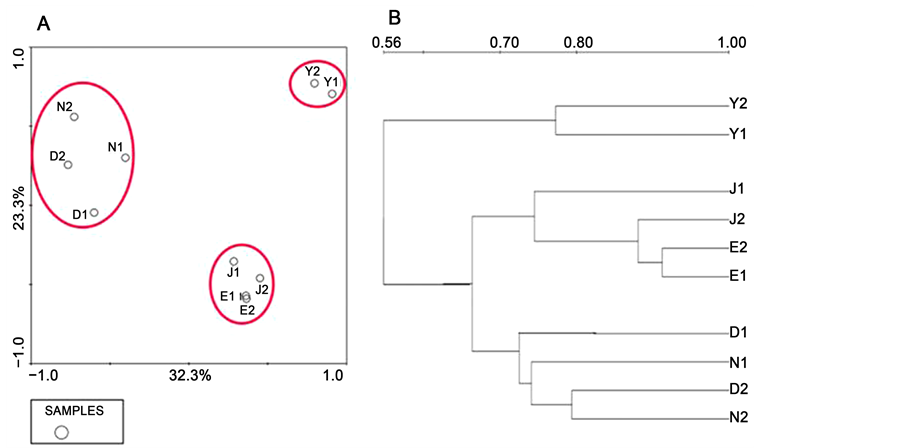
According to the 16S rRNA V3 region PCR-DGGE patterns, the overall composition after the Cy treatment showed a significant difference from that in normal mice (Fig- ure 1(A)). After treated with Cy, the number of bands in model group reduced sharply, and the intensities of bands in some migration locations decreased compared with the normal group. The results indicated that the diversity and structure of the gut microbiota in the model group changed after Cy treatment. The gut microbiota differences in mice treated with A. japonicus were reduced notably. The structures of the gut microbiota in the A. japonicus groups were similar with that in the normal group. The results were also shown clearly in the virtual figure (Figure 2(B)) of PCR-DGGE fingerprints. The diversity indexes of the microbiota in every group showed that the diversity of intestinal flora in the model group decreased, and A. japonicus helped to reverse the changes (Table 1), especially in the dried A. japonicus group.
The principal components analysis (PCA) of the fingerprints showed that mice were classified into three clusters, with the first principal component (PC1) accounting for
Figure 1. PCA and cluster analysis of DGGE fingerprint. (A) The principal components analysis (PCA) scores plot according to the DGGE fingerprint. (B) Clustering of gut microbiota based on distances between different groups. N serves as normal group and Y, D, J, E represent model, dried, instant and enzymatic groups, respectively.
Figure 2. Comparison of gut microbiota composition between the mouse groups. (A) Denaturing gradient gel electrophoresis (DGGE) fingerprinting of V3 region of 16S rRNA genes from faecal bacterial communities. (B) The virtual map of DGGE electrophoresis. Every sample included feces of five mice and feces in 1 and 2 were different five ones. N is for normal group and Y, D, J, E represent model, dried, instant and enzymatic groups, respectively.
Table 1. Analysis of the microbiota diversity of mice intestine.
Each group includes 10 mice. We selected 5 fecal samples randomly into one detecting sample and the other five ones was another sample. Every data was mean of two detecting samples.
32.3% of the variation (Figure 1(A)). The normal and dried A. japonicus group belonged to one cluster, the instant and enzymatic A. japonicus group belonged to another cluster, and the model group was in a third cluster. It confirmed that Cy caused changes in gut microbiota, and A. japonicus helped reverse the changes. The cluster dendrogram showed similar results (Figure 1(B)). These results suggest that A. japonicus could ameliorate the gut microbiota imbalance induced by Cy in mice.
3.2. Identification of the Significantly Different Bands in Denaturing Gradient Gel Electrophoresis (DGGE) Fingerprints
In model mice, the intensities of band 6 (b6), b9, b12, b24, b25 and b28 were significantly reduced. By sequence analysis, these bands were related to Prevotella dentalis DSM 3688 (96% homology), Moryella indoligenes (99%), Barnesiella intestinihominis (99%), Ruminococcaceae bacterium LM158 (98%), Clostridiales bacterium CIEAF 020 (97%), Johnsonella sp. oral taxon 166 (97%) (Figure 2(A) and Table 2). These bacteria belong to Bacteroidetes and Clostridia groups. Bacteroidetes is an intestinally adapted bacterium involved in carbohydrate fermentation, polysaccharide and steroid metabolism, and maintaining intestinal physiological functions, amongst other functions [16] . Most Clostridial bacteria can hydrolyze polysaccharide and protein. The reduction of Bacteroidetes and Clostridia may have an effect on digestion and absorption of polysaccharide and protein. The intensities of bands in A. japonicus groups increased to near normal levels. We speculate that administration of A. japonicus helped to restore normal intestinal physiological function.
3.3. Quantitative Changes in Probiotic Bacteria Revealed by Real-Time Quantitative Polymerase Chain Reaction (RT-PCR)
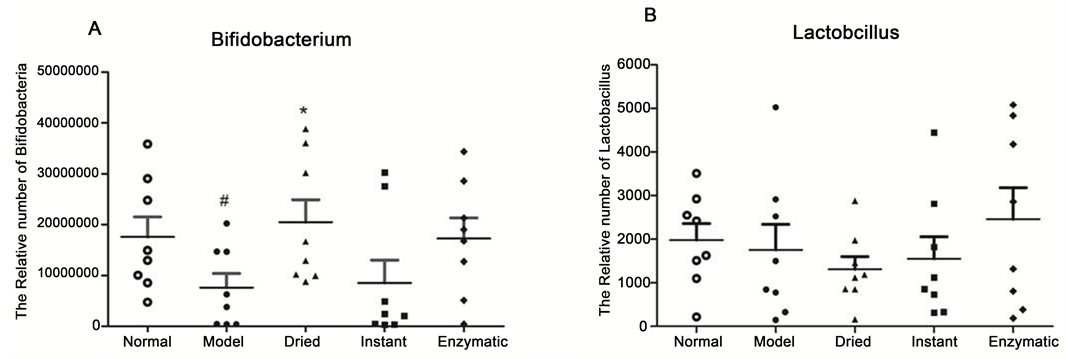
A number of bacteria are friendly and helpful to hosts, but in low abundance, such as probiotic Bifidobacterium and Lactobacillus. Bifidobacterium can reduce intestinal pH to inhibit the growth of harmful bacteria, produce vitamins and amino acids, stimulate immune responses, protect the intestinal barrier, etc. [17] [18] [19] . Lactobacillus can ferment carbohydrates to produce lactic acid and help digestion and absorption, stimulate immunoglobulin production to enhance the host immune function, etc [20] [21] . Hence, the amount of probiotic Bifidobacterium and Lactobacillus was quantified by real-time PCR in this study. The results show that Cy caused a significant reduction of Bifidobacterium (Figure 3(A)). A. japonicus administration increased the quantity of Bifidobacterium and dried A. japonicus showed a significant effect (p < 0.05). However, the amount of Lactobacillus showed no obvious change in this model (Figure 3(B)). These data imply that A. japonicus intake protects the intestine by increasing the quantity of Bifidobacterium.
Table 2. The analysis results of DGGE gel bands recovery sequence.
Ten important bands were cut from the gel, sequenced and then identified in Genbank.
Figure 3. Intestinal microflora compositions in different groups of mice were checked by quantitative real-time PCR analysis of (A) Bifidobacterium, and (B) lactobacillus. Values are expressed as mean ± S.E.M (n = 8 per group). #p < 0.05, different from the normal group; *p < 0.05 different from the model group.
3.4. A. japonicus Ameliorated Intestinal Mucosal Immunity in Immunosuppressive Mice
Lysozyme is an important bactericidal substance in body fluids, which works as a mucosal chemical barrier and represents the innate mucosal immunity effector. SIgA is composed of two immunoglobulin A (IgA) molecules, joined by a J-chain and a secretory component. pIgR transporting dimeric IgA (dIgA) across the epithelial cell function is a necessary condition for the secretion of sIgA to exocrine fluid [22] . Hence, the expression of lysozyme, IgA and pIgR was detected to reflect the mucosal immune function.
The results showed that expression of lysozyme increased with A. japonicus intake, while that in the model group reduced significantly (p < 0.05) (Figure 4(A)). Compared with the normal group, the IgA and pIgR expression of mice in model group decreased significantly (Figure 4(B) and Figure 4(C)). Administration of dried and instant A. japonicas significantly improved the expression of IgA (p < 0.01). The three types of A. japonicus could all increase pIgR expression; however, they showed no statistical significance. All these data indicated that A. japonicus intake could ameliorate intestinal mucosal injury induced by Cy.
4. Discussion
Cy is one of the most common agents in the treatment of cancer [23] [24] . It has higher lethality towards rapidly proliferating cells, and this is not limited to tumor tissue. The intestinal epithelium is composed of rapidly proliferating cells, and is therefore susceptible to Cy. This results in destruction of mucosal barrier and translocation of intestinal microbiota [25] [26] . In this study, the RT-PCR and PCR-DGGE results showed that Cy caused an imbalance in gut microbiota, including decreased abundance of Bifidobacterium, reduced diversity, and altered microflora composition. A. japonicus could reverse these changes. Additionally, the modulation effect of dried A. japonicus on gut
Figure 4. Relative expression of lysozyme, IgA and pIgR in intestine. Lysozyme, IgA and pIgR expression was analyzed by western blotting and GAPDH was the loading control. Each value represents the mean ± S.E.M. of 8 mice in each group. #Significantly different from normal group, p < 0.05; *Significantly different from model group, p < 0.05; **Significantly different from model group, p < 0.01.
microbiota was better. Otherwise, the expression of IgA, pIgR and lysozyme decreased significantly in the model group, which was consistent with previous reports [27] [28] [29] . Dietary A. japonicus could enhance the expression of IgA, pIgR, and lysozyme, and three preparations of A. japonicas prepared using different processing methods did not show significant differences.
The effect of dietary A. japonicus on mucosal immunity was discussed in our previous study [6] . Here, we focused on its effect on intestinal microflora. The human intestinal microflora includes 300 - 500 different species of bacteria [30] . They affect the health of the body, including digestion and absorption, energy metabolism, and immune regulation [31] [32] . Gut microbiota are involved in the digestion of complex macromolecules, particularly fermented polysaccharides, and also provide short-chain fatty acids by enzymatic reaction. They can produce bacteriocins and other antagonists to prevent adhesion and colonization of pathogenic bacteria. Microflora may enhance immune cell reactivity to antigen stimulation, activate lymphoid tissue, and increase sIgA biosynthesis [33] [34] . The RT-PCR results showed that Bifidobacterium decreased in the model group and the alteration of Bifidobacterium in the dried A. japonicus group compared to controls was more significant than the other two groups. Hence, we analyzed changes in gut microbiota using the PCE-DGGE method. DGGE electrophoresis showed that the number and composition of bands changed in the model group, indicating an imbalance of gut microbiota. The electrophoresis patterns of A. japonicus groups were similar to those of the normal group, while the effect of dried A. japonicus was better. We detected polysaccharide and protein in three kinds of A. japonicas, as processing method affects proportions of sea cucumber nutrients. The results showed that the polysaccharide content of dried, instant, and enzymatic A. japonicus was 6.72%, 5.46%, and 5.45%, and the protein content was 73.09%, 65.06%, and 57.42%, respectively. We speculate that the regulatory effect of dietary A. japonicus on gut microbiota was related to amounts of a specific active ingredient. Our study have revealed that dried A. japonicus have better effect on intestinal mucosa protection, but we still didn’t know which component in A. japonicus was related with protection of mucosa damage induced by cy. In the future study, we need to figure out the effective component and how the component interact with intestinal microbial species, if the production of SCFA deprived by microbiota have an effcet on mucosa barrier. These conclusions need further validation.
5. Conclusion
In conclusion, the present study suggests that dietary A. japonicus may regulate gut microbiota and increase the expression of IgA, pIgR and lysozyme to ameliorate small intestinal mucosal disorders caused by Cy. Processing methods affect the nutritional benefits of sea cucumbers, especially its microbiota regulating function. These observations might provide a new mechansm for dietary A. japonicus to reduce possible infectious complications resulting from chemotherapy.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant number: 31330060), and Special funds for the Taishan Scholar construction project. The authors report no conflicts of interest.
Cite this paper
Shi, H.J., Zheng, R., Wu, J., Zuo, T., Xue, C.H. and Tang, Q.J. (2016) The Preventative Effect of Dietary Apostichopus japonicus on Intestinal Mi- croflora Dysregulation in Immunosuppressive Mice Induced by Cyclophosphamide. Journal of Biosciences and Medicines, 4, 24- 35. http://dx.doi.org/10.4236/jbm.2016.411004
References
- 1. Parra, D., Reyeslopez, F.E. and Tort, L. (2015) Mucosal Immunity and B Cells in Teleosts: Effect of Vaccination and Stress. Frontiers in Immunology, 6, 354-357.
https://doi.org/10.3389/fimmu.2015.00354 - 2. Kagnoff, M.F. (1996) Mucosal Immunology: New Frontiers. Immunology Today, 17, 57-59.
https://doi.org/10.1016/0167-5699(96)80579-2 - 3. Shuyu, S., Kang, Z., Hongyan, Z., et al. (2014) Regulatory Effect of Astragalus Polysaccharides on Intestinal Intraepithelial YδT Cells of Tumor Bearing Mice. Molecules, 19, 15224-15236.
https://doi.org/10.3390/molecules190915224 - 4. Pu-Qing, Y.S., Vincent, W., Julie, E., et al. (2012) Expression of Corticotropin Releasing Factor Receptor Type 1 (CRF1) in the Human Gastrointestinal Tract and Upregulation in the Colonic Mucosa in Patients with Ulcerative Colitis. Peptides, 38, 62-69.
https://doi.org/10.1016/j.peptides.2012.07.028 - 5. Kato-Nagaoka, N., Shimada, S.I., Yamakawa, Y., et al. (2015) Enhanced Differentiation of Intraepithelial Lymphocytes in the Intestine of Polymeric Immunoglobulin Receptor-Eficient Mice. Immunology, 146, 59-69.
https://doi.org/10.1111/imm.12480 - 6. Zheng, R., Li, X.M., Cao, B.B., et al. (2015) Dietary Apostichopus japonicus Enhances the Respiratory and Intestinal Mucosal Immunity in Immunosuppressive Mice. Bioscience, Biotechnology, and Biochemistry, 79, 1-7.
https://doi.org/10.1080/09168451.2014.955454 - 7. Quigley, E.M. (2013) Gut Bacteria in Health and Disease. Gastroenterol Hepatol (N Y), 9, 560-569.
- 8. Kuhn, K.A. and Stappenbeck, T.S. (2013) Peripheral Education of the Immune System by the Colonic Microbiota. Seminars in Immunology, 25, 364-369.
https://doi.org/10.1016/j.smim.2013.10.002 - 9. Round, J.L. and Mazmanian, S.K. (2009) The Gut Microbiota Shapes Intestinal Immune Responses during Health and Disease. Nature Reviews Immunology, 9, 313-323.
https://doi.org/10.1038/nri2515 - 10. Goldenberg, O., Herrmann, S., Marjoram, G., et al. (2007) Molecular Monitoring of the Intestinal Flora by Denaturing High Performance Liquid Chromatography. Journal of Microbiological Methods, 68, 94-105.
https://doi.org/10.1016/j.mimet.2006.06.009 - 11. Loh, G. and Blaut, M. (2012) Role of Commensal Gut Bacteria in Inflammatory Bowel Diseases. Gut Microbes, 3, 544-555.
https://doi.org/10.4161/gmic.22156 - 12. Viaud, S., Saccheri, F., Mignot, G., et al. (2013) The Intestinal Microbiota Modulates the Anticancer Immune Effects of Cyclophosphamide. Science, 342, 971-976.
https://doi.org/10.1126/science.1240537 - 13. Leitão, R.F., Brito, G.A., Oriá, R.B., et al. (2011) Role of Inducible Nitric Oxide Synthase Pathway on Methotrexate-Induced Intestinal Mucositis in Rodents. BMC Gastroenterology, 11, 90-100.
https://doi.org/10.1186/1471-230X-11-90 - 14. Xue, C.H., Xue, Y., Wang, J.F., et al. (2007) Sea Cucumber Nutrient and Its Preparing Process. 2007 CN 101016506 A.
- 15. Muyzer, G., Waal, E.C.D. and Uitterlinden, A.G. (1993) Profiling of Complex Microbial Populations by Denaturing Gradient Gel Electrophoresis Analysis of Polymerase Chain Reaction-Amplified Genes Coding for 16s rRNA. Applied and Environmental Microbiology, 59, 695-700.
- 16. Noor, S.O., Ridgway, K., Scovell, L., et al. (2010) Ulcerative Colitis and Irritable Bowel Patients Exhibit Distinct Abnormalities of the Gut Microbiota. BMC Gastroenterology, 10, 134-140.
https://doi.org/10.1186/1471-230X-10-134 - 17. Feng-Ching, H., Chia-Lin, L., Chee-Yin, C., et al. (2013) Oral Administration of Lactobacillus reuteri GMNL-263 Improves Insulin Resistance and Ameliorates Hepatic Steatosis in High Fructose-Fed Rats. Nutrition & Metabolism, 10, 35.
https://doi.org/10.1186/1743-7075-10-35 - 18. Qing, S., Lu, Z. and Tuohy, K.M. (2012) High-Level Dietary Fibre Up-Regulates Colonic Fermentation and Relative Abundance of Saccharolytic Bacteria within the Human Faecal Microbiota in Vitro. European Journal of Nutrition, 51, 693-705.
https://doi.org/10.1007/s00394-011-0248-6 - 19. Salminen, S., Bouley, C., Boutron, M.C., et al. (1998) Functional Food Science and Gastrointestinal Physiology and Function. British Journal of Nutrition, 80, 147-171.
https://doi.org/10.1079/BJN19980108 - 20. Zihler, A., Gagnon, M., Chassard, C. and Lacroix, C. (2011) Protective Effect of Probiotics on Salmonella Infectivity Assessed with Combined in Vitro Gut Fermentation-Cellular Models. BMC Microbiology, 11, 264-270.
https://doi.org/10.1186/1471-2180-11-264 - 21. Sanz, Y., Nadal, I. and Sánchez, E. (2007) Probiotics as Drugs against Human Astrointestinal Infections. Recent Patents on Anti-Infective Drug Discovery, 2, 148-156.
https://doi.org/10.2174/157489107780832596 - 22. Johansen, F.E. And Kaetzel, C.S. (2011) Regulation of the Polymeric Immunoglobulin Receptor and IgA Transport: New Advances in Environmental Factors That Stimulate pIgR Expression and Its Role in Mucosal Immunity. Mucosal Immunology, 4, 598-602.
https://doi.org/10.1038/mi.2011.37 - 23. Palumbo, A. and Rajkumar, S.V. (2010) Multiple Myeloma: Chemotherapy or Transplantation in the Era of New Drugs. European Journal of Haematology, 84, 379-390.
https://doi.org/10.1111/j.1600-0609.2010.01431.x - 24. Watanabe, Y., Etoh, T., et al. (2010) Feasibility Study of Oral Cyclophosphamide Salvage Therapy for the Treatment of Heavily Pretreated Patients with Recurrent Epithelial Ovarian Cancer. International Journal of Clinical Oncology, 15, 468-471.
https://doi.org/10.1007/s10147-010-0094-1 - 25. Trissa, B., Christopher, H., Jonathan, M., et al. (2012) Pseudomonas aeruginosa Virulence Expression Is Directly Activated by Morphine and Is Capable of Causing Lethal Gut-Derived Sepsis in Mice During Chronic Morphine Administration. Annals of Surgery, 255, 386-393.
https://doi.org/10.1097/SLA.0b013e3182331870 - 26. Owari, M., Wasa, M., et al. (2012) Glutamine Prevent Intestinal Mucosal Injury Induced by Cyclophosphamide in Rats. Pediatric Surgery International, 28, 299-303.
https://doi.org/10.1007/s00383-011-3023-0 - 27. Long, L., Yue, X.Q., et al. (2010) Preventive Effects of a Sachet of Chinese Herbs on Influenza and Its Immun Regulation. Journal of Integrative Medicine, 8, 949-954.
- 28. Mansouri, M., Pirouzi, M., et al. (2013) Investigation on the Interaction between Cyclophosphamide and Lysozyme in the Presence of Three Different Kind of Cyclodextrins: Determination of the Binding Mechanism by Spectroscopic and Molecular Modeling Techniques. Molecules, 18, 789-813.
https://doi.org/10.3390/molecules18010789 - 29. Zuo, T., Cao, L., et al. (2014) Dietary Squid Inkpolysaccharide Could Enhance sIgA Secretion in Chemotherapeutic Mice. Food & Function, 5, 3189-3196.
https://doi.org/10.1039/C4FO00569D - 30. Eckburg, P.B., Bik, E.M., et al. (2005) Diversity of the Human Intestinal Microbial Flora. Science, 308, 1635-1638.
https://doi.org/10.1126/science.1110591 - 31. Treangen, T.J., Koren, S., Sommer, D.D., et al. (2013) MetAMOS: A Modular and Open Source Metagenomic Assembly and Analysis Pipeline. Genome Biology, 14, R2.
https://doi.org/10.1186/gb-2013-14-1-r2 - 32. Guarner, F. and Malagelada, J. (2003) Gut Flora in Health and Disease. Lancet, 361, 512-519.
https://doi.org/10.1016/S0140-6736(03)12489-0 - 33. Baumgart, D.C. and Dignass, A.U. (2002) Intestinal Barrier Function. Current Opinion in Clinical Nutrition & Metabolic Care, 5, 685-694.
https://doi.org/10.1097/00075197-200211000-00012 - 34. Lakhdari, O., Tap, J., Béguetcrespel, F., et al. (2011) Identification of NF-κB Modulation Capabilities within Human Intestinal Commensal Bacteria. Journal of Biomedicine & Biotechnology, 2011, Article ID: 282356.
https://doi.org/10.1155/2011/282356
NOTES

*Hongjie Shi and Rong Zheng contributed equally to this paper.