International Journal of Clinical Medicine
Vol.07 No.04(2016), Article ID:66115,12 pages
10.4236/ijcm.2016.74030
Risk Factors, Clinical Features, Baseline Alanine Aminotransferase and CD4+ Count of Children with HIV Co-Infection with Hepatitis B and C at a Tertiary Hospital in Southwest Nigeria
M. O. Durowaye1, S. K. Ernest2, I. A. Ojuawo2
1Department of Paediatrics, Federal Medical Centre, Lokoja, Nigeria
2Department of Paediatrics and Child Health, University of Ilorin, Ilorin, Nigeria

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 25 February 2016; accepted 25 April 2016; published 28 April 2016
ABSTRACT
Background: Human immunodeficiency virus and hepatitis B and C viruses are endemic in sub- Saharan African countries including Nigeria. Researchers have studied the burden of co-infection of HIV with hepatitis B and hepatitis C but the risk factors and clinical presentation have not been much addressed especially in children. Methodology: This was a prospective cross sectional study that determined the prevalence, risk factors, clinical features, baseline CD4+ count, CD4+ percentage, and alanine aminotransferase (ALT) of newly diagnosed, HAART naïve HIV co-infection among children who were managed at a Tertiary Hospital in Ilorin, Nigeria. Result: Of the 60 HIV- infected children recruited, 11.7% had HIV co-infection with HBV or HCV. Children with co-infec- tions (mean age 8.43 ± 2.37 years) were significantly older than their HIV mono-infected counterparts (mean age 5.25 ± 3.96 years) (p = 0.011). There was no significant difference between HIV monoinfection and HIV co-infection with respect to gender (p = 0.758), ethnicity (p = 0.707), religion of parents (p = 0.436), family type (p = 0.184), social class (p = 0.535), previous transfusion (p = 0.053), scarification (p = 0.612), female genital mutilation (p = 0.778), and sharing of clippers (p = 0.806). The mean BMI, immunological staging (p = 0.535), baseline ALT (p = 0.940), and mean baseline CD4+ count (p = 0.928) were comparable. However, the body mass index of HIV co-infec- ted children decreased with age up till age 10 years. Conclusion: There were no risk factors, nor clinical features predictive of co-infection identified in this study. Co-infection did not negatively impact baseline, CD4+ count and ALT.
Keywords:
Co-Infection, Hepatitis B, Hepatitis C, Human Immunodeficiency Virus, Acquired Immunodeficiency Syndrome, HIV, HBV, HCV, Alanine Aminotransferase, ALT, Highly Active Antiretroviral Therapy, HAART, Monoinfection, CD4+, Risk Factors for Co-Infection, Transmission, Hepatitis B Surface Antigen, HBVsAg

1. Introduction
The improvement in access to antiretroviral therapy (ART) has resulted in better survival of people living with HIV/AIDS and has made the obvious burden of chronic complications of HIV infection that manifestation may have been masked by high AIDS-related mortality in the pre-ART era [1] . The understanding of the shared routes of infection and risk factors of HIV, hepatitis B virus (HBV) and hepatitis C virus (HCV) has raised concern of the possibility and effects of co-infection with these viruses [2] - [4] .
Hepatitis C virus infection has been found to accelerate the evolution and progression of liver disease in HIV-infected individuals [4] - [6] . There are also reports that HIV/HBV co-infected patients have higher levels of HBV replication, lower rates of spontaneous resolution of the HBV infection, a higher risk of reactivation of previous infections, and an increased risk of developing cirrhosis [5] [7] [8] .
Co-infection of HIV with HCV and HBV is associated with increased toxicity of antiretroviral medications [5] . Other studies however report no impact of HBV co-infection on HIV disease progression [9] - [11] .
Several studies have reported a significantly higher mean baseline ALT in co-infected patients than patients infected with HIV alone [12] - [14] . The mean CD4+ cell counts in contrast are significantly lower among the HIV/HBV co-infected participants who also have slower rates of CD4+ cell recovery compared with patients with HIV infection alone [8] .
1.1. Risk Factors and Transmission of HIV, HBV and HCV
Vertical transmission also known as mother to child transmission accounts for almost all HIV infections in children younger than 13 years (greater than 90% of paediatric HIV infections in general), although the rate of vertical transmission has reduced significantly in region where there is adequate access to HIV care and support services [14] [15] . Other significant modes of transmission of HIV in children as shared with hepatitis B and C viruses include parenteral exposure to HIV infected blood and blood products, sexual contact with HIV infected persons (especially among adolescents and victims of sexual assaults), and intravenous drug use especially among adolescent boys, [15] believed to be less important in West African children than those in developed countries [14] [16] .
The risk of vertical transmission of HBV which can occur in-utero, intrapartum, or postpartum, is up to 70% - 90% if the mother is hepatitis B envelope antigen (HBeAg) positive, compared to 0% - 30% if mother is HBeAg negative without immunoprophylaxis for the infant [17] [18] . Immunoprophylaxis using hepatitis B immunoglobulin administered soon after birth and HBV vaccination is highly protective, preventing chronic HBV infection in about 95% of infants whose mothers have HBV infection, but the coverage of universal HBV vaccination is very low in developing countries [19] [20] . Some researchers believe that vertical transmission is a less important mode of transmission of HBV in Africa children than their Asian counterparts partly due to lower prevalence of HBeAg in African pregnant women compared to those in Asia [10] [18] . Horizontal transmission of HBV occurs through unapparent blood or blood product exposures from parents, siblings, or playmates who are HBV infected leading to inoculation of HBV into cutaneous scratches, abrasions, or other lesions on mucosal surfaces [21] . Horizontal transmission is believed to be a predominant mode of transmission of HBV in regions endemic for HBV such as Africa, most infections occurring before five year of life [18] . The risk factors of HBV infection in childhood include: maternal HBV infection, transfusion of blood and blood products (largely eliminated where blood and blood products are routinely screened for HBV) [22] [23] , use of unsterile instrument in traditional surgical practices (such as scarification, circumcision and female genital mutilation, uvulectomy), re-use of needles, contaminated house-hold articles (such as, tooth brush, razors, even toy) [19] , known HBV infected family member, rape, and use of intravenous drug.
Hepatitis C virus shares similar routes of transmission and risk factors with both HBV and HIV [24] [25] . However, hepatitis C virus is more efficiently transmitted through direct contact with blood and blood products, thus, infection has been documented to be higher in recipients of contaminated blood and blood products. Sexual activity has been described as inefficient mode of transmission of HCV infection [4] . In childhood, vertical and perinatal transmission remains the most important means of infection with HCV, and has been reported to be enhanced by maternal HIV co-infection [4] [17] .
In Benin, no association was found between risk factors such as circumcision, ear piercing, parenteral injections and blood transfusion with seropositivity for HBsAg or HCV [26] . This is similar to findings in Tanzania [27] . This lack of association of HIV/HBV or HCV co-infection with circumcision, ear piercing, parenteral injections support the believe that horizontal transmission during the first five years of life plays more significant role for HBV transmission in Sub-Saharan Africa than the use of contaminated instrument [4] . A study in Eastern Nigerian reported awareness and use of safety precautions such as use of new razor blade, earring, and surgical consumables by traditional healers that carry out scarification and ear piercing as possible factors that have reduced chances of contamination and transmission of infection through this means [28] . A study in China found high prevalence of HCV infection in HIV-infected children who acquire HIV infection through blood and blood products transfusion, and less HIV/HCV co-infection in children with vertically acquired HIV infection, supporting the role of blood transfusion in the transmission of HCV infection [3] .
Intravenous drug abuse which is a high risk factor for the transmission of HCV is not a common risk behaviour identified in studies from sub-Saharan Africa [5] [13] .
1.2. Effects of HIV Co-Infection with HBV and HCV
Publications have reported the adverse reciprocal interaction of HIV co-infection with HBV, HCV, or both HBV and HCV that affects every aspect of the co-infection, namely: viral replication, clinical presentation, response to therapy, and outcome of patients with the co-morbidities [12] [29] - [31] . Research has evaluated the effect of HIV infection on response to hepatitis B vaccination, and has documented reduced and less durable antibody titre after vaccination [4] [32] . Thus, fewer HIV infected children have protective antibodies against HBsAg after vaccination [33] . The rate of response to HB vaccination is directly proportional to CD4 counts, but has inverse relationship with HIV viral load, although there is no consensus on the immunological threshold at which vaccination becomes futile [4] . Co-infection of HIV with HBV has been reported to be associated with increased risk of chronic HBV infection, higher HBV DNA levels, thus, higher rates of cirrhosis, end stage liver disease, and death from liver diseases, especially in patients with low CD4 count [4] .
It has been recommended that both anti-HBc and HBsAg should be used in screening for HBV infection in HIV/HBV co-infection because about 10% - 45% of patients with co-infection who have negative HBsAg tested positive for anti-HBc, and when evaluated further, have HBV DNA [4] . These patients are referred to as having occult HBV infection, and should be considered infected with HBV. Patients who have positive anti-HBc without HBV DNA should be regarded as having amnestic response and require no treatment [4] [34] . The goal of treatment in HIV co-infection with HBV is maximal suppression of hepatitis B viral replication evident by clearance of HBV DNA or hepatitis B e antigen (HBeAg), and improvement in liver disease [4] . Immune control is rarely achieved in patients with HIV/HBV co-infection [4] .
The unfavourable effects of HIV/HCV co-infection has also been documented but the symptoms are milder and rate of progression of both hepatic and extrahepatic diseases appear to be slower in children. The natural history of HCV is accelerated, with an increase rate of progression to liver cirrhosis, decompensated liver disease, hepatocellular carcinoma, and death [35] . Extra-hepatic manifestations of HIV/HCV co-infection such as cognitive dysfunction and diabetes mellitus may negatively impact on the quality of life of patients with co-in- fection [4] .
Hepatotoxic effects of antiretroviral drugs such as lamivudine and nevirapine, two commonly used first line antiretroviral drugs in developing countries are more likely to develop in HIV-infected children co-infected with HBV or HCV [29] [36] .
Lamivudine is a drug frequently used in the treatment of chronic hepatitis B infection, but resistance to lamivudine is common in HIV/HBV co-infection, occurring in 90% of patients after 4 years of treatment with lamivudine [37] . This potentially, can lead to rapid development of lamivudine resistant HBV infection in resource limited settings, especially in sub-Saharan Africa where horizontal transmission of HBV is a significant mode of transmission.
Immune reconstitution inflammatory syndrome has been described in patients with HIV co-infection with HBV and HCV, associated with significant morbidity and mortality [38] .
Few researches among children have tried to correlate the prevalence of HIV co-infection with HBV or HCV with baseline WHO clinical staging of HIV, because it is logical to expect advanced or severe HIV/AIDS in patients with co-infection [3] [9] . Two studies, one from Nigeria and China did not find significant difference in baseline WHO staging between HIV/HBV or HCV co-infection and HIV-monoinfection [3] [9] . These studies however, differ from the research findings in Cote d’Ivoire where children with HIV/HBV or HCV co-infections presented with Centre for Disease Control (CDC) HIV stage B or C disease compared with children who had HIV-monoinfection [39] . Among adults, a study in Nigeria has reported that the prevalence of HIV co-infection with HBV or HCV increases with HIV disease progression, similar to research finding in India [5] [25] . The finding of higher prevalence of HIV co-infection with HBV or HCV in patients with advanced HIV infection supports a rapid course of HIV disease progression in patients with co-infection.
Rawizza et al. [9] evaluated base line CD4 count, alanine aminotransferase (ALT), and WHO clinical stage of children with HIV infection and compared the results with children with HIV/HBV, HIV/HCV co-infection. They did not find significant difference in baseline CD4 count, WHO clinical stage, and ALT between HIV- monoinfection and HIV/HBV or HIV/HCV co-infections similar to a research finding in Cote d’Ivore [39] . Rawizza et al. [9] however found some HIV infected children who had significant ALT elevation to 5-fold the upper border of normal, this was however, not related to HBV or HCV co-infection [9] . The finding of normal ALT in HIV co-infection with viral hepatitis differs from a Tanzania study that found elevated ALT in children with HIV/HBV or HIV/HCV co-infections [27] .
Rouet et al. [39] in Cote d’Ivoire evaluated the baseline HBeAg in children with HIV/HBV co-infection and followed these children for at least 18 months. It was found that 82% of children with HIV/HBV co-infection had chronic HBeAg positive HBV infection, with persistence of HBeAg beyond 18months in about 80% of these patients. Most of these patients had high level of HBV DNA titre, indicating that they were in the initial immunotolerant stage of HBV infection. It was pertinent to discover that only few of the these children with HIV/HBV co-infection had HBeAg seroconversion, and there was no significant difference at 18 months in terms of HBeAg seroconversion between HIV/HBV co-infected patients on HAART either containing 3TC or not and those without HAART, implying no remarkable benefit from HAART.
Few studies have addressed HIV co-infection with HBV or HCV among children globally, and especially in Nigeria [9] . Studies are required to address the magnitude, risk factors for co-infection and baseline immunological and liver function parameters in children with the co-morbidities. The risk of progression of HBV infection to chronicity in childhood, and the generally high rate of progression of acute HCV infection to chronicity, and their sequelae in adulthood makes determination of the burden of HIV co-infection with viral hepatitis B and C in childhood imperative. This study is a template for subsequent studies that will characterize the features and outcome of the co-infections.
1.3. Methodology
This was a prospective descriptive cross-sectional study of children age 2 months to 13 years who were HIV infected conducted in University of Ilorin Teaching Hospital (UITH), a tertiary institution located in Ilorin, in the North Central geopolitical zone of Nigeria. Recruitment of patients was carried out at the Paediatric Antiretroviral Therapy (ART) Clinic, Emergency Paediatric Unit (EPU), and Paediatric medical ward of the hospital. A total of 60 HIV-infected children were recruited into the study using the Andrew Fisher’s formula to calculate the minimum sample size. The national seroprevalence of HIV used was 4.1%, and the tolerable margin of error was set at 0.05. Ethical clearance was obtained from the Ethical Review Committee of the hospital while individual informed consent was also obtained from the respective parent or care-giver of the child and written assent was also obtained from children aged 7 years and above before subject recruitment.
1.3.1. Subject Recruitment
This study was carried out over eleven month period, from January to October 2011. Consecutive patients who met the recruitment criteria were recruited into the study after informed consent. Recruitment of subjects was done by the investigator.
1.3.2. Inclusion Criteria
All antiretroviral drug naïve HIV infected children aged two months to 14 years whose parent gave consent, and children age 7 years to 10 years who gave assent to participate were recruited.
1.3.3. Exclusion Criteria
All HIV infected children on antiretroviral or antituberculous drugs were excluded from the study.
Potential eligible children whose parents did not give consent, or children 7 years to 14 years who did not give assent to participate in the study.
1.3.4. Data Collection
Semi structured questionnaire was used to obtain information on: socio-demographic characteristics, educational qualifications and occupations of parents, risk factors for transmission of HIV, HBV, and HCV, presenting symptoms of patients, anthropometric measurements, and laboratory parameters.
Socio-economic index scores were awarded to the occupations and educational attainments of parents or caregivers of subjects using the Oyedeji socio-economic classification scheme [40] . The anthropometry of each subject was taken and a detailed medical examination was conducted on each patient.
HBV infection was confirmed using Diaspot HBsAg (AZOG Inc. Phillipsburg, USA), a one step hepatitis B surface antigen test strip [41] , while Hepatitis C infection was screened for using DiaSpot HCV, a one step hepatitis C virus test strip (AZOG Inc. Phillipsburg, USA) [42] [43] . Complete blood count, CD4 count, and Alanine aminotransferase assay were done for all patients recruited to the study by dedicated Laboratory scientists at the ART laboratory following standard laboratory procedure.
1.3.5. Data Analysis
Data was analyzed using Epi-info version 6 software. The results of investigation and other data collected on the proforma were entered into a master sheet using numerical codes. Frequency distribution tables and cross tabulation of variables were generated. Appropriate statistical tests were carried out as necessary. Level of significance was set at 0.05.
2. Results
General Characteristic of Study Population
Sixty HIV-infected children were recruited to the study, of which males were 31 (51.7%), and females were 29 (48.3%) giving a male female ratio of 1.1:1.
Six patients (10%) tested positive for HBsAg, while only one (1.7%) of the subjects tested positive for HCV. The prevalence of HIV co-infection with HBV and HCV among this cohort was thus 11.7%. Among children with HIV co-infection, four (57.1%) were males while 42.9% of the co-infected patients were females, thus, HIV co-infection with HBV and HCV had no relationship with gender (p = 0.925). The mean age of HIV-mon- oinfected children was 5.25 ± 3.96 years, while children with HIV co-infection had a mean age of 8.43 ± 2.37 years (Table 1). Children with HIV co-infection were significantly older than HIV-monoinfected children (t = −3.034, p = 0.011).
Table 2 summarizes the risk factors evaluated in this study. Among HIV-monoinfected children, 38 (72%) had HB vaccination, while only three (42.9%) of children with HIV co-infection with HBV and HCV had HB
Table 1. Age group distribution of children with HIV-monoinfection and HIV co-infection with HBV and HCV.
Table 2. Risk factors for HIV co-infection with HBV or HCV.
*Yate corrected chi.
vaccination. However, there was no significant difference with respect to HB vaccination (x2 = 0.134, p = 0.268). Approximately fifty seven percent (4 out of 7) of HIV co-infected children had been transfused with blood, while 17% (9 out of 53) of HIV-monoinfected children had history of blood transfusion. There was no statistically significant difference between HIV monoinfection and co-infection with respect to previous blood transfusion (x2 = 3.748, p = 0.053). There was no significant difference between HIV-monoinfected children and those with HIV co-infection in terms of: scarification (x2 = 0.306, p = 0.612), circumcision or female genital mutilation (x2 = 0.389, p = 0.777), and sharing of clippers (x2 = 0.403, p = 0.807). Risk factors such as previous surgery, traditional uvulectomy, sexual activity, tattooing, parenteral use of illicit drug, and known family history of hepatitis were not common in this cohort.
Common physical examination findings in the study population included: scarification marks, weight loss, generalized lymphadenopathy, Painless parotid enlargement and hepatomegaly (Table 3). However, when HIV- monoinfected children were compared with those with HIV/HBV and HIV/HCV co-infections, there was no significant difference with respect to physical examination finding of scarification marks (x2 = 0.103, p = 0.206), weight loss (x2 = 0.247, p = 0.499), microcephaly (x2 = 0.211, p = 0.421), generalised lymphadenopathy (p = 0.692), abdominal distension (p = 0.608), and hepatomegaly (x2 = 0.411, p = 0.822). Other physical examination finding whose frequencies were not sufficient for statistical analysis included: pallor, jaundice, painless parotid enlargement, skin rash, ear discharge, lung crepitations, ascites, splenomegaly, and ophthalmopathy.
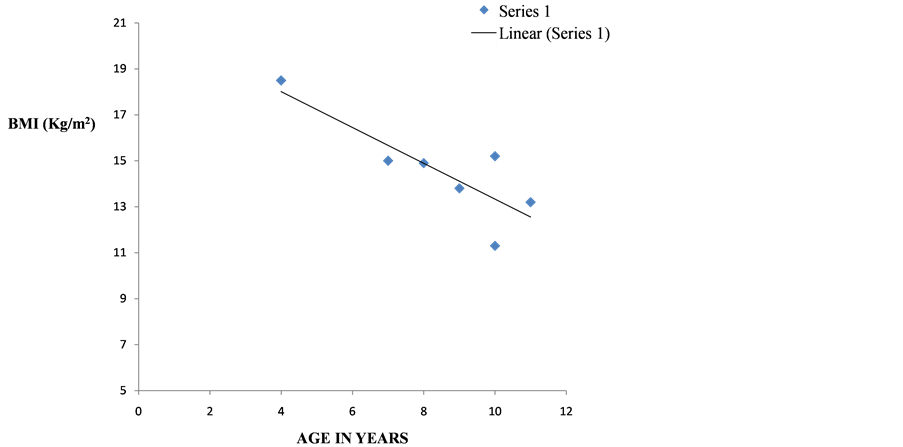
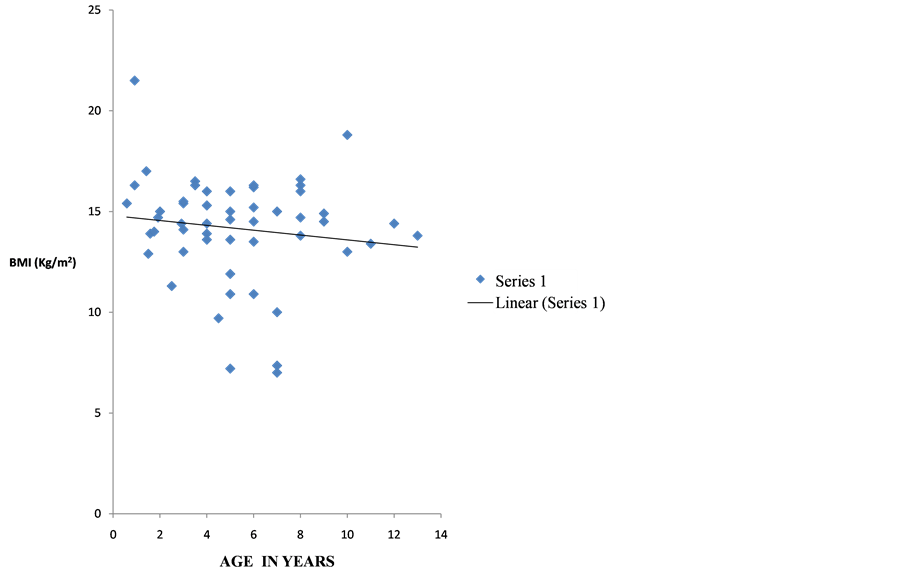
The mean BMI of the study population was 14.21 ± 2.54Kg/m2. Children with HIV monoinfection had a mean BMI of 14.17 ± 2.65 Kg/m2while children with HIV co-infection with HBV and HCV had a mean BMI of 14.56 ± 2.65 Kg/m2. There was no significant difference in mean BMI between children with HIV monoinfection and those with co-infection (p = 0.711). However, Figure 1 shows that the BMI of children with co-infec- tion shows that the BMI of patients with HIV co-infection with HBV and HCV decreased as age advanced up till the age of 10year with a significant negative correlation between ages and BMI (r = −0.83, df = 5, α = 0.05). There was no significant correlation between the ages and BMI of the HIV-monoinfected children (r = −0.1313, df = 51, α= 0.05) (Figure 2).
The mean baseline CD4+ counts of HIV-monoinfected children older than 59 months was not significantly different from that of HIV/HBV and HIV/HCV co-infected children of the same age group (t = 1.022, p = 0.314) (Table 4). Among children younger than five years, there was only one child with HIV/HBV co-infection and the group was not subjected to further statistical analysis.
3. Discussion
This study evaluated the prevalence, risk factors and physical examination findings of children with HIV co-infection with HBV and HCV. The prevalence of HIV co-infection in this study was 11.7%. The prevalence had earlier been reported [44] .
Table 3. Clinical features of HIV-monoinfection and HIV/HBV or HCV co-infection.
*Yate corrected chi
Figure 1. Relationship between BMI and age of HIV co-infected children.
The history of previous blood transfusion was found in 67% of co-infected children in contrast to only 17% of HIV-monoinfected children, however, this difference was not statistically significant.
Risk factors such as circumcision or female genital mutilation, scarification, tattooing, sharing of clippers, had no significant association with HIV/HBV or HIV/HCV co-infections, similar to results of other studies in Nigeria and Africa [26] [27] .
Some risk factors such as traditional uvulectomy, sexual activities, and illicit drug injection were rare in this
Figure 2. Relationship between BMI and age of HIV-monoinfected children.
Table 4. Mean CD4+ count and CD4+ percentages.
cohort. Social stigma associated with risk factors such as sexual activities in our society may make people deny this history. Improvement in heath, and reduction in risky traditional surgical practices may partly explain the low prevalence of risk factors such as genital mutilation, and traditional uvulectomy [26] .
There was only one suspected case of sexual transmission in a sexually active adolescent girl who was living with her maternal grandmother that was a commercial sex worker.
There was no case of known family history of hepatitis B or C in this study. History of hepatitis B in family members living together is a risk factor for horizontal transmission of HBV. However, this information was based on informant’s knowledge and not medical records of parents or family members, and may thus be a reflection of low level of awareness of HBV and HCV infection in the community. Community awareness of hepatitis B or C infection, and the basic knowledge of mode of spread of these viruses have been known to correlate with the prevalence of hepatitis B or C in the community, being relatively lower in communities with high level of awareness and higher in those with low level of awareness [45] .
There was no significant difference between the presenting physical examination findings of HIV-monoin- fected and HIV/HBV or HIV/HCV co-infected children in this cohort. This suggests that co-infection did not negatively affect the clinical presentation of both HBV and HCV infection in this cohort. The few paediatric studies that have evaluated the clinical presentation of children with HIV co-infection with HBV or HCV used the WHO clinical staging, and found no significant difference between children with HIV monoinfection and those with HIV co-infection with HBV or HCV with respect to WHO clinical staging. [3] [9] However, a study in Cote de Ivoire reported that children with HIV co-infection with HBV or HCV presented with Center for Disease Control and Prevention clinical stages B and C compared to stage N or A in the HIV-monoinfected group [39] . Chronic hepatitis B and C viruses in childhood usually run indolent courses until chronic liver diseases manifest, most often in adulthood [46] . This finding of no significant impart of co-infection on presenting symptoms of HIV infection or viral hepatitis B or C infection supports a benign course of HBV and HCV disease in childhood.
The progressive fall in the body mass index of HIV co-infected children as their ages advanced as seen in this study was not observed in the HIV-monoinfected counterparts. This might suggest recent onset of progressive fall in weight relative to height in patients with co-infection. Although, this relationship between BMI and age of children with HIV co-infection with HBV and HCV has not been reported, progressive fall in BMI may be an early sign of HIV co-infection with HBV and HCV.
This study also shows that both HIV-monoinfected and HIV-coinfected children had comparable levels of immunodeficiency, similar to the findings by Telatela et al. in Tanzania [27] . This further confirmed that co-in- fection may not significantly accelerate HIV disease progression in childhood. Other studies in Nigerian children also did not find significant differences in the baseline immunological parameter between HIV co-infection and HIV monoinfection [9] [26] .
The mean baseline alanine aminotransferase of the two groups of children were comparable. Similar finding had been reported by Rawizza et al. [9] in Nigeria, but differs from the findings of the work in Tanzania [27] that found a four-fold risk of ALT elevation in HIV/HBV or HIV/HCV co-infected children compared with HIV-monoinfection. However, the study population in Tanzania is relatively older, and some of the patients were using antiretroviral drugs most of which are potentially hepatotoxic such as nevirapine and efavirenz [47] .
Approximately 9% of HIV-monoinfected children in this study had elevated serum ALT ranging from 1.1 to 2 times the upper limit of reference values. All these five patients were aged 10 years and above, having serum ALT that is directly related to their ages. This supports the fact that advancing ages have correlation with clinical severity. Such elevation of ALT had been reported earlier in Nigeria, and such patients probably have early hepatic manifestation of HIV and should be monitored to identify the evolution and course of HIV infection in them [9] .
4. Conclusion
There was no significant association of risk factors such as blood transfusion, traditional uvulectomy, scarification and tattooing with co-infection in this study. There was no clinical feature predictive of HIV co-infection with HBV and HCV in this cohort. Hepatitis B virus or HCV co-infection did not negatively impact on baseline immunological parameters of HIV infected children. There was no difference between HIV-monoinfected and co-infected children with respect to baseline ALT. The baseline CD4+ count and percentages of HIV-monoin- fected children and HIV infected children with HBV and HCV were similar. There was a need for longitudinal studies that would characterize the evolution and course of HIV-infected children with HBV or HCV co-infec- tion.
5. Limitations
Screening for HBV co-infection in this study was by the use of HBsAg alone, which does not necessarily indicate current infection.
Secondly, HCV infection was not confirmed by plasma PCR for HCV-RNA, making it impossible to distinguish active HCV infection from spontaneously cleared infection.
Cite this paper
M. O. Durowaye,S. K. Ernest,I. A. Ojuawo, (2016) Risk Factors, Clinical Features, Baseline Alanine Aminotransferase and CD4+ Count of Children with HIV Co-Infection with Hepatitis B and C at a Tertiary Hospital in Southwest Nigeria. International Journal of Clinical Medicine,07,280-291. doi: 10.4236/ijcm.2016.74030
References
- 1. Cooley, L. and Sasadeusz, J. (2003) Clinical and Virological Aspects of Hepatitis B Co-Infection in Individuals Infected with Human Immunodeficiency Virus Type-1. Journal of Clinical Virology, 26, 185-193.
http://dx.doi.org/10.1016/S1386-6532(02)00117-8 - 2. Main, J. and McCarron, B. (2005) Hepatitis in HIV-Infected Persons. In: Thomas, H., Lemon, S. and Zuckerman, A., Eds., Viral Hepatitis, 3 Edition, Blackwell Publishing Ltd., Massachusetts, 769-779.
http://dx.doi.org/10.1002/9780470987131.ch50 - 3. Zhou, S., Zhao, Y., He, Y., Li, H., Bulterys, M. and Sun, X. (2010) Hepatitis B and Hepatitis C Seroprevalence in Children Receiving Antiretroviral Therapy for Human Immunodeficiency Virus-1 Infection in China, 2005-2009. Journal of Acquired Immune Deficiency Syndromes, 54, 191-196.
- 4. Koziel, M. and Peters, M. (2007) Viral Hepatitis in HIV Infection. The New England Journal of Medicine, 356, 1445-1454.
http://dx.doi.org/10.1056/NEJMra065142 - 5. Adewole, O.O., Anteyi, E., Ajuwon, Z., Wada, I., Elegba, F., Ahmed, P., et al. (2009) Hepatitis B and C Virus Co-In-fection in Nigerian Patients with HIV Infection. Journal of Infection in Developing Countries, 3, 369-375.
http://dx.doi.org/10.3855/jidc.245 - 6. Rockstroh, J. (2006) Influence of Viral Hepatitis on HIV Infection. Journal of Hepatology, 44, 25-27.
http://dx.doi.org/10.1016/j.jhep.2005.11.007 - 7. Schnuriger, A., Dominguezd, S., Guiguete, M., Harfoucha, S., Samria, A., Ouazeneh, Z., et al. (2009) Acute Hepatitis C in HIV-Infected Patients: Rare Spontaneous Clearance Correlates with Weak Memory CD4 T-Cell Responses to Hepatitis C Virus. AIDS, 23, 2079-2089.
http://dx.doi.org/10.1097/QAD.0b013e328330ed24 - 8. Christian, B., Okuma, J. and Hawkins, H. (2010) Prevalence of Hepatitis B and C Co-Infection and Response to Antiretroviral Therapy among HIV-Infected Patients in an Urban Setting in Tanzania. 17th Conference on Retroviruses & Opportunistic Infections (CROI 2010), San Francisco, 16-19 February 2010.
- 9. Rawizza, H., Ochigb, O.S., Chang, C., MeloniI, M., Oguche, S., Osinusi, I.K., et al. (2010) Prevalence of Hepatitis Co-Infection among HIV-Infected Nigerian Children in the Harvard. PEPFAR ART Program Conference on Retroviruses and Opportunistic Infections, San Francisco.
- 10. Kotzee, T., Pronyk, P., Vardas, E., Heyer, A. and Martinson, N. (2006) HIV and Hepatitis B Co-Infection in Southern Africa: A Review for General Practitioners. Southern African Journal of HIV Medicine, 7, 38-43.
- 11. Law, W.P., Duncombe, C.J., Mahanontharit, A., Boyd, M.A., Ruxrungtham, K., Lange, J.M., et al. (2004) Impact of Viral Hepatitis Co-Infection on Response to Antiretroviral Therapy and HIV Disease Progression in the HIV-NAT Cohort. AIDS, 18, 1169-1177.
http://dx.doi.org/10.1097/00002030-200405210-00010 - 12. Idoko, J., Meloni, S., Muazu, M., Hawkins, C., Bidang, B., Gwamzi, N., et al. (2007) Hepatitis B Virus Co-Infection Impacts Baseline HIV Parameters and HAART-Related Hepatotoxicity Risk in an HIV-Infected Nigerian Cohort. AIDS Prevention Initiative in Nigeria.
- 13. Otegbayo, J., Adewole, I., Taiwo, B., Akingbola, T., Odaibo, G., Adedapo, K., et al. (2008) Prevalence of Hepatitis B and C Seropositivity in a Nigerian Cohort of HIV-Infected Patients. Annals of Hepatology, 7, 152-156.
- 14. Mandala, J., Torpey, K., Kasonde, P., Kabaso, M., Dirks, R., Suzuki, C., et al. (2009) Prevention of Mother-to-Child Transmission of HIV in Zambia: Implementing Efficacious ARV Regimens in Primary Health Centers. BMC Public Health, 9, 314.
http://dx.doi.org/10.1186/1471-2458-9-314 - 15. Yogev, R. and Chadwick, E. (2007) Acquired Immunodeficiency Syndrome (Human Immunodeficiency Virus). In: Kliegman, R., Behrman, R., Jenson, H. and Stanton, B., Eds., Nelson Textbook of Pediatrics, 18th Edition, Saunders Elsevier, Philadelphia, 1427-1442.
- 16. Mboto, C., Fielder, M., Davies-Russell, A. and Jewell, A. (2009) Prevalence of HIV-1, HIV-2, Hepatitis C and Co-Infection in the Gambia. West African Journal of Medicine, 28, 306-309.
http://dx.doi.org/10.4314/wajm.v28i1.48418 - 17. Yazigi, N. and Balistreri, W. (2007) Viral Hepatitis. In: Kliegman, R., Behrman, R., Jenson, H. and Stanton, B., Eds., Nelson Textbook of Pediatrics, 18th Edition, Saunders Elsevier, Philadelphia, 1680-1690.
- 18. Hoffmann, C.J. and Thio, C.L. (2007) Clinical Implications of HIV and Hepatitis B Co-Infection in Asia and Africa. The Lancet Infectious Diseases, 7, 402-409.
http://dx.doi.org/10.1016/S1473-3099(07)70135-4 - 19. Jan-Christian, W. (2010) Hepatitis B—Epidemiology, Transmission and Natural History. In: Mauss, S., Berg, T., Rockstroh, J., Sarrazin, C. and Wedemeyer, H., Eds., Hepatology, 2nd Edition, Flying Publisher, Wuppertal, 7-17.
- 20. Shepard, C., Simard, E., Finelli, L., Fiore, A. and Bell, B. (2006) Hepatitis B Virus Infection: Epidemiology and Vaccination. Epidemiologic Reviews, 28, 112-125.
http://dx.doi.org/10.1093/epirev/mxj009 - 21. Francis, D., Favero, M. and Maynard, J. (1981) Transmission of Hepatitis B Virus. Seminars in Liver Disease, 1, 27-32.
http://dx.doi.org/10.1055/s-2008-1063927 - 22. Polizzotto, M., Wood, E., Ingham, H. and Keller, A. (2008) Reducing the Risk of Transfusion-Transmissible Viral Infection through Blood Donor Selection: The Australian Experience 2000 through 2006. Transfusion, 48, 55-63.
- 23. Dodd, R. (2000) Current Viral Risks of Blood and Blood Products. Annals of Medicine, 32, 469-474.
- 24. La Torre, G., Miele, L., Chiaradia, G., Mannocci, A., Reali, M., Gasbarrini, G., et al. (2007) Socio-Demographic Determinants of Coinfections by HIV, Hepatitis B and Hepatitis C Viruses in Central Italian Prisoners. BMC Infectious Diseases, 7, 100.
http://dx.doi.org/10.1186/1471-2334-7-100 - 25. Saravanan, S., Velu, V., Kumarasamy, N., Nandakumar, S., Murugavel, K.G., Balakrishnan, P., et al. (2007) Coinfection of Hepatitis B and Hepatitis C Virus in HIV-Infected Patients in South India. World Journal of Gastroenterology, 13, 5015-5020.
http://dx.doi.org/10.3748/wjg.v13.i37.5015 - 26. Sadoh, A., Sadoh, W. and Iduoriyekemwen, N. (2011) HIV Co-Infection with Hepatitis B and C Viruses among Nigerian Children in an Antiretroviral Treatment Programme. South African Journal of Child Health, 5, 7-10.
- 27. Telatela, S.P., Matee, M.I. and Munubhi, E.K. (2007) Seroprevalence of Hepatitis B and C Viral Co-Infections among Children Infected with Human Immunodeficiency Virus Attending the Paediatric HIV Care and Treatment Center at Muhimbili National Hospital in Dar-es-Salaam, Tanzania. BMC Public Health, 7, 338.
http://dx.doi.org/10.1186/1471-2458-7-338 - 28. Chukwuka, J., Ezechukwu, C. and Egbuonu, I. (2003) Cultural Influences on Hepatitis B Surface Antigen Seropositivity in Primary School Children in Nnewi. Nigerian Journal of Paediatrics, 30, 140-142.
- 29. Sulkowski, M.S., Thomas, D.L., Mehta, S.H., Chaisson, R.E. and Moore, R.D. (2002) Hepatotoxicity Associated with Nevirapine or Efavirenz-Containing Antiretroviral Therapy: Role of Hepatitis C and B Infections. Hepatology, 35, 182-189.
http://dx.doi.org/10.1053/jhep.2002.30319 - 30. Cheruvu, S., Marks, K. and Talal, A.H. (2007) Understanding the Pathogenesis and Management of Hepatitis B/HIV and Hepatitis B/Hepatitis C Virus Coinfection. Clinical Liver Disease, 11, 917-943.
- 31. Lodenyo, H., Schoub, B., Ally, R., Kairu, S. and Segal, I. (2000) Hepatitis B and C Virus Infections and Liver Function in Aids Patients at Chris Hani Baragwanath Hospital, Johanneburg. East African Medical Journal, 77, 13-15.
- 32. Shire, N., Welge, J. and Sherman, K. (2006) Efficacy of Inactivated Hepatitis A Vaccine in HIV-Infected Patients: A Hierarchical Bayesian Meta-Analysis. Vaccine, 24, 272-279.
http://dx.doi.org/10.1016/j.vaccine.2005.07.102 - 33. Laurence, J. (2005) Hepatitis A and B Immu-Nizations of Individuals Infected with Human Immunodeficiency Virus. American Journal of Medicine, 118, 75S-83S.
- 34. Gandhi, R. (2005) Response to Hepatitis B Vaccine in HIV-1-Positive Subjects Who Test Positive for Isolated Antibody to Hepatitis B Core Antigen: Implications for Hepatitis B Vaccine Strategies. The Journal of Infectious Diseases, 191, 1435-1441.
http://dx.doi.org/10.1086/429302 - 35. Operskalski, E. and Andrea, K. (2011) HIV/HCV Co-Infection: Pathogenesis, Clinical Complications, Treatment, and New Therapeutic Technologies. Current HIV/AIDS Reports, 8, 12-22.
http://dx.doi.org/10.1007/s11904-010-0071-3 - 36. Nyirenda, M., Beadsworth, M., Stephany, P., Hart, C., Hart, I., Munthali, C., et al. (2008) Prevalence of Infection with Hepatitis B and C Virus and Coinfection with HIV in Medical Inpatients in Malawi. Journal of Infection, 57, 72-77.
http://dx.doi.org/10.1016/j.jinf.2008.05.004 - 37. Benhamou, Y., Bochet, M., Thibault, V., Di Martino, V., Caumes, E., Bricaire, F., et al. (1999) Long-Term Incidence of Hepatitis B Virus Resistance to Lamivudine in Human Immunodeficiency Virus-Infected Patients. Hepatology, 30, 1302-1306.
http://dx.doi.org/10.1002/hep.510300525 - 38. Hirsch, H.H., Kaufmann, G., Sendi, P. and Battegay, M. (2004) Immune Reconstitution in HIV-Infected Patients. Clinical Infectious Diseases, 38, 1159-1166.
http://dx.doi.org/10.1086/383034 - 39. Rouet, F., Chaix, M.L., Inwoley, A., Anaky, M.F., Fassinou, P., Kpozehouen, A., et al. (2008) Frequent Occurrence of Chronic Hepatitis B Virus Infection among West African HIV Type-1-Infected Children. Clinical Infectious Diseases, 46, 361-366.
http://dx.doi.org/10.1086/525531 - 40. Oyedeji, G.A. (1984) The Present Day Epidemiology of Severe Protein-Energy Malnutrition in Nigeria. Clinical Pediatrics, 23, 623-628.
http://dx.doi.org/10.1177/000992288402301104 - 41. Blumberg, B. (1971) The Discovery of Australian Antigen and Its Relation to Viral Hepatitis. Vitro, 7, 223.
- 42. Kuo, G., Choo, Q., Alter, H. and Houghton, M. (1989) An Assay for Circulating Antibodies to a Major Etiologic Virus of Human Non-A, Non-B Hepatitis. Science, 244, 362-364.
http://dx.doi.org/10.1126/science.2496467 - 43. Wilber, J. (1993) Development and Use of Laboratory Tests for Hepatitis C Infection: A Review. Journal of Clinical Immunoassay, 16, 204-207.
- 44. Durowaye, M., Ernest, S. and Ojuawo, I. (2014) Prevalence of HIV Co-Infection with Hepatitis B and C Viruses among Children at a Tertiary Hospital in Ilorin, Nigeria. International Journal of Clinical Medicine Research, 1, 42-47.
- 45. Centers for Disease Control, US Department of Health and Human Services (1986) Classification System for Human T-Lymphotropic Virus Type III/Lymphadenopathy-Associated Virus Infections. Annals of Internal Medicine, 105, 234-237.
http://dx.doi.org/10.7326/0003-4819-105-2-234 - 46. Snyder, J.D. and Pickering, L.K. (2010) Viral Hepatitis. In: Kliegman, R.M., Stanton, B., Geme, J.W., Schor, N.F. and Behrman, R.E., Eds., Nelson Textbook of Pediatrics, 19th Edition, Elsevier Inc., Philadelphia, 1324-1331.
- 47. Safrin, S. (2004) Antiviral Agents. In: Katzung, B.G., Masters, S.B. and Trevo, A.J., Eds., Basic and Clinical Pharmacology, 9th Edition, The McGraw-Hill Companies, Inc., China, 1117-1156.