Open Journal of Blood Diseases
Vol.3 No.3(2013), Article ID:36935,4 pages DOI:10.4236/ojbd.2013.33020
Effect of Factor V Leiden on Thrombosis in Childhood Leukemia
![]()
1Biotechnology Institute of Ankara University, Ankara, Turkey; 2TOBB-Economy Technology University Hospital, Ankara, Turkey.
Email: *dilara2684@hotmail.com
Copyright © 2013 Dilara Fatma Akın, Nejat Akar. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received July 8th, 2013; revised August 8th, 2013; accepted August 15th, 2013
Keywords: Thrombosis; Acute Leukemia; Childhood; Factor V Leiden
ABSTRACT
Thromboembolism is an important complication in children undergoing therapy for ALL as it has the potential to impact adversely on both their survival and quality of life. The incidence of thrombosis in children with ALL varies between 1.1% and 36.7% and the actual mean is 3.2%. The aim of our study is to review the available reported data on the effect of FVL on thrombotic risk in pediatric patients with acute leukemia.
1. Introduction
Pediatric patients with acute leukemia are at high risk of both hemorrhage and thromboembolic events (TEs) [1]. Thromboembolic events are among the more frequent and serious complications of acute lymphoblastic leukemia (ALL) and its treatment [2]. Thromboembolic events are thought to result from the interaction of various factors, including effects of disease itself, central venous line, catheterization, infections, dehydration, chemotherapeutic agents such as steroids and L-asparaginase (L-Asp), and acquired or inherited prothrombotic defects (IPDs). However, the risk factors for ALL associated TE are not yet clearly understood [3,4].
Vascular insults reported in children with ALL are discussed mainly in association with acquired quantitative deficiencies of protein C, protein S, or antithrombin associated with enhanced thrombin generation [2,5,6].
Chemotherapy can influence the haemostatic system either through direct effect of the chemotherapeutic agent or through complications such as infections. Corticosteroids activate platelet function, asparaginase reduces the synthesis of natural anticoagulants and in combination they increase the risk of TE in children with ALL. Some studies have shown increased thrombin activation at diagnosis in children with ALL [7,8].
The etiology of thromboembolism is thought to be multifactorial and caused by congenital and acquired risk factors. Common inherited factors are factor V (FV) 1691 G-A and Prothrombin (PT) 20210 G-A mutations [9,10]. Factor V Leiden (FVL) is the leading cause of constitutional thrombophilia. This gene defect includes G-A transition at nucleotide 1691 in exon 10 of the Factor V gene causing APC resistance yielding to a hypercoagulation disorder. The risk of developing thrombotic episodes for persons heterozygous for the FVL mutation is about 5 - 10 folds and for those homozygous is 80 - 100 folds higher than the general population [11].
The association between cancer and thromboembolic disease has been discussed in many reported studies. Many studies suggest that thromboembolism in association with childhood leukemia is a multifactorial entity resulting from the interaction of the disease. Previous reported data on the effect of FVL on leukemia-related thrombosis is controversial [1,12-22].
The aim of the present study is to compile the available data on the thrombotic risk of FVL in pediatric patients with ALL. The data in this review was compiled from PubMed and Science Citation Index databases. “Factor V, Factor V Leiden, FVL, pediatric leukemia, thrombosis” were used as key words. Published studies with their country, number of patients, genotype distribution of FVL and references are given in Table 1.
2. Conclusion
Thromboembolism is a well recognized complication of malignancy. However, the risk factors for ALL associated TE are unclear. The pathogenesis of the cancer-re-
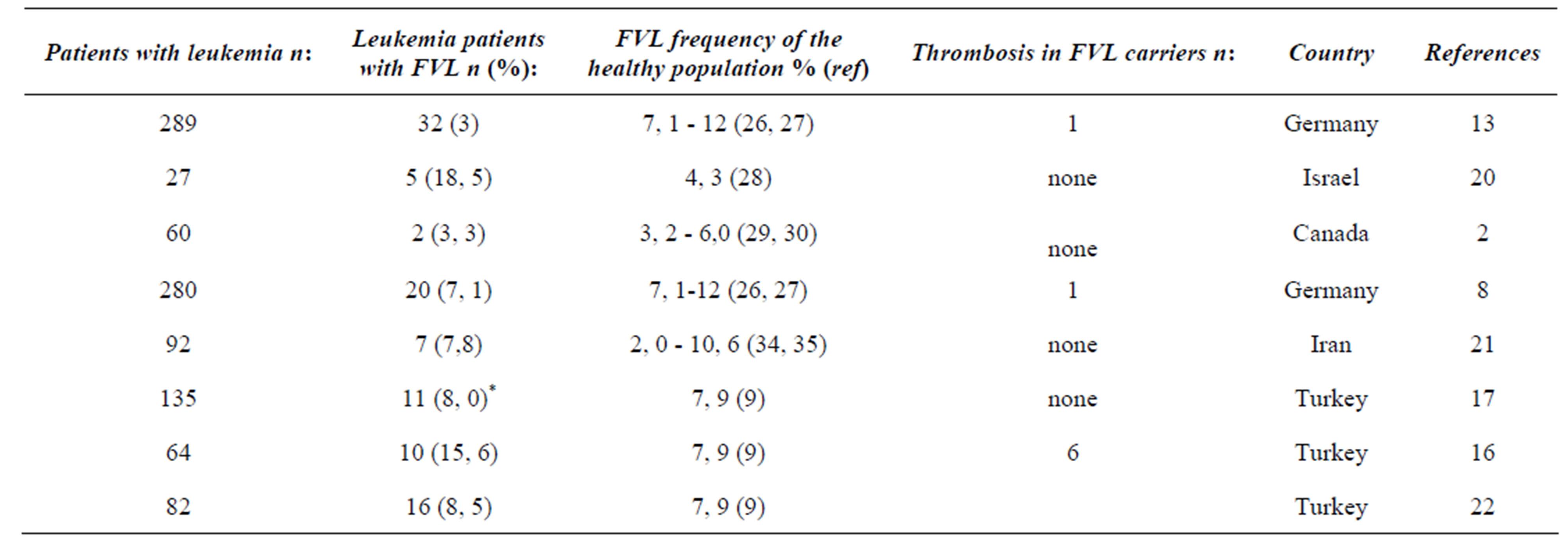
Table 1. The genotype distribution of FVL mutation in children with Leukemia and healty individuals from different country.
lated prothrombotic state is complex and reflects the action of different mechanisms, including activation of blood coagulation via procoagulant substances, impairment of fibrinolytic pathways, alterations of endothelium toward a thrombogenic state detectable in the large majority of patients with cancer. The reported incidence of DVT in children with ALL ranges between 1% and 73%. Several studies on genetic risk factors for thrombosis excluded those patients with cancer-related thrombosis. Although surgery, chemotherapy, central venous catheters, and systemic hypercoagulability were associated with venous thromboembolism (VTE) in cancer, previous reports on the association of FVL and/or prothrombin 20210A with cancer associated VTE were few and present conflicting results [13,23,24].
Nowak-Gottl et al. reported in a prospective multicenter study which focused on the role of prothrombotic risk factors in consecutively admitted newly diagnosed children with ALL carrying prothrombotic risk factors. In this study 32 of 289 consecutively admitted leukemic children (11%) treated according to the ALL-BFM 90/95 study suffered venous thromboembolism [25]. 5 (% 1.73) of them carried thrombosis relevant disease. One of them was heterozygous for the FV 1691 G-A mutation with venous sinus thromboembolism.
Rahimi et al. indicated that the frequency of FVL mutation was higher in ALL patients compared to healty individuals. The frequency of FVL G1691A polymorphism was 7.8% in patients compared to 3.2% in controls (p = 0.052) [21]. None of them carried thrombosis relevant disease.
Caruso et al. demonstrated that inherited thrombophilia increased the risk of VTE in pediatric ALL patients by approximately 8.5 folds (4.4 - 17.4). They indicated that a meta-analysis of prospective studies in childhood ALL and VTE found that symptomatic VTE was diagnosed in the central nervous system (CNS) in 50% of cases, venous VTE in 31%, and cerebral infarction or stroke in 18% [1].
Sivaslıoglu et al. reported that the thrombosis risk was significantly high in cases which had heterozygote FVL mutation. They reported that the screening for FVL mutation would be helpful in children with ALL during the diagnosis period. However they demonstrated that no significant correlation between thrombosis development and increased Lp-a levels, frequencies of MTHFR and PT 20210 A mutations [16].
In our recent study we genotyped 135 pediatric leukemia patients for these mutations. Eleven (8%) of the 135 patients were heterozygous for the FV 1691 G-A mutation. Seven (5.1%) of the patients carried the PT 20210 G-A heterozygous mutation. We suggested, that the FV 1691A and the Prothrombin 20210A mutations are not associated with TE in pediatric leukemias [17]. Akar reported that the frequency of FVL is about 8% in our healthy population [9].
Torun et al. showed that of the eighty two leukemic children, sixteen (8.5%) were carriers of FV mutation [22]. Five (6.0%) of the 82 patients had thromboembolism. One had cerebrovascular thrombosis, the other one with carotids internal thrombosis and other three had Deep Vein Thrombosis.
Elhasid et al. reported that 5 (18.5 %) of the 27 pediatric leukemia patients were carried FV 1691 G-A mutation. None of them carried thrombosis relevant disease [20].
A study by Mitchell et al. reported that none of the 22 children with TEs were positive for Factor V Leiden or prothrombin gene 20201A but 22 of 60 children had TEs, a prevalence of 36.7% without this mutation [2,18]. The North American Prophylactic Antithrombin Replacement in Kids with Acute Lymphoblastic Leukemia Treated with Asparaginase (PARKAA) study failed to show any correlation between the presence of the PT 20210A variant or the FV G1691A mutation and development of asymptomatic VTE [2].
Meta-analysis of 5 studies showed that the presence of at least one genetic prothrombotic factor was associated with an 8 folds increase in the risk of thrombosis in children with ALL [1]. Accurate identification of children with cancer who carry a baseline increased risk of thrombosis could have important therapeutic implications. For example, some drugs which are important components of modern chemotherapy regimens, such as asparaginase, are also known to increase the risk of thrombotic [18].
In conclusion, we believe that FVL mutation does not significantly increase thrombosis risk in leukemic children.
REFERENCES
- V. Caruso, L. Lacoviello, A. D. Castelnuovo, S. Storti, G. Mariani, G. D. Gaetano and M. D. Donati, “Thrombotic Complications in Childhood Acute Lymphoblastic Leukemia: A Meta-Analysis of 17 Prospective Studies Comprising 1752 Pediatric Patients,” Blood, Vol. 108, No. 7, 2006, pp. 2216-2222. http://dx.doi.org/10.1182/blood-2006-04-015511
- L. G. Mitchell, M. Andrew and K. Hanna, “A Prospective Cohort Study Determining the Prevalence of Thrombotic Events in Children with Acute Lymphoblastic Leukemia and a Central Venous Line Who Are Treated with L-Asparaginase: Results of the Prophylactic Antithrombin Replacement in Kids with Acute Lymphoblastic Leukemia Treated with Asparaginase (PARKAA) Study,” Cancer, Vol. 97, No. 2, 2003, pp. 508-516. http://dx.doi.org/10.1002/cncr.11042
- N. Santoro, P. Giordano and G. C. Del Vecchio, “Ischemic Stroke in Children Treated for Acute Lymphoblastic Leukemia: A Retrospective Study,” Journal of Pediatric Hematology/Oncology, Vol. 27, 2005, pp. 153-157. http://dx.doi.org/10.1097/01.mph.0000157379.44167.b5
- T. Celkan, H. Apak, A. Ozkan, V. Guven, T. Erkan, F. Cullu, L. Yüksel and I. Yıldız, “The Etiology of Thromboembolism in Hospitalized Children,” Turkish Pediatric Archive, Vol. 39, 2004, pp. 65-70.
- A. C. Homans, M. E. Rybak, R. L. Baglini, C. Tiarks, M. E. Steiner and E. N. Forman, “Effect of L-Asparaginase Administration on Coagulation and Platelet Function in Children with Leukemia,” Journal of Clinical Oncology, Vol. 5, 1987, p. 811.
- N. Semeraro, P. Montemurro, P. Giordano, F. Schettini, N. Santoro, D. De Mattia, D. Giordano, M. Conese and M. Collucci, “Unbalanced Coagulation-Fibrinolysis Potential during L-Asparaginase Therapy in Children with Acute Lymphoblastic Leukemia,” Thromb Haemost, Vol. 64, 1990, p. 38.
- D. E. Sutherland, I. C. Weitz and H. A. Liebman, “Thromboembolic Complications of Cancer: Epidemiology, Pathogenesis, Diagnosis, and Treatment,” American Journal of Hematology, Vol. 72, No. 1, 2003, pp. 43-52. http://dx.doi.org/10.1002/ajh.10263
- U. Nowak-Göttl, R. Junker, W. Kreuz, A. V. Eckardstein, A. Kosh, N. Nohe, R. Schobess and S. Ehrenforth, “Risk of Recurrent Venous Thrombosis in Children with Combined Prothrombotic Risk Factors,” Blood, Vol. 97, No. 4, 2001, pp. 858-862. http://dx.doi.org/10.1182/blood.V97.4.858
- N. Akar, “Factor V 1691 G-A Mutation Distribution in a Healthy Turkish Population,” Turkish Journal of Hematology, Vol. 26, 2009, pp. 9-11.
- N. Akar, M. Mısıroğlu and E. Akar, “Prothrombin Gene 20210 G-A Mutation in the Turkish Population,” American Journal of Hematology, Vol. 58, No. 3, 1998, p. 249. http://dx.doi.org/10.1002/(SICI)1096-8652(199807)58:3<249::AID-AJH20>3.0.CO;2-3
- R. M. Bertina, B. P. Koeleman and T. Koster, “Mutation in Blood Coagulation Factor V Associated with Resistance to Activated Protein C,” Nature, Vol. 369, 1994, pp. 64-67. http://dx.doi.org/10.1038/369064a0
- A. Redaelli, B. L. Laskin, J. M. Stephens, M. F. Botteman and C. L. Pashos, “A Systematic Literature Review of the Clinical and Epidemiological Burden of Acute Lymphoblastic Leukaemia (ALL),” European Journal of Cancer Care, Vol. 14, 2005, pp. 53-62. http://dx.doi.org/10.1111/j.1365-2354.2005.00513.x
- U. Nowak-Gottl, C. Wermes, R. Junker, H. G. Koch, R. Schobess, G. Fleischhack, D. Schwabe and S. Ehrenforth, “Prospective Evaluation of the Thrombotic Risk in Children with Acute Lymphoblastic Leukemia Carrying the MTHFR TT 677 Genotype, the Prothrombin G20210A Variant, and Further Prothrombotic Risk Factors,” Blood, Vol. 93, 1999, pp. 1595-1599.
- S. Ziegler, W. R. Sperr and P. Knobl, “Symptomatic Venous Thromboembolism in Acute Leukemia: Incidence, Risk Factors, and Impact on Prognosis,” Thrombosis Research, Vol. 115, No. 1-2, 2005, pp. 59-64. http://dx.doi.org/10.1016/j.thromres.2004.07.016
- B. Falanga, “Hemorrhage and Thrombosis in Acute Leukemia,” Haematologica Reports, Vol. 1, No. 9, 2005, pp. 48-51.
- S. Sivaslioglu, T. Gursel, U. Kocak and Z. Kaya, “The Risk Factors for Thrombosis in Children with Acute Lymphoblastic Leukemia,” Clinical and Applied Thrombosis/Hemostasis, 2013, pp. 278-281.
- D. F. Akin, K. Sipahi, T. Kayaalp, Y. Egin, S. Tasdelen, A. E. Kurekci and N. Akar, “Factor V Leiden and Prothrombin 20210A Mutations among Turkish Pediatric Leukemia Patients,” Leukemia Research and Treatment, 2012, pp. 345-349.
- M. Sifontes, R. Nuss, S. P. Hunger, S. Wilimas and L. Jacobsan, “The Factor V Leiden Mutation in Children with Cancer and Thrombosis,” British Journal of Haematology, Vol. 96, No. 3, 1997, pp. 484-489. http://dx.doi.org/10.1046/j.1365-2141.1997.d01-2046.x
- H. G. Ku, R. H. White, H. K. Chew, D. J. Harvey, H. Zhou and T. Wun, “Venous Thromboembolism in Patients with Acute Leukemia: Incidence, Risk Factors, and Effect on Survival,” Blood, 2009, pp. 113-117.
- R. Elhasid, N. Lanir and R. Sharon, “Prophylactic Therapy with Enoxaparin during L-Asparaginase Treatment in Children with Acute Lymphoblastic Leukemia,” Blood Coagulation & Fibrinolysis, Vol. 12, No. , 2001, pp. 367- 370. http://dx.doi.org/10.1097/00001721-200107000-00005
- Z. Rahimi, Akramipour, H. Mozafari, K. Yari, M. R. Golpaygani and A. S. Ahmadi, “Association of Factor V Leiden Mutation with Pediatric Acute lymphoblastic Leukemia in Kermanshah Province,” Journal of Hematology-Oncology and Stem Cell Research, Vol. 6, 2012, p. 10.
- Y. A. Torun, T. Patiroglu, M. A. Ozdemir, Y. Ozkul, A. Ekici and M. Karakukcu, “Inherited Prothrombotic Risk Factors in Turkish Children with Acute Lymphoblastic Leukemia: Significance of Concomitant Genetic Mutation,” Clinical and Applied Thrombosis/Hemostasis, Vol. 18, No. 2, 2012, pp. 218-221. http://dx.doi.org/10.1177/1076029611412366
- H. M. Otterson, J. Mathijssen, H. Ten Cate, M. Soesan, M. Inghels, D. J. Richel and M. H. Prins, “Symptomatic Venous Thromboembolism in Cancer Patients Treated with Chemotherapy: An Underestimated Phenomenon,” Archives of Internal Medicine, Vol. 164, No. 2, 2004, pp. 190- 194. http://dx.doi.org/10.1001/archinte.164.2.190
- E. Ramacciotti, N. Wolosker, P. Puech-Leao, E. A. Zeratti, P. R. Gusson, A. del Giglio and R. F. Franco, “Prevalence of Factor V Leiden, FII G20210A, FXIII Val34Leu and MTHFR C677T Polymorphisms in Cancer Patients with and without Venous Thrombosis,” Thrombosis Research, Vol. 109, No. 4, 2003, pp. 171-174. http://dx.doi.org/10.1016/S0049-3848(03)00179-8
- N. U. Göttl, G. Kenet and L. G. Mitchell, “Thrombosis in Childhood Acute Lymphoblastic Leukaemia: Epidemiology, Etiology, Diagnosis, Prevention and Treatment,” Best Practice & Research Clinical Haematology, Vol. 22, No. 1, 2009, pp. 103-114. http://dx.doi.org/10.1016/j.beha.2009.01.003
NOTES
*Corresponding author.

