Open Journal of Endocrine and Metabolic Diseases
Vol.05 No.07(2015), Article ID:58409,9 pages
10.4236/ojemd.2015.57012
ADRB2, ADRB3, BDKRB2 and MTNR1B Genes Related to Body fat Modulation and Its Interaction with Physical Activity and Blood Pressure
Aline Marcadenti1,2
1Department of Nutrition, Universidade Federal de Ciências da Saúde de Porto Alegre (UFSCPA), Porto Alegre, Brazil
2Postgraduate Studies Program in Cardiology, Instituto de Cardiologia/Fundação Universitária de Cardiologia do Rio Grande do Sul (IC/FUC), Porto Alegre, Brazil
Email: marcadenti@yahoo.com.br
Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 June 2015; accepted 25 July 2015; published 29 July 2015
ABSTRACT
Hypertension (HTN) is the risk factor that most contributes to mortality rates in the world, followed by physical inactivity and obesity. Despite the influence of genetic factors on the genesis of HTN, blood pressure levels are strongly influenced by environmental factors such as physical inactivity and overweight, characterizing it as a polygenic disease. Genetic components and environmental factors such as physical exercise may modulate the phenotype of individuals predisposed to medical conditions such as HTN, independently of modifiable factors such as increased levels of adiposity; however, studies have shown that polymorphic forms detected in genes involved in the mechanisms of blood pressure regulation and also related to body fat modulation may interact with physical activity levels and HTN. The aim of this article is to review the interactions between polymorphisms in ADRB2, ADRB3, BDKRB2 and MTNR1B genes, physical activity and blood pressure.
Keywords:
Receptors, Adrenergic, Beta-2; Receptor, Bradykinin B, Obesity, Hypertension

1. Introduction
Hypertension (HTN) is the leading modifiable risk factor for mortality worldwide [1] , accounting for more than 40% of the deaths caused by cardiovascular diseases (CVDs), renal diseases and type 2 diabetes mellitus [2] . It is estimated that 29% of world population, or 1.56 billion people, will have HTN by 2025, with approximately three-fourths of them living in developing countries [3] . In Brazil, the surveillance of Protective and Risk Factors for Chronic Diseases by Telephone Survey (VIGITEL) [4] , conducted on a probability sample of telephone households drawn from telephone directories of the capitals in the country, and involving 52,929 individuals, showed a prevalence of HTN about 26% (95% CI 23.2 - 28.0) (22.9% of men [95% CI 19.3% - 26.6%] and 27.8% of women [95% CI 24.7% - 30.9%]).
Physical inactivity, considered the fourth leading risk factor for global mortality [1] , caused an estimated 5.3 million premature deaths in the world in 2008, representing approximately 9% (95% CI 5.1% - 12.5%) of global mortality rates [5] . According to VIGITEL, nearly 50% of Brazilian population does not reach the recommended amount of physical activity, especially women (57.4%). The survey classified as insufficiently active those individuals who had not done any leisure time physical activity in the preceding three months, did not do moderate or vigorous activity for work, transportation (walking or cycling for at least 10 minutes) or household chores [4] .
Obesity is one of the main modifiable risk factors that contributes to the global burden of disease and independently associates with cardiovascular mortality and general mortality (fifth major cause) rates [1] [6] . By 2030, the number of overweight (body mass index, BMI ≥ 25 kg/m2) and obese (BMI ≥ 30 kg/m2) adults in the world is projected to be 2.16 billion and 1.12 billion respectively [7] . In Brazil, approximately 51% of individuals are overweight, and 17.5% obese [4] .
HTN and obesity are considered polygenic diseases, and in the last years different genetic approaches have been studied regarding interactions with environment factors (such as physical activity). In studies on associations of candidate genes, gene selection is based on physiopathological mechanisms and biological implications of certain genes in a disease. Genome-wide association studies (GWAS), also known as whole-genome association studies are considered a state of the art tool for genetic screening and identification of genetic variants associated with a condition. In contrast to researches on new candidate genes, in which a restricted number of chromosomal regions are simultaneously studied, GWAS investigate the complete genome, and the approach is therefore said to be non-candidate-driven. Additionally, GWAS focus on associations between a polymorphism (SNP), rather than a gene, and a disease. Thus, the aim of this article is to review the interactions between SNPs in ADRB2, ADRB3, BDKRB2 and MTNR1B genes (related to obesity and body fat modulation), physical activity and blood pressure/HTN.
2. Pathophysiology of HTN
The control of blood pressure is complex and involves hemodynamic, neural, and hormonal mechanisms. Arterial pressure is determined by cardiac output (CO) and systemic vascular resistance (SVR), and increases in both CO and SVR may lead to HTN. The autonomic nervous system (ANS) plays a central role in pressure control: stimulation of the sympathetic nervous system (SNS) can cause vasoconstriction or vasodilation, and malfunction of the ANS may increase CO and SVR via α- and β-adrenergic receptors [8] [9] .
There is increasing evidence that the physiological response to a sodium load is the most important mechanism involved in the genesis of HTN. Studies involving acculturated and unacculturated populations (who use and do not use salt in their diet respectively) have shown an increase in blood pressure with age in acculturated tribes, while in unacculturated tribes, the blood pressure remained relatively constant throughout adult life [10] . However, the influence of salt consumption on blood pressure levels may be influenced by genetic factors, since only a part of the population have increased blood pressure in response to high salt intake [11] .
The renin-angiotensin system is the most important endocrine system involved in the control of blood pressure. Renin converts angiotensinogen into angiotensin I, which is converted to angiotensin II (potent vasoconstrictor) by the enzyme angiotensin-converting enzyme (ACE) in several tissues. In addition to the circulating renin-angiotensin system, locally-expressed renin-angiotensin systems in the kidney, liver, heart and arterial tree, acting in a paracrine manner, may be involved in the control of arterial pressure [8] [9] . The brain renin-angi- otensin system modulates melatonin and angiotensin synthesis in the pineal gland, and may affect both blood pressure levels as well as its circadian rhythm [12] .
In addition, reduced activity of the kallikrein-kinin system has a significant role in the development of HTN. Kallikrein is the main precursor of bradykinin and kallidin (vasodilator kinins), which are degraded by kininases, especially kininase II (or ACE). Therefore, increased levels of ACE may result in increased synthesis of angiotensin II and deactivation of bradykinin [8] [9] .
Obesity is commonly associated with hormonal, renal, hemodynamic and neuroendocrine dysfunctions involved in the etiopathogeny of HTN. Fat deposition (mainly visceral fat) resulting from an imbalance between energy intake and energy expenditure may affect numerous mechanisms, including insulin resistance, sodium retention and increased activity of SNS [13] . The negative effects of visceral fat deposition on cardiometabolic parameters are more pronounced among women than men [14] . The renin-angiotensin-aldosterone system is also altered and may play a role in the development of obesity. A component of this system, angiotensinogen, contributes to adipocyte differentiation (adipogenesis) and is positively associated with fat mass, adipose tissue metabolism and leptin plasma levels [15] , and hence influences mechanisms involved in the regulation of energy expenditure and food intake.
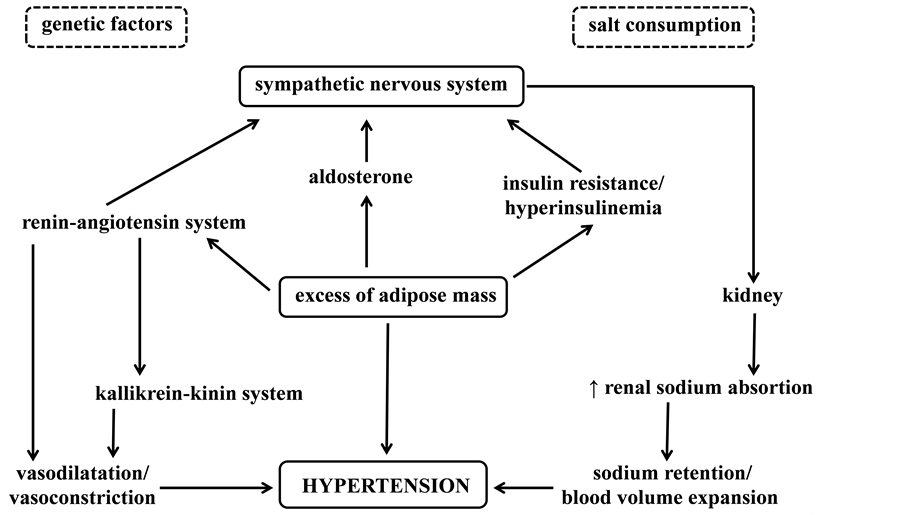
Figure 1 summarizes the main mechanisms involved in the genesis of HTN.
3. Genetics, HTN and Physical Activity
Mutations of specific genes are associated with elevated blood pressure and development of HTN. However, more frequent, common genetic changes are more likely to be involved in the genesis of HTN individually, and the accurate determination of the relative contribution of these genes to the increase of blood pressure is particularly difficult. HTN is twice more common in individuals with one or two hypertensive relatives. Epidemiological studies involving families and twins have suggested that genetic factors account for 30% - 50% of the variation of arterial pressure among populations [8] [16] . The familial aggregation of HTN is partly due to the lifestyle shared by family members living in the same household, especially physical activity and eating patterns. Therefore, HTN is a polygenic, hereditary disease (or multifactorial), caused by the interaction of multiple environmental factors with variations in genes generally located in different chromosomes [17] .
Additionally, similarly to what has been described for eating behavior and appetite, genetic factors may also exert an influence on physical activity patterns [18] . Studies using animal models indicate that mechanisms of the central nervous system (CNS) act on the regulation of daily physical activity [19] , and clinical studies suggest that genetic factors account for 30% - 78% of the variability in daily level of physical activity reported in epidemiological studies [20] [21] . However, although the genetic contribution on levels of physical activity is evident, no loci with robust associations have been described so far.
Besides, the role of physical inactivity and consequent reduction of metabolic rates on the increase in body fat deposition [18] has been questioned, since longitudinal studies have shown that basal levels of physical activity
Figure 1. Schematic illustration about pathophysiology of HTN. Adaptad from Francischetti, E.A et al. [13] .
do not predict adiposity; on the contrary, increased adiposity is associated with reduced physical activity over time [22] [23] . It is estimated that genetic factors account for 40% - 70% of variation in adiposity in humans [24] and, interestingly, changes in lifestyle, especially those related to eating and physical activity habits, may affect susceptible genes and greatly contribute to the development of obesity in predisposing individuals and populations [25] [26] .
4. Beta-Adrenergic Receptors: ADRB2 and ADRB3 Genes
The adrenergic receptors (or adrenoceptors) belong to the class of G protein-coupled receptors, and are activated by endogenous catecholamines, especially epinephrine and norepinephrine. Beta-2 adrenergic receptors are predominant in smooth muscles and their activation lead to relaxation of gastrointestinal smooth muscle, lipolysis in adipose tissue, increased renin secretion from kidney, and dilation of arteries in skeletal muscle [27] . The receptors also mediate bronchodilation and ventricular function, having a direct impact on pulmonary and cardiovascular responses to physical exercise [28] . Beta-3 adrenergic receptors mediate metabolic effects, especially lipolysis in adipose tissue and thermogenesis [29] .
Beta-2 and beta-3 adrenergic receptors are encoded by ADRB2 (adrenoceptor beta 2, surface) and ADRB3 (adrenoceptor beta 3) genes, located on chromosomes 5 and 8 respectively. Different polymorphic forms in ADRB2 detected by GWAS have been associated with nocturnal asthma, type 2 diabetes mellitus (T2DM), obesity [30] , and decrease in mean arterial pressure at rest, during, and after physical activity [31] . Individuals with genetic alterations in ADRB3 have lower energy expenditure and basal metabolic rate as compared with those without genetic alterations [32] .
The most studied polymorphisms in ADRB2 and ADRB3 genes are rs1042713 (Arg16Gly) and rs1042714 (Gln27Glu) in the former, and rs4994 (Trp64Arg) and rs4998 (G > C) in the latter. However, studies investigating interactions between SNPs and gender, physical activity, food intake, adiposity and blood pressure are scarce and inconsistent, making interpretation and generalization of results difficult.
A meta-analysis of 18 observational studies showed that Gln27Glu was associated with 16% increased obesity risk (OR: 1.16, 95% CI: 1.04 - 1.30), whereas no association was found between Arg16Gly and obesity. In addition, neither Gln27Glu nor Arg16Gly was significantly associated with obesity following stratification of the population by gender [30] .
A study conducted on 132 (111 women) severely obese subjects in Brazil, individuals carrying Glu27 allele had 4.7 greater risk of HTN compared to non-carriers [33] . Obese, postmenopausal women, homozygous for Glu27 had increased body fat mass and decreased lean mass measured by bioelectrical impedance analysis [34] . In addition, obese women, carriers of the Glu27 allele, had increased respiratory quotient (determined by indirect calorimetry) and decreased fatty acid oxidation during and after aerobic exercise sessions, suggesting an interaction between body adiposity, energy expenditure and physical exercise [35] .
Significant interactions between Arg16Gly SNP and vigorous physical activity on visceral and subcutaneous abdominal adipose tissue were found among American adolescents. Homozygous for the Gly16 allele benefited less from the effect of physical activity on reduction of fat deposits [36] . A study conducted on Spanish children and adolescents, homozygous for Glu27 (Gln27Glu), showed increased risk for obesity (OR 4.84, 95% CI 1.37 - 17.10) and a significant interaction between hours of TV watching and the Glu27 allele among girls. Watching TV more than 12.5 hours a week increased the obesity risk (OR 6.05, 95% CI 1.31 - 27.71) [37] . In a Japanese population, no significant interaction was found between the presence of Arg64 allele of Trp64Arg in the ADRB3 gene, levels of physical activity and percentage of body fat (detected by bioelectrical impedance) [38] .
In a study investigating the effect of Arg16Gly, Gln27Glu and Trp64Arg polymorphisms on weight loss among adult obese (n = 109) women in Brazil, a seven-week dietary and physical activity intervention did not result in significant differences in weight loss between polymorphisms carriers and non-carriers [39] . Interestingly, the presence of the Arg64 allele in the ADRB3 gene was associated with lower BMI and high density lipoprotein cholesterol levels in individuals with T2DM in Brazil [40] , and the Arg64 allele was negatively associated with HTN in a population of approximately 1800 in China [41] . Results from the DiOGenes (Diet, Obesity and Genes) study on European populations revealed that the presence of G allele of rs4998 in the ADRB3 gene enhanced the effect of waist circumference reduction of high-protein diets [42] .
5. Bradykinin Receptor B2: BDKRB2 Gene
The hormone bradykinin, discovered by Brazilian physiologists, is an inflammatory mediator with vasodilation activity, contributing to prostaglandin and nitric oxide formation. Bradykinin exerts its effects via two receptor subtypes, the B1 receptor and the B2 receptor. B2 receptor is encoded by the BDKRB2 (bradykinin receptor B2) gene located on chromosome 14, and expressed predominantly in smooth muscle. Bradykinin may also stimulate proinflammatory interleukins, e.g. IL-6 and IL-8, in adipose tissue through its action on receptors expressed by adipocytes [43] . Bradykinin is also involved in glucose uptake by skeletal muscle and adipocytes, and the metabolite des-Arg9-bradykinin, which has a high affinity for the B1 receptor, regulates leptin sensitivity in hypothalamus and modulates energy balance [44] .
Changes in BDKRB2 gene expression may also modulate the vascular response to physical activity, which would in part explain the individual variability of the beneficial effect of exercise on cardiometabolic outcomes. Alteration or deletion of alleles is positively associated with metabolic efficacy and performance during exercise [45] [46] . On the other hand, polymorphic changes of the BDKRB2 gene would be strongly associated with blood pressure variations in specific populations [47] .
In spontaneously hypertensive rats, an overexpression of bradykinin B1 and B2 receptors is found in the hypothalamus [48] , and such changes in receptor expression in the hypothalamus are associated with cardiovascular dysfunctions in obese, leptin knockout (ob/ob) rats. Yet, in obese ob/ob mice, B2 receptor activity is also detected in the white adipose tissue, which is the main site of neuroendocrine regulation of energy metabolism, along with the hypothamalus [49] . These findings suggest a connection between the kallikrein-kinin system and obesity, and the involvement of kinin receptors in the etiopathogeny of metabolic syndrome. Animal models also suggest that kallikrein gene expression (bradykinin precursor) in the kidney is regulated by dietary sodium chloride [50] .
Associations between rs1799722 (−58T/C) and rs11847625 (C > G) polymorphisms in the BDKRB2 gene, blood pressure and adiposity have been reported in some populations, especially among Orientals. In addition, the SNP +9/−9 in the BDKRB2 gene has been widely investigated in elite athletes. For instance, in 71 Brazilian athletes undergoing 16 weeks of aerobic training, increased ACE activity was observed in individuals homozygous for the −9 allele, although the genotype did not significantly affect blood pressure levels [51] .
A meta-analysis of 11 studies (including more than 3,800 individuals) showed that the risk of HTN is increased by 24% (OR 1.24, 95% CI 1.05 - 1.46) by the presence of the -58C allele in the BDKRB2 gene, especially in Asian populations [47] . The interaction between -58T/C and rs220721 polymorphisms in the MAS1 gene (also involved in the renin-angiotensin system) increased the risk for obesity by 82% in a Chinese cohort of adults [52] .
The interaction between genetics and sodium sensitivity was evaluated in more than 1900 Chinese subjects who received a 7-day low-sodium diet (3 g/day) followed by a 7-day high-sodium diet (18 g/day). The presence of the rare C allele in the rs11847625 was associated with attenuated systolic blood pressure and mean arterial pressure decreases in response to the low-sodium intervention (z = −3.798) [53] . Interestingly, in the same study group, an interaction between rs945039 and BDKRB2 polymorphisms, blood pressure levels and physical activity level was observed. Also, among physically inactive individuals, the presence of the rare allele was associated with lower diastolic blood pressure, and no significant differences in genotype were found in physically active individuals [54] .
6. Melatonin Receptors: MTNR1B Gene
Melatonin (N-acetyl-5-methoxy tryptamine), a hormone synthesized from the amino acid tryptophan and secreted by the pineal gland, is involved in the regulation of several organic functions, such as sleep and circadian rhythms, anti-inflammatory and antioxidant activities, energy metabolism, with emphasis on modulation of daily energy expenditure and body mass [55] . The main biological effects of melatonin are mediated by MT1 and MT2 receptors, which are members of a subfamily of G protein-coupled receptors, and identified in tissues related to the cardiovascular system, metabolic control and energy expenditure [56] . Administration of melatonin (5 mg/day) for 60 days significantly decreased systolic arterial pressure (−12.3 ± 1.2 mmHg) and diastolic arterial pressure (−6.7 ± 1.4 mmHg) in individuals with metabolic syndrome [57] , supporting the participation of melatonin in blood pressure modulation in CNS.
The MTNR1B (melatonin receptor 1B) gene encodes the MT1 melatonin receptor, which inhibits insulin secretion through its effect on cyclic guanosine monophosphate (cGMP) formation. Plasma glucose levels are significantly lower in MTNR1B knockout animals, and a meta-analysis involving 23 studies (172,963 subjects) suggested a positive association between the presence of polymorphisms in MTNR1B gene and T2DM in Caucasians [G allele: OR = 1.09, 95% CI 1.06 - 1.13, P(Z) < 10−5; dominant model: OR = 1.16, 95% CI 1.07 - 1.25, P(Z) < 10−5; recessive model: OR = 1.19, 95% CI 1.10 - 1.28, P(Z) < 10−5] [58] (insulin resistance and T2DM are risk factors for HTN).
In addition, GWAS have identified associations between rs10830962 (G > C) and rs10830963 (G > C) poly-morphisms in the MTNR1B gene, blood pressure values and adiposity. Results from these studies, however, are still scarce, and available data regarding interactions with energy expenditure, diet and physical activity are limited.
The presence of G alleles in the rs10830962 and rs10830963 SNPs was independently associated with higher mean arterial pressure among individuals with coronary arterial disease and HTN [59] . On the other hand, no interaction between these polymorphisms, arterial pressure and HTN was found in elderly Oriental people in a population-based study [60] .
In the LOGIC-trial (Long-term effects of a lifestyle intervention in obesity and genetic influence in children), adolescents and children participating in the study followed a weight-loss program consisting of reduction in energy intake (by 500 kcal/day), physical activity (11 h/week) and behavioral therapy. In the beginning of the study, the G allele of the rs10830963 SNP was associated with higher BMI values. However, no interaction between genotype, weight loss measures and anthropometric parameters was found after 6 weeks of intervention [61] . In the Preventing Overweight Using Novel Dietary Strategies (POUNDS LOST) trial, 722 overweight and obese adult subjects were assigned to one of four diets (two high-protein diets and two high-fat diets). The presence of the G allele of rs10830963 significantly associated with increased respiratory quotient measured by indirect calorimetry after two years. Additionally, an interaction between genotype, changes in the respiratory quotient and fat intake was found, suggesting a long-term genetic modulation of energy expenditure, which may be affected by dietary fat (the G allele significantly associated with increased respiratory quotient in low-fat diets as compared with high-fat diets). The effect of physical activity was not evaluated in this study [62] .
7. Conclusion
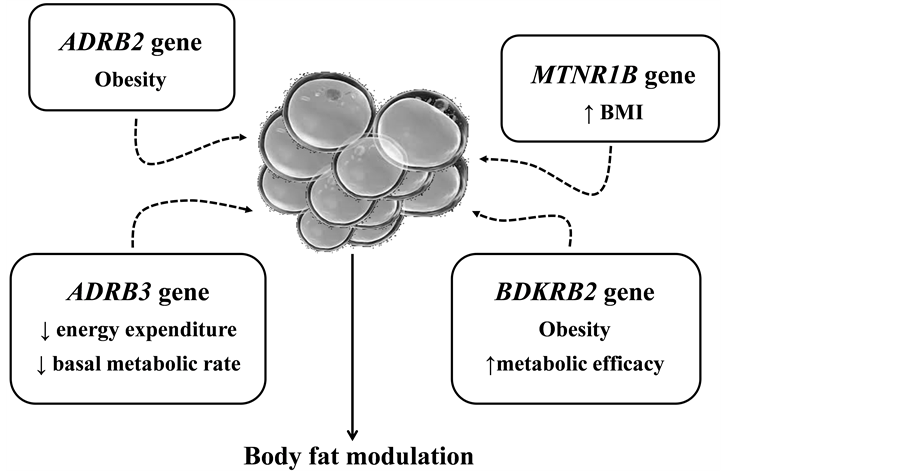
Polymorphism in ADRB2, ADRB3, BDKRB2 and MTNR1B genes seems to interact with physical activity and blood pressure by different mechanisms, mainly by modulating energy expenditure and body fat storage (Figure 2). However, most of the studies that have identified interactions between polymorphic forms of these genes, HTN/obesity phenotypes and physical activity were conducted among specific populations, and some findings
Figure 2. Possible role of ADRB2, ADRB3, BDKRB2 and MTNR1B genes in the body fat modulation.
are based on results obtained from animal models. Thus, results must be interpreted with caution and more studies among different ethnic groups should be conducted in order to replicate them.
Cite this paper
AlineMarcadenti,11, (2015) ADRB2, ADRB3, BDKRB2 and MTNR1B Genes Related to Body fat Modulation and Its Interaction with Physical Activity and Blood Pressure. Open Journal of Endocrine and Metabolic Diseases,05,88-97. doi: 10.4236/ojemd.2015.57012
References
- 1. World Health Organization (WHO) (2009) Global Health Risks: Mortality and Burden of Disease Attributable to Selected Major Risks. WHO Press, Geneva.
- 2. Global Burden of Metabolic Risk Factors for Chronic Diseases Collaboration (2014) Cardiovascular Disease, Chronic Kidney Disease, and Diabetes Mortality Burden of Cardiometabolic Risk Factors from 1980 to 2010: A Comparative Risk Assessment. The Lancet Diabetes & Endocrinology, 2, 634-647.
http://dx.doi.org/10.1016/S2213-8587(14)70102-0 - 3. Kearney, P.M., Whelton, M., Reynolds, K,, Muntner, P., Whelton, P.K. and He, J. (2005) Global Burden of Hypertension: Analysis of Worldwide Data. The Lancet, 365, 217-223. http://dx.doi.org/10.1016/S0140-6736(05)70151-3
- 4. Brasil. Ministério da Saúde. Secretaria de Vigilancia em Saúde. (2014) Vigitel Brazil 2013: Protective and Risk Factors for Chronic Diseases by Telephone Survey. Ministério da Saúde, Secretaria de Vigilancia em Saúde. Ministério da Saúde, Brasília.
- 5. Lee, I.M., Shiroma, E.J., Lobelo, F., Puska, P., Blair, S.N. and Katzmarzyk, P.T., Lancet Physical Activity Series Working Group (2012) Effect of Physical Inactivity on Major Non-Communicable Diseases Worldwide: An Analysis of Burden of Disease and Life Expectancy. The Lancet, 380, 219-229.
http://dx.doi.org/10.1016/S0140-6736(12)61031-9 - 6. Pischon, T., Boeing, H., Hoffmann, K., Bergmann, M., Schulze, M.B., Overvad, K., et al. (2008) General and Abdominal Adiposity and Risk of Death in Europe. New England Journal of Medicine, 359, 2105-2120. http://dx.doi.org/10.1056/NEJMoa0801891
- 7. Kelly, T., Yang, W., Chen, C.S., Reynolds, K. and He, J. (2008) Global Burden of Obesity in 2005 and Projections to 2030. International Journal of Obesity (Lond), 32, 1431-1437.
http://dx.doi.org/10.1038/ijo.2008.102 - 8. Beevers, G., Lip, G.Y.H. and O’Brien, E. (2001) ABC of Hypertension: The Pathophysiology of Hypertension. BMJ, 322, 912-916. http://dx.doi.org/10.1136/bmj.322.7291.912
- 9. Sanjuliani, A.F. (2002) Pathophysiology of Hypertension: Useful Theoretical Concepts for Clinical Practice. Revista SOCERJ, 15, 210-218.
- 10. Carvalho, J.J., Baruzzi, R.G., Howard, P.F., Poulter, N., Alpers, M.P., Franco, L.J., et al. (1989) Blood Pressure in Four Remote Populations in the INTERSALT Study. Hypertension, 14, 238-246.
http://dx.doi.org/10.1161/01.HYP.14.3.238 - 11. Schlaich, M.P., Klingbeil, A.U., Jacobi, J., Delles, C., Schneider, M.P., Schmidt, B.M., et al. (2002) Altered Aldosterone Response to Salt Intake and Angiotensin II Infusion in Young Normotensive Men with Parental History of Arterial Hypertension. Journal of Hypertension, 20, 117-124.
http://dx.doi.org/10.1097/00004872-200201000-00017 - 12. Campos, L.A., Cipolla-Neto, J., Amaral, F.G., Michelini, L.C., Bader, M. and Baltatu, O.C. (2013) The Angiotensin-Melatonin Axis. International Journal of Hypertension, 2013, Article ID: 521783.
- 13. Francischetti, E.A. and Genelhu, V.A. (2007) Obesity-Hypertension: An Ongoing Pandemic. International Journal of Clinical Practice, 61, 269-280. http://dx.doi.org/10.1111/j.1742-1241.2006.01262.x
- 14. Nielsen, S., Guo, Z., Johnson, C.M., Hensrud, D.D. and Jensen, M.D. (2004) Splanchnic Lipolysis in Human Obesity. Journal of Clinical Investigation, 113, 1582-1588. http://dx.doi.org/10.1172/JCI21047
- 15. Hall, J.E., da Silva, A.A., do Carmo, J.M., Dubinion, J., Hamza, S., Munusamy, S., et al. (2010) Obesity-Induced Hypertension: Role of Sympathetic Nervous System, Leptin, and Melanocortins. Journal of Biological Chemistry, 285, 17271-17276. http://dx.doi.org/10.1074/jbc.R110.113175
- 16. Miall, W.E. and Oldham, P.D. (1963) The Hereditary Factor in Arterial Blood-Pressure. BMJ, 1, 75-80. http://dx.doi.org/10.1136/bmj.1.5323.75
- 17. Ehret, G.B. and Caulfield, M.J. (2013) Genes for Blood Pressure: An Opportunity to Understand Hypertension. European Heart Journal, 34, 951-961. http://dx.doi.org/10.1093/eurheartj/ehs455
- 18. Bauman, A.E., Reis, R.S., Sallis, J.F., Wells, J.C., Loos, R.J. and Martin, B.W., Lancet Physical Activity Series Working Group (2012) Correlates of Physical Activity: Why Are Some People Physically Active and Others Not? Lancet, 380, 258-271. http://dx.doi.org/10.1016/S0140-6736(12)60735-1
- 19. Klingberg, F. and Klengel, S. (1993) Lesions in Four Parts of the Basal Forebrain Change Basic Behavior in Rats. NeuroReport, 4, 639-642. http://dx.doi.org/10.1097/00001756-199306000-00009
- 20. Simonen, R.L., Perusse, L., Rankinen, T., Rice, T., Rao, D.C. and Bouchard, C. (2002) Familial Aggregation of Physical Activity Levels in the Quebec Family Study. Medicine & Science in Sports & Exercise, 34, 1137-1142. http://dx.doi.org/10.1097/00005768-200207000-00014
- 21. Aaltonen, S., Ortega-Alonso, A., Kujala, U.M. and Kaprio, J. (2010) A Longitudinal Study on Genetic and Environmental Influences on Leisure Time Physical Activity in the Finnish Twin Cohort. Twin Research and Human Genetics, 13, 475-481. http://dx.doi.org/10.1375/twin.13.5.475
- 22. Ekelund, U., Brage, S., Besson, H., Sharp, S. and Wareham, N.J. (2008) Time Spent Being Sedentary and Weight Gain in Healthy Adults: Reverse or Bidirectional Causality? American Journal of Clinical Nutrition, 88, 612-617.
- 23. Metcalf, B.S., Hosking, J., Jeffery, A.N., Voss, L.D., Henley, W. and Wilkin, T.J. (2011) Fatness Leads to Inactivity, but Inactivity Does Not Lead to Fatness: A Longitudinal Study in Children. Archives of Disease in Childhood, 96, 942-947. http://dx.doi.org/10.1136/adc.2009.175927
- 24. Allison, D.B., Faith, M.S. and Nathan, J.S. (1996) Risch’s Lambda Values for Human Obesity. International Journal of Obesity and Related Metabolic Disorders, 20, 990-999.
- 25. Bouchard, C. (2008) Gene-Environment Interactions in the Etiology of Obesity: Defining the Fundamentals. Obesity, 16, S5-S10. http://dx.doi.org/10.1038/oby.2008.528
- 26. Manolio, T.A. (2010) Genomewide Association Studies and Assessment of the Risk of Disease. New England Journal of Medicine, 363, 166-176. http://dx.doi.org/10.1056/NEJMra0905980
- 27. Chorostowska-Wynimko, J. (2002) Mechanism of B2-Agonists Action and Safety Aspects. Polski Merkuriusz Lekarski, 12, 441-444.
- 28. Snyder, E.M., Johnson, B.D. and Joyner, M.J. (2008) Genetics of β2-Adrenergic Receptors and the Cardiopulmonary Response to Exercise. Exercise and Sport Sciences Reviews, 2, 98-105.
http://dx.doi.org/10.1097/JES.0b013e318168f276 - 29. Balligand, J.L. (2013) β3-Adrenoreceptors in Cardiovascular Diseases: New Roles for an “Old” Receptor. Current Drug Delivery, 10, 64-66. http://dx.doi.org/10.2174/1567201811310010011
- 30. Zhang, H., Wu, J. and Yu, L. (2014) Association of Gln27Glu and Arg16Gly Polymorphisms in β2-Adrenergic Receptor Gene with Obesity Susceptibility: A Meta-Analysis. PLoS ONE, 9, e100489.
http://dx.doi.org/10.1371/journal.pone.0100489 - 31. Snyder, E.M., Beck, K.C., Dietz, N.M., Eisenach, J.H., Joyner, M.J., Turner, S.T., et al. (2006) Arg16Gly Polymorphism of the β2-Adrenergic Receptor is Associated with Differences in Cardiovascular Function at Rest and during Exercise in Humans. The Journal of Physiology, 571, 121-30.
http://dx.doi.org/10.1113/jphysiol.2005.098558 - 32. Sipil?inen, R., Uusitupa, M., Heikkinen, S., Rissanen, A. and Laakso, M. (1997) Polymorphism of the β3-Adrenergic Receptor Gene Affects Basal Metabolic Rate in Obese Finns. Diabetes, 46, 77-80.
http://dx.doi.org/10.2337/diabetes.46.1.77 - 33. Villares, S.M., Mancini, M.C., Gomez, S., Charf, A.M., Frazzatto, E. and Halpern, A. (2000) Association between Polymorphism Gln27Glu of β2-Adrenergic Receptor and Hypertension in Morbid Obese. Arquivos Brasileiros de Endo crinologia e Metabologia, 44, 72-80.
- 34. Rauhio, A., Uusi-Rasi, K., Nikkari, S.T., Kannus, P., Siev?nen, H. and Kunnas, T. (2013) Association of the FTO and ADRB2 Genes with Body Composition and Fat Distribution in Obese Women. Maturitas, 76, 165-171. http://dx.doi.org/10.1016/j.maturitas.2013.07.004�
- 35. Macho-Azcarate, T., Marti, A., González, A., Martinez, J.A. and Iba?ez, J. (2002) Gln27Glu Polymorphism in the beta2 Adrenergic Receptor Gene and Lipid Metabolism during Exercise in Obese Women. International Journal of Obesity and Related Metabolic Disorders, 26, 1434-1441.
http://dx.doi.org/10.1038/sj.ijo.0802129 - 36. Lagou, V., Liu, G., Zhu, H., Stallmann-Jorgensen, I.S., Gutin, B., Dong, Y., et al. (2011) Lifestyle and Socioeconomic-Status Modify the Effects of ADRB2 and NOS3 on Adiposity in European-American and African-American Adolescents. Obesity, 19, 595-603. http://dx.doi.org/10.1038/oby.2010.224
- 37. Ochoa, M.C., Moreno-Aliaga, M.J., Martínez-González, M.A., Martínez, J.A. and Marti, A., GENOI Members (2006) TV Watching Modifies Obesity Risk Linked to the 27Glu Polymorphism of the ADRB2 Gene in Girls. International Journal of Pediatric Obesity, 1, 83-88. http://dx.doi.org/10.1080/17477160600650386
- 38. Nakashima, H., Omae, K., Nomiyama, T., Yamano, Y., Takebayashi, T. and Sakurai, Y. (2013) Beta-3-Adrenergic Receptor Trp64Arg Polymorphism: Does It Modulate the Relationship between Exercise and Percentage of Body Fat in Young Adult Japanese Males? Environmental Health and Preventive Medicine, 18, 323-329. http://dx.doi.org/10.1007/s12199-012-0325-3
- 39. Saliba, L.F., Reis, R.S., Brownson, R.C., Hino, A.A., Tureck, L.V., Valko, C., et al. (2014) Obesity-Related Gene ADRB2, ADRB3 and GHRL Polymorphisms and the Response to a Weight Loss Diet Intervention in Adult Women. Genetics and Molecular Biology, 37, 15-22.
http://dx.doi.org/10.1590/S1415-47572014000100005 - 40. Brondani, L.A., Duarte, G.C., Canani, L.H. and Crispim, D. (2014) The Presence of at Least Three Alleles of the ADRB3 Trp64Arg (C/T) and UCP1-3826A/G Polymorphisms Is Associated with Protection to Overweight/Obesity and with Higher High-Density Lipoprotein Cholesterol Levels in Caucasian-Brazilian Patients with Type 2 Diabetes. Metabolic Syndrome and Related Disorders, 12, 16-24.
http://dx.doi.org/10.1089/met.2013.0077 - 41. Wang, L., Zhang, B., Li, M., Li, C., Liu, J., Liu, Y., et al. (2014) Association between Single-Nucleotide Polymorphisms in Six Hypertensive Candidate Genes and Hypertension among Northern Han Chinese Individuals. Hypertension Research, 37, 1068-1074. http://dx.doi.org/10.1038/hr.2014.124
- 42. Larsen, L.H., Angquist, L., Vimaleswaran, K.S., Hager, J., Viguerie, N., Loos, R.J., et al. (2012) Analyses of Single Nucleotide Polymorphisms in Selected Nutrient-Sensitive Genes in Weight-Regain Prevention: The DIOGENES Study. American Journal of Clinical Nutrition, 95, 1254-1260.
http://dx.doi.org/10.3945/ajcn.111.016543 - 43. Catalioto, R.M., Valenti, C., Liverani, L., Giuliani, S. and Maggi, C.A. (2013) Characterization of a Novel Proinflammatory Effect Mediated by BK and the Kinin B? Receptor in Human Preadipocytes. Biochemical Pharmacology, 86, 508-520. http://dx.doi.org/10.1016/j.bcp.2013.06.005�
- 44. Mori, M.A., Araújo, R.C., Reis, F.C., Sgai, D.G., Fonseca, R.G., Barros, C.C., et al. (2008) Kinin B1 Receptor Deficiency Leads to Leptin Hypersensitivity and Resistance to Obesity. Diabetes, 57, 1491-1500. http://dx.doi.org/10.2337/db07-1508
- 45. Puthucheary, Z., Skipworth, J.R., Rawal, J., Loosemore, M., Van Someren, K. and Montgomery, H.E. (2011) The ACE Gene and Human Performance: 12 Years On. Sports Medicine, 41, 433-448.
http://dx.doi.org/10.2165/11588720-000000000-00000 - 46. Saunders C.J., Xenophontos, S.L., Cariolou, M.A., Anastassiades, L.C., Noakes, T.D. and Collins, M. (2006) The Bradykinin β2 Receptor (BDKRB2) and Endothelial Nitric Oxide Synthase 3 (NOS3) Genes and Endurance Performance during Ironman Triathlons. Human Molecular Genetics, 15, 979-987.
http://dx.doi.org/10.1093/hmg/ddl014 - 47. Li, Y.Y., Zhang, H., Xu, J., Qian, Y., Lu, X.Z., Yang, B., et al. (2012) Bradykinin β2 Receptor-58T/C Gene Polymorphism and Essential Hypertension: A Meta-Analysis. PLoS ONE, 7, e43068.
http://dx.doi.org/10.1371/journal.pone.0043068 - 48. Qadri, F., H?user, W., J?hren, O. and Dominiak, P. (2002) Kinin B1 and B2 Receptor mRNA Expression in the Hypothalamus of Spontaneously Hypertensive Rats. Canadian Journal of Physiology and Pharmacology, 80, 258-263. http://dx.doi.org/10.1139/y02-051�
- 49. Abe, K.C., Mori, M.A. and Pesquero, J.B. (2007) Leptin Deficiency Leads to the Regulation of Kinin Receptors Expression in Mice. Regulatory Peptides, 138, 56-58.
http://dx.doi.org/10.1016/j.regpep.2006.11.018 - 50. Hettinger, U., Lukasova, M., Lewicka, S. and Hilgenfeldt, U. (2002) Regulatory Effects of Salt Diet on Renal Renin-Angiotensin-Aldosterone, and Kallikrein-Kinin Systems. International Immunopharmacology, 2, 1975-1980. http://dx.doi.org/10.1016/S1567-5769(02)00163-7
- 51. Alves, C.R., Alves, G.B., Pereira, A.C., Trombetta, I.C., Dias, R.G., Mota, G.F., et al. (2013) Vascular Reactivity and ACE Activity Response to Exercise Training Are Modulated by the +9/?9 Bradykinin B? Receptor Gene Functional Polymorphism. Physiological Genomics, 45, 487-492.
http://dx.doi.org/10.1152/physiolgenomics.00065.2012 - 52. Zhou, J.B., Liu, C., Niu, W.Y., Xin, Z., Yu, M., Feng, J.P., et al. (2012) Contributions of Renin-Angiotensin System-Related Gene Interactions to Obesity in a Chinese Population. PLoS ONE, 7, e42881.
http://dx.doi.org/10.1371/journal.pone.0042881 - 53. Gu, D., Zhao, Q., Kelly, T.N., Hixson, J.E., Rao, D.C., Cao, J., et al. (2012) The Role of the Kallikrein-Kinin System Genes in the Salt Sensitivity of Blood Pressure: The GenSalt Study. American Journal of Epidemiology, 176, S72-S80. http://dx.doi.org/10.1093/aje/kws277
- 54. Montasser, M.E., Gu, D., Chen, J., Shimmin, L.C., Gu, C., Kelly, T.N., et al. (2011) Interactions of Genetic Variants with Physical Activity Are Associated with Blood Pressure in Chinese: The GenSalt Study. American Journal of Hypertension, 24, 1035-1040. http://dx.doi.org/10.1038/ajh.2011.97
- 55. Srinivasan, V., Ohta, Y., Espino, J., Pariente, J.A., Rodriguez, A.B., Mohamed, M., et al. (2013) Metabolic Syndrome, Its Pathophysiology and the Role of Melatonin. Recent Patents on Endocrine Metabolic & Immune Drug Discovery, 7, 11-25. http://dx.doi.org/10.2174/187221413804660953
- 56. Ekmekcioglu, C. (2006) Melatonin Receptors in Humans: Biological Role and Clinical Relevance. Biomedicine & Pharmacotherapy, 60, 97-108. http://dx.doi.org/10.1016/j.biopha.2006.01.002
- 57. Koziróg, M., Poliwczak, A.R., Duchnowicz, P., Koter-Michalak, M., Sikora, J. and Broncel, M. (2011) Melatonin treatment Improves Blood Pressure, Lipid Profile, and Parameters of Oxidative Stress in Patients with Metabolic Syndrome. Journal of Pineal Research, 50, 261-266.
http://dx.doi.org/10.1111/j.1600-079X.2010.00835.x - 58. Xia, Q., Chen, Z.X., Wang, Y.C., Ma, Y.S., Zhang, F., Che, W., et al. (2012) Association between the Melatonin Receptor 1B Gene Polymorphism on the Risk of Type 2 Diabetes, Impaired Glucose Regulation: A Meta-Analysis. PLoS ONE, 7, e50107. http://dx.doi.org/10.1371/journal.pone.0050107
- 59. Huber, M., Treszl, A., Reibis, R., Teichmann, C., Zergibel, I., Bolbrinker, J., et al. (2013) Genetics of Melatonin Receptor Type 2 Is Associated with Left Ventricular Function in Hypertensive Patients Treated According to Guidelines. European Journal of Internal Medicine, 24, 650-655.
http://dx.doi.org/10.1016/j.ejim.2013.03.015 - 60. Yang, J., Liu, J., Liu, J., Li, W., Li, X., He, Y., et al. (2014) Genetic Association Study with Metabolic Syndrome and Metabolic-Related Traits in a Cross-Sectional Sample and a 10-Year Longitudinal Sample of Chinese Elderly Population. PLoS ONE, 9, e100548. http://dx.doi.org/10.1371/journal.pone.0100548
- 61. Holzapfel, C., Siegrist, M., Rank, M., Langhof, H., Grallert, H., Baumert, J., et al. (2011) Association of a MTNR1B gene Variant with Fasting Glucose and HOMA-B in Children and Adolescents with High BMI-SDS. European Journal of Endocrinology, 164, 205-212. http://dx.doi.org/10.1530/EJE-10-0588
- 62. Mirzaei, K., Xu, M., Qi, Q., de Jonge, L., Bray, G.A., Sacks, F., et al. (2014) Variants in Glucose- and Circadian rhythm-Related Genes Affect the Response of Energy Expenditure to Weight-Loss Diets: The POUNDS LOST Trial. American Journal of Clinical Nutrition, 99, 392-399.
http://dx.doi.org/10.3945/ajcn.113.072066
Abbreviations
ACE: Angiotensin-converting enzyme
ADRB2 gene: Adrenoceptor beta 2, surface
ADRB3 gene: Adrenoceptor beta 3
ANS: Autonomic nervous system
BDKRB2 gene: Bradykinin receptor B2
BMI: Body mass index
cGMP: Cyclic guanosine monophosphate
CNS: Central nervous system
CO: Cardiac output
DiOGenes: Diet, Obesity and Genes study
GWAS: Genome-wide association studies
HTN: Hypertension
LOGIC Trial: Long-term effects of a lifestyle intervention in obesity and genetic influence in children
MTNR1B gene: Melatonin receptor 1B
POUNDS LOST Trial: Preventing Overweight Using Novel Dietary Strategies
SNP: Polymorphism
SNS: Sympathetic nervous system
SVR: Systemic vascular resistance
T2DM: Type 2 diabetes mellitus
VIGITEL: Protective and Risk Factors for Chronic Diseases by Telephone Survey