Open Journal of Gastroenterology
Vol.06 No.10(2016), Article ID:71634,25 pages
10.4236/ojgas.2016.610032
The Accuracy of Transabdominal Ultrasound in Detection of the Common Bile Duct Stone as Compared to Endoscopic Retrograde Cholangiopancreatography (with Literature Review)
Taha Ahmed M. Alkarboly, Salah Mohamad Fatih, Hiwa Abubaker Hussein, Talar M. Ali, Heero Ismael Faraj
Sulaimani School of Medicine, Kurdistan Centre for Gastroenterology & Hepatology (KCGH), Sulaimani, Iraq
Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: April 29, 2016; Accepted: October 25, 2016; Published: October 28, 2016
ABSTRACT
Introduction: Common bile duct stone (CBDS) is a common clinical problem that can cause serious complications, such as acute cholangitis and pancreatitis. It is important to have an accurate, safe, and reliable method for the definitive diagnosis of CBDS before proceeding to therapeutic endoscopic retrograde cholangiopancreatography (ERCP). Objective: To compare the accuracy of trans-abdominal ultrasound (TAUS) as a diagnostic tool at our institution―Kurdistan Centre for Gastroenterology & Hepatology (KCGH)―with invasive tool like ERCP in the diagnosis of bile duct stones, using specificity, sensitivity, and positive and negative predictive values. Patient and Method: After obtaining ethical committee approval & informed consent from every patient. This was a prospective study conducted on 71 patients (24 male patients and 47 females patients) where suspected to have CBDS depending on history, clinical suspicion and blood tests. Their ages range between (2 - 88 years). Both TAUS and ERCP were performed. Final diagnosis was confirmed depending on ERCP as it served as a diagnostic standard in diagnosing CBDS. Result: In 71 patients suspected to have CBDS by TAUS, only 46 patients had stone (65%), and 55 patients had stone by ERCP (77%). In our result, sensitivity, specificity, positive predictive value and negative predictive value for TAUS were 80%, 87.5%, 65.5% and 56%, respectively. Conclusion: TAUS can play an important role as an initial screening procedure for CBDS detection because of the various advantages like easy availability, cost effectiveness, no requirement of contrast material and lack of ionizing radiation but should done with other imaging modality to avoid serious complication of ERCP.
Keywords:
TAUS, ERCP, CBDS, KCGH, Sulaimani

1. Introduction
Common bile duct stone (CBDS) or choledocholithiasis is a common clinical problem that can cause serious complications, such as acute cholangitis and pancreatitis [1] . Therefore, accurately diagnosing CBDS is important for clinical decision making [2] .
CBDS could be primary or secondary; primary stones (10%): arising within the bile duct [3] . The primary stones are associated with biliary stasis and infection and are more commonly seen in Asian populations. The causes of biliary stasis that leads to the development of primary stones include biliary stricture, papillary stenosis, tumors and choledochal cyst [4] [5] .
Secondary stones (retain or recurrent) (90%) [3] , secondary CBDS, are the most common type [6] [7] . The vast majority of duct stones in western countries are formed within the gallbladder [4] [5] . The prevalence of gallbladder stones in the general population is up to 20% [8] , which is twice as common in women as in men, and becomes more prevalent with increasing age [9] [10] ; up to 20% of these patients have synchronous CBDS [11] . As many as 35% of patients with gallstones will ultimately become symptomatic and require cholecystectomy [12] , while approximately 3% - 10% of patients undergoing cholecystectomy will have (CBDS) [13] . These data mean that up to 2% of the general population may have CBDS during their life-span [2] [14] - [17] .
The secondary CBDS forms within the gallbladder and then migrates into the CBD, following gallbladder contractions [18] .
Once in the CBD, stones may reach the duodenum following the bile flow or due to the smaller diameter of the distal CBD at the Vater papilla; they may remain in the choledochus. In the latter case, gallstones may be fluctuant; thus may be asymptomatic [18] , (about 5%) [19] , or cause a variety of bile flow problems, including complete obstruction and jaundice [18] .
The symptom of choledocholithiasis consists of right upper abdominal colicky pain, radiating to the right shoulder with jaundice accompanied by pale stools and dark urine [20] .
Scholastically, Charcot’s triad [21] (jaundice associated with biliary colicky pain, fever and chills), indicates acute cholangitis; as choledocholithiasis is the most frequent etiology of such a clinical picture, it should prompt immediate diagnostic confirmation and CBD drainage [16] , whereas cutaneous itching is rarely present [22] .
Patient with CBDS also may present as acute pancreatitis, showing with transversal abdominal pain potentially radiating to the back and associated with an increase of serum level of amylase/lipase. In the presence of gallstones, the prior cause will be being of biliary origin [16] .
Hepatic abscess may also be a rarer infectious complication of CBDS whereas chronic CBD obstruction may also cause biliary cirrhosis [16] .
Murphy’s sign is commonly negative on physical examination in CBDS, helping to distinguish it from cholecystitis [23] .
1.1. Diagnosis
Diagnosis of choledocholithiasis is not always straightforward and clinical evaluation and biochemical tests are often not sufficiently accurate to establish a firm diagnosis [24] - [26] .
Usually, the diagnosis of choledocholithiasis is based on a combination of clinical suspicion, bio-chemical analysis and imaging findings. Unfortunately, all of these individually have varying diagnostic accuracies and none is a completely reliable method for identifying CBDS [27] .
Liver function tests (LFT) can be used to predict CBDS, Traditionally, an elevated (direct bilirubin, gamma-glutamil-transpherase, alkaline phosphatase) was considered as potentially due to CBDS [25] [26] .
Elevated serum bilirubin and alkaline phosphatase typically reflect biliary obstruction but these are neither highly sensitive nor specific for CBDS [26] . Jaundice and raised GGT level has been suggested to be the most sensitive and specific indicator of CBDS. A value of greater than 90 U/L has been proposed to indicate a high risk of choledocholithiasis [28] , (normal value is about 0 - 30 IU/L) [25] . However, the biochemical predictive models may be affected by inflammatory gallstone disease due to abnormally elevated predictor levels secondary to acute transient hepatocellular injury [28] .
Various imaging tests are used to confirm the diagnosis [24] - [26] . TAUS, endoscopic ultrasonography (EUS), magnetic resonance cholangiopancreatography (MRCP), endoscopicretrograde cholangiopancreatography (ERCP), and intraoperative fluorocholangiography (IOC) are available imaging modalities for the detection of CBDS. The optimal method for investigating suspected CBDS has not been determined [29] .
In order to help select from the various diagnostic and therapeutic options, patients may be classified preoperatively into high, moderate or low risk groups.
The high risk (>50% risk):
Group includes those patients with obvious clinical jaundice or cholangitis with choledocholithiasis or a dilated CBD on ultrasonography.
Moderate risk (10% - 50%):
Patients with a history of pancreatitis or jaundice with elevated alkaline phosphatase levels or multiple small gallstones carry a moderate risk of choledocholithiasis.
Low risk (<5%): Patients with large gallstones, without a history of jaundice or pancreatitis and with normal liver function tests are considered unlikely to have CBD stones and therefore at low risk [30] - [32] .
1.2. Imaging of Biliary Tree
1.2.1. Transabdominal Ultrasound (TAUS)
Represents the first line, non-expensive, non-invasive imaging examination and widely available [2] [19] [24] [33] , for assessing the status of the biliary system, and has been shown to be of value in the differentiation between obstructive and nonobstructive jaundice [34] .
However, is highly operator dependent, but it can provide useful information in experienced hands [35] , it can detect dilated CBD but its accuracy for detecting CBDS is poor [27] [36] - [38] .
Regarding the biliary tree, the right and left hepatic ducts, that is the first-order branches of the of biliary tree, are routinely seen on sonography, and normal second- order branches (intrahepatic) may be visualized [39] . Most of the right and left hepatic ducts are extrahepatic and along with the CHD (common hepatic duct), form the hilar portion of the biliary tree at the portahepatis. The use of spectral and color Doppler US may be needed to distinguish hepatic arteries from ducts [40] .
The intrahepatic ducts (third orders) are tiny structure that run with portal veins radicals in the portal triads [40] . The visualization of third-order or higher-order branches is often an abnormal finding and requires a search for the cause of dilation, the normal diameter of the first-order and second-order branches of the HD(hepatic duct) has been suggested to be 2 mm or less, and no more than 40% of the diameter of the adjacent portal vein [39] .
Enlarged biliary radicles have been described as having a characteristic radiating pattern from the portahepatis [34] .
A characteristic comma shape has been reported as helpful in differentiating the portal veins branches from an enlarged intrahepatic bile duct [41] . However, these are the findings of moderately advanced obstruction [42] in which there is marked dilataion of the biliary tree. In lesser degrees of obstruction these findings are not present [42] - [44] (Figure 1).
Figure 1. Typical ductal branching order. B, Subcostal oblique views foreshorten the right (R) and left (L) hepatic ducts. RA, Right anterior duct; RP, rightposterior duct; 2, segment 2 duct; 3, segment 3 duct; 4, segment 4 duct [24] .
However, minimal dilation of the biliary system due to obstruction can be diagnosed by recognition of the “parallel channel” sign. This sign is due to the concomitant imaging of the right or left portal vein branchs and the adjacent, minimally dilated intrahepatic ducts, this sign has proved highly reliable in differentiating obstructive from nonobstructive jaundice in patients having mild to moderate dilation of intrahepatic biliary tree. Demonstration of the parallel channel sign is a simple means of documenting the presence of obstructive jaundice [45] .
The anatomic basis for this finding is due to the physical contiguity of the biliary system and portal venous system which are bounded by the samefibrous capsule and follow the same anatomic pathway through the liver hilum and liver substance [45] .
The upper limit of normal for diameter of the common bile duct varies with age, is approximately one-tenth of the age of the patient in millimeters about 4 mm at age 40, 7 mm at age 70, etc. [28] or a reasonable rule to follow is to allow 1 mm of internal diameter of the bile duct for each decade after 50 years. A 6 mm bile duct is considered normal in a 60 years old patient [46] .
The hepatic and common bile ducts can sometimes be visualized throughout their course, but part of the common bile duct is often obscured by gas as it passes posterior to the first part of the duodenum [47] (Figure 2).
The classic appearance of CBDS is a rounded echogenic lesion with posterior acoustic shadowing [47] . Although TAUS is considered the gold standard for detecting cholelithiasis [48] having a sensitivity of approximately 99% and a specificity of 99% for detection of GB stone [49] , but sensitivity of TAUS in diagnosis CBDS varies from 20% to 80% depending on the operator [27] [38] , due to the following causes:
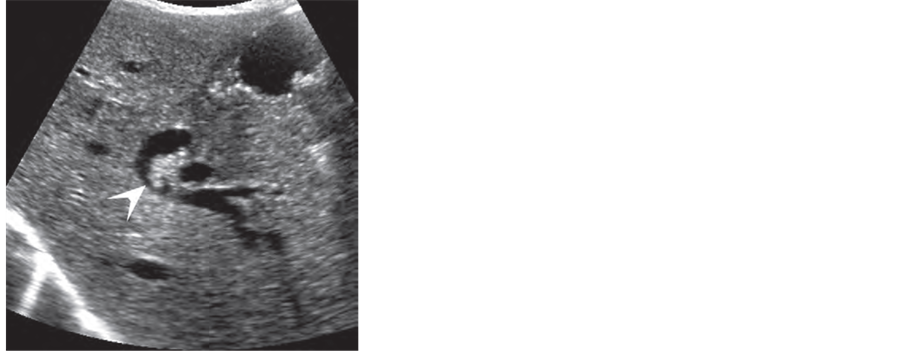
1) Some time no fluid rim will be seen around an impacted distal CBDS because it is compressed against the duct wall. The lateral margins of the stone are therefore not seen, decreasing the conspicuity of the stone, versus a stone seen in the gallbladder or proximal duct, where it is likely to be surrounded by bile [38] (Figure 3(b)).
2) Small stones may lack good acoustic shadows and appear only as a reproducible
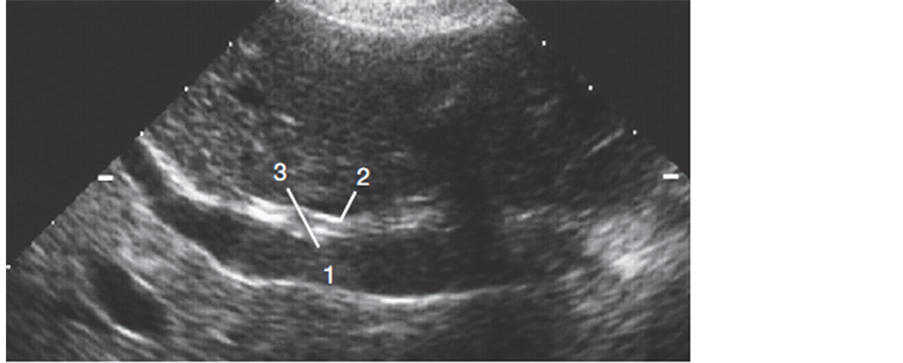
Figure 2. Ultrasound of portahepatis: usual configuration of portal vein, bile duct and hepatic artery. 1: Portal vein; 2: Common bile duct; 3: Hepatic artery [28] .
bright, linear echogenicity, either straight or curved. Awareness of this subtle appearanceof CBDS definitely improves their detection [47] (Figure 3(a)).
3) A majority of stones in the CBD will be in the distal duct right at the ampulla of Vater. Therefore, TAUS evaluation should include assessment of the entire duct, focusing on the periampullary region. Unfortunately, this region is often the most difficult area to see because it may be hidden by bowel gas, making detection of distal CBDS also difficult [47] .
4) CBDS often do not show acoustic shadowing [50] , possible reflection and retraction of the sound beam of the duct wall and the location of the duct beyond the optimal foci of the transducer [51] (Figure 3(a)).
Pitfalls in the diagnosis of CBDS include blood clot (hemobilia) (Figure 4(a) & Figure 4(b)), papillary tumors, and occasionally biliary sludge; none of these will
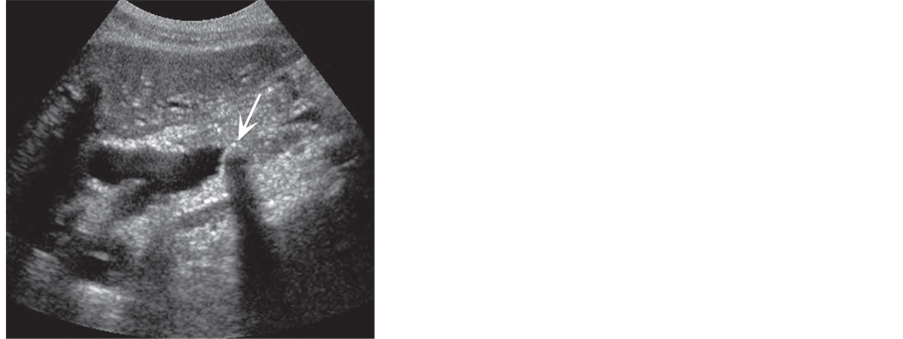
Figure 3. Common bile duct stones. (a) Small stone (arrow) may not show shadowing; (b) Large stone (arrow) has classic findings within a dilated CBD [24] .

Figure 4. Hemobilia: spectrum on sonography. (a) Echogenic blood clot (arrow head) within a dilated duct, after insertion of biliary drainage catheter. Biliary obstruction was caused by pancreatic tumor; (b) Echogenic clot in the common hepatic duct after liver biopsy [24] .
shadow. Surgical clips in the portahepatis, mostly from previous cholecystectomy, appear as linear echogenic foci with shadowing. The short length, the relatively high degree of echogenicity, the lack of ductal dilation, and the absence of the gallbladder should allow differentiation of surgical clips from stones [47] .
In some occasion CBDS diagnosis often relies on indirect signs of CBD obstruction, such as CBD dilation [52] - [54] . Although the CBD dilatation is identified accurately with up to 90% accuracy [51] . The definition of CBD dilation is also a matter of discussion, as suggested “normal limits” vary widely, partly because CBD diameter may increase with age and after cholecystectomy [52] - [54] .
Bile duct size is not largely affected by sex, body weight and height of the patient [55] [56] , but reported to have a major correlation with age > 6 mm + 1 mm per decade above 60 years of age [57] . However, in asymptomatic patients with normal liver function and no evidence of biliary dyskinesia, a duct of up to 12 mm may be observed. This may occur following cholecystectomy, with the duct fulfilling a reservoir function, or in patients with a previous episode of obstruction [29] . On other hand normal CBD not exclude CBDS, because stone may occur in normal duct diameter.
An incomplete, intermittent or early obstruction has been postulated to be the cause for the lack of ductal dilatation [58] . Normal bile duct TAUS has a 95% to 96% negative predictive value [35] [59] .
Also, the TAUS characterization of gallbladder stones harbors some predictive value for CBDS, multiple small (≤5 mm) stones posing a 4-fold higher risk of migration into the duct as opposed to larger and/or solitary stones [33] [60] . As multiple, small sized gallstones are more likely to migrate Into the CBD than single or multiple large stones [61] [62] .
1.2.2. Endoscopic Retrograde Cholangiopancreaticography (ERCP)
ERCP is the standard therapy for of biliary obstruction. However, the success rate is not 100%, depending on various patient and physician related factors [63] . Successful cholangiography by an experienced endoscopist is achieved in greater than 90% of patients. ERCP can have a failure rate of 5% to 10% even in experienced hands [64] .
The main advantage of these techniques, is the ability to sample tissue and perform therapeutic maneuvers, such as biliary drainage, stenting or stone removal [65] .
ERCP has been the gold standard for preoperative diagnosis of CBD calculi. When compared to other tests such as TAUS and MRCP, Stone retrieval and sphincterotomy has supplanted surgical treatment of choledocholithiasis in many institutions [66] [67] . The sensitivity of ERCP with cholangiography alone has been reported as 89% to 93% with a specificity of 100% in studies that used subsequent biliary sphincterotomy and duct sweeping with balloons/baskets as the criterion standard [68] [69] .
ERCP can have false-negative results because of small stones located within dilated biliary ducts, whereas fewer false-negative results were recorded with EUS, and consisted mostly of stones located in the upper common hepatic duct or lying within intrahepatic ducts. Moreover, it is usual to have false-positive results when performing ERCP, because of small air bubbles introduced during cannulation and injection of contrast liquid [70] .
However, it is invasive and may cause several complications [2] . It has been shown that the rate of post-ERCP pancreatitis may be as high as 15%, which includes 1% of patients graded as severe in degree [71] . Biliary tract infection (0.6% - 5%), bleeding (0.3% - 2%), and duodenal perforation (0.1% - 1%) [2] , mortality rate from ERCP is 0.2% - 0.5% [72] [73] .
ERCP is invasive, expensive, and can be complicated by acute pancreatitis [74] . Sphincter problems, Billroth II anastomoses, impacted stones, duodenal diverticula, operator inexperience, and many other factors contribute to failed cannulation in 5% to 10% of cases [64] .
Because the risk of adverse events is higher with ERCP than other modality used in biliary imaging, the use of ERCP as a diagnostic modality is best suited for those patients at high risk of CBDS because they are most likely to benefit from the therapeutic capability [70] [75] .
2. Aim of the Study
The aim of our study was to compare the accuracy of TAUS as a diagnostic tool at our institution (KCGH) with invasive ERCP in the detection of bile duct stones, using specificity, sensitivity, and positive and negative predictive values.
3. Methods
3.1. Patients and Methods
This was a prospective study conducted on 75 patients presenting to Kurdistan center for gastroenterology and hepatology (KCGH); department of ERCP which suspected to have CBS by history, clinical examination and serological test From April 2015 through October 2015.
Four cases were excluded from our study because one of them by CT scan diagnosed to have lower CBD tumor and other papillary cannulation failed because she had periampullary diverticulum, and other 2 cases because by EUS no stone or mass.
Patients with obstructive jaundice, deranged liver function tests (raised total, direct bilirubin and alkaline phosphate and ALT) were included in the study.
For the purposes of this study the hard copy images of ERCP and ultrasound examinations of each patient were collected and the patient details masked.
The patients were fasted overnight (by gastroenterologist because they were prepared for ERCP) and ultrasound examination was done by experience ultrasonologist, in the supine and Left posterior oblique to optimally visualize the biliary ducts. Ultrasound study was performed transabdominally using axial, subcostal and intercostal approach. All ultrasound examinations were performed by using a General Electronic Model Voluson E6 ultrasound unit with (3.5 - 5) MHz curvilinear probe.
The liver parenchyma was scanned to rule out parenchymal pathology and to detect any dilated biliary radicals. Gall bladder was examined to check for Lithiasis, distension, wall thickening, growth or soft tissue lesions. And pancrease examined to exclude pancreatic head mass or pathology that cause biliary obstruction. For the purpose of this study, the common hepatic duct and the common bile duct were considered as one structure, the common duct. This is because of the uncertain site at which the cystic duct joins the common hepatic duct to form the common bile duct. The common duct is arbitrarily divided into two parts: the proximal and the distal parts. The proximal part of the common duct is that segment from the porta down to the first part of the duodenum, whereas the distal part include that segment behind the duodenum and the intrapancreatic portion.
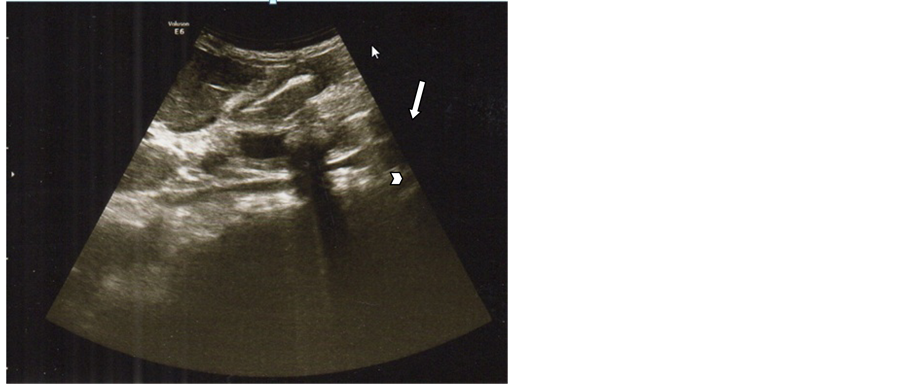
Careful scanning of the entire course and caliber of the duct system whenever possible was done from portahepatis to pancreatic head to trace the extent of the duct dilatation and to localize the level of obstruction. CBD was identified using color Doppler us to differentiate from nearby vessels (portal vein and hepatic artery) (Figure 5). The diagnosis of choledocholithiasis was made when intraluminal echogenic focus with or without acoustic shadowing was demonstrated.
The final diagnosis was based on ERCP. The ERCP examination was performed by one experienced gastroenterologist.
ERCP was done in all patients after regular preparation i.e. overnight fasting, slow IV sedation and precaution of asepsis. Endoscopy was performed using side viewing Endoscope (OLYMPUS EXERA CLV-160) and Videoscope Monitor (Trinitron OEV203). In semiprone position using endoscopy they enter through the mouth until reaching the duodenum and face papilla then using sphicntrotome for biliary cannulation trial done a guide wire passed to bile duct then contrast will be injected to visualize the biliary tree with precautions taking to avoid injecting air. Endoscopicsphinctreotomy done and stone will be extracted using extraction ballon or basket, in case of tumor; plastic or metallic biliary stent will be deployed. A total dose of Buscopan injection (20
Figure 5. Color Doppler Ultrasound of portahepatis show dilated CBD with no stone by ERCP impacted lower CBDS.
mg) was administered toparalyse the duodenum & to relieve the spasm if there is papillary spasm.
Vital signs were monitored continously throughout the procedure and till I hour after the ERCP.
3.2. Exclusion Criteria
・ Heavy daily alcohol intake > 80 g.
・ Hepatotoxic drug intake.
・ Serologic findings of acute viral hepatitis A, C or B.
・ Pancreatic head mass.
3.3. Inclusion Criteria
・ Patients with clinical sign and symptom of jaundice.
・ Patient with elevated liver function test (elevated TBS, direct bilirubin, ALT, serum alkaline phosphate).
・ Patient with dilated CBD by US with or without obvious CBDS.
3.4. Data Entry and Statistical Analysis
Statistical Package for Social Sciences (SPSS) version 22, which is a statistical software program, and Microsoft excel spreadsheets (2013) were used for data entry, calculations, and data interpretations. Descriptive statistics, and up-to-date statistical methods were used in the evaluations.
P-values ≤ 0.05 were considered statistically significant.
4. Results
In this study we have 71 patients who have been screened with TAUS and ERCP, the mean age of the participants was 50.7 years, with a range from 2 to 88 years (Table 1). The males were 24 with a proportion of (34%), and females were 47 (66%) patients. All had different presenting signs and symptoms. Generally, 45 (63%) of patients had jaundice, and the number and percentage of patients with other sign & symptoms such as: fever, RUQ pain, vomiting were: 39 (55%), 70 (99%), and 17 (24%) respectively. 24 cases patient with charcot trait having CBDS by ERCP (82.7%).
Although only RUQ pain was the dominant characteristic among the patients with a high incidence (99%), but patients could be grouped on diagnostic characteristics of Charcot triad (jaundice, fever, RUQ pain), patients with Charcot triad were 29 (41%) patients out of the total number (Table 1).
When it comes to number and percentage of patients with these blood tests, we found that 15 (21.1%) of the patients had a normal reading for serum Alkaline phosphatase, and 56 (78.9%) had abnormal or above normal results. The figures for Alanine aminotransferase (ALT) for normal and above normal results were, 29 (40.8%), and 42 (59.2%) of the patients respectively. Fourteen (19.7%) of the patients had normal readings for TSB, and 59 (80.3%) had above normal results (Table 2).
Table 1. Age and sex distribution of our sample presenting sign and symptom.
Table 2. Frequency and percentage of patients with normal and above normal results for 3 liver function tests (Serum Alkaline Phosphatase, ALT, and TSB).
Table 3, explains detection rates of CBD dilatation and CBD stone by each of TAUS and ERCP as diagnostic tools. TAUS revealed 55 (77%) cases out of 71 with CBD dilatation and ERCP revealed 61 (86%) cases with the same condition. In detection of CBD stones, the frequencies and percentages of detections for each of TAUS and ERCP were: 46 (65%), and 55 (77%) respectively (Figure 6).
It has come to our attention that there are gender differences in the abnormalities that we detected both in TAUS and ERCP. In TAUS, females had higher incidence rates in having dilated CBD and CBD stone, and the incidences were 79% and 72% respectively, and male/female risk ratios were 0.95 and 0.69 respectively, these ratios confirm that females have higher numbers of CBD stones and dilated CBDs (Table 4).
Although ERCP confirmed higher detection rates of abnormalities for both males and females compared to those of TAUS, but incidence of dilated CBD and CBD stones are still higher in females and were: 87%, and 83% respectively, while these incidences in males were; 83%, 67% respectively (Table 4).
Table 3. Detection rates of TAUS and ERCP for CBD dilatation and CBD stones.
Table 4. Male/female differences and risk ratios, as well as diagnostic capability of both TAUS and ERCP in terms of gender differences in abnormalities.
Figure 6. Detection rates of TAUS and ERCP for CBD dilatation and CBDS.
We have decided to find the diagnostic characteristics of TAUS in detecting CBD stones. Sensitivity, specificity, positive and negative predictive values, positive and negative likelihood ratios were all calculated for TAUS, with 95% CIs, by using the findings at ERCP as the reference standard or the gold standard. True positive and true negative results were considered when the results for both tests were similar in detection of CBD stones. A finding was defined as false-positive when TAUS depicted a CBD stone but ERCP setting did not depict the same CBD stone. A false negative finding was considered to be present when a CBD stone was detected in ERCP but the same stone was not detected at TAUS. We could not find any CBD stones in 14 patients in both TAUS and ERCP. The number of true positives were 44 patients, two patients had false positive results and 11 patients had false negative results (Table 5).
In diagnosing CBDS, the sensitivity of TAUS, or the ability of the test to correctly identify patients with the same CBDS as ERCP, was 80% (95% CI: 67% to 89.6%) (Table 6). Specificity or the ability of TAUS to correctly identify those patients without any CBDS was 87.5% (95% CI: 61.6% to 98%).
Positive predictive value, or the probability that a CBDS is present when TAUS test is positive for DBDS, was 95.65% (95% CI: 85% to 99%). Negative predictive value, or the probability that a CBDS is not present in a patient when TAUS test is negative, was 56% (95% CI: 34.9% to 75.6%) (Table 6). And finally, validity (Accuracy) of the test US was 81.7%.
And finally, from the table below (Table 7), we have tried to explain the association
Table 5. True positive, true negative, false positive, and false negative results of CBDS by TAUS (considering ERCP as the gold standard).
Table 6. Diagnostic characteristics of TAUS in detecting CBD stones as compared to ERCP.
Table 7. Relation of dilated CBD to the CBS.
between diagnosed CBD dilatation on ultrasonography and presence of stones in the same patients on ERCP. We have 46 patients who had CBD dilatation on TAUS and tested positive on ERCP, nine patients had CBD dilatation on TAUS and no stones on ERCP. Nine patients had no CBD dilatation on TAUS but ERCP tested positive for CBD stone, and seven patients tested negative on both tests.
It has come to our attention that the patients who had a dilated CBD on TAUS had 4 times higher probability of having a positive result for CBD stone on ERCP than having a positive result for CBD stone on ERCP when TAUS shows no CBD dilatation in the same patients (95% CI: 1.18 to 13.4, P-value = 0.026).
5. Discussion
When evaluating a case of obstructive jaundice, the aim of the radiologist is to confirm the cause and site of obstruction. The success rate in diagnosing specific cause has continued to improve with advancing high resolution equipment, scanning technique and interpretive skills. With the availability of TAUS, MRI including MRCP, ERCP and PTC, diagnostic approach in a patient with biliary tract pathology has been completely revolutionized with accuracy of radiological diagnosis approaching 98% when combined with clinical data [4] .
TAUS has always been used as the initial screening procedure because of the various advantages like easy availability, cost effectiveness, no requirement of contrast material and lack of ionizing radiation. It is well suited to visualize the Common Hepatic Duct (CHD) and proximal CBD [5] [59] , also ultrasound enables sonologist to detect disease outside the biliary tree [75] [76] .
In our study 71 consecutive patients with clinical history and laboratory investigation raising the possibility of choledocholithiasis were examined successively by ultrasound and ERCP.47 female (66.2%) and 24 male (33.8%).
The sex distribution of our sample show female predominant (66.2%) this could be explained by the fact that GS are twice common in female as in male [75] [76] and 90% of CBDS are secondary [3] [77] [78] .
Their ages ranged from 2 - 88 years. Our study showed that nearly all ages are at risk for CBDS. The mean age of patient was (50.7) years.
Regarding the gallstones 19cases of our study had history of cholecystectomy and 52 case no history of cholycystectomy in these 52 cases 10 cases had no gallstone and 42 cases had gall stones.
In our study TAUS correctly detect CBDS in 44 from 55 cases having CBDS by ERCP (Figures 7-12), our PPV was 95% this in agreement with (Humphrey et al. 93%) [77] and (Tandon et al. 90%) [55] and (M.A. Rahim Khan et al. 100%) [56] . Our study show sensitivity of ultrasound was 80% and this in agreement with (LINDSELL D. R. M 84%) [79] and (koenigsberg et al. 82%) [80] , (Faye C. Laing et al. 75%) [81] and (Steffen Rickesa et al. 82%) [82] who found same result. Although most previous study specially in 80th (Barry H. gross et al. 1983 sensitivity 25%) [83] and Einstein DM 1984 sensitivity 18% [50] , show poor sensitivity and wide range (20% - 80%). The studies with poor
Figure 7. Common bile duct stone (arrow) with posterior acoustic shadowing (arrow head).
Figure 8. Normal diameter CBD with 2 small stone (arrow) at distal part with no acoustic shadowing.
Figure 9. Ultrasound of dilated proximal CBD with impacted stone.
Figure 10. Ultrasound of dilated CBD contain stent (arrow) and stone.
Figure 11. TAUS show normal diameter CBD (arrow) no stone; by ERCP small stone extracted.
Figure 12. Dilated CBD distal part obscure by bowel gas.
results are mostly older studies when the resolution of ultrasound machine was not good and it was a new technique still being learnt.
Absence of stones correctly detected in 14 of 25 cases, specificity 87.5%. Our US results had agreement with (Steffen Rickes et al. 88%) [82] & (Rigauts et al. specificity of 91%) [84] , (karki at al 89%) [85] and (Wael A. Shahin et al. 88%) [78] .
Our study had accuracy 81.7%, our result was in agreement with (Steffen Rickesa et al. 83%) [82] , (LINDSELLD. R. M 88%) [79] and (Chak et al. 83%) [86] .
Regarding diagnostic errors in our have 2 false positive cases, one of these cases by US there was dilated duct and multiple small stones, by ERCP there was dilated CBD and no stones, these may be because ERCP could have false-negative results because of small stones located within dilated CBD, where as fewer false-negative results were recorded consisted mostly of stones located in the upper common hepatic duct or lying within intrahepatic ducts [70] , or may due stone passage to duodenum with bile flow [13] .
Second case of the 2 false positive cases by US was suspecting CBDS but by ERCP diagnosed as distal CBD tumor proved by biopsy taken from the mass, these may be due to one of major limitations of US which are assessment of the distal CBD and pancreas, which are often obscured by overlying bowel gas in about 30% - 50% of the patients [5] [34] . The stone detection rate is also influenced by patient factors such as the number, size and site of stones, patient body habitus [37] .
We had 11 false negative cases our negative predictive value about 56%, in these cases stone was not detect by US. 7 of them had dilated CBD (Figure 11 and Figure 12) and 4 had normal CBD by US (Figure 10), by ERCP all had CBD stones, 2of them suspected to had lower CBD tumor by US and ERCP detect impacted lower CBDS, It has been noted that calculi may not cast acoustic shadowing [50] , and absence of surrounding bile makes CBD stones difficult to differentiate from periductal structures [87] . In such circumstances the stone can either be missed or that the intraluminal echogenic foci is miss interpreted as a tumor. Lack of acoustic shadowing is possibly related to the calibre of the duct, depth of duct, refractions and reflections from the duct walls [50] .
TAUS remain a highly operator-dependent and the results are always influenced by the skill of the examiner [88] .
We had 14 case true negative case which had no stone by TAUS and ERCP, 7 of them had normal CBD by TAUS and ERCP, one of them had normal CBD by TAUS and dilated CBD by ERCP 6of them had dilated CBD by TAUS and ERCP. From true negative; 6 of 14 have no abnormally detected by ERCP, 4 of them had sludge, 1 distal CBD stricture and one of them had prominent papilla (stenosis) and biopsy taken to exclude tumor, ultrasound cannot predict such stenosis [89] [90] . The true negative cases also included 2 passed CBD stones, in which the clinical course of the disease strongly suggested a choledochal stone. Endoscopic signs of a passed stone may include an open, reddish, and edemic papilla, but these signs may disappear in a few days. The study design of our investigation offers an explanation for these false-negative results, as ERCP was delayed several days compared to TAUS, If ERCP had been performed immediately after admission to hospital, the detection rate of stones would probably have improved.
So our study show NPV was 56% for CBDS by us so that CBDS not reliably excluded by negative TAUS examination.
Regarding common bile duct dilatation our study show 46cases had CBD dilatation at both TAUS and ERCP, 14 cases had no CBD dilatation at both TAUS and ERCP. 11 cases had no CBD dilatation at US but had CBD dilatation at ERCP this may explain by
1) The site of measurement may differ between both modalities as TAUS may not visualize the most distal part of CBD (due to overlying gas or mass?) while ERCP define the whole duct.
2) Timing difference between both examinations in few days may reveal borderline on TAUS to be mildly dilated on ERCP.
3) By pressure of injecting contrast media in ERCP may cause dilatation of CBD.
Our study show we had 55 cases (77.4%) with dilated CBD by TAUS and 16 cases had no dilated CBD by US (22.5%). in those cases with dilated CBD by TAUS [45] [54] cases proved had CBDS by ERCP and 9 cases had no CBDS by ERCP; because dilatation of CBD can result from other causes: CBD stricture, cholangiocarcinoma, periampullary diverticulum, pancreatic head mass, sphincter of Oddi dysfunction and papillary stenosis, etc. [89] - [91] .
From non dilatedcases [16] , only 7cases proved had no CBDS by ERCP and the 9 cases had CBDS by ERCP (Figure 10), It has come to our attention that the patients who had a dilated CBD on TAUS had 4 times higher probability of having a positive result for CBD stone on ERCP than having a positive result for CBD stone on ERCP when US shows no CBD dilatation in the same patients.
False-negative studies for biliary dilatation may be due to the absence of biliary dilatation in the presence of small non obstructing calculi. Common duct dilatation may not occur for 24 hours after initial stone impaction and intrahepatic duct may not dilate for up 72 hours after impaction [88] .
Limitation of our study:
・ All patient prepared for ERCP so we couldn’t give water for better visualization of distal CBD and pancreas.
・ Another limitation in our study was High BMI because affect the resolution of the ultrasound image.
・ Bowel gas which obscure distal CBD and pancreas.
6. Conclusions
・ In conclusion transabdominal ultrasound is still a valuable non-invasive preliminary procedure for detection of common duct stones.
・ A positive diagnosis is useful because patients will be spared from unnecessary preoperative invasive and expensive cholangiographic examinations.
・ In the presence of a negative or indeterminate result, further evaluation by other imaging modalities like MRCP and EUS may be required.
7. Recommendation
・ We recommend other study to include larger sample size.
・ More awareness of the observers because subtle finding may be over looked.
・ If available it will be better if all patients have TAUS examination at same day when they undergo ERCP to prevent false positive ultrasound finding.
Cite this paper
Alkarboly, T.A.M., Fatih, S.M., Hussein, H.A., Ali, T.M. and Faraj, H.I. (2016) The Accuracy of Transabdominal Ultrasound in Detection of the Common Bile Duct Stone as Compared to Endoscopic Retrograde Cholangiopancreatography (with Literature Review). Open Jour- nal of Gastroenterology, 6, 275-299. http://dx.doi.org/10.4236/ojgas.2016.610032
References
- 1. National Institutes of Health (1993) National Institutes of Health Consensus Development Conference Statement on Gallstones and Laparoscopic Cholecystectomy. American Journal of Surgery, 165, 390-396.
http://dx.doi.org/10.1016/S0002-9610(05)80929-8 - 2. ASGE Standards of Practice Committee, Maple, J.T., Ben-Menachem, T., Anderson, M.A., Appalaneni, V., Banerjee, S., et al. (2010) The Role of Endoscopy in the Evaluation of Suspected Choledocholithiasis. Gastrointestinal Endoscopy, 71, 1-9.
http://dx.doi.org/10.1016/j.gie.2009.09.041 - 3. Grant, L. and Griffin, N. (2013) Grainger & Allison’s Diagnostic Radiology Essentials: Expert Consult: Online and Print. Churchill Livingstone, London.
- 4. Fulcher, A.S. and Turner, M.A. (2002) MR Cholangiopancreatography. Radiologic Clinics of North America, 40, 1363-1376.
http://dx.doi.org/10.1016/s0033-8389(02)00046-5 - 5. Ferruci Jr., J.T. (1979) Body Ultrasonography. The New England Journal of Medicine, 300, 590-602.
http://dx.doi.org/10.1056/NEJM197903153001104 - 6. Cuschaieri, A. (2002) Disorders of the Biliary Tract. In: Cuschieri, A., Steele, R.J. and Mossa, A.R., Eds., Essential Surgical Practice A, 4th Edition, Arnolds, London, 375-452.
- 7. Saharia, P.C., Zuidema, G.D. and Cameron, J.L. (1997) Primary Common Bile Stones. Annals of Surgery, 185, 598-604.
- 8. Aerts, R. and Penninckx, F. (2003) The Burden of Gallstone Disease in Europe. Alimentary Pharmacology & Therapeutics, 18, 49-53.
http://dx.doi.org/10.1046/j.0953-0673.2003.01721.x - 9. Dandan, I.S., Soweid, A.M. and Abiad, F. (2008) Choledocholithiasis: Overview. eMedicine Gastroenterology.
http://emedicine.medscape.com/article/172216-overview - 10. Catalana, O.A., Sahani, D.V., Kalva, S.P., Cushing, M.S., et al. (2008) MR Imaging of the Gallbladder: A Pictorial Essay. RadioGraphics, 28, 135-155.
http://dx.doi.org/10.1148/rg.281065183 - 11. Borzellino, G., Rodella, L., Saladino, E., Catalano, F., et al. (2010) Treatment for Retrieved Common Bile Duct Stones during Laparoscopic Cholecystectomy: The Rendezvous Technique. Archives of Surgery, 145, 1145-1149.
http://dx.doi.org/10.1001/archsurg.2010.261 - 12. Schirmer, B.D., Winters, K.L. and Edlich, R.F. (2005) Cholelithiasis and Cholecystitis. Journal of Long-Term Effects of Medical Implants, 15, 329-338.
http://dx.doi.org/10.1615/JLongTermEffMedImplants.v15.i3.90 - 13. Freitas, M.L., Bell, R.L. and Duffy, A.J. (2006) Choledocholithiasis: Evolving Standards for Diagnosis and Management. World Journal of Gastroenterology, 12, 3162-3167.
- 14. Paul, A., Millat, B., Holthausen, U., Sauerland, S., et al. (1998) Diagnosis and Treatment of Common Bile Duct Stones (CBDS): Results of a Consensus Development Conference. Surgical Endoscopy, 12, 856-864.
http://dx.doi.org/10.1007/s004649900729 - 15. Treckmann, J., Sauerland, S., Frilling, A. and Paul, A. (2006) Common Bile Duct Stones— Update 2006. In: Neugebauer, E.A.M., Sauerland, S., Fingerhut, A., Millat, B. and Buess, G., Eds., EAES Guidelines for Endoscopic Surgery, Springer, Berlin Heidelberg, 329-333.
http://dx.doi.org/10.1007/978-3-540-32784-4_16 - 16. Williams, E.J., Green, J., Beckingham, I., Parks, R., Martin, D. and Lombard, M. (2008) Guidelines on the Management of Common Bile Duct Stones (CBDS). Gut, 57, 1004-1021.
http://dx.doi.org/10.1136/gut.2007.121657 - 17. Maple, J.T., Ikenberry, S.O., Anderson, M.A., Appalaneni, V., Decker, G.A., Early, D., Evans, J.A., Fanelli, R.D., Fisher, D., Fisher, L., et al. (2011) The Role of Endoscopy in the Management of Choledocholithiasis. Gastrointestinal Endoscopy, 74, 731-744.
http://dx.doi.org/10.1016/j.gie.2011.04.012 - 18. Acosta, J.M. and Ledesma, C.L. (1974) Gallstone Migration as a Cause of Acute Pancreatitis. The New England Journal of Medicine, 290, 484-487.
http://dx.doi.org/10.1056/NEJM197402282900904 - 19. Topal, B., Van de Moortel, M., Fieuws, S., Vanbeckevoort, D., van Steenbergen, W., Aerts, R. and Penninckx, F. (2003) The Value of Magnetic Resonance Cholangiopancreatography in Predicting Common Bile Duct Stones in Patients with Gallstone Disease. British Journal of Surgery, 90, 42-47.
http://dx.doi.org/10.1002/bjs.4025 - 20. Schwartz, S.I. (1974) Gallbladder and Extrahepatic Biliary System. In: Schwartz, S.I., Lillehei, R.C., Shires, G.T., Spencer, F.C., Storer, E.H., et al., Eds., Principles of Surgery, 2nd Edition, McGraw-Hill, New York, 1221-1254.
- 21. Charcot, J.M. (1877) Leconssur les maladies du foie, des voiesbiliaireset des reins. Faculté de Médecine de Paris, Paris.
- 22. Kondo, S., Isayama, H., Akahane, M., et al. (2005) Detection of Common Bile Duct Stones: Comparison between Endoscopic Ultrasonography, Magnetic Resonance Cholangiography, and Helical-Computed-Tomographic Cholangiography. European Journal of Radiology, 54, 271-275.
http://dx.doi.org/10.1016/j.ejrad.2004.07.007 - 23. National Institute of Diabetes and Digestive and Kidney Diseases (2007) “Gallstones” (PDF). National Digestive Diseases Information Clearinghouse, National Institutes of Health, United States Department of Health and Human Services, Bethesda, Maryland. Retrieved 6 November 2010.
- 24. Rumack, C.M., Wilson, S.R., Charboneau, J.W. and Levine, D. (2011) Diagnostic Ultrasound. 4th Edition, Mosby, Maryland Heights.
- 25. Peng, W.K., Sheikh, Z., Paterson-Brown, S. and Nixon, S.J. (2005) Role of Liver Function Tests in Predicting Common Bile Duct Stones in Acute Calculous Cholecystitis. British Journal of Surgery, 92, 1241-1247.
http://dx.doi.org/10.1002/bjs.4955 - 26. Sgourakis, G., Dedemadi, G., Stamatelopoulos, A., Leandros, E., Voros, D. and Karaliotas, K. (2005) Predictors of Common Bile Duct Lithiasis in Laparoscopic Era. World Journal of Gastroenterology, 11, 3267-3272.
http://dx.doi.org/10.3748/wjg.v11.i21.3267 - 27. Varghese, J.C., Liddell, R.P., Farrell, M.A., Murray, F.E., Osborne, D.H. and Lee, M.J. (1999) The Diagnostic Accuracy of Magnetic Resonance Cholangiopancreatography and Ultrasound Compared with Direct Cholangiography in the Detection of Choledocholithiasis. Clinical Radiology, 54, 604-614.
http://dx.doi.org/10.1016/S0009-9260(99)90023-5 - 28. Chang, C.W., Chang, W.H., Lin, C.C., Chu, C.H., Wang, T.E. and Shih, S.C. (2009) Acute Transient Hepatocellular Injury in Cholelithiasis and Cholecystitis without Evidence of Choledocholithiasis. World Journal of Gastroenterology, 15, 3788-3792.
- 29. Dalton, S.J., Balupuri, S. and Guest, J. (2005) Routine Magnetic Resonance Cholangiopancreatography and Intra-Operative Cholangiogram in the Evaluation of Common Bile Duct Stones. Annals of the Royal College of Surgeons of England, 87, 469-470.
- 30. Abboud, P.A., Malet, P.F., Berlin, J.A., Staroscik, R., Cabana, M.D., Clarke, J.R., Shea, J.A., Schwartz, J.S. and Williams, S.V. (1996) Predictors of Common Bile Duct Stones Prior to Cholecystectomy: A Meta-Analysis. Gastrointestinal Endoscopy, 44, 450-457.
http://dx.doi.org/10.1016/S0016-5107(96)70098-6 - 31. Barkun, A.N., Barkun, J.S., Fried, G.M., Ghitulescu, G., Steinmetz, O., Pham, C., Meakins, J.L., Goresky, C.A. and the McGill Gallstone Treatment Group (1994) Useful Predictors of Bile Duct Stones in Patients Undergoing Laparoscopic Cholecystectomy. Annals of Surgery, 220, 32-39.
http://dx.doi.org/10.1097/00000658-199407000-00006 - 32. Cohen, M.E., Slezak, L., Wells, C.K., Andersen, D.K. and Topazian, M. (2001) Prediction of Bile Duct Stones and Complications in Gallstone Pancreatitis Using Early Laboratory Trends. The American Journal of Gastroenterology, 96, 3305-3311.
http://dx.doi.org/10.1111/j.1572-0241.2001.05330.x - 33. Costi, R., Sarli, L., Caruso, G., Iusco, D., Gobbi, S., et al. (2002) Preoperative Ultrasonographic Assessment of the Number and Size of Gallbladder Stones: Is It a Useful Predictor of Asymptomatic Choledochallithiasis? Journal of Ultrasound in Medicine, 21, 971-976.
- 34. Taylor, K.J.W., Carpenter, D.A., McCready, V.A. and Hill, C.A. (1975) Grey Scale Ultrasonography in the Differential Diagnosis of Obstructive Jaundice. In: White, D., Ed., Ultrasound in Medicine, Vol. 1, Proceedings of the 19th Annual Meeting of the American Institute of Ultrasound in Medicine, Plenum, New York.
- 35. Liu, T.H., Consorti, E.T., Kawashima, A., Tamm, E.P., Kwong, K.L., et al. (2001) Patient Evaluation and Management with Selective Use of Magnetic Resonance Cholangiography and Endoscopic Retrograde Cholangiopancreatography before Laparoscopic Cholecystectomy. Annals of Surgery, 234, 33-40.
http://dx.doi.org/10.1097/00000658-200107000-00006 - 36. Kaltenthaler, E., Walters, S., Chilcot, J., Blakeborough, A., Vergel, Y.B. and Thomas, S. (2006) MRCP Compared to Diagnostic ERCP for Diagnosis When Biliary Obstruction Is Suspected: A Systemic Review. BMC Medical Imaging, 6, 2-10.
http://dx.doi.org/10.1186/1471-2342-6-9 - 37. Morgan, S. and Traverso, L.W. (2000) Intraoperative Cholangiography and Postoperative Pancreatitis. Surgical Endoscopy, 14, 264-266.
http://dx.doi.org/10.1007/s004640000078 - 38. Scheiman, J., Carlos, R., Barnett, J., et al. (2001) Can Endoscopic Ultrasound or Magnetic Resonance Cholangiopancreatography Replace ERCP in Patients with Suspected Biliary Disease? A Prospective Trial and Cost Analysis EUS versus MRCP for Suspected Biliary Disease. The American Journal of Gastroenterology, 96, 2900-2904.
http://dx.doi.org/10.1111/j.1572-0241.2001.04245.x - 39. Bressler, E.L., Rubin, J.M. and McCracken, S. (1987) Sonographic Parallel Channel Sign: A Reappraisal. Radiology, 164, 343-346.
http://dx.doi.org/10.1148/radiology.164.2.3299485 - 40. Bret, P.M., de Stempel, J.V., Atri, M., Lough, J.O. and Illescas, F.F. (1988) Intrahepatic Bile Duct and Portal Vein Anatomy Revisited. Radiology, 169, 405-407.
http://dx.doi.org/10.1148/radiology.169.2.3051116 - 41. Taylor, K.J.W. and Carpenter, D.A. (1975) The Anatomy and Pathology of the Portal Hepatis Demonstrated by Gray Scale Ultrasonography. Journal of Clinical Ultrasound, 3, 117-119.
http://dx.doi.org/10.1002/jcu.1870030208 - 42. Doust, B.D. (1976) The Use of Ultrasound in the Diagnosis of Gastroenterological Disease. Gastroenterology, 70, 602-610.
- 43. Taylor, K.J.W., Carpenter, B.E. and McCready, V.A. (1974) Ultrasound and Scintigraphy in the Differential Diagnosis of Obstructive Jaundice. Journal of Clinical Ultrasound, 2, 105-116.
http://dx.doi.org/10.1002/jcu.1870020205 - 44. Oldberg, B.B. (1976) Ultrasonic Cholangiography: Gray-Scale B-Scan Evaluation of the Common Bile Duct. Radiology, 118, 401-404.
http://dx.doi.org/10.1148/118.2.401 - 45. Gray, H. (1973) Anatomy of the Human Body. 29th American Edition, edited by C. M. Gross, Lea & Febiger, Philadelphia.
- 46. Jeffrey, R.B. and Ralls, J. (1995) Sonography of the Abdomen. 1st Edition, Raven Press, Ltd., New York, 204.
- 47. Baron, R.L., Tublin, M.E. and Peterson, M.S. (2002) Imaging the Spectrum of Biliary Tract Disease. Radiologic Clinics of North America, 40, 1325-1354.
http://dx.doi.org/10.1016/S0033-8389(02)00045-3 - 48. Kothari, S.N., Obinwanne, K.M., Baker, M.T., Mathiason, M.A. and Kallies, K.J. (2013) A Prospective, Blinded Comparison of Laparoscopic Ultrasound with Transabdominal Ultrasound for the Detection of Gallbladder Pathology in Morbidly Obese Patients. Journal of the American College of Surgeons, 216, 1057-1062.
http://dx.doi.org/10.1016/j.jamcollsurg.2013.02.009 - 49. Yeh, B.M., Breiman, R.S., Taouli, B., Qayyum, A., Roberts, J.P. and Coakley, F.V. (2004) Biliary Tract Depiction in Living Potential Liver Donors: Comparison of Conventional MR, Mangafodipir Trisodium-Enhanced Excretory MR, and Multi–Detector Row CT Cholangiography—Initial Experience. Radiology, 230, 645-651.
http://dx.doi.org/10.1148/radiol.2303021775 - 50. Einstein, D.M., Lapin, S.A., Ralls, P.W. and Halls, J.M. (1984) The Insensitivity of Sonography in the Detection of Choledocholithiasis. American Journal of Roentgenology, 142, 725-728.
http://dx.doi.org/10.2214/ajr.142.4.725 - 51. Turner, M.A. and Fulcher, A.S. (2001) The Cystic Duct: Normal Anatomy and Disease Processes. RadioGraphics, 21, 3-22.
http://dx.doi.org/10.1148/radiographics.21.1.g01ja093 - 52. Sarli, L., Costi, R., Gobbi, S., Sansebastiano, G. and Roncoroni, L. (2000) Asymptomatic Bile Duct Stones: Selection Criteria for Intravenous Cholangiography and/or Endoscopic Retrograde Cholangiography Prior to Laparoscopic Cholecystectomy. European Journal of Gastroenterology & Hepatology, 12, 1175-1180.
http://dx.doi.org/10.1097/00042737-200012110-00002 - 53. Majeed, A.W., Ross, B., Johnson, A.G. and Reed, M.W. (1999) Common Duct Diameter as an Independent Predictor of Choledocholithiasis: Is It Useful? Clinical Radiology, 54, 170-172.
http://dx.doi.org/10.1016/S0009-9260(99)91008-5 - 54. Lacaine, F., Corlette, M.B. and Bismuth, H. (1980) Preoperative Evaluation of the Risk of Common Bile Duct Stones. Archives of Surgery, 115, 1114-1116.
http://dx.doi.org/10.1001/archsurg.1980.01380090080019 - 55. Tandon, B.N., Rana, S. and Acharya, S.K. (1987) Bedside Ultrasonography: A Low-Cost Definitive Diagnostic Procedure in Obstructive Jaundice. Journal of Clinical Gastroenterology, 9, 353-356.
http://dx.doi.org/10.1097/00004836-198706000-00023 - 56. Khan, M.A., Khan, A.A. and Shafqat, F. (1996) Comparison of Ultrasonography and Cholangiography (ERCP/PTC) in the Differential Diagnosis of Obstructive Jaundice. Journal of the Pakistan Medical Association, 46, 188-190.
- 57. Weerakkody, Y., Gaillard, F., et al. (2013) Choledocholithiasis. Radiopedia.
- 58. Taylor, K.J.W., Rosenfield, A.T. and de Graaff, C.S. (1979) Anatomy and Pathology of the Biliary Tree as Demonstrated by Ultrasound. In: Taylor, K.W.J., Ed., Clinics in Diagnostic Ultrasound. Ultrasound in Gastrointestinal Disease, Vol. 1, Churchill Livingstone, New York, 103-121.
- 59. Yang, M.H., Chen, T.H., Wang, S.E., et al. (2008) Biochemical Predictors for Absence of Common Bile Duct Stones in Patients Undergoing Laparoscopic Cholecystectomy. Surgical Endoscopy, 22, 1620-1624.
http://dx.doi.org/10.1007/s00464-007-9665-2 - 60. Wilson, T.G., Hall, J.C. and Watts, J.M. (1986) Is Operative Cholangiography Always Necessary? British Journal of Surgery, 73, 637-640.
http://dx.doi.org/10.1002/bjs.1800730820 - 61. Huguier, M., Bornet, P., Charpak, Y., Houry, S. and Chastang, C. (1991) Selective Contraindications Based on Multivariate Analysis for Operative Cholangiography in Biliary Lithiasis. Surgery, Gynecology & Obstetrics, 172, 470-474.
- 62. Taylor, T.V., Armstrong, C.P., Rimmer, S., Lucas, S.B., Jeacock, J. and Gunn, A.A. (1988) Prediction of Choledocholithiasis Using a Pocket Microcomputer. British Journal of Surgery, 75, 138-140.
http://dx.doi.org/10.1002/bjs.1800750216 - 63. Puspok, A. (2007) Biliary Therapy: Are We Ready for EUS Guidance? Minerva Medica, 98, 379-384.
- 64. Cotton, P.B. (1993) Endoscopic Retrograde Cholangiopancreatography and Laparoscopic Cholecystectomy. American Journal of Surgery, 165, 474-478.
http://dx.doi.org/10.1016/S0002-9610(05)80944-4 - 65. Loperfido, S., Angelina, G., Benetti, G., Chilovi, F., Costan, F., et al. (1998) Major Early Complications from Diagnostic and Therapeutic ERCP a Prospective Multicenter Study. Gastrointestinal Endoscopy, 48, 1-10.
http://dx.doi.org/10.1016/S0016-5107(98)70121-X - 66. Hallal, A.H., Amortegui, J.D., Jeroukhimov, I.M., et al. (2005) Magnetic Resonance Cholangiopancreatography Accurately Detects Common Bile Duct Stones in Resolving Gallstone Pancreatitis. Journal of the American College of Surgeons, 200, 869-875.
http://dx.doi.org/10.1016/j.jamcollsurg.2005.02.028 - 67. Nathanson, L.K., O’Rourke, N.A., Martin, I.J., et al. (2005) Postoperative ERCP versus Laparoscopic Choledochotomy for Clearance of Selected Bile Duct Calculi: A Randomized Trial. Annals of Surgery, 242, 188-192.
http://dx.doi.org/10.1097/01.sla.0000171035.57236.d7 - 68. Prat, F., Amouyal, G., Amouyal, P., et al. (1996) Prospective Controlled Study of Endoscopic Ultrasonography and Endoscopic Retrograde Cholangiography in Patients with Suspected Common Bile Duct Lithiasis. The Lancet, 347, 75-79.
http://dx.doi.org/10.1016/S0140-6736(96)90208-1 - 69. Tseng, L.J., Jao, Y.T., Mo, L.R. and Lin, R.C. (2001) Over-the-Wire US Catheter Probe as an Adjunct to ERCP in the Detection of Choledocholithiasis. Gastrointestinal Endoscopy, 54, 720-723.
http://dx.doi.org/10.1067/mge.2001.119255 - 70. Putzer, G., Paal, P., Chemelli, A.P., Mark, W., Lederer, W. and Wiedermann, F.J. (2013) Resolution of Biliary Stricture after Living Donor Liver Transplantation in a Child by Percutaneous Trans-Hepatic Cholangiography and Drainage: A Case Report. Journal of Medical Case Reports, 7, 160.
http://dx.doi.org/10.1186/1752-1947-7-160 - 71. Cheng, C.L., Sherman, S., Watkins, J.L., et al. (2006) Risk Factors for Post-ERCP Pancreatitis: A Prospective Multicenter Study. The American Journal of Gastroenterology, 101, 139-147.
http://dx.doi.org/10.1111/j.1572-0241.2006.00380.x - 72. Ong, T.Z., Khor, J.L., Selamat, D.S., Yeoh, K.G. and Ho, K.Y. (2005) Complications of Endoscopic Retrograde Cholangiography in the Post-MRCP Era: A Tertiary Center Experience. World Journal of Gastroenterology, 11, 5209-5212.
- 73. Rhodes, M., Sussman, L., Cohen, L. and Lewis, M.P. (1998) Randomised Trial of Laparoscopic Exploration of Common Bile Duct versus Postoperative Endoscopic Retrograde Cholangiography for Common Bile Duct Stones. The Lancet, 351, 159-161.
http://dx.doi.org/10.1016/S0140-6736(97)09175-7 - 74. Neitlich, J.D., Topazian, M., Smith, R.C., Gupta, A., Burrell, M.I. and Rosenfield, A.T. (1997) Detection of Choledocholithiasis: Comparison of Unenhanced Helical CT and Endoscopic Retrograde Cholangiopancreatography. Radiology, 203, 753-757.
http://dx.doi.org/10.1148/radiology.203.3.9169700 - 75. Leopold, G.R. (1979) Ultrasonography of Jaundice. Radiologic Clinics of North America, 17, 127-136.
- 76. Cooperberg, P.L., Pon, M.S., Wong, P., Stoller, J.L. and Burhenne, H.J. (1979) Real-Time High Resolution Ultrasound in the Detection of Biliary Calculi. Radiology, 131, 789-790.
http://dx.doi.org/10.1148/131.3.789 - 77. O’Connor, H.J., Hamilton, I., Ellis, W.R., Watters, J., Lintott, D.J. and Axon, A.T. (1986) Ultrasound Detection of Choledocholithiasis: Prospective Comparison with ERCP in the Postcholecystectomy Patient. Gastrointestinal Radiology, 11, 161-164.
- 78. Shahin, W.A. and Bokhary, H.A. (2012) Characteristics of Endoscopic Retrograde Cholangiopancreatography (ERCP) Patients and Predictors of Common Bile Duct (CBD) Stones in a Tertiary Hospital, Saudi Arabia. International Journal of Public Health and Epidemiology, 1, 14-19.
- 79. Lindsell, D.R.M. (1990) Ultrasound Imaging of Pancreas and Biliary Tract. The Lancet, 335, 390-393.
http://dx.doi.org/10.1016/0140-6736(90)90217-S - 80. Koenigsberg, M., Wiener, S.N. and Waizer, A. (1979) The Accuracy of Sonography in the Differential Diagnosis of Obstructive Jaundice: A Comparison with Cholangiography. Radiology, 133, 157-165.
http://dx.doi.org/10.1148/133.1.157 - 81. Laing, F.C., Jeffrey, R.B. and Wing, V.W. (1984) Improved Visualization of Choledocholithiasis by Sonography. American Journal of Roentgenology, 143, 949-952.
- 82. Rickes, S., Treiber, G., Monkemüller, K., Peitz, U., Csepregi, A., Kahl, S., Vopel, A., Wolle, K., Ebert, M.P., Klauck, S. and Malfertheiner, P. (2006) Impact of the Operator’s Experience on Value of High-Resolution Transabdominal Ultrasound in the Diagnosis of Choledocholithiasis: A Prospective Comparison Using Endoscopic Retrograde Cholangiography as the Gold Standard. Scandinavian Journal of Gastroenterology, 41, 838-843.
- 83. Gross, B.H., Harter, L.P., Gore, R.M., Callen, P.W., Filly, R.A., Shapiro, H.A. and Goldberg, H.I. (1983) Ultrasound Evaluation of Common Bile Duct: Prospective Comparison with Endoscopic Retrograde Cholangiography. Radiology, 146, 471-474.
http://dx.doi.org/10.1148/radiology.146.2.6849097 - 84. Rigauts, H., Marchel, G., Van Steenbergen, W. and Ponette, E. (1992) Comparison of Ultrasound and E.R.C.P. in the Detection of the Cause of Obstructive Biliary Disease. Fortschr Roentgenstr & Nuklearmed, 156, 252-257.
- 85. Karki, S., Joshi, K.S., Regmi, S., Gurung, R.B. and Malla, B. (2013) Role of Ultrasound as Compared with ERCP in Patient with Obstructive Jaundice. Kathmandu University Medical Journal, 11, 237-240.
- 86. Chak, A., Hawes, R., Cooper, G., et al. (1999) Prospective Assessment of the Utility of EUS in the Evaluation of Gallstone Pancreatitis. Gastrointestinal Endoscopy, 49, 599-604.
http://dx.doi.org/10.1016/s0016-5107(99)70388-3 - 87. Callen, P.W. and Filly, R.A. (1981) Ultrasound in the Evaluation of Patients with Right Upper Quadrant Pain. Clinical Diagnostic Ultrasound, 7, 21-32.
- 88. Nurman, A. (2009) Imaging of Common Bile Duct Stones. Universa Medicina, 28, 59-68.
- 89. Holm, A.N. and Gerke, H. (2010) What Should Be Done with a Dilated Bile Duct? Current Gastroenterology Reports, 12, 150-156.
http://dx.doi.org/10.1007/s11894-010-0094-3 - 90. Godfrey, E.M., Rushbrook, S.M. and Carroll, N.R. (2010) Endoscopic Ultrasound: A Review of Current Diagnostic and Therapeutic Applications. Postgraduate Medical Journal, 86, 346-353.
http://dx.doi.org/10.1136/pgmj.2009.096065 - 91. Niederau, C., Sonnenberg, A. and Mueller, J. (1984) Comparison of the Extrahepatic Bile Duct Size Measured by Ultrasound and by Different Radiographic Methods. Gastroenterology, 87, 615-621.