Open Journal of Gastroenterology
Vol. 2 No. 4 (2012) , Article ID: 25061 , 4 pages DOI:10.4236/ojgas.2012.24032
Barrett’s esophagus following treatment of achalasia with botulinum toxin
![]()
1Division of Gastroenterology, Mayo Clinic, Rochester, USA
2Department of Surgery, Mayo Clinic, Rochester, USA
Email: *harold.kristi@mayo.edu
Received 21 July 2012; revised 22 August 2012; accepted 30 August 2012
Keywords: Barrett’s Esophagus; Achalasia; Botulinum Toxin
ABSTRACT
Achalasia is an uncommon primary motor disorder of the esophagus with an annual incidence of 0.8/100,000. Very few cases of coexistent Barrett’s esophagus (BE) and achalasia in patients without prior surgical myotomy or pneumatic dilation have been reported. We report the case of a 65 year old female who was diagnosed with achalasia in June 2002. Endoscopy at that time revealed biopsy-confirmed normal esophageal mucosa. The patient subsequently underwent two trials of botox injection with progressively worsening dysphagia. A repeat endoscopy two years later showed a short segment of salmon-colored mucosa in the distal esophagus which was biopsy-confirmed Barrett’s epithelium with no dysplasia. The patient eventually underwent laparoscopic Heller myotomy and Toupet fundoplication. Postopera tively, she recovered well and with significant alleviation of her dysphagia. This study reviews reported cases of coexistent achalasia and BE, and discusses possible etiologies of concurrent BE and achalasia, and implications for treatment.
1. INTRODUCTION
Achalasia is a primary motor disorder of the esophagus first recognized greater than 300 years ago when it was thought to be due to an obstruction of the distal esophagus at the cardiac sphincter and was termed “cardiospasm” [1]. In 1937 Lendrum proposed that the syndrome was due to incomplete relaxation of the lower esophageal sphincter (LES), and he renamed it achalasia (literally, “failure to relax”) [1]. The manometric characteristics of achalasia are aperistalsis of the lower twothirds of the esophagus and incomplete relaxation of the LES [2]. Pathologically, there is a marked decrease or even absence of myenteric ganglion cells in the esophageal body as well as collagen replacement of myenteric nerves [3]. Similar findings are noted at the LES. Excitatory neurons (predominantly cholinergic) are unaffected while inhibitory neurons are dramatically reduced in number [4].
Barrett’s esophagus (BE) is characterized by the replacement of the normal squamous epithelium in the distal esophagus by columnar epithelium which demonstrates specialized intestinal metaplasia. This condition seems to occur most often as a consequence of gastroesophageal reflux disease (GERD). Repeated acid exposure leads to a chronic inflammatory state prompted by cycles of mucosal injury and healing which ultimately initiates a metaplastic process whereby columnar cells replace squamous cells [5]. This epithelium is characterized by mucin-containing goblet cells, and it is only with these pathologic changes that a diagnosis of BE can be made. BE is a concerning finding given the risk of progression to dysplasia and adenocarcinoma [6].
Simply put, achalasia results from a hypertensive LES while BE results from a hypotensive LES. This article examines the coexistence of these disease processes in one patient who developed BE after diagnosis of achalasia prior to either surgical therapy or endoscopic dilation, and reviews relevant literature and implications for treatment.
2. CASE PRESENTATION
A 65 year-old female patient originally presented with a one-year history of dysphagia. Esophageal motility studies demonstrated aperistalsis, a resting LES pressure of 27 mmHg, and incomplete LES relaxation. An esophagogastroduodenoscopy (EGD) revealed retained food in a dilated esophagus and mild inflammatory changes of the distal esophagus. Biopsies showed squamous esophageal mucosa without diagnostic abnormality. The patient was diagnosed with achalasia and treated with endoscopic injection of botulinum toxin.
This intervention provided her relief for one year, but a second endoscopic injection of botulinum toxin was required, and this relieved her symptoms for three months. Several months later, an EGD at another institution demonstrated a short segment of salmon-colored mucosa in the distal esophagus which was biopsy-confirmed Barrett’s epithelium. One year after the second botulinum toxin injuection, the patient underwent an EGD which demonstrated retained food in the esophagus and a one centimeter segment that was both endoscopically and pathologically consistent with BE (Figure 1). She received a third endoscopic treatment with botulinum toxin at the time of this EGD, but unfortunately symptoms recurred two weeks later.
The patient underwent a laparoscopic Heller esophageal myotomy with Toupet fundoplication in November 2004. At her postoperative visit two weeks later, the patient reported resolution of dysphagia and gastroesophageal reflux, and improved quality of life.
3. DISCUSSION
Coexistent BE and achalasia in this patient may be explained by several different theories. The exact pathogenesis is not certain in this case, but we know that the patient had close endoscopic follow-up and that the short segment BE was observed two years after the diagnosis of achalasia. Endoscopic observations are imperfect, but

Figure 1. Endoscopic photograph of distal esophagus demonstrating retained food and salmon-colored mucosa typical of Barrett’s esophagus.
this patient had repeated exams which lessens the likelihood of BE being missed earlier. Additionally, this patient had not received definitive treatment for achalasia at the time of diagnosis of BE, thus the presence of BE was not unmasked by dilation or myotomy. Finally, the contribution of botulinum toxin into the equation may be relevant. The patient did have two separate treatments with symptomatic improvement prior to the diagnosis of BE, which suggests that botulinum toxin was effective in reducing the pressure of the LES, and possibly contributing to the development of BE.
This patient developed BE only after the onset of achalasia. During an interval of approximately two years, the patient underwent two EGDs at our institution, and at least one more at another hospital without evidence of BE. The patient had received two separate treatments with botulinum toxin during that same interval. The known cases of coexistent achalasia and BE will be reviewed, followed by theories regarding their coexistence, and finally, implications for treatment.
Previously published cases of the coexistent BE and achalasia in patients without prior surgical myotomy or pneumatic dilation is presented in Table 1 [7-13]. Based on English literature review, this appears to be the tenth case reported. In three cases, no information is available regarding age, presence of GERD, segment length, or malignancy. Wide variability in patient demographics and treatments were noted. In three patients, the syndrome of achalasia occurred in the presence of a malignancy, and this is more accurately known as secondary achalasia or pseudoachalasia [2]. Other possible causes include, GERD leading to BE with subsequent independent development of achalasia. Moreover, achalasia could lead to BE via local effects of retained food with fermentation and lactate production. Achalasia could lead to BE due to prolonged contact time of any acid that refluxes across the LES. Finally, treatment of achalasia could lead to GERD and subsequently BE.
Coexistent BE and achalasia may simply occur coincidentally. The prevalence of BE varies widely in the literature. Some of this variation may be attributed to the definition of BE used by that particular study. An autopsy study of 223 consecutive patients demonstrated a prevalence of 0.9% [14]. Another study which performed upper endoscopy on patients presenting for colonoscopy found a prevalence of 6.8% in 961 patients [15]. The annual incidence of achalasia has been reported to be 0.8 per 100,000 in a Scottish population study [16]. It has been well-described that patients with symptomatic GERD who later develop achalasia may become asymptomatic with respect to GERD as symptoms of dysphagia progress [9]. This pattern of symptoms occurs as the LES pressure increases, preventing reflux and producing dysphagia. This theory does not explain the onset of
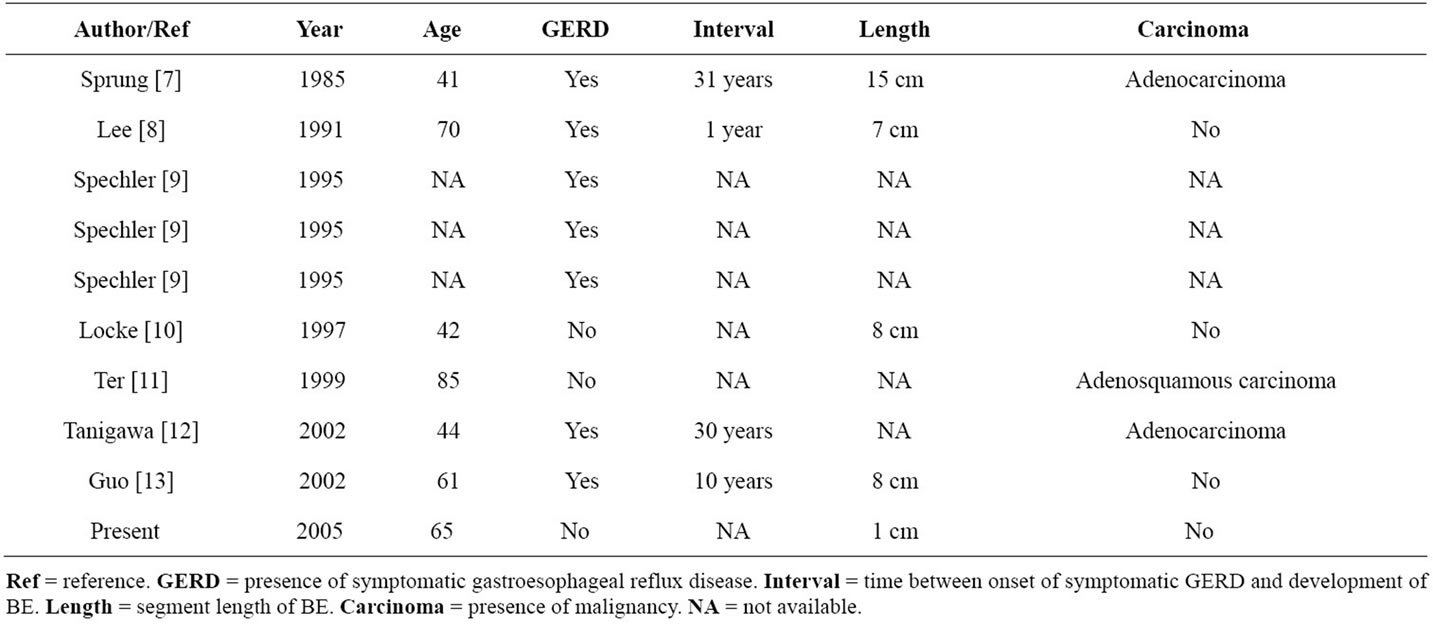
Table 1. Reported cases of BE and achalasia without prior myotomy or pneumatic dilation for achalasia.
achalasia but recognizes that co-existence need not imply causation.
Alternatively, BE could be caused by chronic irritation. For instance, retained food in the esophagus due to incomplete relaxation of LES is often be encountered in patients with achalasia [2]. If food remains in the esophagus long enough, fermentation may occur. Intraesophageal pH has been shown to be lower in achalasia patients with retained food compared to achalasia patients without retained food. The lower pH is thought to be due to lactic acid, which has been demonstrated in the esophagus [17]. It is conceivable that this lactic acid formed through fermentation of food could injure the esophageal mucosa and induce the changes seen in BE. Whether or not this occurs is unknown, although some ex vivo models suggest that BE is due to the episodic presence of acid in the distal esophagus rather than continuous exposure [18].
One final explanation is that patients with achalasia exhibit significant variability in the mean LES pressure and may even have episodes of complete relaxation of the LES [9,19]. This transient relaxation may lead to reflux, and subsequently the acid is cleared less effectively due to aperistalsis and elevated pressures at the LES that are typical of achalasia. It is difficult to interpret pH data in these patients because of the possible contribution of fermentation, but in one study 10 of 48 patients with untreated achalasia had evidence of abnormal acid exposure without any difference in the presence of retained food. This study did not note any difference in mean LES pressures between patients with and without abnormal pH readings [20].
The idea that treatment for achalasia can, in rare cases, lead to BE has implications for clinical decision-making and increased level of suspicion and post-myotomy surveillance for BE. With laparoscopic techniques increasingly utilized, most patients without serious comorbidities will elect to undergo a laparoscopic surgical myotomy and fundoplication which would decreased incidence of post-operative GERD, but not eliminate the risk of BE [21,22].
![]()
![]()
REFERENCES
- Spiess, A.E. and Kahrilas, P.J. (1998) Treating achalasia: From whalebone to laparoscope. The Journal of the American Medical Association, 280, 638-642. doi:10.1001/jama.280.7.638
- Reynolds, J.C. and Parkman, H.P. (1989) Achalasia. Gastroenterology Clinics of North America, 18, 223-255.
- Goldblum, J.R., Whyte, R.I., Orringer, M.B., et al. (1994) Achalasia. A morphologic study of 42 resected specimens. The American Journal of Surgical Pathology, 18, 327- 337. doi:10.1097/00000478-199404000-00001
- Singaram, C., Koch, J., Gaumnitz, E.A., et al. (1996) Nature of neuronal loss in human achalasia. Gastroenterology, 110, A259.
- Spechler, S.J. (2002) Clinical practice. Barrett's Esophagus. The New England Journal of Medicine, 346, 836- 842. doi:10.1056/NEJMcp012118
- Goldblum, J.R. (2003) Barrett’s esophagus and Barrett’srelated dysplasia. Modern Pathology, 16, 316-324. doi:10.1097/01.MP.0000062996.66432.12
- Sprung, D.J. and Gibb, S.P. (1985) Barrett’s esophagus in a patient with achalasia. American Journal of Gastroenterology, 80, 330-333.
- Lee, F.I. and Bellary, S.V. (1991) Barrett’s esophagus and achalasia: A case report. Journal of Clinical Gastroenterology, 13, 559-561. doi:10.1097/00004836-199110000-00017
- Spechler, S.J., Souza, R.F., Rosenberg, S.J., et al. (1995) Heartburn in patients with achalasia. Gut, 37, 305-308. doi:10.1136/gut.37.3.305
- Locke, M., Bank, S., Gross, C., et al. (1997) Long segment Barrett’s esophagus in a young patient with achalasia. American Journal of Gastroenterology, 92, 1596.
- Ter, R.B., Govil, Y.K., Leite, L., et al. (1999) Adenosquamous carcinoma in Barrett’s esophagus presenting as pseudoachalasia. American Journal of Gastroenterology, 94, 268-270. doi:10.1111/j.1572-0241.1999.00813.x
- Tanigawa, H., Kida, Y., Kuwao, S., et al. (2002) Hepatoid adenocarcinoma in Barrett’s esophagus associated with achalasia: First case report. PATH International, 52, 141- 146. doi:10.1046/j.1440-1827.2002.01328.x
- Guo, J.-P., Gilman, P.B., Thomas, R.M., et al. (2002) Barrett’s esophagus and achalasia: A report of two cases and review of the literature. Journal of Clinical Gastroenterology, 34, 439-443. doi:10.1097/00004836-200204000-00011
- Ormsby, A.H., Kilgore, S.P., Goldblum, J.R., et al. (2000) The location and frequency of intestinal metaplasia at the esophagogastric junction in 223 consecutive autopsies: Implications for patient treatment and preventive strategies in Barrett’s esophagus. Modern Pathology, 13, 614- 620. doi:10.1038/modpathol.3880106
- Rex, D.K., Cummings, O.W., Shaw, M., et al. (2003) Screening for Barrett’s esophagus in colonoscopy patients with and without heartburn. Gastroenterology, 125, 1670-1677. doi:10.1053/j.gastro.2003.09.030
- Howard, P.J., Maher, L., Pryde, A., et al. (1992) Five year prospective study of the incidence, clinical features, and diagnosis of achalasia in Edinburgh. Gut, 33, 1011- 1015. doi:10.1136/gut.33.8.1011
- Smart, H.L., Foster, P.N., Evans, D.F., et al. (1987) Twentyfour-hour acidity in achalasia before and after pneumatic dilation. Gut, 28, 883-887. doi:10.1136/gut.28.7.883
- Fitzgerald, R.C., Omary, M.B. and Triadafilopoulos, G. (1996) Dynamic effects of acid on Barrett’s esophagus. An ex vivo proliferation and differentiation model. The Journal of Clinical Investigation, 98, 2120-2128. doi:10.1172/JCI119018
- Katz, P.O., Richter, J.E., Cowan, R., et al. (1986) Apparent complete lower esophageal relaxation in achalasia. Gastroenterology, 90, 978-983.
- Shoenut, J.P., Micflikier, A.B., Yaffe, C.S., et al. (1995) Reflux in untreated achalasia patients. Journal of Clinical Gastroenterology, 20, 6-11. doi:10.1097/00004836-199501000-00004
- Richards, W.O., Torquati, A., Holzman, M.D., et al. (2004) Heller myotomy versus Heller myotomy with Dor fundoplication for achalasia: A prospective randomized double-blind clinical trial. Annals of Surgery, 240, 405-415. doi:10.1097/01.sla.0000136940.32255.51
- Harold, K.L., Matthews, B.D., Kercher, K.W., et al. (2003) Surgical treatment of achalasia in the 21st century. Southern Medical Journal, 96, 1-5.
NOTES
*Corresponding author.

