Open Journal of Metal
Vol.2 No.1(2012), Article ID:18316,7 pages DOI:10.4236/ojmetal.2012.21004
Cobalt Recovery from Waste Catalysts (Petroleum Refining Industry from Gujarat)
Department of Electro-Hydro Metallurgy, Central Electrochemical Research Institute, Karaikudi, India
Email: *sivachemist@rediffmail.com
Received December 30, 2011; revised February 2, 2012; accepted February 15, 2012
Keywords: Hydrometallurgical; Electrowinning; Cobalt; Flux Material; Waste Catalysts; H2SO4 Leaching
ABSTRACT
A hydrometallurgical process has been developed for cobalt recovery from a waste catalyst (petroleum refining Industry). This waste catalyst containing about 2.18 weight % of Co, is highly contaminated by Mg, Al, Si, Ca, Fe, Ni, Cu, Zn, Mo. The major steps are: 1) The spent catalyst is roasted with flux material in an electrical furnace at very high temperature (700˚C) for a specific duration; 2) The roasted sample is leached with sulphuric acid to bring the metal contents into solution form; 3) For separating cobalt values from the leach solution, the solution pH is raised by NaOH addition, where all cobalt content is precipitated at a pH of about 12; 4) This cobalt hydroxide precipitate is filtered and dissolved in minimum amount of sulphuric acid to get cobalt sulphate solution which is used as the electrolyte for the electrolytic recovery of cobalt. For optimizing various parameters like 1) H2SO4 concentration; 2) Duration; 3) Cobalt concentration; 4) Current density; 5) Temperature; 6) Stirring etc. The particle surface morphology and deposited layers have been characterized by scanning electron microscopy (SEM). A compact metallic deposit containing 70% cobalt was obtained.
1. Introduction
Cobalt compounds have been used for centuries to impart a rich blue colour to glass, glazes and ceramics. Cobalt has been detected in Egyptian sculpture and Persian jewelry from the third millennium BC, in the ruins of Pompeii (destroyed AD 76), and in China dating from the Tang dynasty (AD 618-907) and the Ming dynasty (AD 1368-1644). Cobalt glass ingots have been recovered from shipwrecks dating to the time of the Minoans.
Cobalt is a strategic and critical metal used in many diverse industrial applications [1,2]. Cathode cobalt is used primarily to make superalloys for gas turbine engines, but is also extensively used in chemicals, carbide and diamond tools and magnets [3]. In addition, cobalt finds application in medical implants, oil desulphurization, ceramics and the computer industry, each requiring high purity cathode cobalt which is 99.8% pure. It was stated that the production of refined cobalt in 1997 amounted to 24.471 t from a product mixture of metal and chemicals.
Various forms of cobalt metal, including briquettes, cathode (electrolytic cobalt), fines, granules (shot), ingot, powder, and have been produced and marketed [4]. The most popular form is cathode cobalt involving sulfate medium from a pressure leaching operation. Cobalt is typically associated with heavy metals such as nickel, zinc and copper; hence cobalt is usually extracted in small quantities as a byproduct from non-ferrous metal production. Most cobalt production is associated with nickel production from sulfide ores as well as laterites. Pressure leaching, solvent extraction followed by the electro winning of nickel and cobalt, is the preferred route for nickel and cobalt processing, replacing the reduction roasting and electrorefining process route. This processing route allows recovery rates of over 90% Co and Ni much higher than 50% for the reduction roasting route, much higher recovery rates of cobalt require novel technology in cobalt electrowinning.
The main media for cobalt electrowinning are sulfate or chloride solutions. In the electrowinning of cobalt from a sulfate solution, cobalt is deposited on the cathode coupled with oxygen evolution at the anode and in chloride solutions and the deposition is coupled with chlorine gas evolution at the anode, from chloride medium [5].
Catalysts for the Petroleum and Chemical Industries
Several cobalt compounds are used in several chemical reactions as catalysts [1]. Cobalt acetate is used for the production of terephthalic as well as dimethyl terephthalic acids, which are the key compounds for the production of PET. The steam reforming and hydrodesulfurization for the production of petroleum, which uses mixed cobalt-molybdenum aluminum oxides as catalyst is another important application. Cobalt and its compounds especially cobalt carboxylates, known as cobalt soaps are good oxidation catalysts. These are used in paints, varnishes, and ink as drying agents in which the oxidation of certain compounds leads to the drying. The same carboxylates are used to improve the adhesion of the steel to rubber in steel belted radial tires [1,2].
2. Experimental
Initially synthetic cobalt solution is employed for the electrolytic recovery. After optimizing various parameters like acid concentration, duration, cobalt concentration, current density, temperature, stirring etc., for maximum cobalt recovery, commercial waste Petroleum industry catalyst is used to recover cobalt content by roastingleaching step. From the leach solution cobalt was precipitated and redissolved in sulphuric acid which was used for electrolysis of cobalt recovery.
2.1. Estimation of Cobalt in Electrolyte
10 - 20 ml (exactly measured) of cobalt solution is taken in 250 ml conical flask. EDTA solution in excess over the amount of cobalt is added and then 5 ml of buffer (pH5) and 3 - 5 drops of PAN indicator. The solution is diluted to about 100 ml and titrated with the copper solution until the colour just change to violet at the end point [6,7].
2.2. Electrolytic Cell Set up
The experiments are carried out in a 250 ml glass beaker using cobalt sulfate and sulphuric acid as the electrolyte. The stainless steel is used as cathode (one side) while lead metal with silver as impurity (1%) is used as the anode. Electrowinning experiments are carried out under different conditions to optimize various parameters for maximum cobalt recovery [5].
The electrodes are connected to the DC power supply (rectifier). The cathode is connected to the –ve terminal while anode is connected to the +ve terminal of the rectifier. The cell voltage between the two electrodes is measured employing a multimeter.
The cathode is polished one side insulated and weighed before electrolysis. After the electrolysis the cathode with cobalt deposit is removed from the cell, washed dried and accurately weighed. The difference in the cathode weight gives the weight of the cobalt deposited from which the current efficiency is calculated.
3. Results and Discussion
Cobalt is considered as a speciality metal. In India cobalt is mainly produced as by-product during the electrolytic recovery of copper and nickel. Cobalt is used for making: super alloys (for parts in aircraft engines), corrosion and wear resistant alloy, high speed stock, catalysts in petroleum industries cemented carbides and diamond tools, magnetic materials etc. [3].
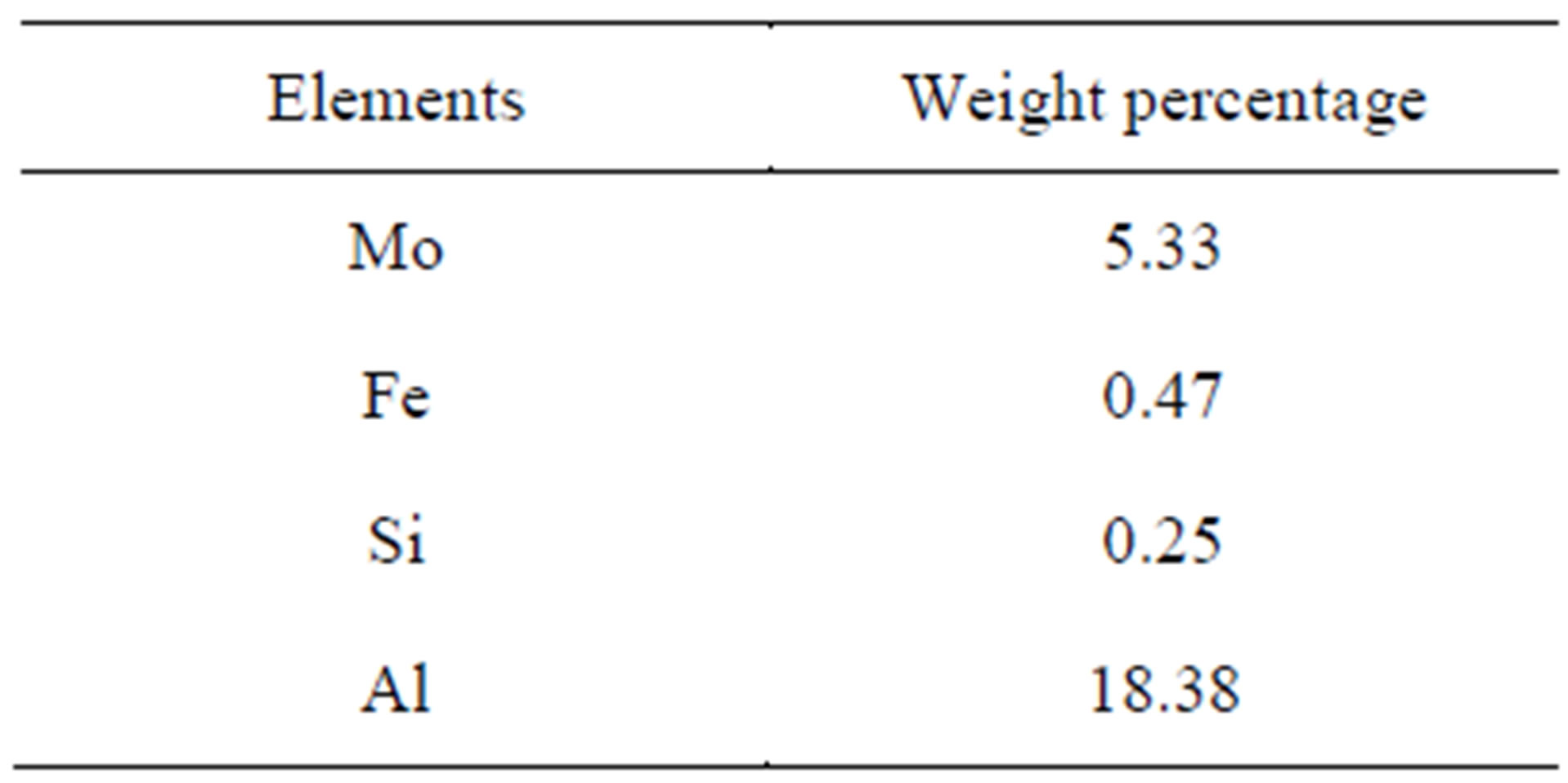
In India cobalt consumption is more than its production. Hence to bridge the gap between production and consumption of cobalt, it is very important to recover cobalt from various secondary sources [8]. The cobalt sources mentioned above become secondary sources after their useful activity. One such example is waste catalyst used in petroleum industry for hydrodesulfurization of crude oil. The project work constitutes the recovery of cobalt from the waste catalyst of petroleum industries whose reported wt% is between 3% - 9% depending on the source. In this investigation spent catalyst from Gujarat is employed as the raw material.
This spent catalyst mostly consists of Mo, Co, Ni, Fe, Si, Al as their oxides. To bring the cobalt content into solution from the fine powder, spent catalyst 5 gm of this powder catalyst is roasted with 10 gm of flux material like sodium carbonate taken in a clay crucible, and heated in an electric furnace at 700˚C for 3 hour duration where in this roasting unwanted impurities are removed as slags. The melt is removed, powdered and leached with 30% H2SO4 solution for 60 minutes under stirring at 70˚C and filtered. Since along with cobalt, nickel is also leached, this nickel is removed by adding 10% DMG (dimethyl glyoxime) solution to the filtrate to precipitate nickel as nickel-dimethyl glyoxime. This is warmed and the precipitate is filtered and the clear filtrate is made up to a known volume in a standard volumetric flask. Its cobalt content is estimated volumetrically and the solution is concentrated to raise the cobalt level to about 30 g/L Figure 1.
The optimizing various parameters like 1) H2SO4 concentration; 2) duration; 3) cobalt concentration; 4) current density; 5) temperature and 6) stirring for maximum cobalt recovery through electrowinning and finally at these optimized conditions, cobalt metal is recovered from waste catalyst leach solution.
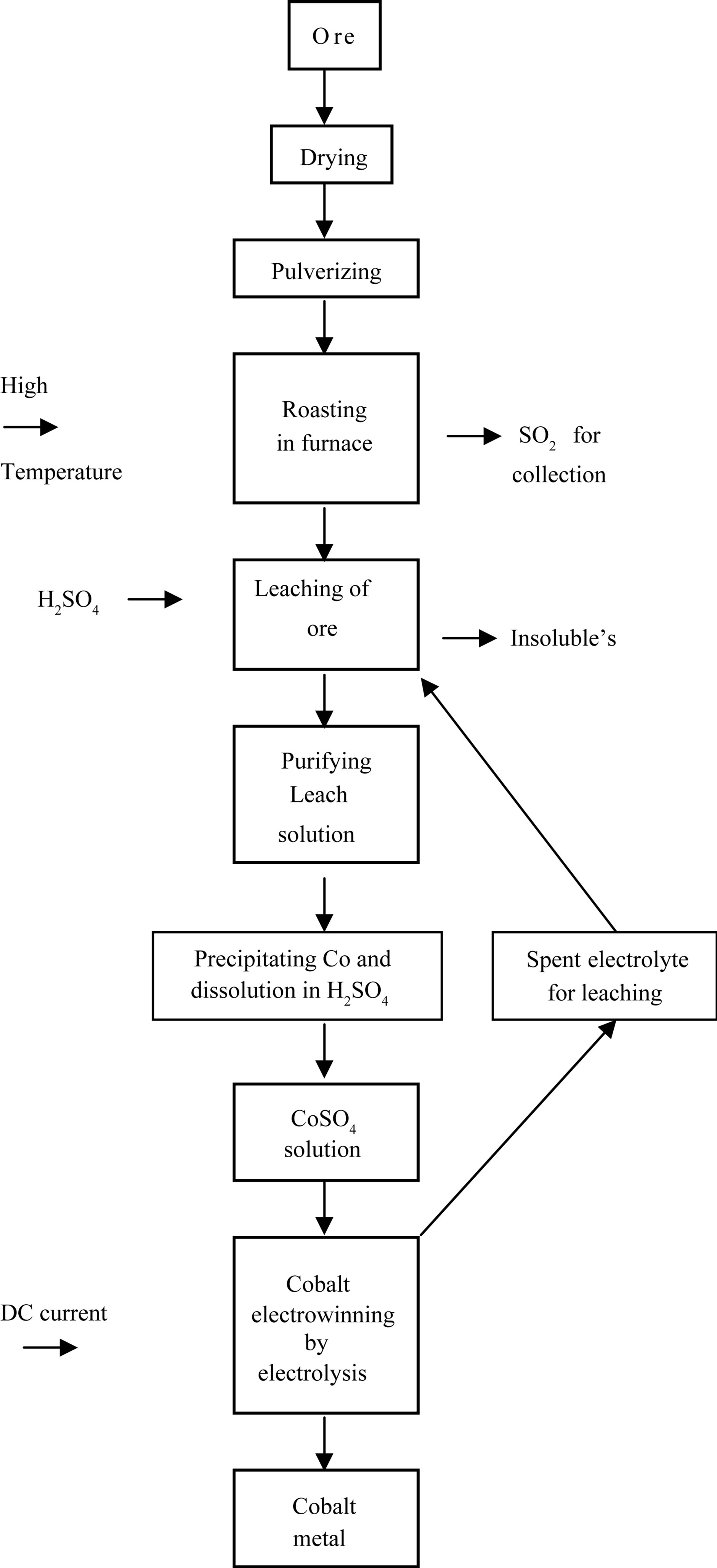
Figure 1. Flow chart on cobalt recovery.
3.1. Effect of H2SO4 Concentration
Effect of H2SO4 concentration on cobalt recovery from synthetic 30 g/L Co as CoSO4 solution is given in Table 1 (Figure 2). The H2SO4 concentration studied is in the range of 0 - 100 g/L. As the H2SO4 concentration increases from 0 to 100 g/L the current efficiency on cobalt recovery decreases from 85.5% to 14.2%. The decrease in current efficiency may be due to the redissolution of cobalt deposit which increase with H2SO4 concentration. i.e. As the concentration of H2SO4 increases, decreases in
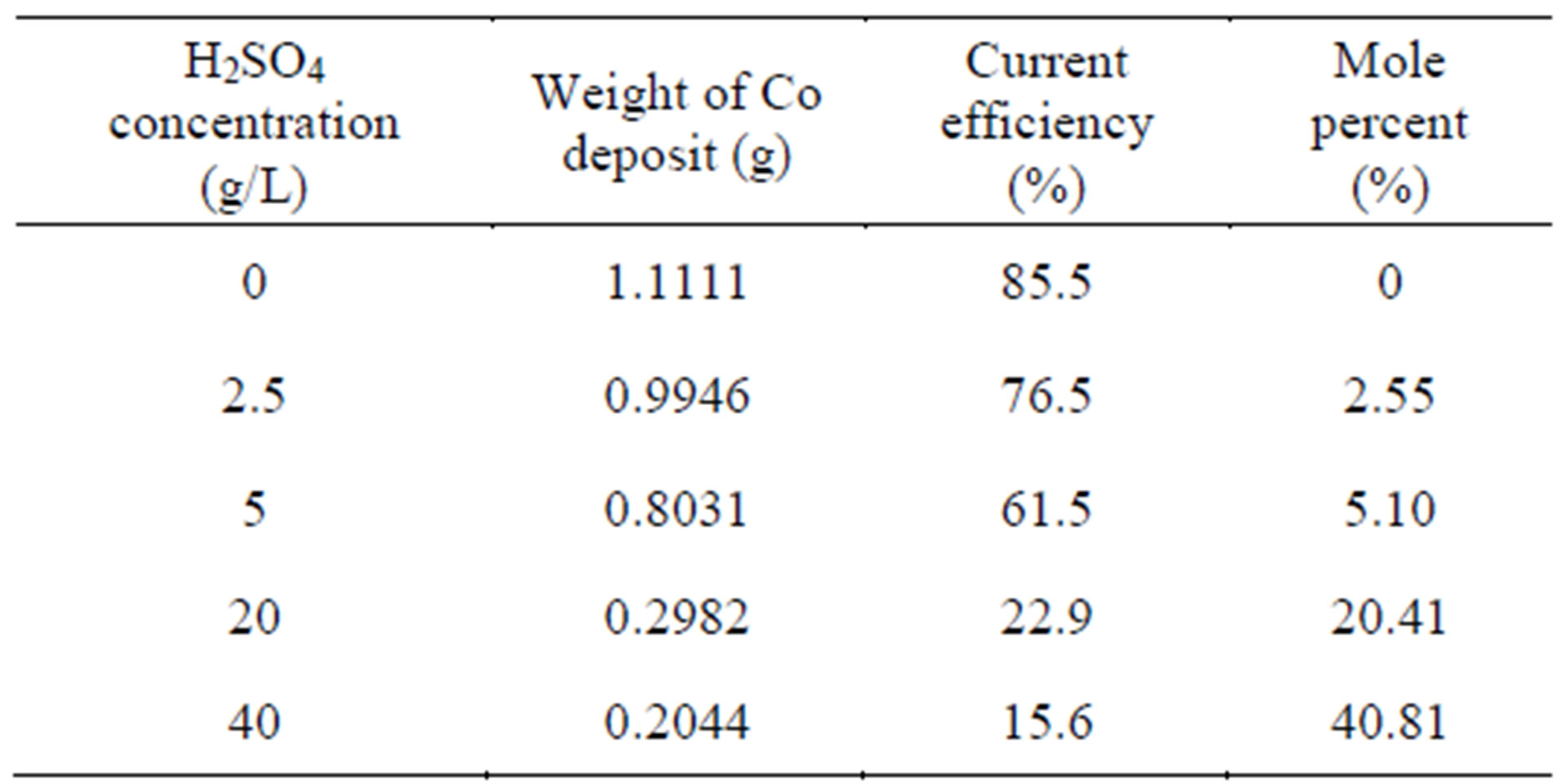
Table 1. Effect of acid concentration on cobalt electro winning.
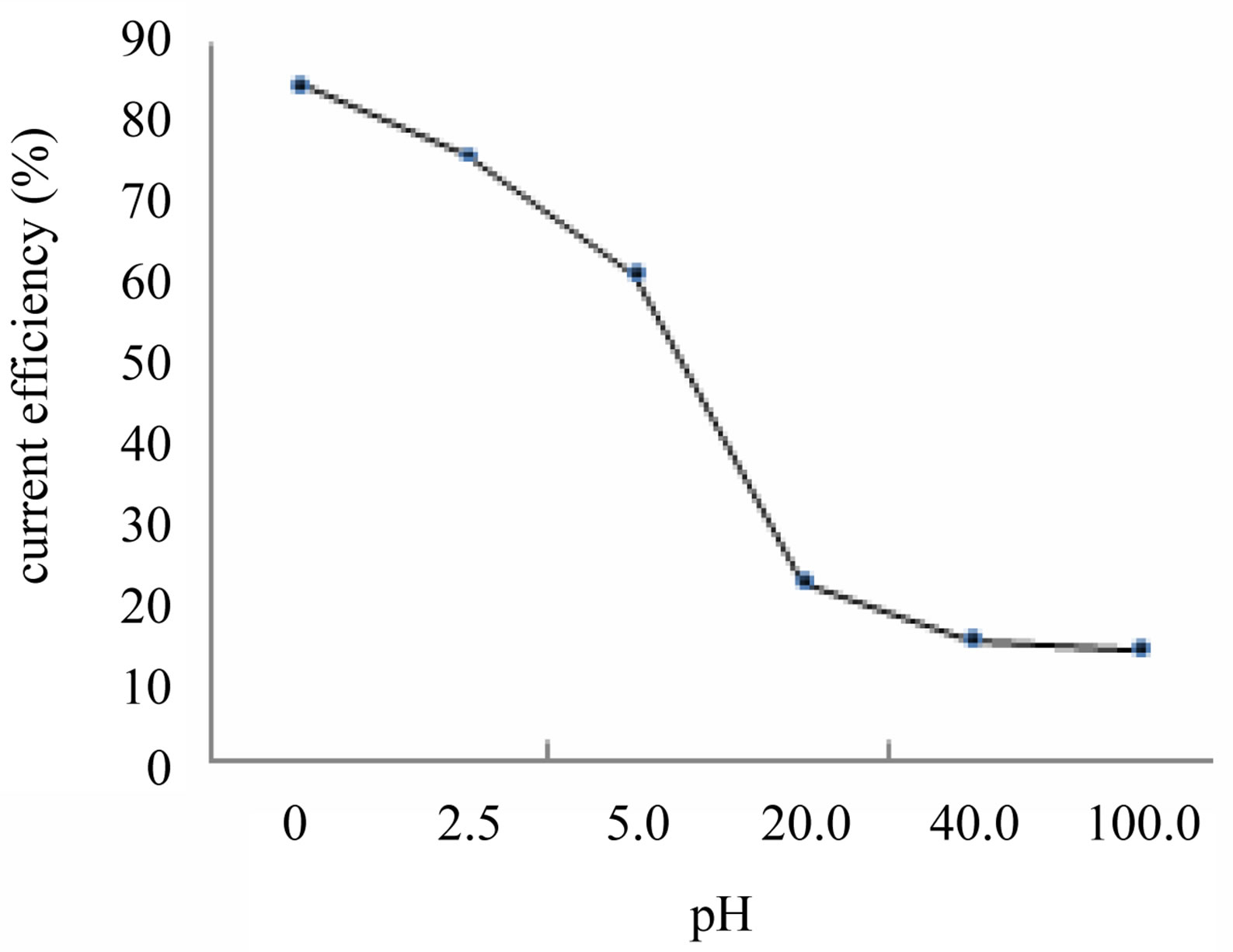
Figure 2. Effect of acid concentration (pH) on cobalt recovery.
the current efficiency on cobalt recovery. Whereas, the concentration of H2SO4 is in 0 g/L, the current efficiency and deposition of cobalt recovery are raised at a maximum level.
For any electrolysis, the cell voltage is a critical factor and it is directly related to power consumption. Only this 5 g/L H2SO4 case has moderate cell voltage of 4.43 V and hence 5 g/L H2SO4 is taken as optimum concentration.
3.2. Effect of Duration
Table 2 and Figure 3 show the effect of duration (30 - 180 min) on cobalt recovery. All experiments are carried out at optimized H2SO4 concentration of 5 g/L. As the duration increases from 30 minute, the current efficiency for cobalt recovery also increases and it reaches a maximum value of 61.5% at 120 minutes duration after which the current efficiency starts decreasing. From this table it seems that 120 minutes duration is enough for the cobalt recovery under the specified electrolytic conditions and therefore 120 minutes is found to be optimum duration, In all theses cases cobalt metal foil in gray colour is deposited.
3.3. Effect of Cobalt Concentration
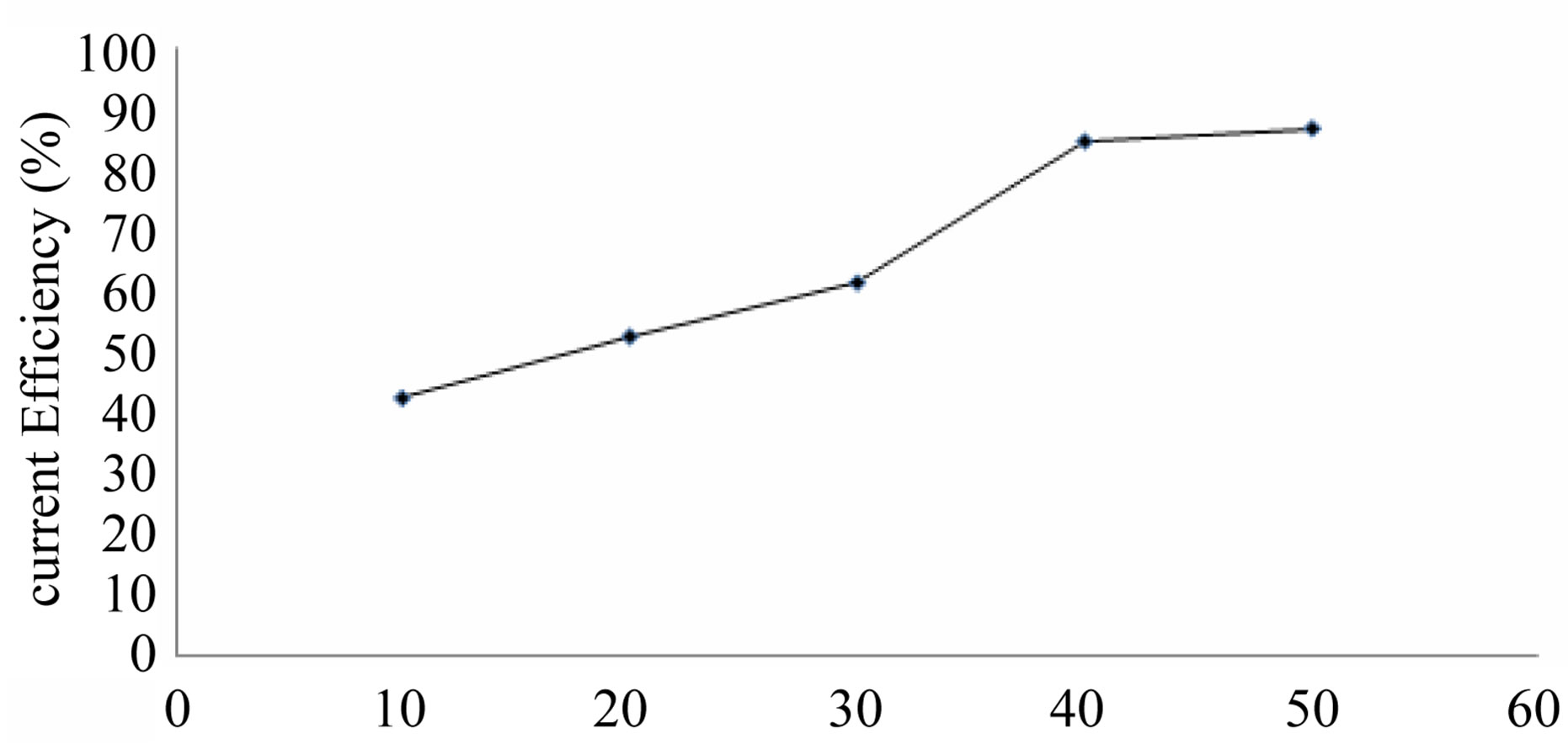
Effect of cobalt concentration (10 - 50 g/L) on cobalt
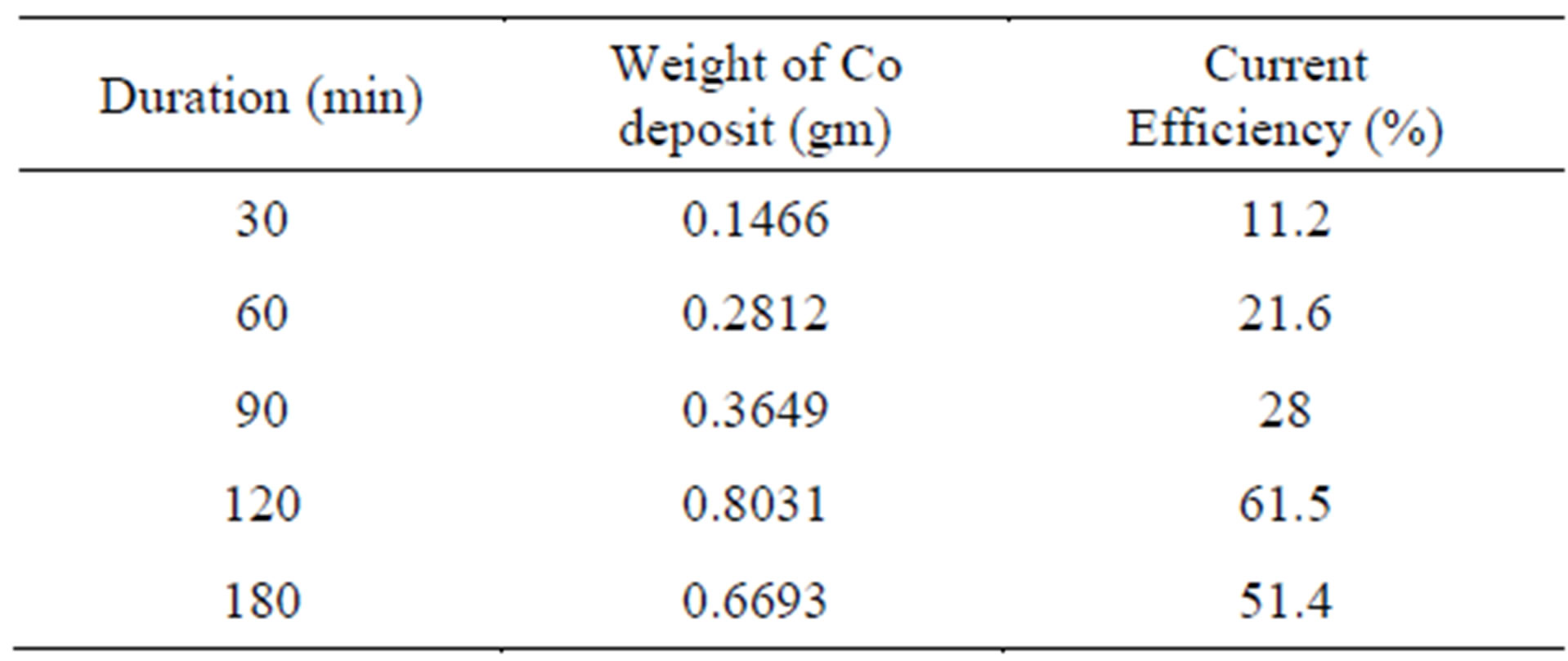
Table 2. Effect of duration on cobalt electrowinning.
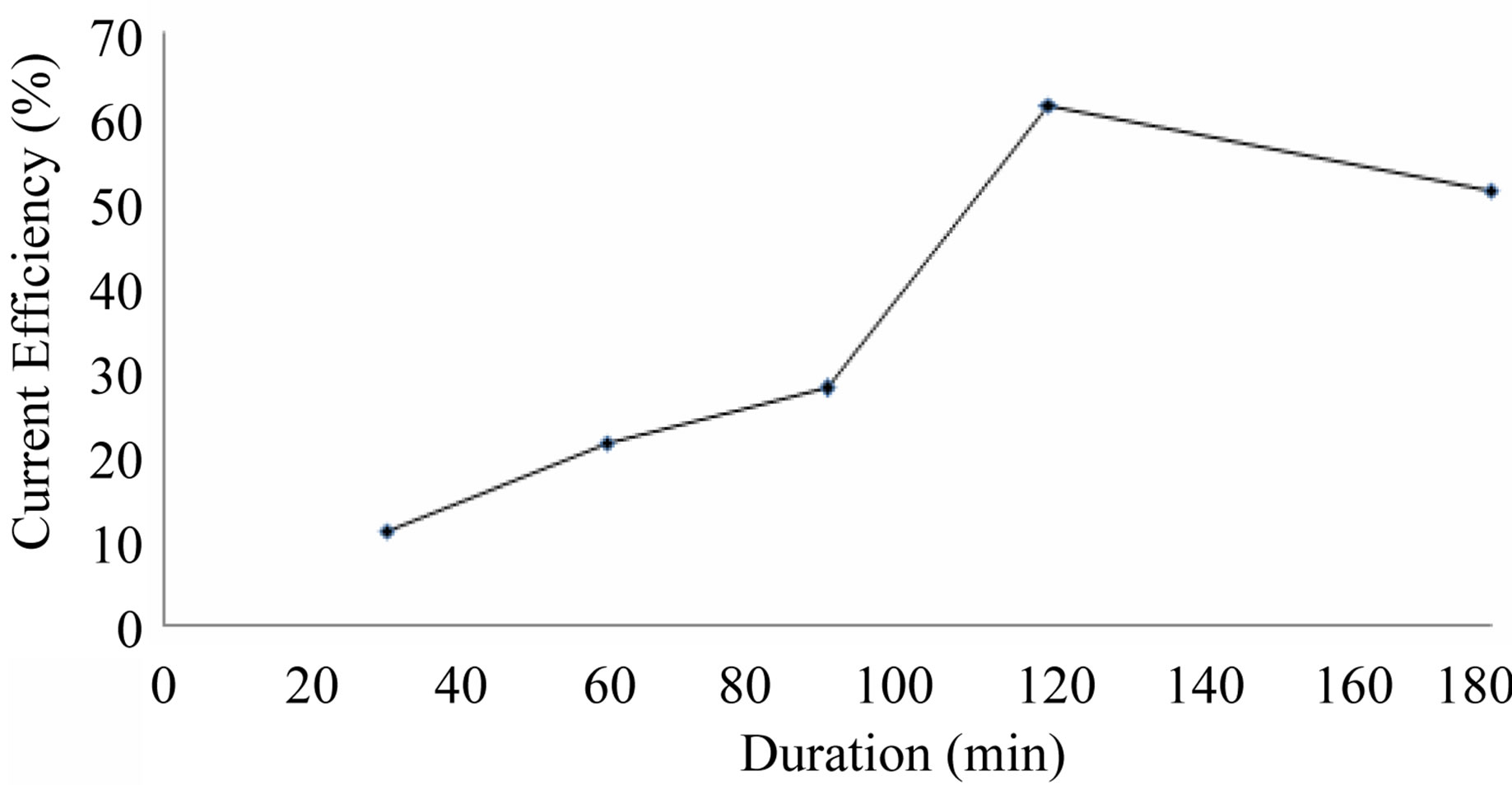
Figure 3. Effect of duration on cobalt recovery.
recovery is shown in Table 3 and Figure 4. As in expected with increase in cobalt concentration the current efficiency on cobalt recovery also increases and from this table 30 g/L Co is chosen as best optimum level and above and below this there is a rise in all voltage, which may be due to decrease in conductivity of the electrolyte.
3.4. Effect of Current Density
Current density is directly related to production/recovery of product. Table 4 and Figure 5 show the effect of current density (150 - 400 A/M2) on cobalt recovery.
Generally with low current density the rate of metal deposit will be less and hence at 150 A/M2 current density, very low cobalt weight is observed with low current efficiency of 64.5%. With increase in current density the metal recovery increases and therefore current efficiency increases. But after 200 A/M2 case, the current efficiency decreases due to the more liberation of hydrogen gas at the cathode besides the cobalt metal deposition. Hence from this table, 200 A/M2 is found to be optimum current density.
3.5. Effect of Temperature
The effect of temperature (30˚C - 60˚C) on cobalt recovery/current efficiency is provided in the Table 5 and Figure 6. At 30˚C, the current efficiency is 85.1%. This reaches little high value of 97.4% at 60˚C. But for conducting experiments at 60˚C more electric power is required. Hence in order to reduce cost of cobalt recovery, the room temperature 30˚C is arrived as optimum temperature on cobalt recovery.

Table 3. Effect of cobalt concentration on cobalt electro winning.
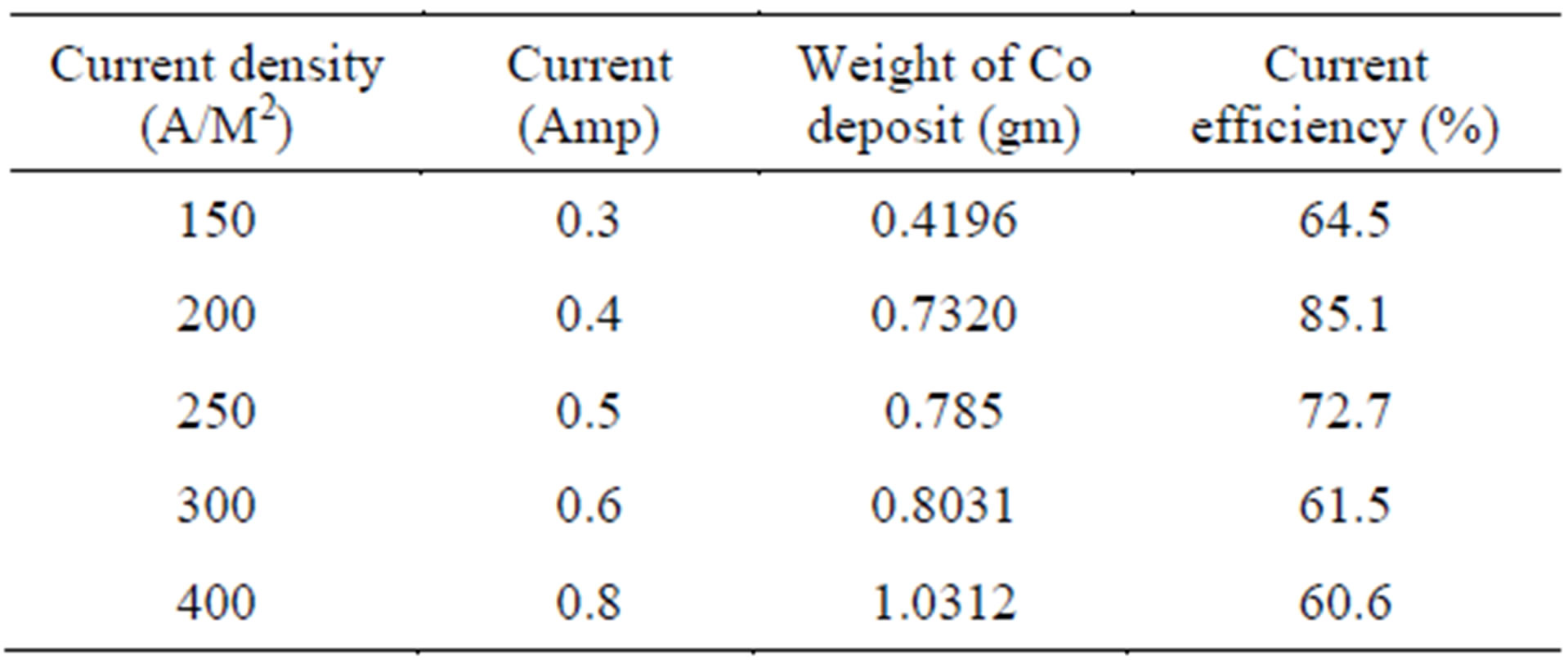
Table 4. Effect of current density on cobalt electrowinning.
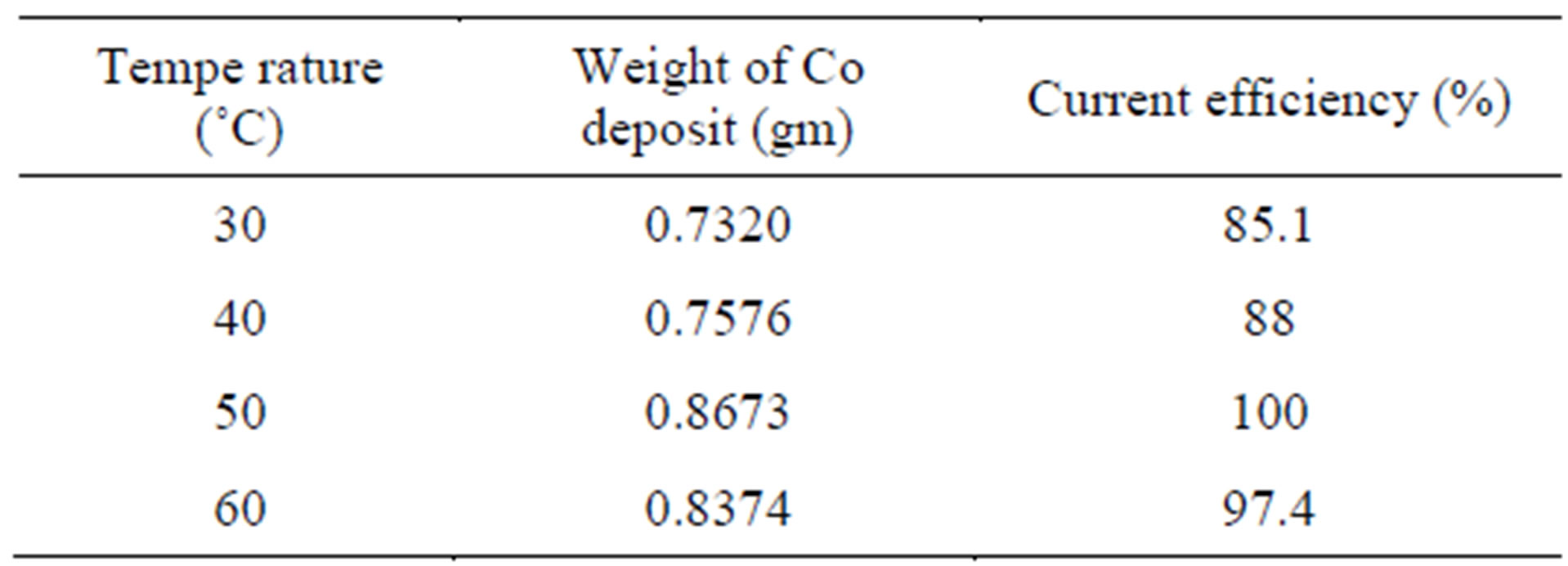
Table 5. Effect of temperature on cobalt electrowinning.
 cobalt concentration (g/L)
cobalt concentration (g/L)
Figures 4.Effect of cobalt concentration on cobalt recovery.
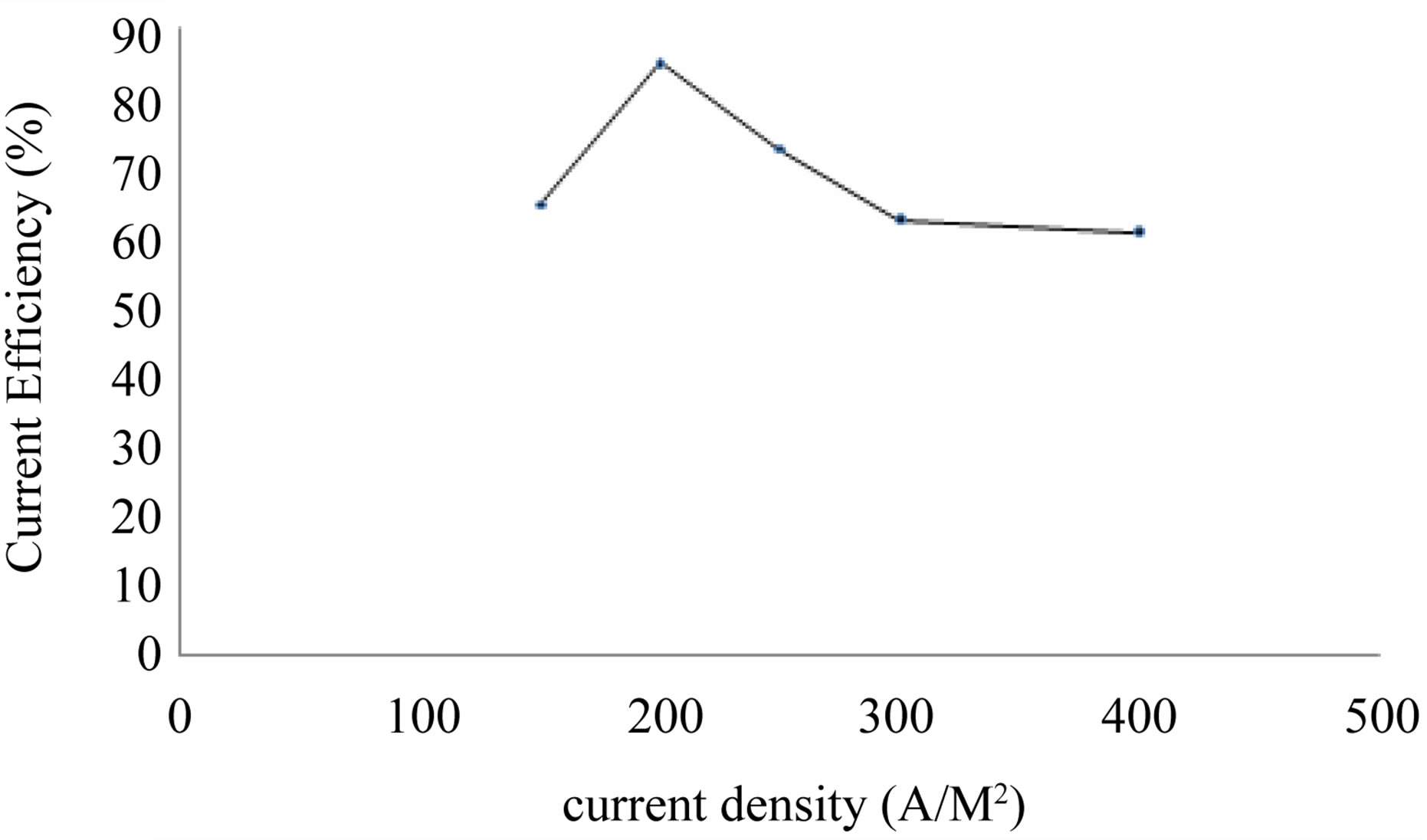
Figure 5. Effect of current density on cobalt recovery.
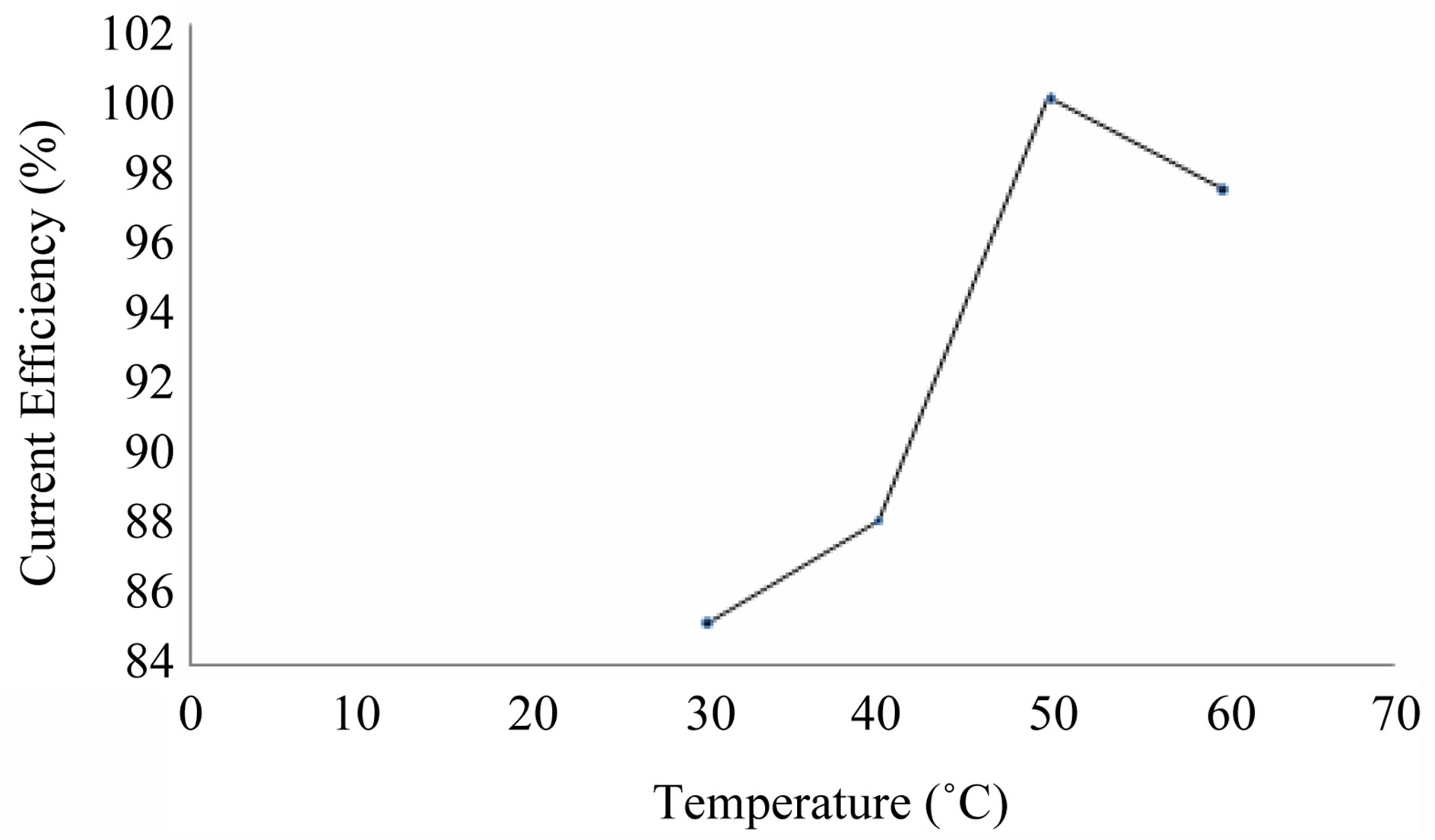
Figure 6. Effect of temperature on cobalt recovery.
3.6. Effect of Stirring
Since stirring/agitation has critical effect on current efficiency of cobalt recovery, effect of stirring between 0 - 700 RPM is carried out and the result is shown in Table 6 and Figure 7.
From the table it is observed that with increase in stirring the current efficiency is decreased Whereas, the 0 RPM of stirring, the current efficiency is high and deposition of cobalt recovery is also highest compared to other RPM condition which is an abnormal behaviour. This may be due to the redissolution of the cobalt deposit with increase of stirring. Hence solution need not be agitated for cobalt recovery under the specified electrolytic conditions. The optimum parameters for the electrowinning of cobalt is shown in Table 7.
3.7. SEM (EDAX) Analysis and Characterization of Cobalt Deposit
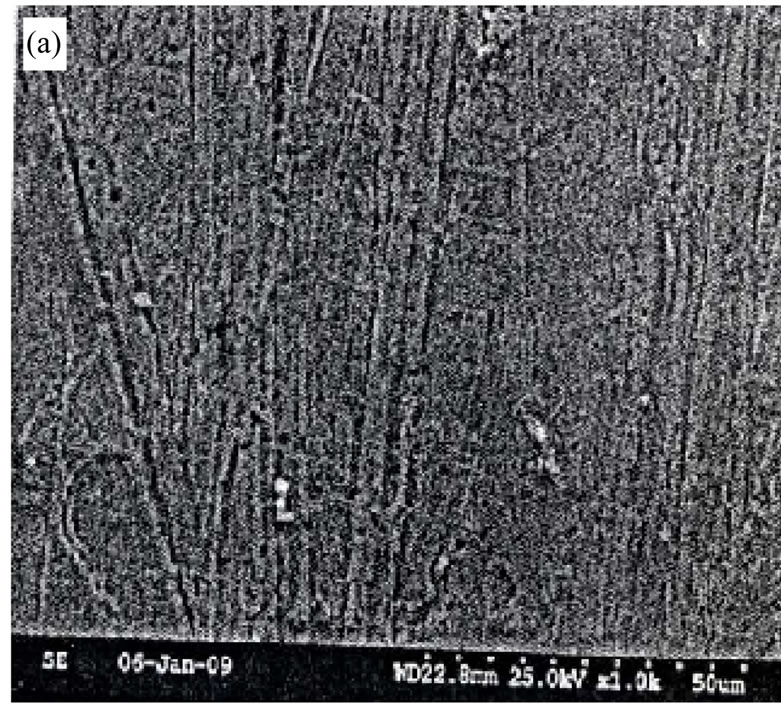
Figure 8 show the quantitative composition of waste catalyst by SEM (EDAX). From this figure the Co concentration is found to be 2.18 wt%. Figures 9(a) and (b) show the SEM figures of waste catalyst before roastingleaching. Magnification is 100 in Figure 9(a) and it is 1000 in Figure 9(b). In both figures the white areas denote Al2O3 while the remaining grey and dark colours are due to the presence of other oxides. Figures 9(c) and (d) is the SEM figures of catalyst after roasting-leaching at different magnifications.
Figures 10(a) and (b) shows the SEM figure of cobalt deposit with H2SO4 and without H2SO4 respectively recovered at a current density of 300 A/M2 in the electrolysis. In Figure 10(a) there are many small holes due to the liberation of H2 during Co deposit leading to decrease in current efficiency (value is: 61.5%), while the Figure 9(b) has only few holes of H2 indicating high current efficiency (value is 85.5%).
4. Conclusions
This project work is based on the recovery of cobalt from petroleum industry where it is used as hydrodesulphuri-
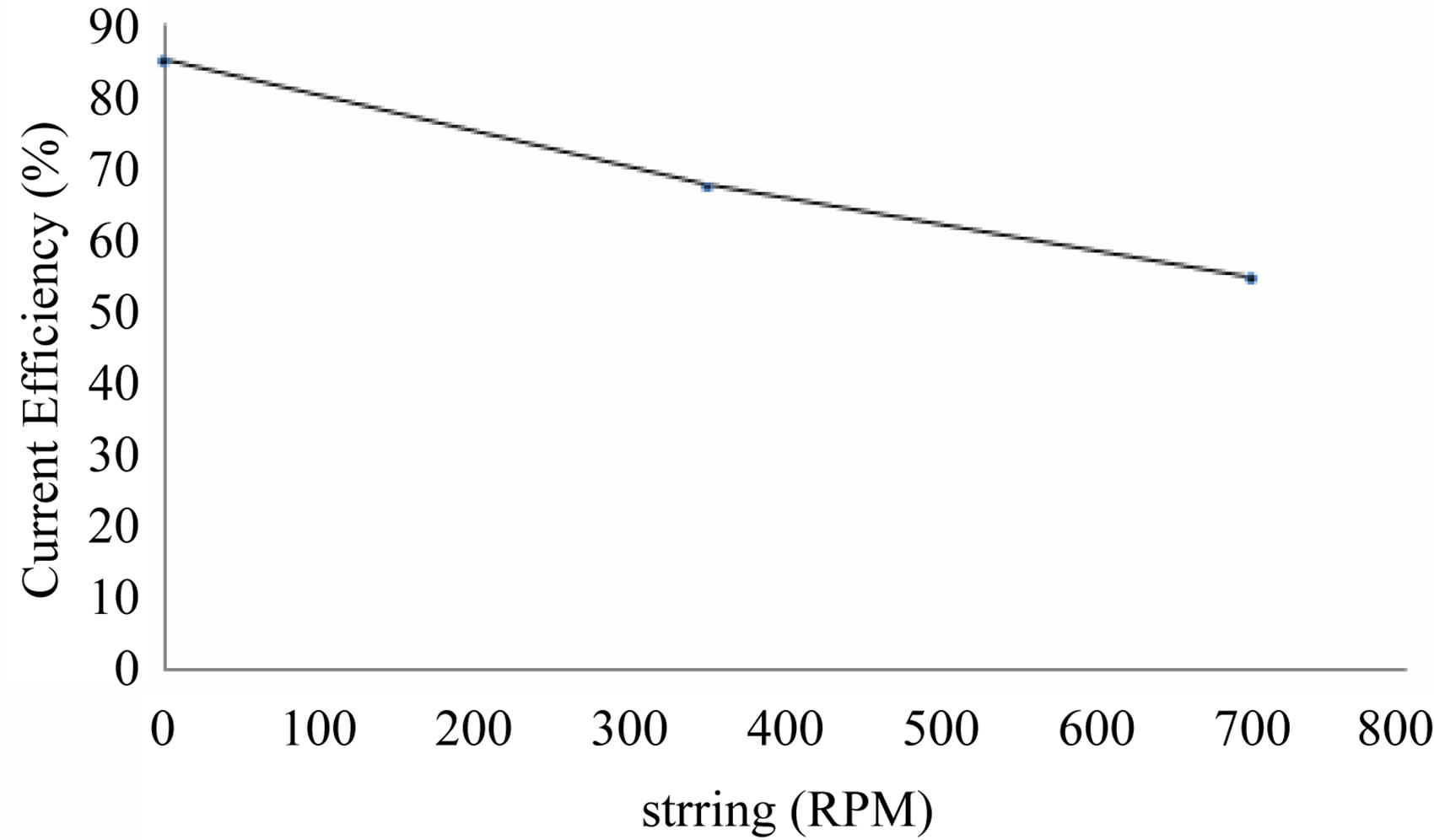
Figure 7. Effect of stirring on cobalt recovery.
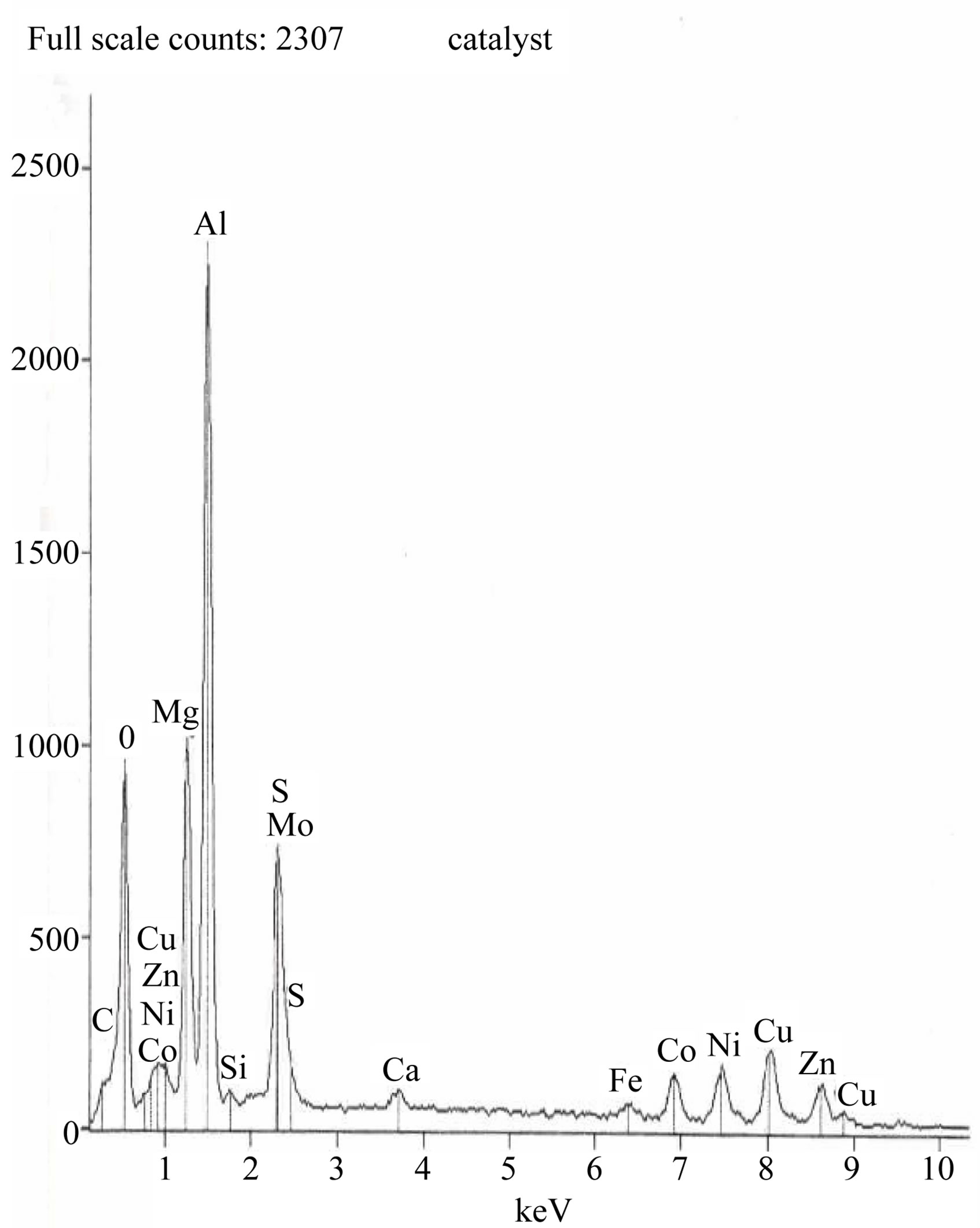
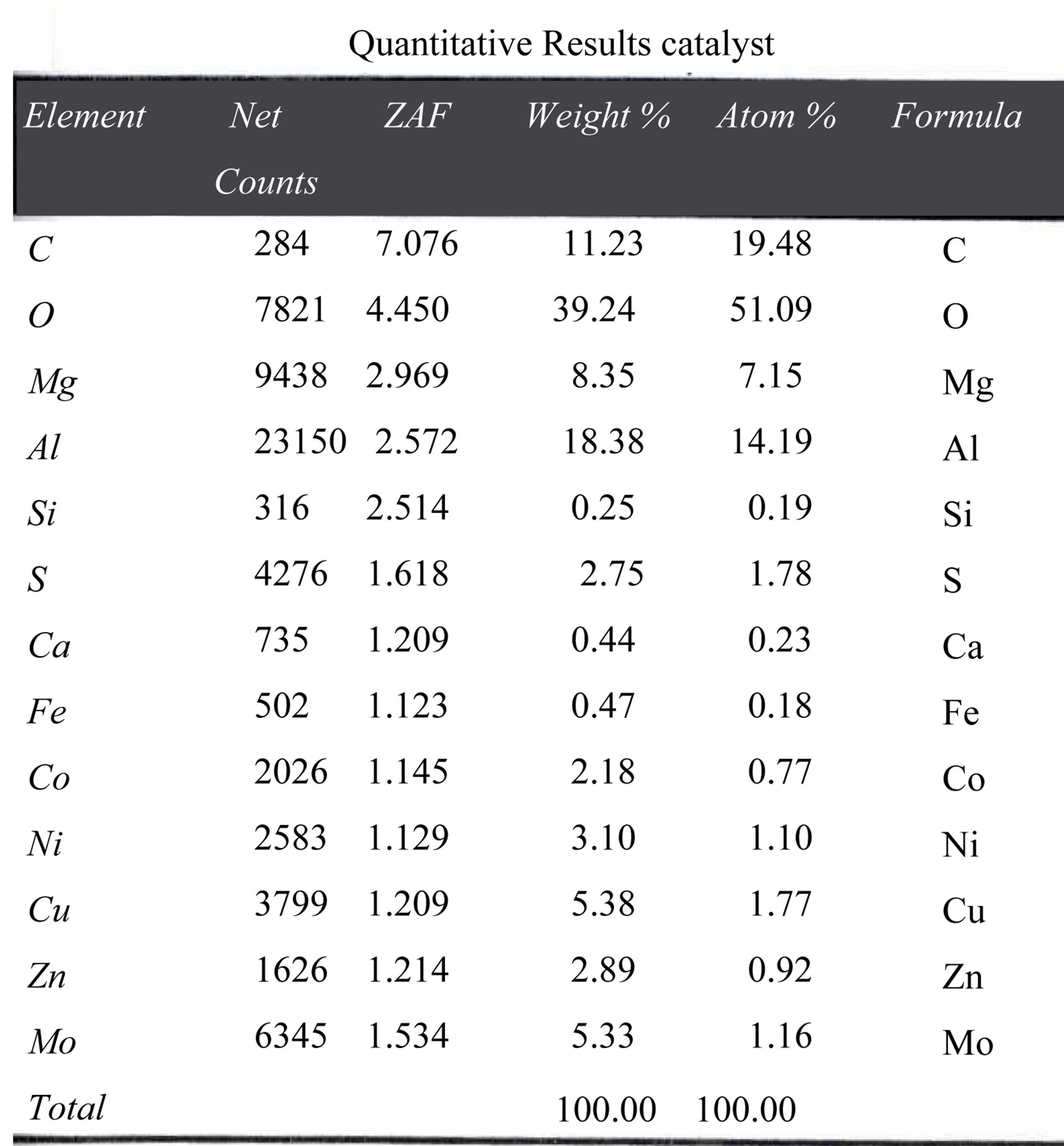
Figure 8. Quantitative composition of waste catalyst by SEM (EDAX).

Figures 9. SEM figure of (a) waste catalyst (×100 magnification) before roasting; (b) waste catalyst (×1000 magnification) before roasting; (c) waste catalyst (×100 magnification) after roasting-leaching; (d) waste catalyst (×1000 magnification) after roasting-leaching.

Figures 10. SEM figures of (a) Co deposit (with H2SO4 in bath) at 300 A/M2 current density; (b) Co deposit (without H2SO4 in bath) at 300 A/M2 current density.
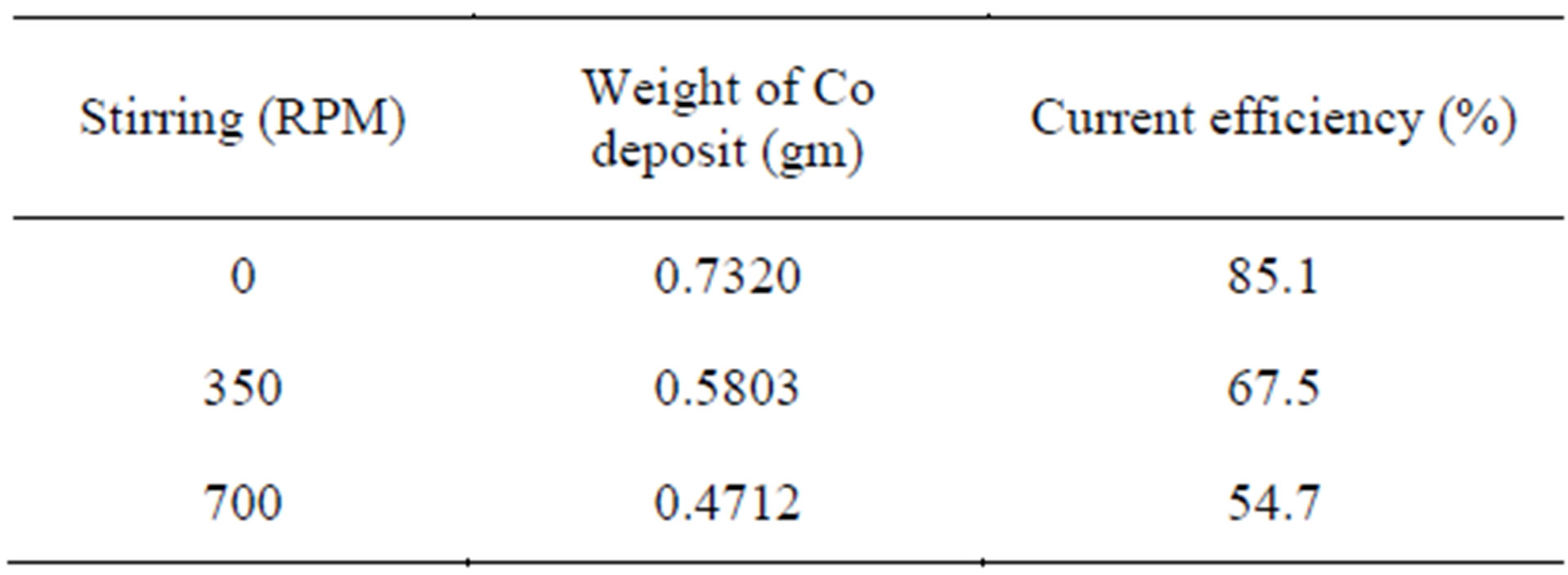
Table 6. Effect of stirring on cobalt electrowinning.
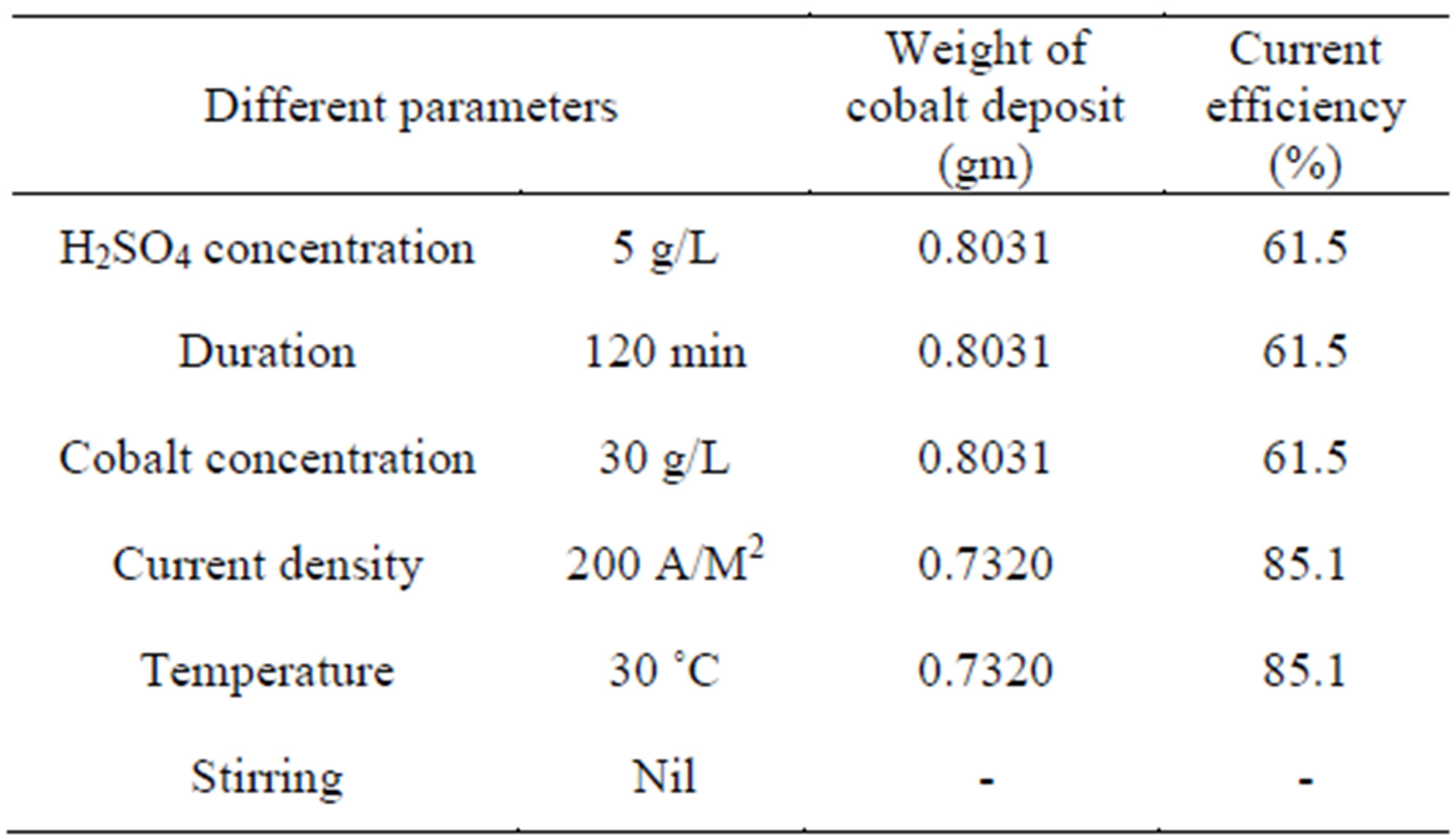
Table 7. Optimum electrowinning parameters.
zation catalyst for the purification of petroleum oil. (like removal of sulphur/addition of hydrogen)
Cobalt recovery from synthetic cobalt solutions are studied and the optimized values for these parameters for the specified electrolytic condition are H2SO4 concentration: 5 g/L (2.13 pH), Duration: 120 minutes, Cobalt concentration: 30 g/L, Current density: 200 A/M2, Temperature: 30˚C, Stirring: Nil.
At the optimized conditions, cobalt metal foil is electrolytically recovered (about 70%) from the waste catalyst roasted leached solution (after concentration).
5. Acknowledgements
This work was supported by the Department of EHM division, Central Electrochemical Research Institute, Karaikudi-630 006, Government of India.
REFERENCES
- N. L. Piret, “Enhancing Cobalt Recovery from Primary and Secondary Resources,” Journal of the Minerals, Metals and Materials Society, Vol. 50, No. 10, 1998, pp. 42-43. doi:10.1007/s11837-998-0351-1
- M. J. Hawkins, “Recovering Cobalt from Primary and Secondary Sources,” Journal of the Minerals, Metals and Materials Society, Vol. 50, No. 10, 1998, pp. 46-50. doi:10.1007/s11837-998-0353-z
- S. J. Wang, “Cobalt—Its Recovery, Recycling and Application,” Journal of the Minerals, Metals and Materials Society, Vol. 58, No. 10, 2006, pp. 47-50. doi:10.1007/s11837-006-0201-y
- P. Searle, “Forecasting the Cobalt Market through 2015,” Journal of the Minerals, Metals and Materials Society, Vol. 50, No. 10, 1998, pp. 42-43. doi:10.1007/s11837-998-0352-0
- S. C. Das and T. Subbaiah, “Electrowinning of Cobalt, Winning from Pure Cobalt Sulphate Bath,” Hydrometallurgy, Vol. 12, No. 3, 1984, pp. 317-333. doi:10.1016/0304-386X(84)90004-5
- A. I. Vogel, “A Text Book of Quantitative Inorganic Analysis Including Elementary Instrumental Analysis,” The English Language Book Society and Longman, London, 1978, pp. 569-573.
- H. A. Flaschka, “EDTA Titrations, an Introduction to Theory and Practice,” Pergamon Press, London, 1964.
- R. P. Das, “Recovering Cobalt from Secondary Sources in India,” Journal of the Minerals, Metals and Materials Society, Vol. 50, No. 10, 1998, pp. 46-50. doi:10.1007/s11837-998-0354-y
- I. M. Valverde Jr., J. F. Paulino and J. C. Afonso “Hydrometallurgical Route to Recover Molybdenum, Nickel, Cobalt and Aluminum from Spent Hydro Treating Catalyst in Sulphuric Acid Medium,” Journal of Hazardous Materials, Vol. 160, No. 2-3, 2008, pp. 310-317. doi:10.1016/j.jhazmat.2008.03.003
- V. S. Bagotzky, “Fundamentals of Electrochemistry,” Plenum Press, New York, 1993, p. 404.
- Elsevier, “Metal Finishing Guidebook and Dictionary,” Elsevier Science Publication, New York, Published Annually.
- A. Brenner, “Electrodeposition of Alloys, Principles and Practice,” Academic Press, New York, 1963.
- B. B. Kar, B. V. R. Murthy and V. N. Misra, “Extraction of Molybdenum from Spent Catalyst by Salt-Roasting,” International Journal of Mineral Processing, Vol. 76, No. 3, 2005, pp. 143-147. doi:10.1016/j.minpro.2004.08.017
- R. G. Busnardo, N. G. Busnardo, G. N. Salvato and J. C. Afoxiso, “Processing of Spend NiMo and CoMo/Al2O3 Catalysts via Fusion with KHSO4,” Journal of Hazardous Materials, Vol. 139, No. 2, 2007, pp. 391-398.
- C. Kargl-Simard, J. H. Huang and A. M. Alfantazi, “Electrical Conductivity and Density of CoSO4/H2SO4 Solutions in the Range of Modern Cobalt Electrowinning Electrolytes,” Minerals Engineering, Vol. 16, No. 6, 2003, pp. 529-
 535. doi:10.1016/S0892-6875(03)00059-1
535. doi:10.1016/S0892-6875(03)00059-1 - W. J. Basirun, W. J. Basirun and Y. Alias, “Influence of Magnetic Field of the Electro Deposition of Ni-Co Alloy,” Journal of Chemical Sciences, Vol. 122, No. 2, 2010, pp. 279-285. doi:10.1007/s12039-010-0032-9
NOTES
*Corresponding author.

