Open Journal of Animal Sciences
Vol.2 No.3(2012), Article ID:21366,7 pages DOI:10.4236/ojas.2012.23022
Single Layer Centrifugation with Androcoll-ETM improved progressive motility and percentage of live spermatozoa with intact acrosome of chilled stallion semen but did not have an effect on DNA integrity*
![]()
1Centro de Ciência Animal e Veterinária (CECAV), University of Trás-os-Montes e Alto Douro, Quinta de Prados, Vila Real, Portugal; alcosta@utad.pt
2Veterinary Degree, Universty of Trás-os-Montes e Alto Douro, Quinta de Prados, Vila Real, Portugal
3Centro de Estudos em Ciência Animal (CECA), Instituto de Ciências Biomédicas Abel Salazar (ICBAS), University of Porto, Oporto, Portugal
4Coimbra Higher School of Agriculture, São Martinho do Bispo, Coimbra, Portugal
5Alter Real Foundation, Alter do Chão, Portugal
Received 14 May 2012; revised 18 June 2012; accepted 30 June 2012
Keywords: Stallion Sperm; Single Layer Centrifugation; Androcoll-ETM; Membrane Integrity; Comet Assay Component; Formatting; Style; Styling
ABSTRACT
A significant number of stallions produce low quality ejaculates with high sensibility to chilling. Single Layer Centrifugation (SLC) with Androcoll-ETM has been presented as an efficient method of selecting good quality spermatozoa. In the current study, changes in sperm quality (motility, viability, acrosome integrity and DNA damage) occurring during storage at 5˚C for a maximum of 72 h, were investigated. For that, one ejaculate from 12 stallions was split in two aliquots: control and SLC-selected. Both aliquots were chilled and stored at 5˚C and spermatozoa were evaluated for motility, viability and acrosome integrity at 24, 48 and 72 h post collection. DNA damage was evaluated at 48 h post collection using the comet assay. In the SLC-selected aliquots, there was a significant improvement in terms of progressive motility (0 h: P = 0.005; 24 h: P < 0.001; 48 h: P < 0.001; 72 h: P < 0.001) and percentage of live spermatozoa with intact acrosome (24 h: P = 0.003; 48 h: P = 0.003; 72 h: P = 0.004). The DNA damage (in Arbitrary Units) was not different between SLCselected and control samples (P > 0.05). SLC with Androcoll-ETM improved semen quality prolonging sperm longevity of chilled semen (P = 0.012). This positive effect was more evident in ejaculates most sensitive to chilling that had a sharp decrease in motility in the first 24 h of refrigeration and for all ejaculates at 72 h postchilling. Therefore, this method reveals to be a useful technique for selecting spermatozoa and maintain sperm quality during storage.
1. INTRODUCTION
Stallions are selected mainly based on their pedigree, athletic performance or other phenotypic characteristics [1] rather than on semen quality. In many European countries, AI with chilled semen is the preferred breeding method for mares [2] but a significant percentage of stallions produce semen highly sensitive to chilling [3].
In order to improve the quality of chilled semen it is important to maximize its resistance to refrigeration. It has been suggested that the removal of the seminal plasma by centrifugation, previously to dilution in the extender, could enhance the sperm survival [4,5]. However, this procedure still does not consistently select “good quality” spermatozoa [6]. Recently, a method involving a single layer centrifugation (SLC) through a colloid of silica with a species-specific formulation (Androcoll-ETM) was developed at the Swedish University of Agricultural Sciences. This procedure permitted the selection of a sub-population of highly motile spermatozoa with normal morphology and good DNA integrity [7-9].
The basic features of semen evaluation consist on conventional laboratory tests, such as estimation of spermatozoa motility and morphologic characteristics, as well as concentration, volume and colour of the ejaculate. However, the predictive value of these examinations can be limited and the pregnancy rates can decrease, especially when the artificial insemination (AI) is performed with chilled or frozen semen [8]. The ability to accurately determine the quality of semen will better enable the clinician to predict the expected fertility of a stallion [10]. Fluorescent probes have been used in many species to evaluate the integrity of the sperm membrane, acrosome and mitochondria [10]. In the SYBR-14/PI staining, the SYBR-14 penetrates the membranes of all sperm, whereas PI penetrates only membranes of damaged sperm. Both probes bind to DNA nucleic acids with SYBR-14 fluorescing green while PI fluoresces red, allowing for the differentiation of live and dead cells [10- 12].
One factor that could play an important role in the fertilizing capacity of sperm is the deposition of abnormal sperm chromatin and/or DNA in the oocyte [13]. A few assays have been used over the last years to access changes in sperm chromatin or DNA, including the terminal deoxynucleotidyl transferase mediated-deoxyuridine triphosphate nick end labelling (TUNEL) assay, the Sperm Chromatin Structure Assay (SCSA) and the single-cell electrophoresis assay (SCGE) [14].
The SCGE, or comet assay [15] has been recognized as one of the most sensitive techniques for measuring DNA strand breaks [16] in various cells including sperm [17]. Determination of DNA damage at the single cell level can aid the improvement of semen preservation protocols, optimizing fertility by making appropriate adjustments for adequate protection of the cells [17].
The objectives of this study were to 1) evaluate the changes in sperm motility, viability and acrosome integrity in extended semen (control) and in SLC-selected spermatozoa, stored at 5˚C over 72 h; 2) evaluate the DNA integrity in control and SLC-selected spermatozoa stored at 5˚C at 48 h post collection using the comet assay and 3) evaluate the longevity of control and SLCselected samples.
2. MATERIALS AND METHODS
2.1. Animals
A single ejaculate of 12 stallions 4 to 22 years old was collected, in three different locations, between April and August of 2011. Stallions mounted a phantom or a mare in heat and ejaculated into a warmed artificial vagina. Semen was collected into a warm plastic bottle, fitted with a filter to separate the gel fraction. Ejaculates were kept at 37˚C until they were extended 1:1. After assessment of subjective progressive motility, 15 mL of extended semen was processed with Androcoll-ETM (see 2.4) and the resulting sperm cells pellet was diluted to a concentration of 50 × 106 spermatozoa/mL. The remaining ejaculate (control) was directly diluted to a concentration of 50 × 106 spermatozoa/mL.
2.2. Media
Kenney’s extender [18] was prepared with 1000 mL of distilled water, 27 g of glucose (G6152 Sigma-Aldrich, St. Louis USA), 40 g of sacarose (S1888 Sigma-Aldrich, St Louis USA) and 24 g of skimmed milk powder (Molico, Nestlé®). INRA96® was purchased commercially (IMV, l’Aigle Cedex France) as was EquiPro® (Minitub Ibérica S.L., Tarragona, Spain). Androcoll-ETM is composed of a silanecoated silica colloid in a buffer salt solution, optimized for stallion spermatozoa (SLU, Uppsala, Sweden).
2.3. Fresh Semen Analysis
Fresh semen motility was estimated by light microscopy evaluation (× 200), after placement of a drop on a previously heated slide (37˚C), which was covered by a cover slip. Fresh sperm concentration was accessed with Spermacue® (Minitub Ibérica S.L., Tarragona, Spain). For ejaculates with a concentration reading in the Spermacue® lower than 150 × 106 spermatozoa/mL, the concentration was re-calculated using a Neubauer chamber.
2.4. Single Layer Centrifugation
Fifteen mL of extended semen diluted to a concentration of 100 × 106 spermatozoa/mL in Kenny’s extender were carefully pipetted into a 50 mL centrifuge tube already containing 15 mL of Androcoll-ETM. After this procedure, a sharp interface between the colloid and the sample was obtained. Centrifugation at 500 × g for 20 min was then performed. The supernatant was removed until it was just 1 - 2 mm above the pellet. The pellet was then re-suspended and transferred to a clean tube. Using a Neubauer chamber to assess the concentration, the pellet was extended to a 50 × 106 spermatozoa/mL concentration with either INRA96® or EquiPro®.
2.5. Chilling Process
Two samples per stallion (control and SLC-selected) were chilled in a neopor container (Minitub Ibérica S.L., Tarragona, Spain) for transport during a maximum of 24 h. Then they were placed in a refrigerator at 5˚C until 72 h after collection.
2.6. Subjective Estimation of Motility
Aliquots (7.5 µL) of each sample were placed on a previously heated slide (37˚C), covered with a cover slip and examined by light microscopy (× 200). Several fields of the slide were evaluated before making a definite estimation of the progressive motility. Samples were examined at 24, 48 and 72 h post collection or until they reached a progressive motility inferior to 10%. Progressive motility of fresh semen was estimated by an experienced operator, different for each participating centre. After 24 h chilling, estimates of progressive motility were done by the same operator.
2.7. Sperm Viability and Acrosome Integrity
Live-Dead® Sperm Viability Kit (L-7011; Invitrogen, Eugene, OR, USA), which consists in a combination of the fluorochromes SYBR-14 and propidium iodide (PI), as well as FITC-PNA (L7381, Sigma-Aldrich, St. Louis USA) (0, 1 mg/1mL PBS) were used for this purpose. Two hundred µL of each sample was extended in 600 µL of Hepes buffer (10 nM HEPES, 150 nM NaCl, 10% BSA, pH 7.4) and centrifuged at 400 × g for 3 min. The supernatant was then discarded and 5 µL of SYBR-14 were added in a dark chamber. After 5 min of incubation at room temperature, 5 µL of FITC-PNA were added. Finally, 3 µL of PI were added, also in a dark chamber. After another 5 min of incubation, an aliquot of 7.5 µL was placed on a slide and observed at the fluorescent microscope (Nikon Microphot-FXA®, UV filter, X 400). One hundred to 200 spermatozoa per slide were counted and classified into one of four categories: live spermatozoa with intact acrosome (SYBR+/PNA–); live spermatozoa with damaged acrosome (SYBR+/PNA+); dead spermatozoa with intact acrosome (PI+/PNA–); dead spermatozoa with damaged acrosome (PI+/PNA–).
2.8. Single Cell Gel Electrophoresis Assay (Comet Assay)
It was performed 48 h post collection, since DNA damage is usually significantly different from fresh semen after 48 h of refrigeration [17]. The protocol was based on Sipinen et al. [19]. Slides were precoated with agarose by dipping into a vertical staining jar of melted 1% standard agarose in water. Excess agarose was drained and the slides were dried at room temperature. Samples were pipetted to a tube containing PBS and mixed 1:1 with 2% low melting point agarose. The samples were cast onto precoated glass slides, each one containing two gels with a volume of 70 µL and 3 × 104 cells each. Cell lysis was achieved submerging the slides in two consecutive lysis solutions: 1) lysis buffer (2.5 M NaCl, 0.1 M EDTA 10 mM Tris, 1% Triton X-100, pH 10) containing 10 mM dithiothreitol (D0632 Sigma-Aldrich, St Louis USA) for 90 min at 4˚C and 2) lysis buffer containing 0.05 mg/mL proteinase K (P2308 SigmaAldrich, St Louis USA) for 90 min at 4˚C. Two slides (with two gels each), were prepared per animal, one to measure basal DNA strand8/breaks (SBs) and the other for incubation with formamidopyrimidine DNA glicolase (FPG) (purchased from Professor A. Collins, University of Olso, Norway) which during incubation after lysis creates a strand break at 8-oxoguanines and other altered purines (FPG sites). After lysis, slides were washed in 3 changes of enzyme reaction buffer (40 mM Hepes, 0.1 M KCl, 0.5 mM EDTA, 0.2 mg/mL BSA, pH 8 with KOH) in staining jar for 5 min each at 4˚C. Meanwhile the enzyme formamidopyrimidine glycosylase (FPG) was prepared by adding 300 µl of buffer to 10 µL of enzyme ready to use. Then, 50 µL of enzyme solution (or buffer alone, as control) were placed onto each gel and covered with 22 × 22 mm cover slip. The slides were then incubated at 37˚C for 30 min in a moist box. After removal of the cover slips, the slides were incubated in electrophoresis buffer (0.3 M NaOH, 1 mM EDTA) at 4˚C for 30 min and then subjected to electrophoresis, run at 25 V and 300 mA for 30 min. The slides were then neutralized with a 10 min wash in PBS in staining jar at 4˚C, followed by 10 min in water. They were left at room temperature overnight to dry. Comet visualization was performed by visual analysis and the results expressed in Arbitrary Units, accordingly to Collins [20].
2.9. Longevity
Longevity (hours) was accessed based on estimation of the progressive motility at 24 h intervals. When samples reached a progressive motility lower than 10%, they were discarded.
2.10. Statistics
The statistical analyses were performed using Statistical Package for the Social Sciences (SPSS, IBM SPSS Statistics, Rel. 19.0.0.2010. Chicago: SPSS Inc., 2010) [21]. We fitted an ANOVA model with two main effects: time (four levels for sperm motility, 0, 24, 48 and 72 h and three levels for sperm viability, 24, 48 and 72 h) and semen processing (two levels, control and SLC-selected). The interactions were verified and, afterwards when there was no statistical difference, means were compared within time, checking variance heterogeneity (Levene test). In the cases where time was not a source of variation (ex.: DNA damage, done only at 48 h post chilling) the Independent-Samples T-test was applied directly. In all cases significance was set at P < 0.05.
3. RESULTS
The ANOVA analysis revealed that interaction Time vs. SLC-selected samples was not statistically significant (P > 0.05) in any case. Subjective progressive motility estimations (0 h: n = 12; 24 h: n = 12; 48 h: n = 11; 72 h: n = 6) were higher (P < 0.01) in SLC-treated samples for all time points (Figure 1). A sharper (P < 0.001) decrease of motility was observed in the first 24 h of refrigeration in the control samples, than in the SLC-treated (P = 0.004) ones (Figure 2). Three stallions had a fresh semen
progressive motility lower than 35%. After SLC treatment, progressive motility of these ejaculates improved to 50% (n = 1) and 60% (n = 2). The Independent-Samples T-test analysis for sperm viability and acrosome integrity is shown on Table 1. Sperm DNA integrity (n = 9) did not improve significantly (P > 0.05) in basal or oxidative damage (Table 2), even though there was a decrease of the mean values in SLC-selected samples, especially considering basal damages. Longevity (n = 12) of control and SLC-selected samples showed significant differences (Table 3). In SLC-selected semen all (n = 12; 100%) samples reached 72 h post collection with a pro-
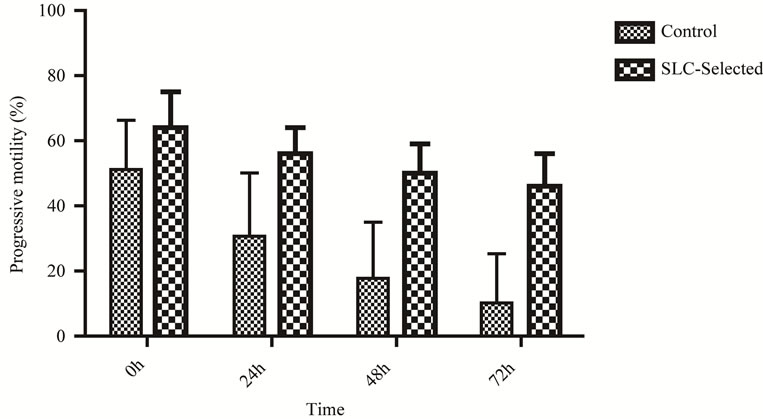
Figure 1. Subjective progressive motility (mean ± S.D.) for stallion sperm samples (0 h: n = 12; 24 h: n = 12; 48 h: n = 11; 72 h: n = 6) with and without SLC treatment. Samples were stored at 5˚C. Differences between means of control and SLCselected samples were statistically significant (P < 0.05) at all time points.
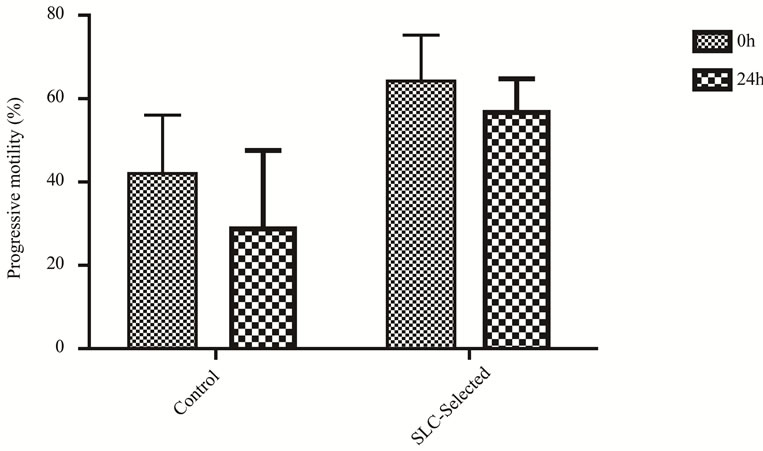
Figure 2. Subjective progressive motility (mean ± S.D.) for stallion sperm samples (0 h: n = 12; 24 h: n = 12) with and without SLC treatment in the first 24 h. Samples were stored at 5˚C. The decrease of sperm motility was sharper in control samples (P < 0.001) than in SLC-selected samples (P = 0.004).
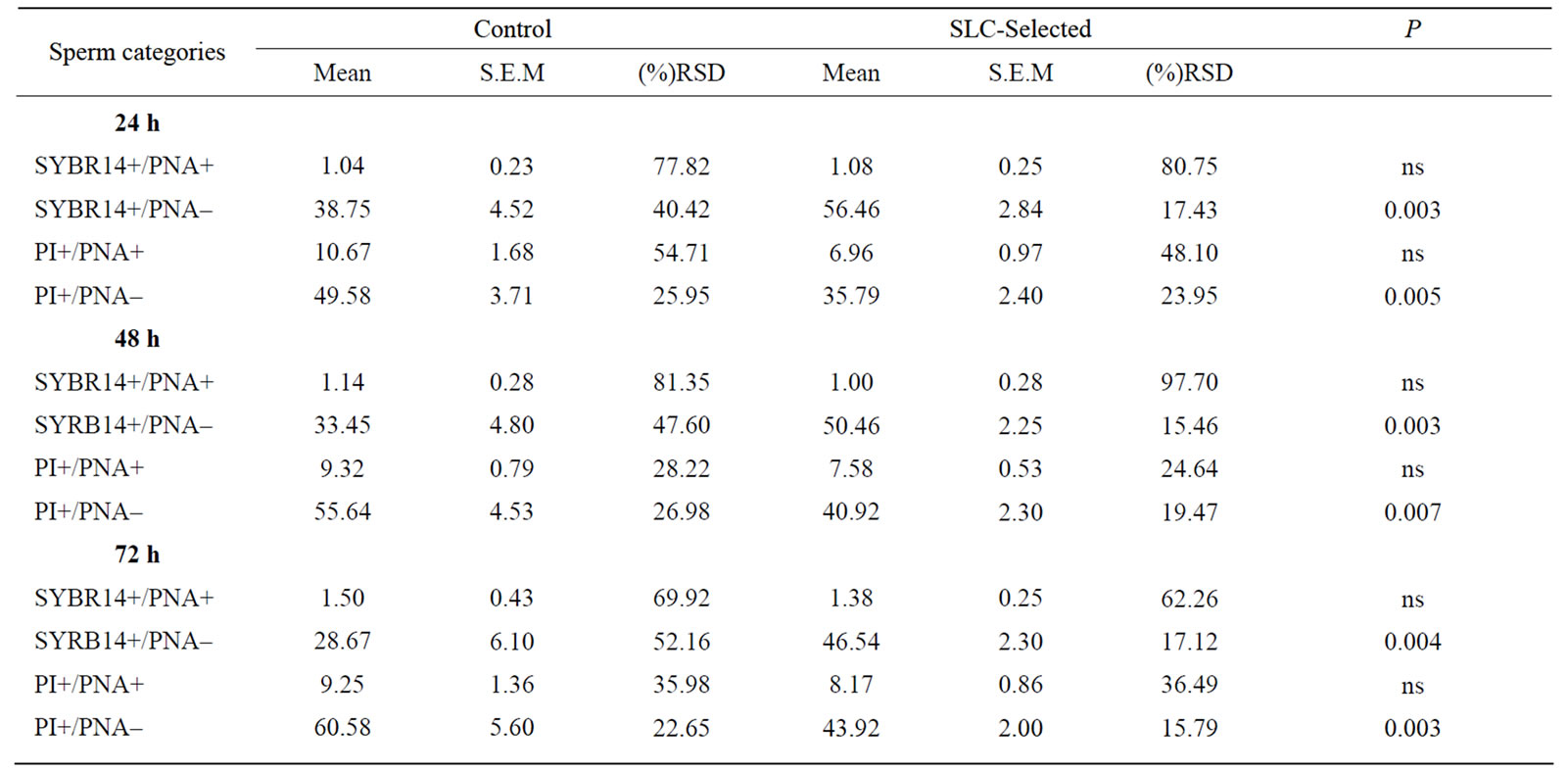
Table 1. Means, S.E.M. and relative standard deviation (RSD) of different sperm sub-populations after staining with fluorochromes SYBR-14, PI and PE-PNA measured at 24, 48 and 72 h after collection, after being stored at 5˚C (24 h: n = 12; 48 h: n = 11; 72 h: n = 6): live sperm with damaged or reacted acrosome (SYBR-14+PNA+); live sperm with intact acrosome (SYBR-14+PNA–); dead sperm with damaged or reacted acrosome (PI+PNA+) and dead sperm with intact acrosome (PI+PNA–).
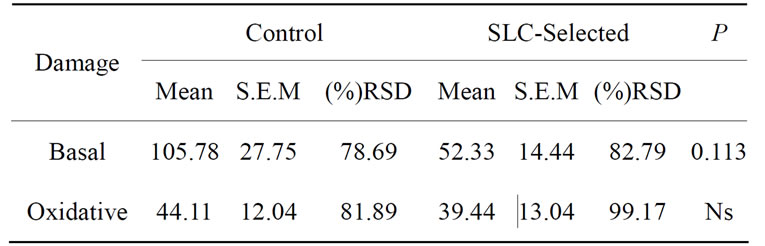
Table 2. Mean Arbitrary Units (AU) values of DNA integrity at 48 h post collection for stallion sperm samples (n = 9) preserved at 5˚C.
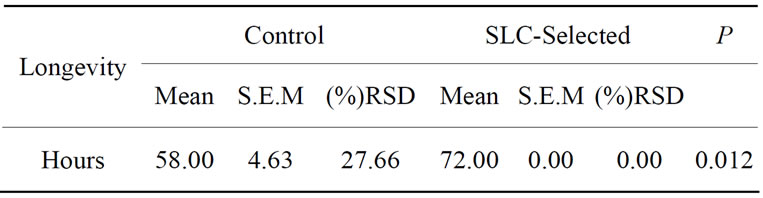
Table 3. Means (±S.E.M) of sperm longevity measured in hours, for control and SLC-selected samples (n = 12).
gressive motility higher than 10%, while only half (n = 6; 50%) samples of the control semen had motility above 10% at 72 h post-chilling.
4. DISCUSSION
As it should be expected, there was a progressive deterioration of all studied parameters of sperm quality, over time. However, this was more evident in control, rather than in SLC-treated samples. Previous studies have shown that SLC significantly improves the quality of fresh, as well as of chilled semen up to 48 h [7-9]. In the present study, samples were stored for 72 h, as long as they preserved progressive motility above 10%.
A sharper progressive motility decrease after 24 h of chilling was evident in control samples, while it was discrete in SLC-selected semen. After 72 h of refrigeration all SLC-selected samples maintained their progressive motility above 10%, compared to only half of the control samples.
A higher proportion of viable spermatozoa was found in the SLC-selected samples when compared to control samples, at all time periods. This is also consistent with results from previous studies [7-9,22] which suggested that removal of unviable spermatozoa by SLC results in better longevity of the viable population. The reason for this increased longevity has been attribute to the fact that dead spermatozoa are a source of reactive oxygen species (ROS) that leaking through unviable membranes may cause damage to other spermatozoa [23] attacking the polyunsaturated fatty acids (PUFA) of the sperm membrane [24,25]. Since spermatozoa contain high concentrations of PUFA, they are particularly sensitive to oxidative stress [24] and as a consequence of lipid peroxidation the plasma membrane may lose fluidity and integrity [25].
The acrosome membrane can be damaged during semen handling and processing [26,27] and that may lead to sub-fertility in some stallions. In the present study, acrosome damage was not statistically different in control and SLC-selected samples, even though there was a numerical decrease after treatment with Androcoll-ETM. Considering that semen handling can damage the acrosome membrane [27], and that as previously mentioned ROS leaking through unviable membranes and cause damage to other spermatozoa [23], damaged acrosome membranes, both in dead and live cells, may be responsible for damaging other sperm cells in the ejaculate.
Previous studies [8,9] have tested sperm chromatin integrity, showing a significant improvement in SLC-selected samples. In our study, we used the comet assay that detects DNA strand breaks, instead of chromatin damage. Unexpectedly, DNA damage was not significantly different in control and SLC-selected samples. Johannisson et al. [8] and Morrell et al. [9] suggested that the beneficial of SLC may be two-fold: it selects spermatozoa with intact chromatin due to the fact that immature spermatozoa or those with damaged chromatin have a lower density and, removal of seminal plasma along with dead spermatozoa and leukocites may prevent further damaged caused by ROS, in samples preserved at 5˚C. The latter is supported by data from Baumber et al. [23] in which the antioxidants catalase and glutathione reduced equine sperm DNA damage. The lack of difference in DNA damage between control and SLC-treated samples in our study could be, at least in part, a consequence of a limited number of samples analysed.
In conclusion, single layer centrifugation with Androcoll-ETM significantly improved the quality of semen samples of spermatozoa stored at 5˚C. This procedure selected spermatozoa that were motile, possessed good membrane integrity and that maintained these characteristics during storage significantly better than control samples, prolonging its longevity. The overall advantages of SLC-selected samples over the control group were particularly evident at 72 h of refrigeration. Thus, the use of Androcoll-ETM may be particularly useful when AI has to be performed with semen refrigerated for more than 48 h, which often occurs in mares with delayed ovulation after pharmacological induction of ovulation, or when using stallions that produce ejaculates particularly sensitive to chilling, e.g. with obvious decrease in progressive motility after 24 h of refrigeration. Field trials with AI using chilled, SLC-selected and control semen are warranted to assess the effect of these positive in vitro laboratory results on pregnancy rates of mares. Recent data from Morrell et al. [28] based in a restricted number of observations, suggest that in fact SLC can be used to increase the fertility of “problem” ejaculates.
5. ACKNOWLEDGEMENTS
The authors gratefully acknowledge Professor Teresa Rangel, Head of Physiology Laboratory (Animal Sciences Department, UTAD) for allowing the utilization of the laboratory and equipment.
REFERENCES
- Brito, L.F.C. (2007) Evaluation of stallion sperm morphology. Clinical Techniques in Equine Practice, 6, 249- 264. doi:10.1053/j.ctep.2007.09.004
- Aurich, J. and Aurich, C. (2006) Developments in European horse breeding and consequences for veterinarians in equine reproduction. Reproduction in Domestic Animals, 41, 275-279. doi:10.1111/j.1439-0531.2006.00719.x
- Morrell, J.M. (2006) Update on semen technologies for animal breeding. Reproduction in Domestic Animals, 40, 1-5.
- Loomis, P.R. (2006) Advanced methods for handling and preparation of stallion semen. Veterinary Clinics of North America: Equine Practice, 22, 663-676. doi:10.1016/j.cveq.2006.07.002
- Aurich, C. (2008) Recent advances in cooled-semen technology. Animal Reproduction Science, 107, 268-275. doi:10.1016/j.anireprosci.2008.04.015
- Morell, J.M., Rodriguez-Martinez, H. and Johanninson, A. (2010) Single layer sentrifugation of stallion spermatozoa improves sperm quality compared with sperm washing. Reproductive BioMedicine Online, 21, 429-436. doi:10.1016/j.rbmo.2010.03.027
- Morrell, J.M., Dalin, A.M. and Rodriguez-Martinez, H. (2008) Prolongation of stallion sperm survival by centrifugation through coated sílica colloids: A preliminary study. Animal Reproduction, 5, 121-126.
- Johannisson, A., Morrell, J.M., Thorén, J., Jönsson, M., Dalin, A.M. and Rodriguez-Martinez, H. (2009) Colloidal centrifugation with Androcoll-ETM prolongs stallion sperm motility, viability and chromatin integrity. Animal Reproduction Science, 116, 119-128. doi:10.1016/j.anireprosci.2009.01.008
- Morrell, J.M., Johannisson, A., Strutz, H., Dalin, A.M. and Rodriguez-Martinez, H. (2009) Colloidal centrifugation of stallion semen: Changes in sperm motility, velocity, and chromatin integrity during storage. Journal of Equine Veterinary Science, 29, 24-32. doi:10.1016/j.jevs.2008.11.006
- Love, C.C., Thompson, J.A., Brinsko, S.P., Rigby, S.L., Blanchard, T.L., Lowry, V.K. and Varner, D.D. (2003) Relationship between stallion sperm motility and viability as detected by two fluorescence staining techiniques using flow cytometry. Theriogenology, 60, 1127-1138. doi:10.1016/S0093-691X(03)00122-5
- Garner, D.L. and Johnson, L.A. (1995) Viability assessment of mammalian sperm using SYBR-14 and propidium iodide. Biology of Reproduction, 53, 276-284. doi:10.1095/biolreprod53.2.276
- Foster, M.L., Love, C.C., Varner, D.D., Brinsko, S.P., Hinricks, K., Teague, S., LaCaze, K. and Blanchard, T.L. (2011) Comparison of methods for accessing integrity of equine sperm membranes. Theriogenology, 76, 334-341. doi:10.1016/j.theriogenology.2011.02.012
- Love, C.C. and Kenney, R.M. (1998) The relationship of increased susceptibility of sperm DNA to denaturation and fertility in the stallion. Theriogenology, 50, 955-972. doi:10.1016/S0093-691X(98)00199-X
- Ball, B.A. (2008) Diagnostic methods for evaluation of stallion subfertility: A review. Journal of Equine Veterinary Science, 28, 650-665. doi:10.1016/j.jevs.2008.10.003
- Ostling, O. and Johanson, K.J. (1984) Microelectrophoretic study of radiation-induced DNA damages in individual mammalian cells. Biochemical and Biophysical Research Communications, 123, 291-298. doi:10.1016/0006-291X(84)90411-X
- Collins, A.R., Dobson, V.L., Dusinska, M., Kennedy, G. and Stetina, R. (1997) The comet assay: What can it really tell us? Mutation Research, 29, 183-193. doi:10.1016/S0027-5107(97)00013-4
- Linfor, J.J. and Meyers, S.A. (2002) Detection of DNA damage in response to cooling injury in equine spermatozoa using single-cell gel electrophoresis. Journal of Andrology, 23, 107-113.
- Kenney, R.M., Bergman, R.V., Cooper, W.L. and Morse, F.W. (1975) Minimal contamination techniques for breeding mares: Techniques and preliminary findings. Proceedings of American Association of Equine Practitioners, 21, 327-336.
- Sipinen, V., Laubenthal, J., Baumgartner, A., Cemeli, E., Linchooten, J.O., Godschalk, R.W.L., Van Schooten, F.J., Anderson, D. and Brunborg, G. ( 2010) In vitro evaluation of baseline and induced DNA damage in human sperm exposed benzo[a]pyrene or its metabolite benzo[a]pyrene- 7,8-diol-9,10-epoxide, using the comet assay. Mutagenesis, 25, 417-425. doi:10.1093/mutage/geq024
- Collins, A.R. (2004) The comet assay for DNA damage and repair. Molecular Biotechnology, 26, 249-261. doi:10.1385/MB:26:3:249
- Statistical Package for the Social Sciences (SPSS) (2010) IBM SPSS Statistics, Rel. 19.0.0.2010. SPSS Inc., Chicago.
- Morrell, J.M., Macias Garcia, B., Pena, F.J. and Johannisson, A. (2011) Processing stored stallion semen doses by Single Layer Centrifugation. Theriogenology, 76, 1424-1432. doi:10.1016/j.theriogenology.2011.06.011
- Baumber, J., Ball, B.A., Linfor, J.J. and Meyers, S.A. (2003) Reactive oxygen species and cryopreservation promote DNA fragmentation in equine spermatozoa. Journal of Andrology, 24, 621-627.
- Jones, R., Mann, T. and Sherins, R. (1979) Peroxidative breakdown of phospholipids in human spermatozoa, spermicidal properties of fatty acid peroxides, and protective action of seminal plasma. Fertility and Sterility, 31, 531-537.
- Storey, B.T. (1997) Biochemistry of the induction and prevention of lipoperoxidative damage in human spermatozoa. Molecular Human Reproduction, 3, 203-2013. doi:10.1093/molehr/3.3.203
- Pommer, A.C., Linfor, J.J. and Meyers, S.A. (2002) Capacitation and acrosome exocytosis are enhanced by incubation of stallion spermatozoa in a commercial semen extender. Theriogenology, 57, 1493-1501. doi:10.1016/S0093-691X(02)00659-3
- Bosard, T., Love, C.C., Brinsko, S., Blanchard, T., Thompson, J. and Varner, D. (2005) Evaluation and diagnosis of acrosome function/dysfunction in the stallion. Animal Reproduction Science, 89, 215-217.
- Morrell, J.M., Mari, G., Kútvölgyi, G., Meurling, S., Mislei, B., Iacono, E. and Rodriguez-Martinez, H. (2011) Pregnancies following artificial insemination with spermatozoa from problem stallion ejaculates processed by single layer centrifugation with Androcoll-E. Reproduction in Domestic Animals, 46, 642-645. doi:10.1111/j.1439-0531.2010.01721.x
NOTES
*Funding source: This work was partially financed by Projects PTDC/ CVT/108456/2008 (FCT) and COMPETE: FCOMP-01-0124-FEDER- 009565 (FEDER), “Development of methods to increase the fertilizing ability of chilled and frozen stallion semen: a multidisciplinary approach”.

