World Journal of Neuroscience
Vol.2 No.1(2012), Article ID:17725,9 pages DOI:10.4236/wjns.2012.21004
Immunocytochemical identification and distribution of the cell types in the pituitary gland of Bagrus bayad (Teleostei, Bagridae)
![]()
1Fish Reproduction Laboratory, National Institute of Oceanography and Fisheries, Alexandria, Egypt
2Zoology Department, Faculty of Science, Zagazig University, Zagazig, Egypt
Email: *mostafa_mousa2002@yahoo.com
Received 24 October 2011; revised 14 November 2011; accepted 13 December 2011
Keywords: Immunocytochemistry; Pituitary Gland; Bagrus Bayad (Teleostei)
ABSTRACT
Immunocytochemical identification of the different cell types in the pituitary gland of Bagrus bayad was performed using antisera against mammalian (human and rat) and piscine hormones. The adenohypophysis was composed of rostral pars distalis (RPD), proximal pars distalis (PPD) and pars intermedia (PI). Prolactin and adrenocorticotrophic cells were located in the rostral pars distalis of the pituitary. Gonadotrophic and growth hormone cells were distributed in the proximal pars distalis, but gonadotrophic cells appear also at the border of the pars intermedia. Somatolactin cells, as well as alpha-melanotrophic cells were located in the pars intermedia of B. bayad pituitary. The prolactin (PRL) cells were distributed in the RPD stained with orange G and showed strong immunoreactivity with antiserum to chum salmon. The adrenocorticotrophic (ACTH) cells were lead hematoxylin-positive (PbH+) and showed strong immunoreactivity with anti-human ACTH; these cells bordered the neurohypophysis and grouped in islets between PRL cells in the RPD. Growth hormone (GH) cells were densely distributed with the gonadotrophic (GTH) cells in the PPD. They were orange G positive and reacted with antiserum to chum salmon. GTH cells were located in the central area of the PPD and in the external border of the PI. These cells were Alcyan Blue and PAS positive, and immunostained with anti-chum salmon GTH Iβ and anti-chum salmon GTH IIβ. In addition, antiserum to rat thyrotropin stimulating hormone β (TSHβ) reacted positively to the GTH cells. These results suggest that GTH I, GTH II and TSH are synthesized in the same cells in the pituitary of B. bayad. The PI was composed mainly of PbH+ cells and a PAS+ cell adjacent to the neurohypophysis. The PAS+ cells from the PI bound specifically to anti-chum somatolactin. Anti-alphamelanin stimulating hormone (MSH) stained only the PbH+ (alpha-melanotrophic) cells of the PI.
1. INTRODUCTION
The pituitary gland of teleosts has already been the subject of research for many years. The identification and the distribution of the different cell types in the pituitary gland of teleosts have been studied using histochemical and different physiological techniques [1- 4]. In addition, the adenohypophyseal cells have been characterized by immunocytochemical techniques using antisera against mammalian and piscine hormones [4-9]. Seven different classes of hormones, grouped into three main families have been described: 1) growth hormone (GH)/prolactin (PRL) family, containing PRL, GH and somatolactin (SL); 2) glycoprotein hormones including gonadotrophins (GTHs) and thyrotropin (TSH); and 3) proopiomelanocortin-derived hormones such as adrenocorticotropic (ACTH) and melanotropic hormone (MSH) [2,4,6,8,10].
B. bayad (Forsskål, 1775) is an important food fish in tropical and semitropical waters. It grows fast and attains 112 cm in length with maximum weight of 12.5 kg [11]. B. bayad is locally known as “Bayad” and is so far one of the most important economic species as food fish in Egypt and finds good marketing in the different parts of Egypt. During the past years with the development of industries and modern civilization, large amount of pollutants have been discharged into the Nile and the surrounding lakes. These pollutants not only affect the growth, health, and nutritional value of fishes but also the distribution (i.e., survival) and reproduction of economically important fishes. B. bayad is one of those fishes, which affected by environmental factors and its distribution was restricted only to Lake Nasser and the Nile River. On the other hand it disappeared from the most of north lakes; Manzalah and Borollus and most of the Nile River.
In spite of the extensive importance of B. bayad, it is somewhat surprising that such limited information concerning its reproduction is available. [12] studied the breeding biology, the life cycle and mating behavior of B. bayad. In addition, [13] investigated the sexual maturity of B. bayad macropterus (synonym of B. bayad) in both wild and under captivity. They obtained that gonad development was faster under captivity than in wild. However, a detailed description of the cell types in the pituitary of this species has not yet been done. The anatomical localization of different cell types in the pituitary is important in understanding their functional role and physiological relevance during the reproductive cycle of this economic species. The aim of the present study was to localize and characterize the different pituitary cell types using histochemical and immunocytochemical approaches.
2. MATERIAL AND METHODS
2.1. Animals
From Nile River, 20 fish of adult B. bayad (mature males and females) with standard length larger than 30 cm, were collected alive during the prespawning and spawning season (March to June).
2.2. Histological and Histochemical Methods
Prior to dissection, the fishes were anesthetized in a solution (20 mg/l) of clove oil (Sigma) and then perfused via the ascending aorta with 20 ml of normal saline, followed by 50 ml of Bouin`s fluid at 4˚C. Immediately after the dissection, the pituitary gland, attached to the brain, was postfixed in Bouin's fluid for 24 hr at 4˚C. The fixed brain and pituitaries were thereafter dehydrated through graded ethanol solution, cleared and embedded in paraplast (mp: 56˚C - 58˚C). Consecutive median sagittal sections of the pituitary gland were made at 4 μm thickness. For each specimen, selected sections were stained with the following techniques:
1) Harris’s alum hematoxylin, according to [14] and aqueous solution of eosin (1%) was used as a counter stain.
2) Periodic Acid-Schiff-Lead hematoxylin (PAS-PbH): Lead hematoxylin “PbH” [15] combined with PAS [16].
3) Performic acid-Alcyan Blue (PFAAB)-Periodic Acid-Schiff (PAS)-Orange G (OG) stain [17].
2.3. Immunocytochemical Procedures
Antibodies: Rabbit antisera directed against human ACTH and rat thyrotropin (rBetaTSH) was obtained from National Institute of Health. The α-MSH antiserum was kindly provided by Dr. R. M. Dores (University of Denver, USA). Antisera to chum salmon (Oncorhynchus keta) hormones; chum salmon GTH Iβ subunit (Lot No. 8707), chum salmon GTH IIβ subunit (Lot No. 8506), chum salmon somatotropin (chum GH) (Lot No. 8208), chum salmon prolactin (Lot No. 8502) and chum salmon somatolactin (Lot No. 8906) were obtained from Dr. H. Kawauchi (School of Fisheries Science, Kitasato University, Iwate, Japan).
Immunocytochemical reactions: Immunocyto-chemical staining for the sections of the pituitary gland was performed with a vectastain ABC (Avidin-biotin peroxidase complex) Kit (Vector Laboratories) as described previously [18]. In brief, sections were deparaffinized in xylene, rehydrated through graded ethanol, washed in 10 mM phosphate-buffered saline (PBS; pH 7.4) for two times 10 min each. All incubations were done at room temperature and PBS was used for washing after each step. Sections were incubated with the antisera to the various hormones for 12 - 18 hr. The dilution of the hormone antisera was determined empirically and their working dilutions were shown in Table 1. Thereafter, the sections were incubated with the biotinylated secondary antibody (Vector Laboratories) for 1 h. and with avidinbiotin-conjugated peroxidase for 45 min. Finally, the sections were washed and stained with 3, 3’-diaminobenzidine tetrahydrochloride (DAB) (Sigma) including 0.01% H2O2 in 0.05 M Tris-buffered saline (pH 7.6) for 3 - 5 min. After the enzyme reaction, the sections were washed in tap water, dehydrated in alcohol, cleared in xylene and mounted in DPX.
In order to confirm the specificity of the immunoreactive procedures, adjacent sections were stained according
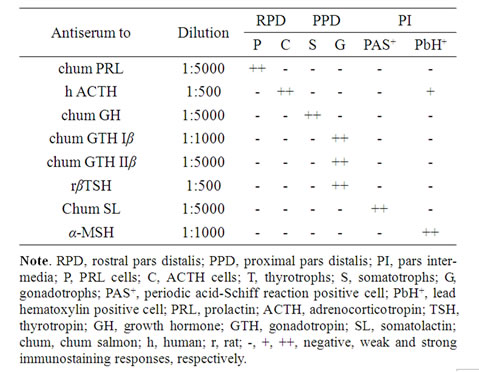
Table 1. Immunocytochemical staining of the adenohypophysis of B. bayad.
to the above described protocol but incubation in the primary antisera was omitted. In addition, normal bovine serum was used instead of primary antiserum. No positive structures or cells were found in these sections.
3. RESULTS
The pituitary gland of B. bayad consists of the neurohypophysis, and the adenohypophysis, which showed the three major subdivisions typical of teleost; an anterior rostral pars distalis (RPD), a medium proximal pars distalis (PPD) and posterior pars intermedia (PI) (Figures 1 (a)-(c)). Moreover, neurohypophysial processes penetrated the different adenohypophyseal areas. According to the histochemical and immunocytochemical behavior, six endocrine cell types were identified in the pituitary gland of B. bayad.
3.1. Prolactin (PRL)-Secreting Cells
The PRL cells occupy the major part of the RPD (Figures 1(a), (b)). These cells have variable sizes, with irregular shapes and have spherical nuclei with a distinct nucleolus (Figure 2(a)). They are frequently arranged in cords bordering the neurohypophysis. The PRL cells form a compact mass being stained orange with orange G stain (Figure 2(b)) and are accordingly termed “orangeophilous acidophils”, and also stained with eosin (Figure
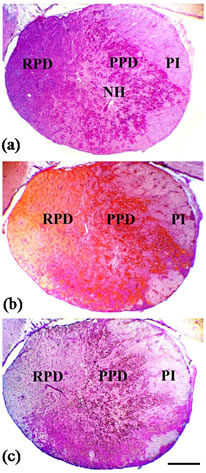
Figure 1. Sagittal section of the pituitary gland of mature female B. bayad stained with Harris’s hematoxylin and eosin (a), AB-PAS-OG (b) and PASPbH combined stain (c) showing the rostral pars distalis (RPD), proximal pars distalis (PPD) and pars intermedia (PI) which comprise the adenohypophysis, and neurohypophysis (NH). Scale bar = 300 µm.
2(a)). The PRL cells showed strong immunoreactivity to anti-chum salmon PRL (Table 1 and Figure 2(c)).
3.2. Adrenocorticotropin (ACTH)-Secreting Cells
The ACTH cells appear as cords bordering the PRL cells or as islets among PRL cells and the neurohypophysis (Figures 3(a) and 3(b)). They are varied in size, spherical or oval in shape and with small centric nuclei (Figures 2(a) and 3(a)). These cells stained positively with lead hematoxylin (PbH+) and have no affinity for PAS (Figures 2(b) and 3(a)). Anti-serum to human ACTH bound strongly to the ACTH cells (Figure 3(b)). Also, anti-hACTH showed cross-reaction with the PbH+ cells of the PI (Table 1).
3.3. Somatolactin (SL) and Alpha-Melanin Stimulating Hormone (α-MSH)-Secreting Cells
Two types of cells were found in the PI of B. bayad; one
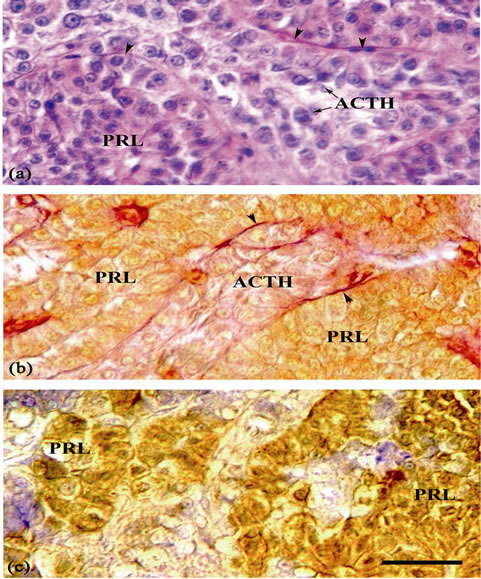
Figure 2. Sagittal section of the pituitary gland of mature female B. bayad. (a) A magnified portion of (Figure 1(a)) displaying the prolactin (PRL) cells with irregular shapes and have spherical nuclei with a distinct nucleolus, ACTH cells and nerve fibers (arrowheads). (b) A magnified portion of (Figure 1(b)), showing the PRL cells stained with orange G, in addition to ACTH cells and nerve fibers (arrowheads). (c) A magnified portion of sagittal section immunostained with anti-chum salmon prolactin antiserum. PRL positive cells are restricted in the rostral pars distalis. Scale bar = 25 µm.
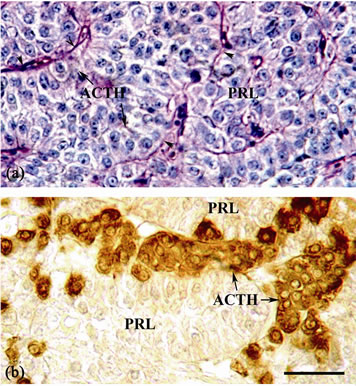
Figure 3. Sagittal section of the pituitary gland of mature female B. bayad: (a) Stained with PAS-PbH combined stain, showing the ACTH cells, the PRL cells and the neurohypophysis (arrowheads). (b) Immunostained with anti-human ACTH antiserum. The ACTH cells appear as cords bordering the PRL cells or as islets between PRL cells. Scale bar = 25 µm.
is stained with periodic acid-Schiff (PAS+ cells) and the other cell type is stained with lead hematoxylin (PbH+ cells) (Figure 4(b)). Anti-αMSH stained only the PbH+ cells of the PI (Figure 4(c)). In addition, anti-αMSH positive cells showed cross-reaction with the anti-hACTH (Table 1). Alpha-melanotropin (MSH)-immunoreactive cells were PAS negative in the PAS-PbH-stained section. These cells showed variable shapes and sizes (Figures 4(a)-(c)).
The PAS+ cells of the PI have variable sizes and shapes and were found either singly or in-groups (Figures 4(a), (b), (d)). The PAS+ cells showed strong immunoreactivity to anti-chum salmon SL (Figure 4(d)).
3.4. Somatotropin (STH) or Growth Hormone (GH)-Secreting Cells
The GH cells are distributed in the PPD and are in close contact with neurohypophysis (Figures 1(b), 5(a), 5(b)). They are dispersed singly or in groups between the basophils. The cytoplasm of these cells is densely granulated and their nuclei were oval or rounded (Figure 5(a)). These acidophilic cells were strongly stained with eosin and orange G (Figures 5(a), 5(b)) but negative to PAS. The somatotrops showed strong and specific immunoreactivity to anti-chum salmon GH which showed no cross-reaction with other cell types (Figure 5(c)).
3.5. Gonadotropins (GTH Iβ and GTH IIβ) and Thyrotropin (TSH)-Secreting Cells
The GTH cells are present in the central part of the PPD
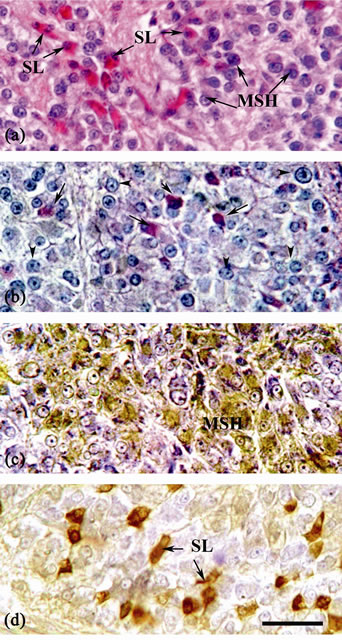
Figure 4. Sagittal section of the pituitary gland of mature female B. bayad: (a) Stained with Harris’s hematoxylin and eosin, showing the cell types of the pars intermedia; SL cells stained with eosin and have small sizes and MSH cells stained with haematoxylin and have large variable sizes and shapes. (b) Stained with PAS-PbH, showing the cell types in the pars intermedia; the PAS-positive cells (arrows) and PbHpositive cells (arrowheads). (c) Immunostained with anti-α-MSH antiserum, displaying the intensely stained MSH cells in the PI. (d) Immunostained with anti-chum salmon somatolactin (SL) antiserum, showing SL immunoreactive cells in the PI. Scale bar = 25 µm.
(Figures 1(b), 1(c)), and the external border of the PI. These basophilic cells manifest variable shapes and sizes and exhibit spherical nuclei (Figures 5(a), 5(b), 6(a)). The GTH cells contained numerous basophilic granules, which were positive to PAS and Alcyan blue (Figures 5 (b) and 6(a)). They also exhibited a large unstained intra-cytoplasmic vacuole. Anti-sera to chum salmon GTH Iβ and chum salmon GTH IIβ bound strongly and specifically to the GTH cells (Figures 6(b), 6(c)). In addition, antiserum to rat TSHβ subunit showed strongly and specifically immunoreaction to the gonadotropins (GTH Iβ and GTH IIβ)-secreting cells (Figure 6(d) and Table 1).
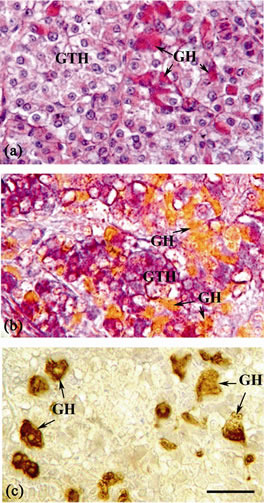
Figure 5. Sagittal section of the pituitary gland of mature female B. bayad: (a) Stained with Harris’s hematoxylin and eosin, displaying the growth hormone (GH) cells stained red with eosin. (b) Stained with AB-PAS-OG, showing the growth hormone (GH) cells stained orange with OG in the PPD, beside GTH cells. (c) Immunostained with anti-chum salmon growth hormone (antichum GH) antiserum, showing GH immunoreactive cells in the PPD. Scale bar = 25 µm.
4. DISCUSSION
B. bayad adenohypophysis was studied by both histochemical and immunocytochemical methods, the general structure conforming to that described for other teleosts. Six cell types were identified in the adenohypophysis of B. bayad. In the RPD two cell types could be distinguished, the prolactin (PRL) cells and the ACTH cells. Somatotrophs (GH cells) and gonadotropins (GTH Iβ and GTH IIβ) and thyrotropin (TSH)-secreting cells have been identified in the PPD and in the external border of the PI. The PI was mainly composed of two cell types; the somatolactin (SL) cells and the melanotrops (MSH cells). The distribution of the different types of cells identified in the present work is in a good agreement with previous studies in teleost fish [19-22]. The secretory cells show segregation into the different zones of the adenohypophysis.
4.1. PRL Cells
In the present study we have used an antiserum against chum salmon PRL that showed a quite good and specific immunoreaction with PRL cells of B. bayad, as has been reported for other teleosts [20-22]. Moreover, in contrast
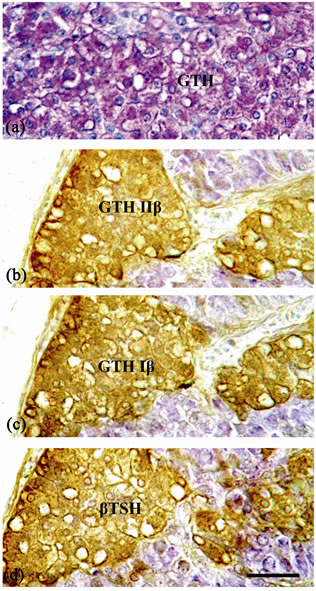
Figure 6. Sagittal section of the pituitary gland of mature female B. bayad: (a) Stained with PAS-PbH, showing GTH cells stained positively with PAS. (b)-(d) Three successive sections through the external border of the PI immunostained with antichum salmon GTH IIβ subunit (b), antichum salmon GTH Iβ subunit (c) and anti-βTSH (d). GTH Iβ, GTH IIβ and βTSH immunoreactivity are localized in the same cell types. Scale bar = 25 µm.
to other PRL antisera [23], it did not cross-react with GH cells. As for other freshwater and marine teleosts, B. bayad PRL cells are exclusively localized in the RPD [21, 22,24,25]. The osmoregulatory role of PRL in hypoosmotic environments is well established in teleosts, and especially in euryhaline species [26-28].
4.2. GH Cells
In this study we used anti-chum salmon GH that has been utilized to reveal GH cells of salmonids [29] and non-salmonid species [20-22,30,31]. This antiserum strongly stained GH cells of B. bayad that were restricted, similarly to other teleost fish, to the dorsal and ventral parts of the PPD [20,21,24,25,31].
The physiological role of GH as a growth-promoting hormone has been well established in teleosts [32]. Also GH has been involved in metabolism, reproduction and immune response [33]. In addition, an osmoregulatory role of GH has been reported in salmonids and nonsalmonid species [34,35,28].
4.3. SL Cells
Two types of cells were found in the PI of B. bayad; one is stained with lead hematoxylin (PbH+ or MSH cells) and the other cell type is stained with periodic acidSchiff (PAS+ cells). Immunocytochemical studies indicated that PAS-negative were MSH cells while PASpositive were SL cells.
The SL is the latest pituitary hormone of the GH/PRL family described [36]. In the last few years, several studies have analyzed the structure, localization and physiological role of SL. In this study, we used antiserum against chum salmon SL. The SL genes seem to be highly conserved and the protein homology is high among the different teleost species [36,37]. Our results showed that anti-SL reacted only with the SL cells of B. bayad. The distribution and localization of SL in this species is similar to those in other teleosts [18,38-41].
The physiological function of SL is still unknown. This hormone has been related to reproductive maturation, calcium metabolism, stress, acid-base regulation, fat metabolism, background adaptation and osmoregulation [37,40,41]. The possible involvement of SL in gonadal maturation was determined by correlating changes in cell number and distribution, and intensity of staining (using immunocytochemistry) with gonadal maturation and spawning of Oncorhynchus nerka and O. Keta, Oreochromis niloticus, Mugil cephalus and Latus niloticus [18,40-42].
4.4. ACTH and α-MSH Cells
The ACTH cells of B. bayad were immunostainesd by an antiserum against human ACTH, the same one used for other teleost species [22-24,26,29]. This antiserum also showed cross-reactivity with the MSH cells present in the PI of these species and also in B. bayad as depicted by our results. The family of proopiomelanocortin (POMC)- derived hormones includes ACTH, MSH, β-endorphin and LPH. These hormones proceeded from differential processing of a common precursor molecule POMC and the amino acidic sequence of α-MSH is identical to the 13 first amino acids of the ACTH molecule [43,44]. This may account for the cross-reactivity observed in B. bayad using anti-human ACTH.
The ACTH cells were located in the RPD, forming a palisade between PRL cells and the branches of neurohypophysial tissue. According to their locations, both ACTH cells in the RPD and MSH cells in the PI stained with lead hematoxylin (PbH+) and were PAS-negative. This result agrees with previous reports for other teleosts and suggests that teleosts do not have the capacity to glycosylate the precursor proopiomelanocortin [45].
With respect to the melanotropic cells, immunocytochemical studies have shown that α-MSH antiserum specifically immunostained melanotropic (MSH) cells in all the teleost species so far studied [19,21] including B. bayad. In B. bayad the MSH cells distributed in the PI, intermingled with SL cells. This is the typical distribution for MSH and SL cells reported for other teleosts [6,8,20- 22]. However, in Solea senegalensis [46] and Thalassoma duperry [31] MSH cells appear to surround the SL cells.
The physiological role of ACTH is the stimulation of synthesis and release of cortisol from the inter-renal tissue [47]. On the other hand, MSH has been related to adaptation to a different background colour [48,49] and also in stress response [26].
4.5. TSH and GTH Cells
The family of adenohypohyseal glycoprotein hormones includes TSH and GTH. Both hormones have an identical α-subunit but different β-subunit [4,50]. Thus, the use of specific antiserum against β-subunit of TSH and GTH is necessary for the specific immunocytochemical detection of TSH or GTH cells. To our knowledge, there is no specific antibody against the piscine β-subunit of TSH. However, antiserum raised against human-β-TSH have been demonstrated to cross-react selectively with the TSH-producing cells of several teleost species [19,21], although a weak immunoreactivity to this antiserum was observed in the gonadotrophs of some teleosts [51,52]. In our study, anti-human β-TSH showed a strong immunoreactivity with the gonadotrophs of B. bayad.
The present study showed that GTH Iβ and GTH IIβ immunoreactivities were colocalized in the same cells in the pituitary of B. bayad. Similarly, in Seriola dumerilii, from which GTH I and GTH II have been obtained, chum salmon βGTHI and βGTHII immunoreactivities were colocalized in the same cells [19]. The use of antisera against human-β-TSH and chum salmon β- GTHs (GTH I and GTH II) on consecutive sections showed that β-TSH and β-GTHs (GTH I and GTH II) immunoreactivities were colocalized in the same cells located in PPD and PI in the pituitary of B. bayad. These results suggest that GTH I, GTH II and TSH may be synthesized in the same cells in the pituitary of B. bayad. The distribution of GTH cells in B. bayad is similar to that reported in other telesosts, the GTH cells being located in the dorsal and ventral portions of PPD, as well as were found around the PI of B. bayad [6,8,19-22].
Gonadotropin produced by gonadotrophs is most closely associated with reproduction, stimulating steroid production, uptake of vitellogenin, oocyte maturation and ovulation, and spermiation [53-58]. Furthermore, [59] added that FSH may trigger the female-to-male sex change in Epinephelus merra.
5. ACKNOWLEDGEMENTS
The authors are extremely grateful to Drs. H. Hideshi Kawauchi and R. M. Dores for kindly donating the antisera used in this study.
REFERENCES
- Holmes, R.L. and Ball, J.N., Eds. (1974) The pituitary gland: A comparative account. Cambridge University Press, London, 170-220.
- Ferrandino, I., Pica, A. and Consiglio, G.M. (2000) Immunohistochemical detection of ACTH and MSH cells in the hypophysis of the hermaphroditic teleost, Diplodus sargus. European Journal of Histochemistry, 44, 397-406.
- Borella, M.I., Venturieri, R. and Mancera, J.M. (2009) Immunocytochemical identification of adenohypophyseal cells in the pirarucu (Arapaima gigas), an Amazonian basal teleost. Fish Physiology and Biochemistry, 35, 3-16. doi:10.1007/s10695-008-9254-x
- Ohkubo, M., Katayama, S. and Shimizu, A. (2010) Molecular cloning and localization of the luteinizing hormone β subunit and glycoprotein hormone α subunit from Japanese anchovy Engraulis japonicus. Journal of Fish Biology, 77, 372-387. doi:10.1111/j.1095-8649.2010.02683.x
- García-Hernández, M.P., García-Ayala, A., Zandbergen, M.A. and Agulleiro, B. (2002) Investigation into the duality of gonadotropic cells of Mediterranean yellowtail (Seriola dumerilii, Risso 1810): Immunocytochemical and ultrastructural studies. General Comparative Endocrinology, 128, 25-35. doi:10.1016/S0016-6480(02)00052-7
- Mousa, M.A. (2002) Immunocytochemical and histochemical study on oogenesis in thin-lipped grey mullet, Liza ramada. Journal of the Egyptian German Society of Zoology, 39, 549-567.
- García-Ayala, A., Villaplana, M., García-Hernández, M.P., Chaves Pozo, E. and Agulleiro, B. (2003) FSH-, LH-, and TSH-expressing cells during development of Sparus aurata L. (Teleostei). An immunocytochemical study. General Comparative Endocrinology, 134, 72-79. doi:10.1016/S0016-6480(03)00198-9
- Mousa, M.A., Khalil, N.A. and Gaber, S.A. (2006) Distribution of immunoreactivities for adeno-hypophysial hormones in the pituitary gland of the Nile mormyrid, Mormyrus kannume (Teleostei, Mormyridae). Journal of the Egyptian German Society of Zoology, 51, 33-56.
- Shimizu, A., Hamaguchi, M., Ito, H., Ohkubo, M., Udagawa, M., Fujii, K., Kobayashi, T. and Nakamura, M. (2008) Appearances and chronological changes of mummichog Fundulus heteroclitus FSH cells and LH cells during ontogeny, sexual differentiation, and gonadal development. General Comparative Endocrinology, 156, 312-322. doi:10.1016/j.ygcen.2008.01.022
- Batten, T.F.C. and Ingleton, P.M. (1987) The hypothalamus and pituitary gland. The structure and function of the hypothalamus and pituitary gland. In: Chester-Jones, I., Ingleton, P.M. and Phillips, J.G., Eds., Fundamentals of Comparative Vertebrate Endocrinology, Plenum Press, New York, 283-409.
- Abdel-Latif, A.F. (1974) Fisheries of lake nasser. Aswan Regional Planning, Lake Nasser Development Centre, Aswan, 235.
- Hashem, M.T. (1981) The breeding biology of Bagrus bayad. Bulletin Institute Oceanography and Fisheries, 7, 416-428.
- Tsadu, S.M., Lamai, S.L. and Oladimeji, A.A. (2003) Sexual maturity, fecundity and egg size of wild and cultured samples of Bagrus bayad macropterus. 16th Annual Conference of the Fisheries Society of Nigeria (FISON), Maiduguri, 4-9 November 2001, 66-73.
- Conn, H.J. (1953) Biological stains. Williams and Wilkins Company, Baltimore.
- McConial, M.A. (1947) Staining of the central nervous system with lead hematoxylin. Journal of Anatomy, 81, 371-372.
- Pearse, A.G.E. (1949) The cytochemical demonstration of gonadotropic hormone in the human anterior hypophysis. Journal of Pathology and Bacteriology, 61, 195. doi:10.1002/path.1700610206
- Heath, E.H. (1965) Application of the performic acidalcian blue periodic acid-Schiff-orange G stain to sections of pituitary glands from domestic mammals. American Journal of Veterinary Research, 26, 36.
- Mousa, M.A. and Mousa, S.A. (1999) Immunocytochemical study on the localization and distribution of the somatolactin cells in the pituitary gland and the brain of Oreochromis niloticus (Teleostei, Cichlidae). General Comparative Endocrinology, 113, 197-211. doi:10.1006/gcen.1998.7200
- García-Hernández, M.P., García-Ayala, A., Elbal, M.T. and Agulleiro, B. (1996) The adenohypophysis of Mediterranean yellowtail, Seriola dumerilii (Risso, 1810): An immunocytochemical study. Tissue Cell, 18, 577-585.
- Mousa, M.A. (1998) Immunocytochemical and histochemical studies on the reproductive endocrine glands of the Nile tilapia, Oreochromis niloticus (Teleostei, Cichlidae). Journal of the Egyptian German Society of Zoology, 27, 109-134.
- Segura-Noguera, M.M., Laíz-Carrión, R., Martín del Río, M.P. and Mancera, J.M. (2000) An immunocytochemical study of the pituitary gland of the white seabream (Diplodus sargus). Histochemical Journal, 32, 733-742. doi:10.1023/A:1004101127461
- Mousa, M.A. and Khalil, N.A. (2004) Immunocytochemical study of cell type distribution in the pituitary gland of the common sole, Solea solea (Linnaeus, 1758). Journal of Union of Arab Biologists Cairo, 22, Zoology, 51-74.
- Kawauchi, H. and Yasuda, A. (1989) Evolutionary aspects of growth hormones from non-mammalian species. In: Muller, E.E., Cocchi, D. and Locatelli, V., Eds., Advances in Growth Hormone and Growth Factor Research, Springer-Verlag, Berlin and Heidelberg, 51-68.
- Pandolfi, M., Paz, D.A., Maggese, C., Meijide, F.J. and Vissio, P.G. (2001) Immunocytochemical localization of different cell types in the adenohypophysis of the cichlid fish Cichlasoma dimerus (Heckel, 1840). Biocell, 25, 35-42.
- Rodriguez-Gomez, F.J., Rendon-Unceta, M.C., Pinuela, C., Munoz-Cueto, J.A., Jimenez-Tenorio, N. and Sarasquete, C. (2001) Immunocytohistochemical characterization of pituitary cells of the bluefin tuna. Thunnus thynnus L. Histology and Histopathology, 16, 443-451.
- Mancera, J.M., Fernandez-Liebrez, P., Grondona, J.M. and Perez-Figares, J.M. (1993) Influence of the environmental salinity on prolactin and corticotropic cells in the euryhaline gilthead sea bream (Sparus aurata L.). General Comparative Endocrinology, 90, 220-231. doi:10.1006/gcen.1993.1077
- Auperin, B., Leguen, I., Rentier-Delrue, F., Smal, J. and Prunet, P. (1995) Absence of a tiGH effect on adaptability to brackish water in tilapia (Oreochromis niloticus). General Comparative Endocrinology, 97, 145-159. doi:10.1006/gcen.1995.1014
- Mousa, M.A., El-Shebly, A.A. and Khalil, M.B.A. (1999) Effect of salinity on prolactin and growth hormone cell activity in Mugil cephalus. Egyptian Journal of Aquatic Biology and Fisheries, 3, 85-101.
- Kawauchi, H., Moriyama, S., Yasuda, A., Yamaguchi, K., Shirahata, K., Kato, J. and Hirano, T. (1986) Isolation and characterization of chum salmon growth hormone. Archive of Biochemistry and Biophysics, 244, 542-552. doi:10.1016/0003-9861(86)90622-3
- Mancera, J.M., Fernandez-Liebrez, P. and Perez-Figares, J.M. (1995) Effect of decreased environmental salinity on growth hormone cells in the gilthead sea bream (Sparus aurata L.). Journal of Fish Biology, 46, 494-500. doi:10.1111/j.1095-8649.1995.tb05990.x
- Parhar, Y.S., Nagahama, Y., Grau, E.G. and Ross, R.M. (1998) Immunocytochemical and ultrastructural identification of pituitary cell types in the protogynous Thalassoma duperrey during adult sexual ontogeny. Zoological Science, 15, 263-276. doi:10.2108/zsj.15.263
- Mclean, E. and Donaldson, E.M. (1993) The role of growth hormone in growth of poikilotherms. In: Schreibman, M.P., Scanes, C.G. and Pang, P.K.T., Eds., The Endocrinology of Growth, Development and Metabolism in Vertebrates, Academic Press, New York, 43-71.
- Björnsson, B.Th. (1997) The biology of salmon growth hormone: From daylight to dominance. Fish Physiology and Biochemistry, 17, 9-24. doi:10.1023/A:1007712413908
- Sakamoto, T., McCormick, S.D. and Hirano, T. (1993) Osmoregulatory actions of growth hormone and its mode of action in salmonids: A review. Fish Physiology and Biochemistry, 11, 155-164. doi:10.1007/BF00004562
- Mancera, J.M. and Mccormick, S.D. (1998) Osmoregulatory actions of the GH/IGF axis in non-salmonids teleosts. Comparative Biochemistry and Physiology B, 121, 43-48. doi:10.1016/S0305-0491(98)10112-8
- Rand‑Weaver, M. and Kawauchi, H. (1993) Growth hormone, prolactin and somatolactin: A structural overview. In: Hochachka, P.W. and Mommsen, T.P., Eds., Biochemistry and Molecular Biology of Fishes, Elsevier, Amsterdam, 2, 39-56.
- Kaneko, T. (1996) Cell biology of somatolactin. International Review of Cytology, 169, 1-14. doi:10.1016/S0074-7696(08)61983-X
- Rand‑Weaver, M., Baker, J.B. and Kawauchi, H. (1991) Cellular localization of somatolactin in the pars intermedia of some teleost fishes. Cell Tissue Research, 263, 207‑215. doi:10.1007/BF00318762
- Kaneko, T., Kakizawa, S., Yada, T. and Hirano, T. (1993) Gene expression and intra cellular localization of somatolactin in the pituitary of rainbow trout. Cell Tissue Research, 272, 11-16. doi:10.1007/BF00323565
- Mousa, M.A. and Mousa, S.A. (2000) Implication of somatolactin in the regulation of sexual maturation and spawning of Mugil cephalus. Journal of Experimental Zoology, 287, 62-73. doi:10.1002/1097-010X(20000615)287:1<62::AID-JEZ8>3.0.CO;2-0
- Khalil, N.A., El-Gamal, A.S., Gaber, S.A. and Mousa, M.A. (2007) Immunohistochemical localization of gonadotropin-releasing hormone and somatolactin during sexual maturation and spawning of Lates niloticus. Journal of Biological Science, 7, 1102-1111. doi:10.3923/jbs.2007.1102.1111
- Olivereau, M. and Rand‑Weaver, M. (1994) Immunocytochemical study of the somatolactin cells in the pituitary of pacific salmon, Oncorhynchus nerka, and O. keta at some stages of the reproductive cycle. General Comparative Endocrinology, 93, 28‑35. doi:10.1006/gcen.1994.1004
- Follenius, E. and Dubois, M.P. (1980) Localization of anti-ACTH, anti-MSH and anti-alpha-endorphin reactive sites in the fish pituitary. In: Jutisz, M. and McKerns, K.W., Eds., Synthesis and Release of Adenohypophyseal Hormones, Plenum, New York, 197-208.
- Dores, R.M. (1990) The proopiomelanocortin family. In: Epple, A., Scanes, C.G. and Stetson, M.H., Eds., Progress in Comparative Endocrinology, Wiley-Liss, New York, 22-27.
- Iturriza, F.C. and Estivariz, F.E. (1986) Lack of glycolsilation of proopiomelanocortin might account for periodic acid-Schiff-negative reaction in ACTH cells of teleost fishes. General Comparative Endocrinology, 61, 229-236. doi:10.1016/0016-6480(86)90200-5
- Rendón, C., Rodríguez-Gómez, F.J., Muñoz-Cueto, J.A., Piñuela, C. and Sarasquete, C. (1997) An immunocytochemical study of pituitary cells of the Senegalese sole, Solea senegalensis (Kaup, 1858). Histochemical Journal, 29, 813-822. doi:10.1023/A:1026481521916
- Henderson, I.W. and Garland, H.O. (1980) The interrenal gland in pisces: Part 1 Physiology. In: Chester-Jones, I. and Henderson, I.W., Eds., A General, Comparative and Clinical Endocrinology of the Adrenal Cortex, 3, Academic Press, New York, 473-523.
- Baker, B.I., Wilson, J.F. and Bowley, T.J. (1984) Changes in pituitary and plasma levels of MSH in teleosts during physiological colour change. General Comparative Endocrinology, 55, 142-149. doi:10.1016/0016-6480(84)90138-2
- Zhu, Y. and Thomas, P. (1996) Elevations of somatolactin in plasma and pituitaries and increased alpha-MSH cell activity in red drum exposed to black background and decreased illumination. General Comparative Endocrinology, 101, 21-31. doi:10.1016/S0016-6480(96)90191-4
- Pierce, J.G. and Parsons, T.F. (1981) Glycoprotein hormones: Structure and function. Annual Review of Biochemistry, 50, 465-495. doi:10.1146/annurev.bi.50.070181.002341
- Nozaki, M., Naito, N., Swanson, P., Mijata, K., Nakai, Y., Oota, Y., Suzuki, K. and Kawauchi, H. (1990) Salmonid pituitary gonadotrophs I. Distinct cellular distributions of two gonadotropins, GTH I. and GTH II.. General Comparative Endocrinology, 77, 348-357. doi:10.1016/0016-6480(90)90224-A
- Yan, Y.H. and Thomas, P. (1991) Histochemical and immunocytochemical identification of the pituitary cell types in three Sciaenid fishes: Atlantic croaker (Micropogonias undulatus), spotted seatrout (Cynoscion nebulosus) and red drum (Sciaenops ocellatus). General Comparative Endocrinology, 84, 389‑400. doi:10.1016/0016-6480(91)90086-L
- Wallace, R.A. and Selman, K. (1981) Cellular and dynamic aspects of oocyte growth in teleosts. American Zoologist, 21, 325-343.
- Goetz, F.W. (1983) Hormone control of oocyte final maturation and ovulation in fishes. In: Hoar, W.S., Randall, D.J. and Donaldson, E.M., Eds., Fish Physiology, Academic Press, New York, 117-170.
- Nagahama, Y. (1987) Gonadotropin action on gametogenesis and steroidogenesis in teleost gonads. Zoological Science, 4, 209-222.
- Tyler, C., Sumpter, J. and Bromage, N. (1987) The hormonal control of vitellogenic uptake into cultured ovarian follicles of the rainbow trout. In: Idler, D.R., Crim, L.W. and Walsh, J.M., Eds., Proceedings of the Third International Symposium on Reproductive Physiology of Fish, St. John’s, 2-7 August 1987, 220.
- Mommsen, T.P. and Walsh, P.J. (1988) Vitellogenesis and oocyte assembly. In: Hoar, W.S. and Randall D.J., Eds., Fish Physiology, 11A, Academic Press, Inc., New York, 347-406.
- Shimizu, A., Hamaguchi, M., Ito, H., Ohkubo, M., Udagawa, M., Fujii, K., Kobayashi, T. and Nakamura, M. (2008) Appearances and chronological changes of mummichog Fundulus heteroclitus FSH cells and LH cells during ontogeny, sexual differentiation, and gonadal development. General Comparative Endocrinology, 156, 312-322. doi:10.1016/j.ygcen.2008.01.022
- Kobayashi, Y., Alam, M.A., Horiguchi, R., Shimizu, A. and Nakamura, M. (2010) Sexually dimorphic expression of gonadotropin subunits in the pituitary of protogynous honeycomb grouper (Epinephelus merra): Evidence that follicle-stimulating hormone (FSH) induces gonadal sex change. Biology of Reproduction, 82, 1030-1036. doi:10.1095/biolreprod.109.080986
NOTES
*Corresponding author.

