Open Journal of Genetics
Vol.3 No.2(2013), Article ID:33617,9 pages DOI:10.4236/ojgen.2013.32013
A realized Tuber magnatum niche in the upper Sinni area (South Italy)
![]()
Dipartimento delle Culture Europee e del Mediterraneo (DICEM), Università degli Studi della Basilicata, Matera, Italy
Email: giovanni.figliuolo@unibas.it
Copyright © 2013 Giovanni Figliuolo et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 7 January 2013; revised 20 February 2013; accepted 1 May 2013
Keywords: Genetic Diversity; Conservation; Niche; White Truffle
ABSTRACT
A realized Tuber magnatum niche, in the upper Sinni area (Serrapotamo, south Italy), was studied using geographic information system, direct in situ survey and genetic diversity at DNA marker loci. Truffières were north facing, associated with a fine loamy-sandy soil bearing high carbonate content over a conglomerate bead-rock substrate. Target area is located in Pavari's cold and medium-cold Lauretum phithoclimatic belts. Ascoma genomic DNA was analyzed and ITS sequences, microsatellites (MA2, MA4 and MA7), along with M13 minisatellite loci, did show low heteozygosity (HE = 0.153). Serrapotamo genotypes did not express localized markers when compared with Piemonte (north Italy) genetic material. ITS marker, monomorphic within T. magnatum, was efficient at distinguishing T. borchii in a mixture of both. Serrapotamo niche represents a unique T. magnatum diversity spot in the southernmost side of the Mediterranean region. Maintaining Serrapotamo T. magnatum diversity by undertaking conservation measures is adviced.
1. INTRODUCTION
The range of favorable environmental conditions under which an organism can survive and reproduce itself (fundamental niche) can be predicted for the most important wood tree and domesticated crop species [1-3]. Nonetheless, because of the combination of negative (e.g. species competition, dispersal constraints, etc.) with positive interactions (e.g. symbiosis), the realized niche [4], for a given species, can be more or less extended than the predicted one [2]. Genetic variation will ensure species resilience and persistence within a changing environment [5]. The knowledge of hypogean mushroom species that live in symbiosis with the surface roots of many plant species, collectively known as truffles, would be enhanced by integrating the niche concept and genetic diversity data. Niche modeling can improve the management of both natural and artificial truffières. The niche concept has been used primarily for Tuber melanosporum Vitt. (Périgord black) and subsequently for less important species (T. aestivum Vitt., T. brumale var. moschatum Ferry, T. macrosporum Vitt., T. borchii Vitt. and T. mesentericum Vitt.) whose environmental optimal growing conditions (e.g. soil, climate and vegetation) show a wider range of variation than the more specialist T. magnatum Pico. Artificial truffières are dictated by validated protocols of plant inoculation, cultivation and truffle harvesting with trained dogs [6]. In contrast, predicting T. magnatum fundamental niche is not so far possible. Growing areas are not fully known while ecological and genetic baseline data are often insufficient. T. magnatum, the Italian white truffle (“tartufo bianco pregiato”), is one of the top most delicacies in the culinary list. In the “good” years T. magnatum price oscillates between 1200 and 2500 EUR/Kg, but it can rise up to 3000 or 4000 EUR/Kg in years with difficult environmental conditions. Truffle market involves about 400 millions of euro and hundreds of harvesters holding a regular license [7].
T. magnatum ascocarps are harvested from very localized natural or quasi-natural ecosystems mapped in Italy [8,9], Croatia [10], Slovenia [9,11], Ticino Canton [8], Serbia and Hungary [12]. Italy is the main producer of “tartufo bianco pregiato”. Surprisingly, T. magnatum producing areas are reported as located in central and northern Italy and only “rarely” south of Umbria [8,9]. For the first time Pomarico et al. [13] brought to light Basilicata as the southern Italian Region rich in Tuber species. Among the edible species, T. magnatum is common and locally distributed in “Serrapotamo”, an area connected with Pollino National Park [13]. Rubini et al. [9] assessed genetic distance of some T. magnatum specimens from Basilicata. Overall, genetic analyses supported a self-fertilization breeding system consistent with the estimates of the within and between population genetic diversity [9,11,13]. However, differentiation due to habitat fragmentation should not be excluded. Delineating geographic areas characterized by a suitable climate for perennial symbiotic species is a consolidated method in forestry since the early 20th century [1]. The Lauretum cold belt, associated with T. magnatum distribution [13], requires an annual mean temperature from 12˚C to 17˚C, a temperature higher than 3˚C in the coldest month and the average of the yearly minimum temperatures higher than −9˚C [1]. When phytoclimatic conditions are met, geological maps represent an excellent additional tool to identify truffle production areas given that suitable soils (regolithic or residual) depend on the bed-rock type [14]. T. magnatum, whose mycelium is more widespread than can be inferred from the truffles and ectomycorrhiza distribution [15], prefers alkaline, moist, very well drained, and aerated soil [10,16]. In opposite, assessing soil micro-flora composition and prevalence is difficult; α-Proteobacteria is the most represented bacterial component, associated with the ascoma [17].
Although artificial mycorrhization of plant roots with T. magnatum ascospores is feasible [18,19], rarely massive ascomata productions have been reported in artificial plantations. Thus, the actual marketed ascomata are from natural or semi-natural realized niches. Expert harvesters are aware that overexploitation in conjunction with climatic impacts will produce quick T. magnatum genetic erosion. The ascoma intrinsic value depends on its authenticity, freshness, optimal readiness, fragrance, size, shape regularity, integrity and weight. Truffles from less important species (e.g. T. borchii) can be visually confused with T. magnatum. Furthermore, T. borchii, as a generalist species, is more abundant and can be sympatric to T. magnatum. In south Italy, Greece and Turkey T. borchii is ripening earlier than February-March. It follows that late-ripening T. magnatum ascocarps can be phenotypically confused with earlier-ripening T. borchii. Ecologic niche studies using geographic information system integrated with molecular marker technology and in situ monitoring are appropriate to carry out preliminary assessment of areas suited to T. magnatum conservation.
We attempted to promote in situ conservation of such a magnificent gift of the nature by characterizing one of the most important producing niches in south Italy and assessing its level of genetic diversity.
2. MATERIALS AND METHODS
2.1. Biological Sample
Fifty-three T. magnatum specimens (whole ascocarp or a fragment of it) were collected with the help of trained dogs, in different sites from natural or quasi-natural truffle fields located in the territory of the following municipalities (Serrapotamo area): Chiaromonte, Calvera, Castronuovo, Fardella, Francavilla S., Teana, Carbone, Episcopia and S. Chirico R. Sampling sites are mostly included within the Pollino National Park. Eight ascomata from Montemagno and Mombercelli d’Asti (Piemonte) were used as out-group (Table 1). As reference, three different species (T. borchii, T. mesentericum and T. brumale) were used in molecular analyses (Table 1).
2.2. DNA Analysis
Tissue scrap (0.25 1 g), from each ascoma, was cleaned with distilled water to eliminate soil residues, before total genomic DNA extraction. The collection (No = 54) was pre-screened with the ITS molecular fragment which allows easily the distinction of different species, including T. borchii from T. magnatum. T. borchii produced a lower molecular weight band (~560 bp) in comparison with T. magnatum (~660 bp) (Table 1). T. magnatum total genomic DNA, according to the cited Authors’ protocol [20-23] with minor laboratory-specific modifications, was used to screen the following different marker types:
• Minisatellite M13 using the universal primer M13 (GAGGGTGGCGGTTCT) [20];
• Three microsatellite loci (MA2, MA4, MA7) [21];
• ITS1 (5’TCCGTAGGTGAACCTGCGG-3’) and ITS4 (5’TCCTCCGCTTATTGATATGC3’) universal primers [22];
• ITS sequence of seven specimens from Piemonte and seven from different Basilicata sampling sites was obtained throughout direct amplicon sequencing [23].
ITS sequences were compared using the NCBI online standard Basic Local Alignment Search Tool (BlastN).
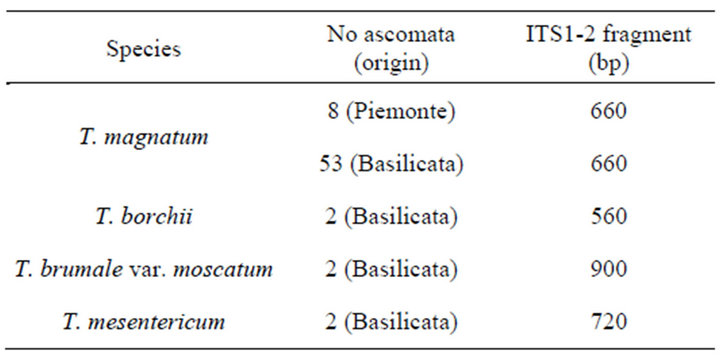
Table 1. Truffle DNA specimens from different ascomata analyzed with molecular markers along with ITS fragment discriminating different species.
2.3. Genetic Data Analysis
Genetic diversity was assessed using the expected heterozygosity (HE) calculated as HE = 1 − (p2 + q2), where for haploid binary data, p is the frequency of the “present” band and q = 1 − p the frequency of the “absent” band [24]. Genetic profiles were analyzed with Nei’s genetic distance [25] to generate a distance matrix with all pair-wise comparisons used as input for Principal Coordinates Analysis (Figure 1) [26].
2.4. Ecologic Characterization
Map layers for geology, soil [27], vegetation [28], bioclimate [29], and truffle-sampling sites were overlaid to find associations between macro-ecologic sources of variation and T. magnatum harvesting sites (Figure 2). In addition, on site survey was carried out to assess flora and vegetation. Vegetation was characterized during the truffle search by scoring species dominance and distribution pattern, using as sampling unit a representative area of about 1 hectare. About 132 Km2 of the investigated territory included producing sites. Using either the world climate database at 30 arc second spatial resolution (~750 m) (http://www.worldclim.org/) or the average precipitation and temperature values from local weather stations reported in Cantore et al. [29] were derived climate data. Bioclimatic variables represent annual temperature and precipitation trends, seasonality indices (e.g. annual range in temperature and precipitation) and extreme or limiting environmental factors (e.g. temperature of the coldest and warmest month, and precipitation of
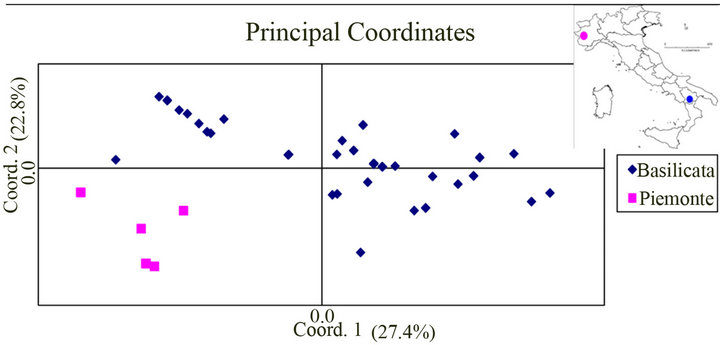
Figure 1. Genetic relationships among 61 T. magnatum ascomata from Basilicata (♦ No = 53) and Piedmont (■ No = 8) detected using minisatellite (M13) and microsatelites (MA2, MA4, and MA7) loci. The first two coordinates explain 50.2% of the total variance. The two symbols represent genotypes with different geographical origin. Twenty-one observations are hidden.
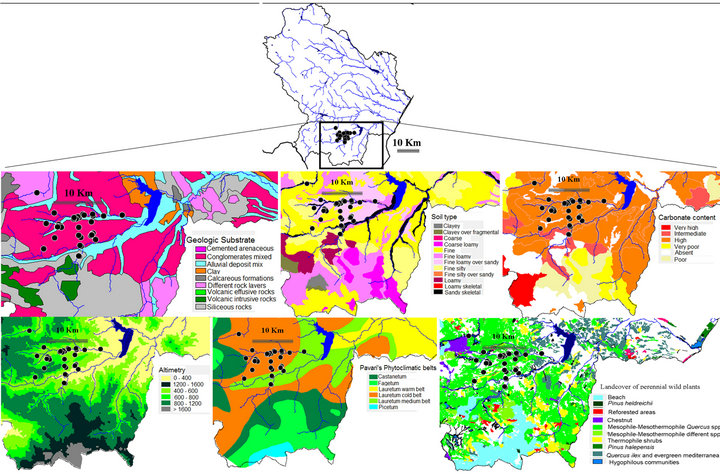
Figure 2. Ascomata sampling sites (●), relationships of T. magnatum vs geologic substrate, soil type, carbonate content, altimetry, phyto-climatic belts and land cover type specific to the ecologic niche of the upper Sinni catchment basin in the south-west side of Basilicata Region (IT).
the wet and dry quarters). The quarter is a period of three months (1/4 of the year) [30]. The average bioclimatic variables were estimated with the values of the climatic surface for each truffle harvesting area.
It was adopted a frame of five main phytoclimatic belts (Lauretum, Castanetum, Fagetum, Picetum and Alpinetum) the first time proposed by E. Mayr in the XIX century, adopted by Pavari [1] and updated for Basilicata Region by Cantore et al. [29] using the best local climatic record series. GIS analysis was conducted using Diva-Gis software (http://www.diva-gis.org/).
3. RESULTS
3.1. T. magnatum Genetic Diversity
Define abbreviations and acronyms the first time they Mini and microsatellites loci were polymorphic (Table 2). Minisatellite M13 marker produced 13 polymorphic fragments followed by microsatellites MA2, MA4, and MA7 with three or four polymorphic fragments. T. magnatum overall expected heterozygosity was HE = 0.153. Piemonte sample showed HE = 0.148 and Basilicata HE = 0.158. Genetic distances within sub-group were lower than distance between sub-groups. Scatter diagram of first and second canonical variables shows a wide variation of Basilicata gene pool without any neat distinction from Piedmont genetic material (Figure 1). Two main interconnected genetic groups, along the first canonical variable, can be distinguished in the Serrapotamo area. The first two main components explain 27.4% and 22.8% of the variation, respectively. The second coordinate depicted a more specific discrimination of Piemonte ascomata. This type of specimen partition is due to frequency dependent alleles. Population specific bands were not detected. Within Piemonte group 5 out of 8 specimens were genetically distinct and within Basilicata group 32 different ascomata genotypes out of 53 specimens were scored (Figure 1). Genetic discrimination makes it possible to infer that less than half genotypes

Table 2. Expected heterozygosity (HE) in Tuber magnatum estimated with three different molecular markers.
(21 ascomata out of 53) probably share their parental primary hyphae with those of the genetically distinct ascomata. It follows that population size of genetically distinct primary hyphae, across the entire territory, should be below the critical size necessary to avoid the deleterious effects of random genetic drift.
3.2. Discrimination of T. magnatum vs T. borchii
ITS amplified fragment distinguished different Tuber species. More specifically, the lowest band (~560 bp) was noticed in T. borchii; T. magnatum showed ~660 bp band; T. mesentericum a higher band (~720 bp) and the highest one (~900 bp) was generated by T. brumale var. moschatum (Table 1).
Identifying cross-contamination of one T. borchii ascoma out of 54 ascomata, morphologically recognized as T. magnatum, was allowed by 560 bp band specific to T. borchii. ITS sequenced amplicons including internal transcribed spacer 1, partial sequence, 5.8S ribosomal RNA gene complete sequence and internal transcribed spacer 2 partial sequence of T. magnatum either from northern or southern origin did not show any sequence variation (GenBank No: JX087950, JX087951, JX- 087952, JX087953, JX087954, JX087955, JX087956, JX087957, JX087958, JX087959, JX087960, JX087961, JX087962).
3.3. Climate
The bioclimatic variables, here reported, based on temperatures and rainfall monthly values, can meaningful indicate the relationships between the studied area and its mild climate (Table 3 and Figure 3). Lauretum cold belt captures T. magnatum realized niche with few sites falling within the Lauretum medium belt (Figure 2). The third trimester (July, August and September) performs either as the driest or warmest quarter (Table 3 and Figure 3). In this period, precipitations are often the outcome of thunderstorms, while during the coldest quarter (January, February, and March) the snow can persist on the soil for one or more days. Furthermore minimum temperature of the coldest month (=3.16˚C in January) will not coincide with the average of the annual minimum temperature (=−5.7˚C), given that the minimums can also be captured by December and February (data not shown).
Temperature data recorded by weather stations from 1926 to 1984 in the target zone along with phytoclimatic Pavari's belt, retrived from Cantore et al. [29] agree with data from the world database. By comparing climatic patterns of the upper Sinni valley vs northern sites (Montemagno and Mombercelli d’Asti) (Table 3 and Figure 3) it is noticeable a slight shift from a Mediterranean climate to a slight Continental one. Both sites share highest precipitations in October and November, with evenly distributed annual mean temperatures, mean monthly temperature range, isothermality and temperature of the warmest month (July). Nonetheless, in Piemonte minimum temperature (January) can drop below 2˚C (between 0.5˚C and 1.1˚C), winter is drier and, spring wetter than in Basilicata (Table 3 and Figure 3).
3.4. Vegetation
T. magnatum is mainly associated with the Lauretum cold belt. Few sampling sites are included in Lauretum medium belt. Dogs did find ascomata within and between vegetation patches of mesophile-mesothermophile perennial plant species. The ecological niche was not impacted by human activities with the exception of traditional agriculture and low input forest management practices dictated by domestic owners. Perennial wild plants most likely associated with T. magnatum are: Quercus pubescens Willd., Q. cerris L., oak hybrids, Populus canescens (Aiton) Sm., P. tremula L., P. alba L., Crategus oxyacantha L. and Salix caprea L. (Table 4). These findings were based on the in situ perceptions (e.g. plant host root proximity to ascomata), not on scientific proofs (e.g. symbiosis histological evidence).
Overall, vegetation was not evenly distributed: linear formations (at the margins of cultivated fields or walking paths), forested patches composed of mesophile-mesothermophile trees (north faced slopes), mixed with dense thermophile shrubs (south, southeast faced slopes) were present. Perennial plant species richness was high at the collecting site (Table 4). Piemonte (Montemagno d’Asti) white truffles were associated with the following species: Populus nigra L., P. alba L., P. tremula L., Quercus robur L., Salix alba L. and Tilia cordata Mill. in moist soils and with few signs of anthropic activity [11]. Mature ascomata of T. magnatum can be harvested, in Basilicata, from mid September until the end of December. High species richness of commercially relevant truffles has been found in Serrapotamo niche: During the winter and early spring, T. borchii can be harvested; along with T. magnatum it grows, from November till March, T. brumale var. moscatum and, during the summer, T. aestivum (syn. uncinatum if harvested in autumn).
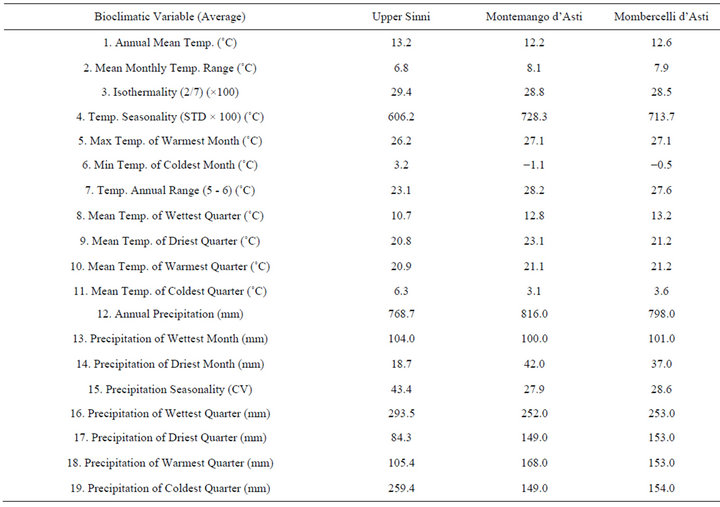
Table 3. Bioclimatic variables, from world climate database, specific to the target area of the upper Sinni catchment basin (Basilicata Region) and two areas of Montemagno and Mombercelli d’Asti located in Piedmont Region.
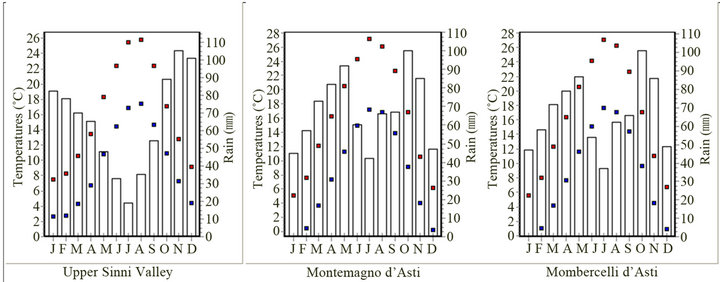
Figure 3. Minimum (■), maximum (■) temperatures (˚C), and monthly precipitations (mm) (bar chart) specific to upper Sinni catchment basin (Basilicata Region), Montemagno and Mombercelli d’Asti (Piemonte Region).

Table 4. Perennial plant species scored over a representative site within the area reported in Figure 2.
3.5. Geology and Pedology
T. magnatum ascomata have been found in soils having a fine loamy structure over sandy, with high carbonate content. The geological substrate was of conglomerates made of fine cement mixing round stones of different size.
3.6. Landscape
Serrapotamo territory produces a significant quantity of T. magnatum ascomata. T. magnatum is distributed across a variable landscape (~150 Km2) buffering Serrapotamo creek and upper Sinni catchment basin in Basilicata (9992.24 km2) (Figure 2). Overall, the area has a variable geomorphology and a difficult accessibility. Mountains (from 1000 to 2000 m) on the north-north-west side protect the target area from the Atlantic and northern storms, while Pollino range acts as a barrier against dry winds coming from south. Producing truffle sites were all located on northern faced lands and at different altitudes (from 350 to 750 m) (Figure 2). The regional main area is excluded from the details of Figure 2 because do not meet the whole set of environmental associations found in Serrapotamo area with the exception of few point areas (data not shown). Although soil, geology, and climate are suited in north-west, side of the Region, at present the ancient natural ecosystem has been replaced by agro-ecosystems.
4. CONCLUSIONS
Accurate niche modeling for T. magnatum, given the unsatisfactory available ecological and genetic data, would be unrealistic. At present, we can provide no more than management guidelines. The effect of basic a-biotic factors such as climate, soil type, pedo-genetic substrate as well as the effect of radiation, electric potential, chemical stimuli and, CO2 vs O2 or N2 soil content on species life cycle is still not completely clarified [31]. The expert harvesters ask assessing the role of absolute minimum and maximum soil temperatures together with the effect of snow and hailstorms. Symbiotic vs competition relationships among biotic factors, especially bacteria, may play a key role [17]. The lack of ascomata production from successful T. magnatum mycorrhized plants does not provide any insight about “species × environment” interaction. The role of genetic diversity on sexual reproduction and mycorrhization, as well as the evolutionary responses to environmental and demographic changes need to be investigated. Dog is condicio sine qua non for all truffle-related activities, included science. Monitoring is difficult, also in the computer age, given that biosensors are not yet able to mimic dog biological capabilities in scents distinction [32]. For a diagnostic screening it is necessary the isolation of sub-population specific markers. In opposite, at inter-specific level, T. borchii can be efficiently discriminated from T. magantum using ITS marker. With such easy to use marker, we estimated a contamination level of one T. borchii ascoma out of 64 ascomata. Such type of mixture could be problematic for inocula preparation (in mycorrhization nurseries) since T. borchii is more competetive than T. magnatum in plant root mycorrhization [19].
The assessed not optimal heterozygosity is consistent with the reproduction system of an inbreeder (homothallic fungi) rather than out-breeder (heterothallic fungi). However, we do not exclude that T. magnatum could be an out-breeder affected by genetic drift. Habitat isolation and locally low effective population size could have lead to inbreeding. Genetic diversity (HE) at three microsatellites loci (MA2, MA4, and MA7) agreed with that reported by Rubini et al. [21] except for the lower heterozygosity here detected at MA4 locus. It is clear that genetic diversity, due to frequency dependant alleles, is geographically structured. According to the migration model from south to north (after the last glaciation) [33], we expect genetic richness in southern areas, for both host plants and fungi. It follows that we should consider Serrapotamo niche as a key site for T. magantum conservation.
Almost all the realized T. magnatum niches are overexploited. Site-specific anthropogenic impacts are mainly due to soil compaction by tracks, systematic sylviculture and loss of traditional land cover management. Nonetheless, across the studied Region, in the last two decades, following boars’ population size increase, Tuber spp. dispersal ability should be higher than in the past. Fungal mycelium does not tolerate soil compaction and aridification. Tuber ascomata were rarely found in woods impacted by systematic sylviculture. In addition, the current climatic change probably will promote altitudinal and latitudinal shift of the niche. Populations from low to medium density, even if they exhibit adaptive evolution, after abrupt environmental change, have high extinction probability [34]. Conservation measures, such as the efforts to increase size, quality, and connectivity of species’ habitat, could at least reach the goal to maintain (if not to increase) genetic diversity and, therefore, the adaptive potential within and among truffle beads.
Genetic diversity and localized distribution, discovered in this case-study, are basic conditions for planning T. magnatum conservation in one of the southernmost distribution area of the species.
Management Guidelines
Based on this preliminary study, at the present, it is possible to outline the following general statements:
• Keep local climate uniform by managing appropriately the land-cover in order to decrease the daily variance for humidity and temperature;
• Maintain linear high trunk tree formations (preferably host plants), north-south oriented, because produce shadow, perform as a barrier against winds and reduce topsoil vapor-transpiration;
• Avoid systematic forest management (e.g. coppicing, thinning and systematic cuttings);
• Apply very selective cuttings and protect symbiont species;
• Manage coppice vs high trunk with gradualism, year after year. Reduce the number of shoots without damaging the source-sink plant equilibrium;
• Remove, without reducing species-richness, shrubs in excess that belong to not symbiotic plant species;
• Maintain an open wood structure, with ground protected by the tree shadow;
• Allow canopy development to ensure a fresh undercanopy climate, especially during the third quarter;
• Remove and prevent bulk of raw organic matter, such as fallen wood;
• Do not compact soil with machineries;
• Increase, nearby productive symbionts (e.g. poplar, willow, etc.), the number of host plants using clonal propagation;
• Monitor truffle production and other measurable parameters.
5. ACKNOWLEDGEMENTS
CNR (Istituto per la Protezione delle Piante, Torino) kindly provided Piemonte DNA specimens. We are deeply grateful to local experts who assisted our field survey and to Università degli Studi della Basilicata for its institutional support. This paper is dedicated to the honor of Fly and to the memory of Lola.
REFERENCES
- Pavari, A. (1916) Preliminary assessment of the cultivation of exotic forestry species in Italy. General section. Annales of the Regio Istituto Superiore Forestale Nazionale, Florence, Italy, 1-221.
- Puliam, H.R. (2000) On the relationship between niche and distribution. Ecology Letters, 3, 349-361. doi:10.1046/j.1461-0248.2000.00143.x
- Jones, P.G., Diaz, W. and Cock, J.H. (2005) Homologue: A computer system for identifying similar environments throughout the tropical world. Version beta a.0. Centro Internacional de Agricoltura Tropical (CIAT), Cali.
- Hutchinson, G.E. (1957) Concluding remarks. Cold Spring Harbor Symposia on Quantitative Biology, 22, 415- 427. doi:10.1101/SQB.1957.022.01.039
- Reed, E.T., Schindler, D.E. and Waples, R.S. (2010) Interacting effects of phenotypic plasticity and evolution on population persistence in a changing climate. Conservation Biology, 25, 56-63. doi:10.1111/j.1523-1739.2010.01552.x
- Hall, I.R., Brown, G.T. and Zambonelli, A. (2007) Taming the truffle: The history, lore, and science of the ultimate mushroom. Timber Press, Portland, 304 pp.
- Ciani, S. (2008) White truffle, the good sleeping in the forest. Origine. Special Issue of the Agric. Informer. Year III, September-October 2008, 30-34.
- Hall, I.R., Zambonelli, A. and Primavera, F. (1998) Ectomycorrhizal fungi with edible ascomata. 3. Tuber magnatum, Tuberaceae. Economic Botany, 52, 191-200. doi:10.1007/BF02861209
- Rubini, A., Paolocci, F., Riccioni, C., Vendramin, G. and Arcioni, S. (2005) Genetic and phylogeographic structures of the symbiotic fungus Tuber magnatum. Environmental Microbiology, 71, 6584-6589. doi:10.1128/AEM.71.11.6584-6589.2005
- Bragato, G., Vignozzi, N., Pellegrini, S. and Sladonja, B. (2010) Physical characteristics of the soil environment suitable for Tuber magnatum production in fluvial landscapes. Plant Soil, 329, 51-63. doi:10.1007/s11104-009-0133-8
- Mello, A., Murat, C., Vizzini, A., Gavazza, A. and Bonfante, P. (2005) Tuber magnatum Pico, a species of limited geographical distribution: Its genetic diversity inside and outside a truffle ground. Environmental Microbiology, 7, 55-65. doi:10.1111/j.1462-2920.2004.00678.x
- Bratek, Z., Gògàn, A., Halàsz, K., Bagi, I., Erdei, V. and Bujàki, G. (2003). The northernmost habitats of Tuber magnatum known from Hungary. http://www.kert.szie.hu/resources/217.pdf
- Pomarico, M., Figliuolo, G. and Rana, G.L. (2007) Tuber spp. Biodiversity in one of the southernmost European distribution areas. Biodiversity Conservation, 16, 3447- 3461. doi:10.1007/s10531-006-9013-1
- Costa, M. (2006) Geological, geomorphologic traits and truffle genesis. Italian Journal of Geosciences, 6, 229- 233.
- Zampieri, E., Murat, C., Cagnasso, M., Bonfante, P. and Mello, A. (2010) Soil analysis reveals the presence of an extended mycelial network in a Tuber magnatum truffle ground. FEMS Microbiology Ecology, 71, 43-49. doi:10.1111/j.1574-6941.2009.00783.x
- Rondelli, F. (1998) Truffle bearing soils in the Central Apennine: Observation of hierarchic structures of the pedologic traits. Monti e Boschi, 5, 44-49.
- Barbieri, E., Guidi, C., Bertaux, J., Frey-Klett, P., Garbaye, J., Ceccaroli, P., Saltarelli, R., Zambonelli, A. and Stocchi, V. (2007) Occurrence and diversity of bacterial communities in Tuber magnatum during truffle maturation. Environmental Microbiology, 9, 2234-2246. doi:10.1111/j.1462-2920.2007.01338.x
- Granetti, B. (1987) Mycorrhization of some Salix species with Tuber magnatum Pico. Annali della Facoltà di Agraria, 41, 875-888.
- Bencivenga, M., dal Savio, D. and Venanzi, G. (1990) Development of symbiotic plants and mycorriza evaluation in a truffle bed of Tuber magnatum Pico after four years since plantation. Annali della Facoltà di Agraria, 43, 817-828.
- Latouche, G.N., Daniel, H.M., Lee, O.C., Mitchell, T.G., Sorrel, T.C. and Meyer, W. (1997) Comparison of use of phenotypic and genotypic characteristics for identification of species of the anamorph genus Candida and related teleomorph yeast species. Journal of Clinical Microbiology, 35, 3171-3180.
- Rubini, A., Topini, F., Riccioni, C., Paolocci, F. and Arcioni, S. (2004) Isolation and characterization of polymorphic microsatellite in white truffle (Tuber magnatum). Molecular Ecology Notes, 4, 116-118. doi:10.1111/j.1471-8286.2004.00587.x
- White, T.J., Bruns, T., Lee, S. and Taylor, J. (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis, M.A., Gelfand, D.H., Sninsky, J.J. and White, T.J., Eds., PCR Protocols. A Guide to Methods and Applications, Academic Press, San Diego, 315-332.
- Mang, S.M. and Figliuolo, G. (2010). Species delimitation in Pleurotus eryngii species-complex inferred from ITS and EF-1α gene sequences. Mycology, 1, 269-280.
- Weir, B.S. (1996) Genetic data analysis II. Sinawer Associates, Inc., Sunderland.
- Nei, M. and Li, W.H. (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proceedings of the National Academy of Sciences of the United States of America, 76, 5269-5273. doi:10.1073/pnas.76.10.5269
- Peakall, R. and Smouse, P.E. (2006) GENALEX 6: Genetic analysis in Excel. Population genetic software for teaching and research. Molecular Ecology Notes, 6, 288- 295. doi:10.1111/j.1471-8286.2005.01155.x
- Regione Basilicata (2003) Pedologic map of Basilicata Region. Workshop on Pedologic Survey to Know, Safeguard and Valorise Basilicata Landscape and Agriculture, 7 April 2003, Basilicata Region, Department Agriculture and Rural Development, 1-54.
- Costantini, G., Bellotti, A., Mancino, G., Borghetti, M. and Ferrara, A. (2006) Basilicata forest map. Basilicata Region. Department Environment, Landscape and Sustainability Policies, INEA, Potenza.
- Cantore, V., Iovino, F. and Pontecorvo, G. (1987) Climatic features and phytoclimatic zones of Basilicata. National Research Council (CNR), Institute of Ecology and Forest Hydrology, Rende, Cosenza,
- Hijmans, R.J., Cameron, S.E., Parra, J.L., Jones, P.G., Jarvis, A. (2005) Very high resolution interpolated climate surfaces for global land areas. International Journal of Climatology, 25, 1965-1978. doi:10.1002/joc.1276
- Pacioni, G. and Comandini, O. (1999) Tuber. In: Cairney, J.W.G. and Chambers, S.M., Eds., Ectomycorrhizal fungi. Key Genera in Profile. Springer, Berlin, Heidelberg, 163- 186. doi:10.1007/978-3-662-06827-4_6
- Habib, M.K. (2007) Controlled biological and biomimetic systems for landmine detection. Biosensors and Bioelectronics, 23, 1-18. doi:10.1016/j.bios.2007.05.005
- Murat, C., Diez, J., Luis, P., Delaruelle, C., Dupré, C., Chevalier, G., Bonfante, P. and Martin, F. (2004) Polymorphism at the ribosomal DNA ITS and its relation to postglacial re-colonization routes of the Perigord truffle Tuber melanosporum. New Phytologist, 164, 401-411. doi:10.1111/j.1469-8137.2004.01189.x
- Boulding, E.G. and Hay, T. (2001). Genetic and demographic parameters determining population persistence after a discrete change in the environment. Heredity, 86, 313-332. doi:10.1046/j.1365-2540.2001.00829.x

