Open Journal of Preventive Medicine
Vol.3 No.4(2013), Article ID:33740,8 pages DOI:10.4236/ojpm.2013.34047
Application of causal model to maternal smoking cessation intervention in pregnancy
![]()
1Department of General Practice, School of Primary Health Care, Monash Univeristy, Melbourne, Australia; *Corresponding Author: ansarirm@yahoo.com
2Department of General Practice, School of Primary Health Care, Monash Univeristy, Melbourne, Australia
3Department of Endocrinology & Diabetes, Dallah Hospital, Riyadh, KSA
Copyright © 2013 Rashid M. Ansari et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 12 May 2013; revised 12 June 2013; accepted 24 June 2013
Keywords: Intervention; Pregnant Women; Maternal Smoking; Causal Modelling; Offspring
ABSTRACT
The adverse effects of maternal smoking during pregnancy on both the offspring and women are well known. The main objective of this research article is to provide health professional causal modelling approach to make a more comprehensive assessment of major determinants of smoking behaviour during and after pregnancy and consequently the outcomes of pregnant women smoking which are adversely affecting both the offspring and pregnant women. The causal model based on theory and evidence was modified and applied to material smoking cessation intervention to control the adverse effects of smoking on offspring obesity and neurodevelopment. In this approach a generic model links behavioural determinants, causally through behaviour, to physiological and biochemical variables, and health outcomes. It is tailored to context, target population, behaviours and health outcomes. The model provides a rational guide to appropriate measures, intervention points and intervention techniques, and can be tested quantitatively. The causal modelling approach showed promising results which can be used to help maternal smoking women to understand the risk of smoking and help them to quit smoking. The regression analysis of maternal smoking women BMI (n = 1000) on offspring BMI was statistically significant, p < 0.05, 95% CI (0.28 - 0.38) and so was the analysis of offspring SBP on maternal BMI for male offsprings, p < 0.05, 95% CI (0.06 - 0.43) but not for female offsprings (p > 0.05). This supported the hypothesis that maternal smoking women BMI during pregnancy is an important determinant of offspring obesity and consequently the risk factors of cardiovascular development. The causal modelling approach is unique as it provides an incentive to health professional to use these models to target any important and modifiable determinants of the maternal smoking behaviour and decrease the risk of adverse pregnancy outcomes for the offspring and the mother.
1. INTRODUCTION
Maternal smoking during pregnancy has been associated with a wide range of adverse outcomes in offspring [1,2]. The main adverse effects act through the respiratory and vascular systems—babies of smokers are more likely to have respiratory problems as well as circulatory effects. Despite a large body of evidence from samples of geographically diverse populations, a direct causal link between maternal smoking during pregnancy and neurodevelopment has not been established [3]. Maternal smoking is also linked to childhood obesity [4,5]. The epidemiological studies have identified smoking during pregnancy as exerting an independent, adverse effect on a variety of reproductive and other health outcomes [6].
However, there remains considerable debate regarding the impact of maternal smoking during pregnancy on the physical and cognitive development of children [7]. Therefore, establishing the control strategies to address these effects can provide insight into the aetiology of adverse child outcomes ranging from birth complications [8,9] to behaviour problems [10], psychological disturbances [11] asthma [12], obesity [13,14], and cognitive delays [15].
The maternal smoking cessation interventions designed over the past 25 years to reduce smoking during pregnancy have not been very successful. Therefore, interventions should be developed to address the key factors to motivate maternal smoking women in quitting smoking [16] and to help smoking women in behaviour change approach [17]. In that direction, Becker [18] presented the health belief model, Maddux and Rogers [19] presented protection motivation theory and the theory of reasoned action was proposed by Fishbein and Azjen [20]. All these authors suggested that the impact of a negative health outcome and the desire to avoid it or reduce its impact creates a motivation for self-protection.
In a Cochrane literature review, interventions with high intensity and high quality yielded the greatest cessation rates in late pregnancy [21] and the lower intensity treatments increased abstinence rates compared with usual care [22,23]. In addition, even the most effective interventions seldom exceeded cessation rates of 20% among maternal smoking women [16]. Therefore, these interventions should be linked to design issues during early stages of intervention development [24].
These links become more important when interventions are complex with a number of components [25]. These interventions approach apply mapping techniques including logic models [26] and matrices for interventions methods and strategies [27]. The causal models also complement these approaches and since maternal smoking occurs along with a broader constellation of social and behavioural factors that also influence child growth and development [28].
In this research project, causal modelling approach is applied using a simple generic model which links disease and behavioural determinants in a causal pathway. The model has four levels: behavioural determinants, behaviour (smoking cessation), physiological and biochemical variables, and health outcomes and for each application, a generic model is also specified. The specific model is tailored to characteristics of the target population (pregnant smoking population and pregnant women of low socioeconomic status), social context, and target behaviour and health or disease outcomes. The selection of behavioural determinants, intervention and measurement points, and behaviour change techniques were guided by theory and evidence [28].
The causal modelling approach is similar to the FourModel Approach proposed by Bauman et al. [29], which works from a theoretical model to an implementation model for the programme. The causal modelling is unique as it assists in the choice of behaviour change techniques and help to control the adverse effects of maternal smoking on offspring obesity and neurodevelopment.
The challenging research question is that what intervention techniques might be most effective to help pregnant women to understand the risk of smoking and to reinforce positive beliefs in them to quit smoking. The hypothesis of causal relationship would be supported if maternal smoking cessation could be shown to reduce the risk of obesity and neurodevelopment in offsprings. The hypothesis that the maternal BMI during and after pregnancy is an important factor of offspring obesity and consequently the cardiovascular risk factors development will also be tested in this study.
2. MATERIALS AND METHODS
2.1. Framework of Interventions
A framework of the complex interventions was initially developed by Medical Research Council (MRC) of UK [25,30], which emphasized the importance of the early stages of developing the intervention approach. Table 1 provides the framework for developing and designing the complex interventions for maternal smoking cessation [28,30]. The modelling approach in Phase 2 is illustrated by using the causal modelling techniques to guide the design of a programme to support behaviour change (maternal smoking behaviour during pregnancy) for this intervention.
The development of causal model for maternal smoking drew iteratively on epidemiology and psychology. The methods used in developing the causal model for maternal smoking are summarized in Table 2. Epidemiology informed the causal generic model from behaviour to health outcome in four ways [28,30]: define the health outcome (childhood obesity and neurodevelopment), define the target group (pregnant smoking women), iden-

Table 1. Framework for designing the complex interventions for smoking cessation adapted from Campbell et al. [30] and Hardeman et al. [28].

Table 2. Methods used in the development of the causal model for maternal smoking.
tification of the target behaviour and its impact on health outcome and developing the measures of the target behaviour. The main method used to inform the causal model was a review of epidemiological evidence. The psychology informed the maternal smoking model from the behavioural point of view and developed measures of change in behavioural determinants.
The methods of theory-based causal modelling used in conducting an expert meeting with the stakeholders, systematic reviews of intervention aimed at smoking cessation, conducting interviews of individuals and the focus groups with the target population (smoking women in pregnancy; pregnant women with low socio-economic status), selection of intervention points and the team meetings.
2.2. Techniques to Support Behaviour Change
This research project used the techniques to support behaviour change based on the underlying theory and applications proposed by Hardeman et al. [28] and based on that theory, the final causal model was developed which represents the maternal smoking cessation intervention. The Planned Behaviour-TPB theory presented by Ajzen [32] was selected for the causal model which specifies causal links and helps to test the theory’s causal pathways [33-35]. The theory proposes persuasive messages as the main technique to change beliefs [36]. In maternal smoking model, information is provided about the potential to control the outcome (adverse effects on offspring) by means of maternal smoking cessation. In addition, other techniques to impact directly on intention and behaviour, not covered by the TPB approach, have also been selected. These include goal setting, self-monitoring and reinforcement and building family support [31].
2.3. Target Population (Settings)
The smoking cessation and reduction intervention targeted women who were pregnant and the theory-based causal modelling concepts were applied to two types of population, one was related to the “general” pregnant smoking population and the other one was related to pregnant sub-populations, women of low socioeconomic status based on the National Health Survey data of Pakistan [45]. The efficacy of medical clinic based smoking cessation interventions is well established [46,47]. However, women belonging to low socioeconomic status have various problem such as transportation, time, cost and child care to participate in such settings [48,49]. In this project, the smoking cessation intervention was specifically considered in respect to clinical settings as similar clinical-based intervention studies were appeared to be effective in reaching and enhancing the cessation among female smokers of low socioeconomic status [40, 50].
The interventions targeted at general pregnant smokers involved multiple components, but in most cases, the effectiveness of individual components was not tested. The interventions specifically targeting women in this group are lacking, but several strategies have been tested in this area and one intervention, using a self-help guide [51] showed promising results. The self help guide is based on the Trans-theoretical Model [43], and motivational interviewing techniques. Self-help guides may be important for supporting cessation efforts in the “general” pregnant smoker population.
2.4. Data Collection and Analysis
This study used the specific data of pregnant women (sub-sample) related to two types of population—general pregnant smoking women and pregnant smoking women of low socioeconomic status from the National Health Survey of Pakistan (NHSP) conducted between 1990 and 1994 by the Pakistan Medical Research Council (PMRC) with technical assistance from the US Centers for Disease Control (CDC). The sampling details, design, components, survey instruments and quality control have previously been reported [45]. Ethical approval for the survey was obtained from the Institutional Review Board at the Pakistan Medical Research Council [52].
In this study, analysis was limited to 1000 mothers for whom prospective data on self-reported smoking status from pre-pregnancy to 14 years post delivery was available from the survey [45] including their offspring who has attended a follow-up examination at age of 14 years. The body mass index of offspring was included as timedependent variables in these models, along with age at baseline. The baseline questionnaire was based on the determinants on maternal smoking behaviour. The two major outcomes, obesity and systolic blood pressure (SBP) were considered as the two important risk factors for the cardiovascular disease among offsprings and STATA was used for statistical analysis.
2.5. Statistical Analysis
We examined the association between maternal smoking by categorizing it in two groups of off-spring male and female and the outcomes in these exposure groups were compared by one-way analysis of variance, using an F test when the outcome was based on continuous BMI and a chi-squared test when the outcome was based on BMI categories. The data was analyzed with regards to height, weight and SBP. The off-spring BMI was classified into normal, overweight and obese according to the standard definitions and based on the surveys by Coleman [53].
3. RESULTS
Descriptive Statistics
The descriptive analysis was carried out for each measured variable such as SBP and maternal BMI and derived variable such as offspring BMI was calculated as BMI = Weight (kilograms)/height (square meters). The histograms of offspring BMI and SBP were plotted to check the range of variables and box plots were plotted for offspring BMI and SBP for distribution of data in the sub-sample. The mean, median and standard deviation of offspring BMI and SBP were calculated and shown in Table 3. The regression analysis was performed to see
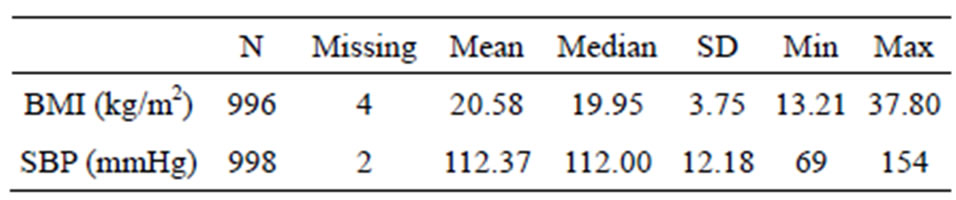
Table 3. Summary statistics for offspring BMI and SBP at age 14 subsample of maternal smoking population.
the strength of association between the two groups.
The two sample t-test found a statistically significant difference between male and female offsprings in terms of BMI and SBP (Tables 4 and 5). A test of the equality of variances found that the variances were unequal between male and female for BMI (p = 0.005) but not for SBP, therefore, the t-test used the unequal variances version for BMI but not for SBP.
The regression analysis of offspring BMI on maternal BMI showed that for every unit increase in maternal BMI, the offspring BMI at age 14 years increased by 0.33 kg/m2. The 95% confidence interval CI (0.28 - 0.38) and the value of p < 0.05 suggested that the increase in offspring BMI is statistically significant. Similarly, the regression analysis of offspring SBP on maternal BMI showed that for every unit increase in maternal BMI, the offspring SBP at age 14 years increased by 0.24 mmHg. The 95% confidence interval CI (0.06 - 0.43) and the value of p < 0.05 suggested that the increase in offspring SBP is statistically significant.
The regression slope was statistically significant for male offsprings (p-value < 0.05) but not for female offsprings (p-value > 0.05). These results are in agreement with the findings of Paradis et al. [54] which have indicated that maternal BMI is positively associated with SBP in all age-gender group and that the mean SBP of 13 years old was higher in males than in females.
4. DISCUSSIONS
4.1. Theory-Based Behavioural Determinants
Focus groups and interviews with pregnant women in our study showed that few were aware that maternal smoking would increase their risk of adverse effects on their offsprings. Experts proposed at their first meeting that the theory should specify determinants of intention to change, as risk awareness and associated motivation to adopt preventative measures could not be assumed among the offspring of maternal smoking women.
Behavioural models and cognitive-behavioural models were not selected as the organizing theory, because they do not clearly specify how to strengthen motivation and primarily focus on problem behaviours. Social cognition models that specify determinants of motivation were chosen for further consideration.
After the expert meeting the project team reviewed the

Table 4. Summary statistics and two-sample t-test of body mass index by sex in the sample of maternal smoking population.
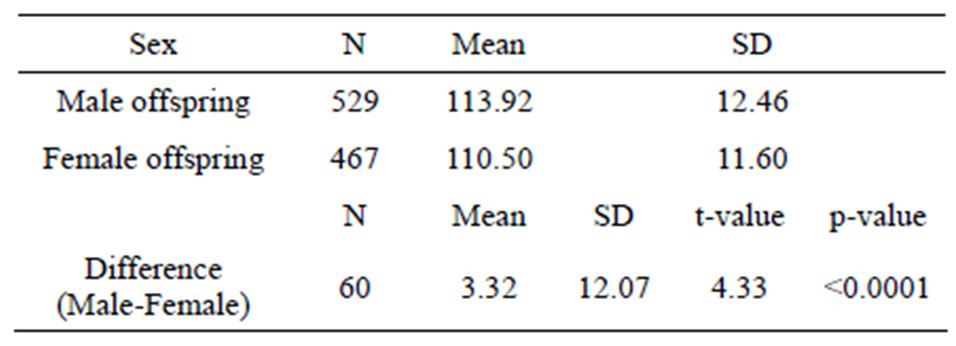
Table 5. Summary statistics and two-sample t-test of systolic blood pressure by sex in the sample of maternal smoking population.
social cognition theories in more detail. The Theory of Planned Behaviour was selected to underpin the causal model, as it clearly specifies causal links between determinants of intention and behaviour, there is good evidence to support the theory’s predictive validity, and construct measurement is clearly specified, which allows testing of the theory’s causal pathways.
4.2. Intervention Points
In our study, TPB was used in a novel, although theoretically appropriate, way. The importance of building on individuals’ own reasons for change was corroborated by interviews with at-risk individuals. Experts identified a limitation of the TPB: a strong intention alone does not always lead to behaviour change therefore; intention and behaviour were identified as further intervention points.
4.3. Techniques to Support Behaviour Change
At the second meeting experts identified additional techniques with evidence of effectiveness to change beliefs: reinforcing positive beliefs in pregnant women and problem solving in relation to negative beliefs. To bridge the ‘gap’ between intention and behaviour, other techniques, not informed by the TPB, were selected to impact directly on intention and behaviour. The experts identified additional techniques to support individuals in moving from intention to action: action planning building social support, facilitating habit formation and preventing relapse.
The final causal model, including intervention points, behaviour change techniques and associated measures, is shown in Table 6. The behavioural determinants are derived from the TPB and the model specifies that change in specific maternal smoking behaviours will impact on physiological and biochemical variables, either directly via self-belief in giving-up smoking or indirectly via information about the adverse effects on offsprings. Changes in these variables would result in reinforcement of positive beliefs and consequently the reduction in the risk of adverse pregnancy outcomes for the offspring and the mother.
4.4. Limitations of Intervention Approach
A questionnaire method was used in this intervention approach to obtain information on smoking status. This approach did not take into consideration biochemical measures to verify information on smoking habits, therefore, the potential for misclassification bias cannot be ruled out [55]. The other limitation of the study would be from the point of view of overall methodological concern on theory-based causal modelling and its specific roles on the various components in the interventions, and the difficulties of assessing these components independently [28,30], therefore their impact in these maternal smoking interventions will be difficult to assess.
5. CONCLUSIONS
This research article has presented a causal modelling approach to the development of a theory-based programme for evaluation of maternal smoking cessation intervention. It outlined the steps followed in the translation of a generic causal model into a specific model, in the context of maternal smoking and behavioural programme for its prevention in that population and indicated testable pathways from behavioural determinants to health outcomes and logical intervention points, and linked the pathways to behaviour change techniques and measures.
The contribution of this research article has come from the causal modelling approach which showed promising results and supported the research question that causal modelling approach can help pregnant smoking women to understand the risk of smoking and can provide an incentive to health professional to use these models to target any important and modifiable determinants of the maternal smoking behaviour and decrease the risk of adverse pregnancy outcomes and promote the best possible health outcomes for the offspring and the mother during and after the pregnancy. A set of instructions on how to apply causal model in clinical settings was developed for health professional to be used in clinical settings and a short duration video was developed for maternal smoking women providing details of risk of smoking as an outcome of the model.
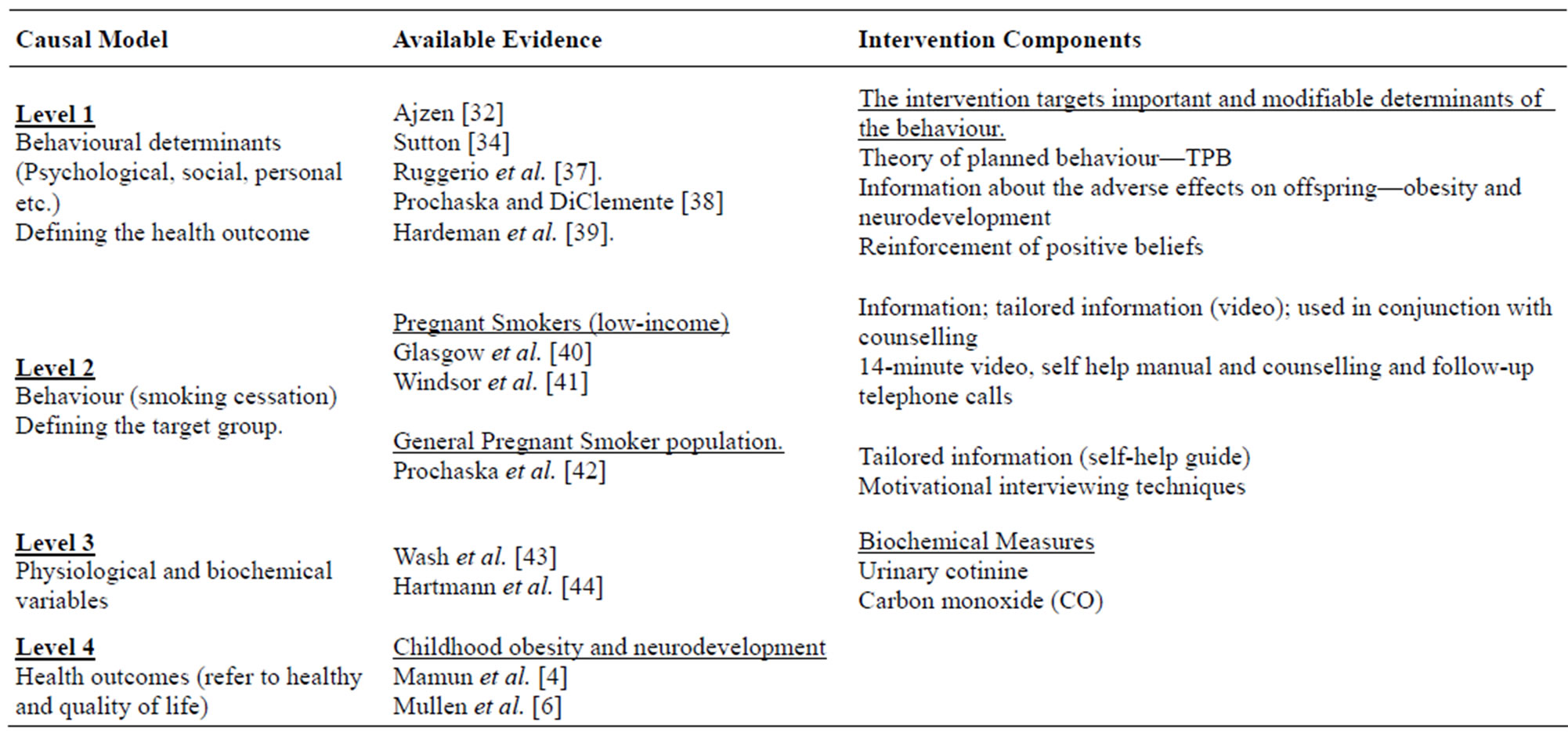
Table 6. Final maternal smoking causal model for pregnant smoking population and pregnant smoking women with low socio-economic status.
The statistical analysis has also supported the hypothesis that maternal BMI during and after the pregnancy is an important determinant of offspring obesity and consequently the risk factors of cardiovascular development. Although, the hypothesis that the causal modelling approach will help to control the adverse effects of maternal smoking on offspring neurodevelopment was not supported by this study, but there is some evidence that cigarette smoking during pregnancy contributes to some extent a variety of short-and long-term effects on the neurodevelopment of offspring [56].
The future research in this area should explore the adverse effects of maternal smoking on offspring neurodevelopment and address the intervention approaches that target women in lower social classes and the involvement of smoking partners with multi-component interventions [57,58]. The biochemical measures should also be taken into consideration to verify the information on maternal smoking habits [43,44]. In addition, broad epidemiological studies should be considered in future which can help to inform environmental, social, political, economic and geographical determinants of behaviours, and genetic determinants of risk.
6. ACKNOWLEDGEMENTS
The authors are highly thankful to Dr. Akif Ullah Khan, medical director of Ibn-Al-Nafees Medical Center, Peshawar-Pakistan for providing extensive help and support to acquire the specific data of maternal smoking women and their offspring attending that clinic. The authors also extend their appreciation to Pakistan Medical Research Council for providing linkage to major national health studies, digital and electronic databases for educational and research purposes. The authors have no conflicts of interest to declare.
REFERENCES
- Olds, D. (1997) Tobacco exposure and impaired development: A review of the evidence. Mental Retardation and Developmental Disabilities Research Reviews, 3, 257-269. doi:10.1002/(SICI)1098-2779(1997)3:3<257::AID-MRDD6>3.0.CO;2-M
- Royal College of Physicians (1992) Smoking and the young. Royal College of Physicians, London.
- Matthews, T.J. (2001) Smoking during pregnancy in the 1990’s. National Center for Health Statistics, Hyattsville.
- Mamun, A.A., Lawlor, D.A., Alati, R., et al. (2006) Does maternal smoking during pregnancy have a direct effect on future offspring obesity? Evidence from a prospective birth cohort study. American Journal of Epidemiology, 164, 317-325. doi:10.1093/aje/kwj209
- Power, C. and Jefferis, B.J.M.H. (2002) Fetal environment and subsequent obesity: A study of maternal smoking. International Journal of Epidemiology, 31, 413-419. doi:10.1093/ije/31.2.413
- Mullen, P., Richardson, M. and Quinn, V. (1997) Postpartum return to smoking: Who is at risk and when. American Journal of Health Promotion, 11, 323-330. doi:10.4278/0890-1171-11.5.323
- Gilman, S.E., Breslau, J., Subramanian, S.V., et al. (2008) Social factors, psychopathology, and maternal smoking during pregnancy. American Journal of Public Health, 98, 448-453. doi:10.2105/AJPH.2006.102772
- Kramer, M.S. (2003) The epidemiology of adverse pregnancy outcomes. An overview. Journal of Nutrition, 133, 1592S-1596S.
- Haustein, K.O. (1999) Cigarette smoking, nicotine and pregnancy. International Journal of Clinical Pharmacology and Therapeutics, 37, 417-427.
- Wakschlag, L.S. and Hans, S.L. (2002) Maternal smoking during pregnancy and conduct problems in high-risk youth: A developmental framework. Development and Psychopathology, 14, 351-369. doi:10.1017/S0954579402002092
- Thapar, A., Fowler, T., Rice, F., et al. (2003) Maternal smoking during pregnancy and attention deficit hyperactivity disorder symptoms in offspring. American Journal of Psychiatry, 160, 1985-1989. doi:10.1176/appi.ajp.160.11.1985
- Davey, S.G. (2008) Assessing intrauterine influences on offspring health outcomes: Can epidemiological studies yield robust findings? Basic & Clinical Pharmacology & Toxicology, 102, 245-256. doi:10.1111/j.1742-7843.2007.00191.x
- Chen, A., Pennell, M.L., Klebanoff, M.A., et al. (2006) Maternal smoking during pregnancy in relation to child overweight: Follow-up to age 8 years. International Journal of Epidemiology, 35, 121-130. doi:10.1093/ije/dyi218
- Linnnet, K.M., Dalsgaard, S., Obel, C., et al. (2003) Maternal lifestyle factors in pregnancy risk of attention deficit hyperactivity disorder and associated behaviour: Review of the current evidence. American Journal of Psychiatry, 160, 1028-1040.
- Knopik, V.S. (2009) Maternal smoking during pregnancy and child outcomes: Real or spurious effect? Developmental Neuropsychology, 34, 1-36. doi:10.1080/87565640802564366
- Orleans, C.T., Barker, D.C., Kaufman, N.J., et al. (2000) Helping pregnant smokers quit: Meeting the challenge in the next decade. Tobacco Control, 9, 6-11. doi:10.1136/tc.9.suppl_3.iii6
- Weinstein, N.D. (1993) Testing four competing theories of health-protective behaviour. Health Psychology, 12, 324-333. doi:10.1037/0278-6133.12.4.324
- Becker, M.H. (1974) The health belief model and personal health behaviour. Health Education Monographs, 2, 324-473.
- Maddux, J.E. and Rogers, R.W. (1983) Protection motivation and self efficiency: A revised theory of fear appeals and attitude change. Journal of Experimental Social Psychology, 19, 469-479. doi:10.1016/0022-1031(83)90023-9
- Fishbein, M. and Azjen, I. (1975) Belief, attitude, intention, and behaviour. An introduction to theory and research. Addison-Wesley, Reading.
- Lumley, J., Oliver, S. and Waters, E. (2003) Interventions for promoting smoking cessation during pregnancy (Cochrane Review). The Cochrane Library, Oxford Update Software.
- Fiore, M.C., Bailey, W.C., Cohen, S.J., et al. (2000) Treating tobacco use and dependence. Clinical practice guideline. US Department of Health and Human Services, Public Health Service, Rockville.
- Windsor, R.A., Woodby, L.L. and Miller, T.M. (2000) Effectiveness of agency for healthcare policy and research clinical practice guideline and patient education methods for pregnant smokers in medicaid maternity care. American Journal of obstetrics and Gynecology, 182, 68- 75. doi:10.1016/S0002-9378(00)70492-3
- Ory, M.G., Jordan, P.J. and Bazarre, T. (2002) The behavior change consortium: Setting the stage for a new century of health behavior-change research. Health Education Research, 17, 500-511. doi:10.1093/her/17.5.500
- Medical Research Council (2000) A framework for the development and evaluation of RCTs for complex interventions to improve health. Medical Research Council, London.
- Conrad, K.J., Randolph, F.L. and Kirby, M.W.J. (1999) Creating and using logic models: Four perspectives. Alcoholism Treatment Quarterly, 17, 17-31. doi:10.1300/J020v17n01_02
- Bartholomew, L.K., Parcel, G.S. and Kok, G. (2001) Intervention mapping: Designing theoryand evidencebased health promotion programs. Mountain View, Mayfield.
- Hardeman, W., Sutton, S. and Griffin, S. (2005) A causal modeling approach to the development of theory-based behaviour change programmes for trial evaluation. Health Education Research, 20, 676-687. doi:10.1093/her/cyh022
- Bauman, L.J., Stein, R.E.K. and Ireys, H. (1991) A framework for conceptualizing interventions. Sociological Practice Review, 2, 241-251.
- Campbell, M., Fitzpatrick, R., Haines, A. et al. (2000) Framework for design and evaluation of complex interventions to improve health. British Medical Journal, 321, 694-696. doi:10.1136/bmj.321.7262.694
- McLean, N., Griffin, S., Toney, K., et al. (2003) Family involvement in weight control, weight maintenance and weight loss interventions: A systematic review of randomized trials. International Journal of Obesity and Related Metabolic Disorders, 27, 987-1005. doi:10.1038/sj.ijo.0802383
- Azjen, I. (1991) The theory of planned behaviour. Organizational Behaviour and Human Decision Processes, 50, 179-211. doi:10.1016/0749-5978(91)90020-T
- Godin, G. and Kok, G.J. (1996) The theory of planned behavior: A review of its applications to health-related behaviors. American Journal of Health Promotion, 11, 87-98. doi:10.4278/0890-1171-11.2.87
- Sutton, S. (1998) Predicting and explaining intentions and behavior: how well are we doing? Journal of Applied Social Psychology, 28, 1317-1338. doi:10.1111/j.1559-1816.1998.tb01679.x
- Armitage, C.J. and Conner, M. (2001) Efficacy of the theory of planned behaviour: A meta-analytic review. British Journal of Social Psychology, 40, 471-499. doi:10.1348/014466601164939
- Ajzen, I. and Fishbein, M. (1980) Understanding attitudes and predicting social behavior. Prentice Hall, Englewood Cliffs.
- Ruggiero, L., Tsoh, J.Y., Everett, K., et al. (2000) The trans-theoretical model of smoking: Comparison of pregnant and non-pregnant smokers. Addictive Behaviour, 25, 239-251. doi:10.1016/S0306-4603(99)00029-5
- Prochaska, J.O. and DiClemente, C.C. (1982) Trans-theoretical therapy: Toward a more integrative model of change. Psychotherapy: Theory, Research & Practice, 19, 276-288. doi:10.1037/h0088437
- Hardeman, W., Johnston, M., Johnston, M., et al. (2002) Application of the theory of planned behaviour in behaviour change interventions: A systematic review. Psychology and Health, 17, 123-158. doi:10.1080/08870440290013644a
- Glasgow, R.E., Vogt, T.M. and Boles, S.M. (1999) Evaluating the public health impact of health promotion interventions: The RE-AIM framework. American Journal of Public Health, 89, 1322-1327. doi:10.2105/AJPH.89.9.1322
- Windsor, R.A., Woodby, L.L. and Miller, T.M. (2000) Effectiveness of agency for healthcare policy and research clinical practice guideline and patient education methods for pregnant smokers in Medicaid maternity care. American Journal of Obstetrics and Gynecology, 182, 68-75. doi:10.1016/S0002-9378(00)70492-3
- Prochaska, J.O., DiClemente, C.C. and Norcross, J.C. (1992) In search of how people change: Applications to addictive behaviours. American Psychological Association, 47, 1102-1114. doi:10.1037/0003-066X.47.9.1102
- Walsh, R.A., Redman, S. and Adamson, L. (1996) The accuracy of self-report of smoking status in pregnant women. Addictive Behaviours, 21, 675-679.
- Hartmann, K.E., Thorp Jr, J.M., Pahel-Short, L., et al. (1996) A randomized controlled trial of smoking cessation intervention in pregnancy in an academic clinic. Obstetrics and Gynecology, 84, 621-626. doi:10.1016/0029-7844(95)00492-0
- Pakistan Medical Research Council (1998) National health survey of Pakistan. Health profile of the people of Pakistan, 1990-1994. Pakistan Medical Research Council, Islamabad.
- King, T.K., Borrelli, B., Black, C., et al. (1997) Minority women and tobacco: Implications for smoking cessation interventions. Annals of Behavioral Medicine, 19, 301- 313. doi:10.1007/BF02892295
- Groner, J.A., Ahijevych, K., Grossman, L.K., et al. (2000) The impact of a brief intervention on maternal smoking behaviour. Pediatrics, 105, 267-271.
- Orlandi, M.A. (1987) Promoting health and preventing disease in health care settings: An analysis of barriers. Preventive Medicine, 16, 119-130. doi:10.1016/0091-7435(87)90011-9
- Stewart, M.J., Brosky, G., Gillis, A., et al. (1996) Disadvantaged women and smoking. Canadian Journal of Public Health, 87, 257-260.
- Eakin, E.G., Glasgow, R.E., Whitlock, E.P. and Smith, P. (1998) Reaching those most in need: Participation in a planned parenthood smoking cessation program. Annals of Behavioral Medicine, 20, 216-220. doi:10.1007/BF02884963
- Valbo, A. and Schioldborg, P. (1994) Smoking cessation in pregnancy: The effect of self-help manuals. Journal of Maternal-Fetal Investigation, 4, 167-170.
- Qureshi, A.A., Wajid, G., Shaikh, I.A., et al. (1992) Ethical considerations for human investigation in Mallick MD. Sample design for the National Health Survey of Pakistan. Pakistan Journal of Medical Research, 31, 289-290.
- Coleman, T. (2004) Special groups of smokers. BMJ, 328, 575. doi:10.1136/bmj.328.7439.575
- Paradis, G., Lambert, M., O’Loughlin, J., et al. (2004) Blood pressure and Adiposity in children and adolescents. Circulation, 110, 1832-1838. doi:10.1161/01.CIR.0000143100.31752.B7
- Apseloff, G., Ashton, H., Friedman, H., et al. (1994) The importance of measuring cotinine levels to identify smokers in clinical trials. Clinical Pharmacology and Therapeutics, 56, 460-462. doi:10.1038/clpt.1994.161
- Ernst, M., Moolchan, E.T. and Robinson, M.L.A. (2001) Behavioural and neural consequences of prenatal exposure to nicotine. Journal of the American Academy of Child and Adolescent Psychiatry, 40, 630-641. doi:10.1097/00004583-200106000-00007
- Floyd, R.L., Rimer, B.K., Giovino, G., et al. (1993) A review of smoking in pregnancy: Effects on pregnancy outcomes and cessation efforts. Annual Reviews of Public Health, 14, 379-411. doi:10.1146/annurev.pu.14.050193.002115
- Dolan-Mullen, P., Ramirez, G. and Groff, J.Y. (1994) A meta-analysis of randomized trials of prenatal smoking cessation interventions. American Journal of Obstetrics and Gynecology, 171, 1328-1334. doi:10.1016/0002-9378(94)90156-2
ABBREVIATIONS
BMI = Body Mass Index;
SBP = Systolic Blood Pressure;
MRC = Medical Research Council;
CDC = Centre for Disease Control;
NHSP = National Health Survey of Pakistan;
PMRC = Pakistan Medical Research Council;
SD = Standard Deviation;
TPB = Theory of Planned Behaviour.

