Journal of Surface Engineered Materials and Advanced Technology
Vol.3 No.1(2013), Article ID:27323,7 pages DOI:10.4236/jsemat.2013.31004
Preparation and Characterization of Clay (Maghnite-H)/Poly(3,4-Ethylenedioxythiophene) Composites
![]()
Laboratory of Polymers Chemistry, Department of Chemistry, Faculty of Sciences, University of Oran, Oran, Algeria.
Email: *rachidmeghabar@yahoo.fr
Received September 2nd, 2012; revised October 2nd, 2012; accepted October 8th, 2012
Keywords: Poly(3,4-Ethylenedioxythiophene); PEDOT; Clay; Maghnite-H; Montmorillonite
ABSTRACT
Composites of Maghnite-H, a Montmorillonite sheet silicate clay, exchanged with protons, and Poly(3,4-ethylenedioxythiophene) (PEDOT) were prepared by in situ chemical polymerization of the 3,4-ethylenedioxythiophene, without the use of solvent or oxidant. The effect of changing monomer/clay ratio was studied and the resultant composite structures were characterized by Inferred spectroscopy, 27Al and 13C Solid-State NMR spectroscopy, scanning electron microscopy and powder X-ray diffraction. All analyses are consistent with a structure were the polymer is (partially) intercalated into the clay structure, which in favourable cases lead to exfoliation. The presence of the clay in the polymer leads to a desired increase in thermal stability as witnessed by thermogravimetry.
1. Introduction
Polythiophene and its derivatives have attracted much interest over recent decades due to exceptional electrical properties and their environmental stability [1-6]. From this class of conducting polymers with conjugated π- bonds, especially poly 3,4-ethylenedioxythiophene (PEDOT) possess unique properties such as high environmental stability [7,8] and a small band gab (1.6 - 1.7 eV); a direct consequence of the electron donation properties of the ether functionality [9]. In addition, the electronic conductivity can reach as high as 300 S/cm after the polymer has been oxidized [10]. These characteristics have lead to several applications, such as antistatic paints [11,12], electrochromic devices [13,14], electrochemical capacitors [15], biosensors [16-18], polymer solar cells (PSCs) [19,20], polymer light-emitting diodes (PLEDs) [21,22] etc. Polymerisation of EDOT may be accomplished by either electrochemical or chemical oxidation. Both techniques lead to a material which is in his native state is insoluble and cannot be melted below the decomposition temperature [6,23]. This problem has been resolved by researchers at Bayer AG., Germany, by persulfate oxidative polymerization in a solution containing a polyelectrolyte (polystyrene sulfonic acid) [24]. More recently, aqueous solvated nanoparticles of PEDOT have been prepared using ammonium perdisulfate and the surfactant dodecylbenzene [25]. Another approach to polymer structures with nanometer dimensions is to form clay/polymer hybrids. These hybrids have found numerous applications due to their mechanical properties, their thermal stability and their reduced permeability to gases [26]. Recent examples include hybrids of clay and polyaniline [27,28], and clay and polypyrrole [29]. An especially elegant approach to these composites is the solvent-free direct reaction between the clay and the monomer, where the clay serves the double role of reactant/ catalyst for the polymerization and inorganic component of the final hybrid material. In this manner Biswas et al. [30,31] have prepared composites of Poly(N-vinylcarbazole) and polypyrrole by direct reaction with montmorillonite. Sadok et al. [32] have prepared polypyrrole composites by using a clay montmorillonite naturally rich in ions or synthetically doped with ions (Saponite substituted with Fe3+ in tetrahedral sites). Surprising little clay composite studies have been performed on polymers which contain the thiophene functionality. A notable exception is Ballav et al. [33] who have succeeded in preparing composites polythiophene/montmorillonite, by solvent-free direct reaction between the monomer and montmorillonite-Na+. The composites produced showed that the polythiophene was intercalated in the interlayer space and that the resultant material showed a modest but non-zero electronic conductivity on the order of 10−4 S/cm.
In this article, we examine the use of Maghnite-H+ (MagH), a Montmorillonite sheet silicate, in the synthesis of composites with PEDOT. The choice of MagH is based on related work where it acts as a catalyst of cationic polymerization of vinyl [34-37] and heterocylic [38-42] monomers. Here the synthesis takes place in a one pot reaction between the clay and the monomer without the use of solvent or oxidant. Specifically, we examine the effect of the relative ratios between the monomer and the clay on the intercalation and possible exfoliation of the clay structure.
2. Experimental
2.1. Materials
MagH was prepared using the method of Belbachir et al. [34]. EDOT was obtained from Bayer AG. (Leverkusen, Germany) and used without further purification.
2.2. Synthesis
All reactions were performed using the same methodology described below, with however, a variation of time, for the 10% by weight of MagH/monomer 24 h reaction and for 50% 96 h reaction. The reaction times and relative concentration were altered according to Table 1.
Hybrides from 10% by weight of MagH/monomer reaction mixtures.
In a round bottom flask fitted with a condenser was mixed 1 g (0.007 mol) EDOT and 0.5 g MagH previously dried at 100˚C overnight. The mixture was heated to 90˚C, under constant stirring using a magnetic stir bar. Upon the addition of the MagH a colour change was witnessed from clear over red to brown before finally becoming dark green. After 24 h of heating a back solid was formed, and the suspension was cooled to room temperature. Residual unreacted monomer and shorter solvable oligomeres were removed by washing with dichloromethane. The residual black solid (the hybrid material) was dried at 60˚C overnight to remove any solvent traces. (Calculate a real yield based on incorporated polymer relative to the initial amount). The amount of incorporated polymer was determined to 21 m/m-% based on the mass gain.
2.3. Characterization
Infrared spectra (400 - 4000 cm−1) were collected from samples ground with dry KBr and pressed into pellets. Spectral peaks around 2350 cm−1 stem from residual CO2 in the beam path. 13C and 27Al Solid state NMR was preformed using a BRUKER ASX 400 CP-MAS. Thermogravometric analysis (TGA) was performed on a Standon Redcroft STA780 thermal analyser and in air at a heating rate of 5 deg/min within the temperature range of 25˚C - 1000˚C. The powder X-ray diffraction (XRD) patterns of the samples were recorded using Philips XRD X’PERT PRO II diffractometer (2 goniometers, Montpellier, French) using Cu-Kα radiation and (λ = 1.5404 Å) produced at 40 kV and 40 mA. Scanning electron microscopy micrographs where collected on a Hitachi S-2600N with a 25 kV working potential, a working distance of 7.0 - 7.4 mm and a SE detector.
3. Results and Discussion
3.1. Characterization by Infrared Spectra (IR)
Figure 1 shows, respectively, the spectra IR of MagH, the composites MagH-PEDOT (1): B and MagH-PEDOT (2): C and of polymer PEDOT: D Vibrations around 1337 and 1518 cm−1. (B, C, D) are due respectively to the elongation of C-C and C=C bonds of the thiophene cycle, these peaks do not appear in the IR spectrum of MagH. Peaks located at 980, 833 and 794 cm−1 are due to the vibrations of the CS bond in thiophene, and signals around 1080 cm−1 indicate C-O-C bond of the ethylenedioxy group. The weak peaks from 2854 to 2925 cm−1 show the CH elongations of ethylene related to the thiophene cycle. All IR peaks found in the composites, compared with the peaks of MagH and PEDOT, confirm the presence of PEDOT in the composites. Kvarnstrom and et al. [9] and Jeong et al. [25] have obtained same results in the synthesis of the PEDOT.
3.2. Characterization by 13C Solid State NMR
The solid 13C NMR spectrum of the composite MagHPEDOT (1) shown in Figure 2, shows three peaks characteristic of PEDOT, a peak at 110 ppm of C1 carbon related to the sulphur atom, a second at 140 ppm of sp2 carbon (C2), and a third at 65.87 ppm of the sp3 carbon (C3) of the cyclic ethylene related to thiophene. The
Table 1. Operating conditions of the synthesis of the MagH-PEDOT hybrids.
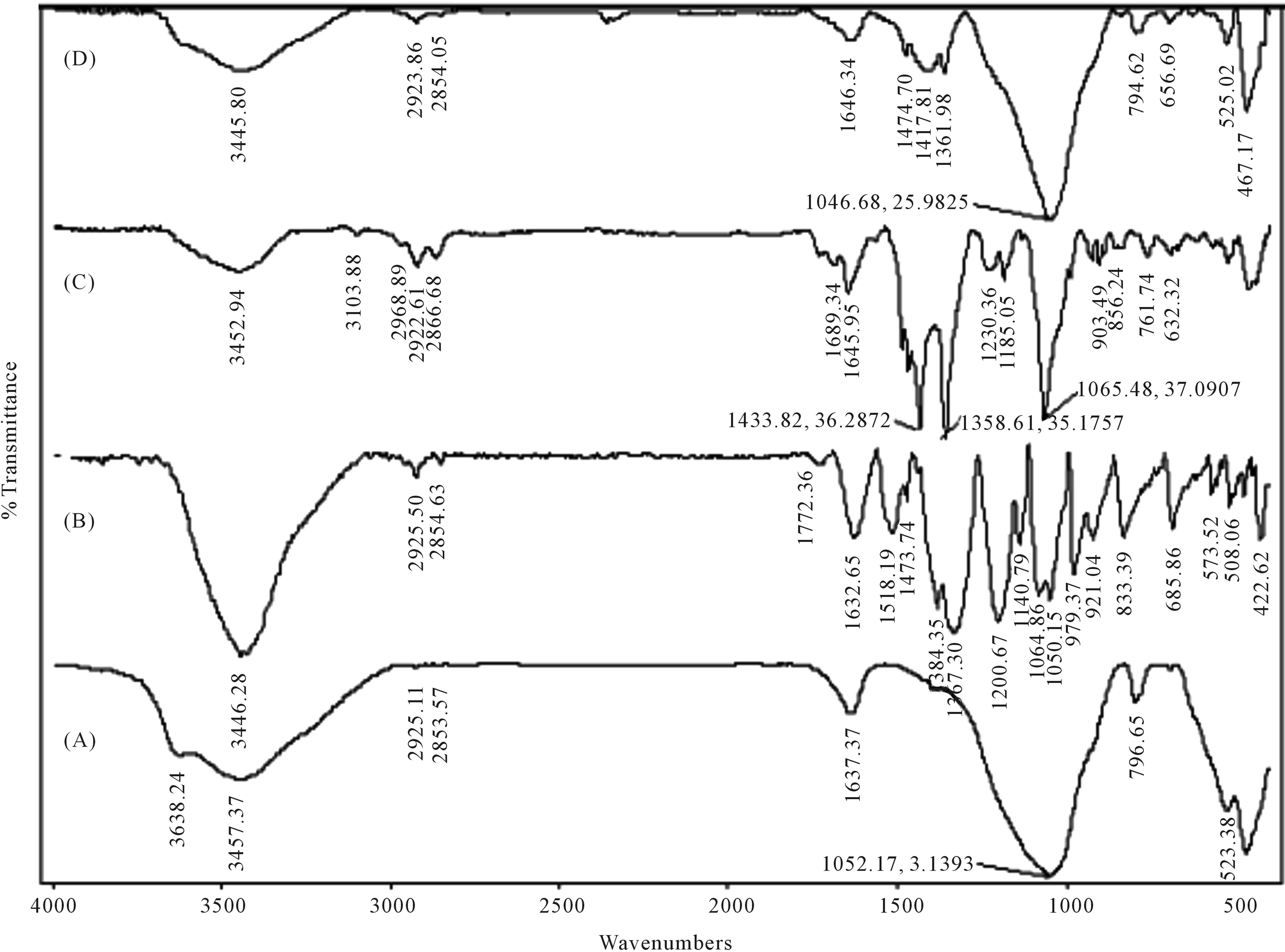
Figure 1. IR spectrums of (A) Maghnite-H+ (MagH); (B) PEDOT, prepared with FeCl3; (C) Hybrid MagH-PEDOT (10% MagH, 96 h, 90˚C); (D) Hybrid MagH-PEDOT (1) (50% MagH, 24 h, 90˚C).
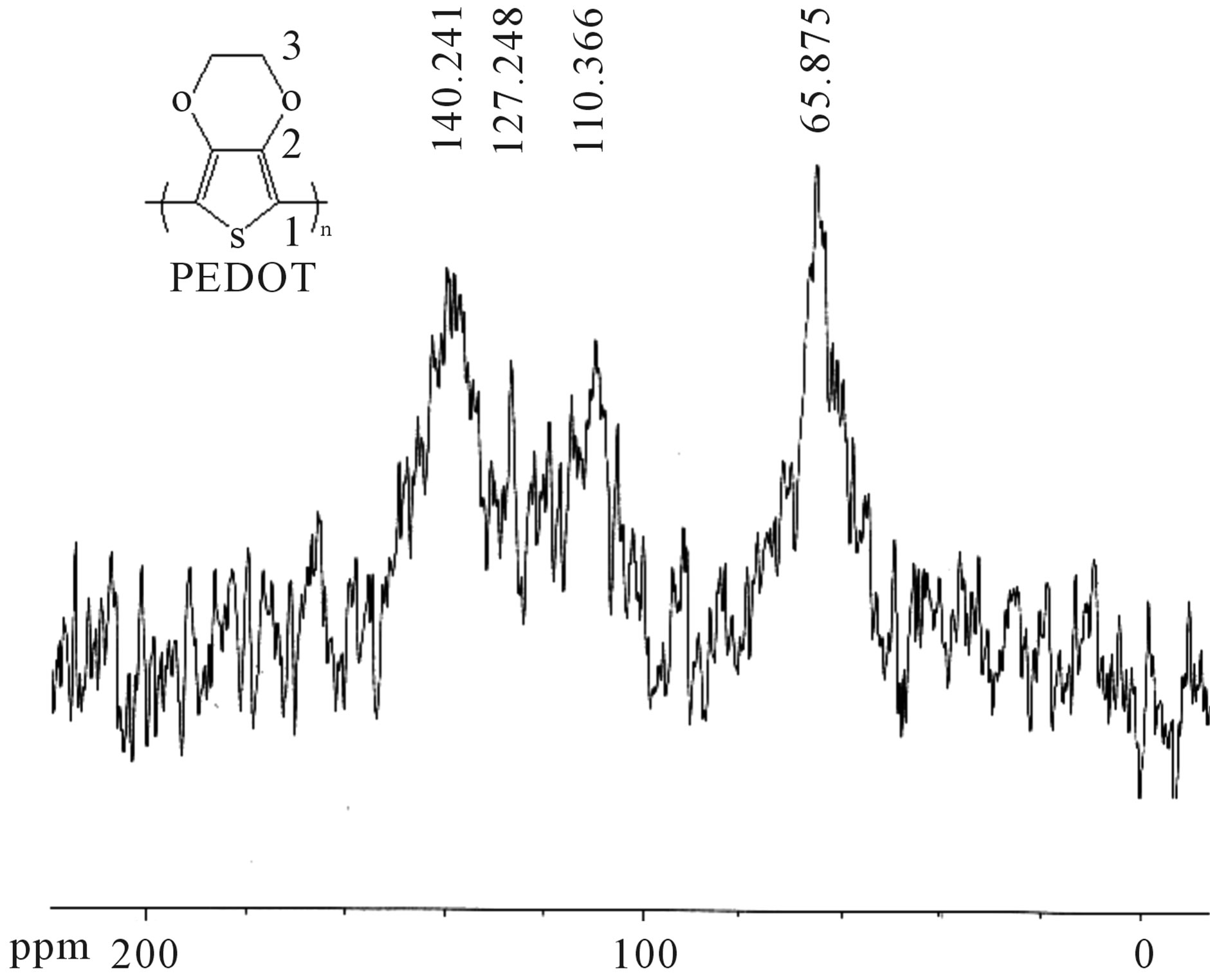
Figure 2. 13C solid state NMR spectrum of hybrid MagHPEDOT (1), obtained with 50% MagH, 24 h, 90˚C.
structure obtained is similar to that found by Meng et al. [43], which confirms the presence of PEDOT in the MagH-PEDOT composite.
3.3. Characterization by X-Ray Diffraction (XRD)
The reticular distances d001 of interfoliaceous space, were calculated from X-ray diffractograms of the hybrids and Monmorillonite (MagH), by using Kα radiation of the copper anode with λ (Cu) = 1.5404 Å, and in the MagHPEDOT (b) hybrid. The characteristic montmorillonite peak reaches 13.26 Å for 2q = 6.65 (Figure 3(A)), the montmorillonite peak in MagH appears to an interfoliaceous distance of 12.87 Å (Figure 3(A)). The Figure 3(B) shows the displacement of the montmorillonite peak in the composite MagH-PEDOT (b) towards the weak angles, compared with that of MagH. Thus, in the case of the MagH-PEDOT (b) composite, the intercalation of PEDOT between the layers of montmorillonite (MagH) has taken place. The same results were observed in the case of the composites of PNVC-MMT with an interfoliaceous space d001 = 14.6 per 2q = 6.9, which indicates the intercalation of polymer PNVC in montmorillonite MMT [24]. On the other hand, the diffractogram of the MagH-PEDOT (c) hybrid (Figure 3(A)), presents an ab-
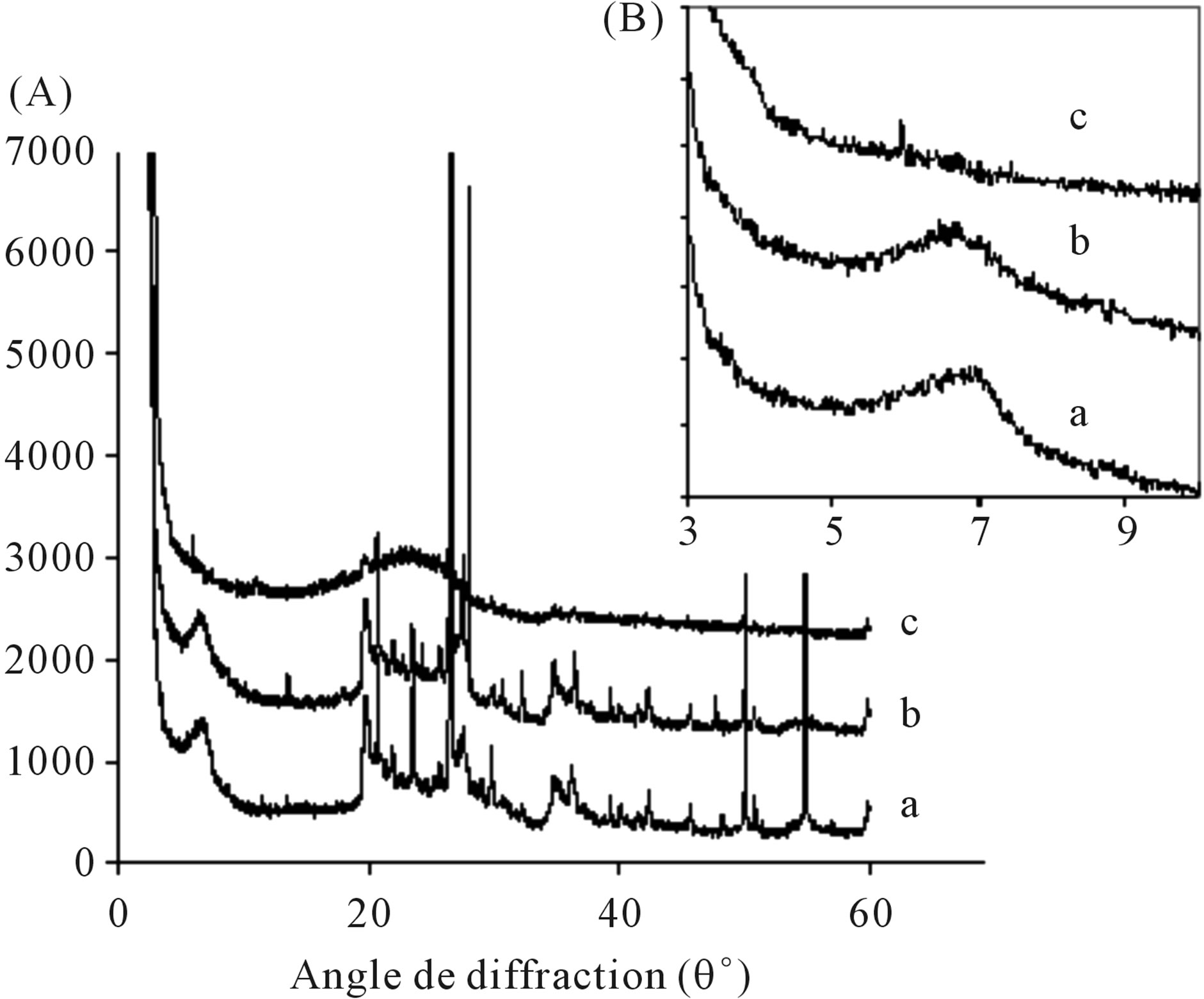
Figure 3. (A) XRD patterns of MagH (a) and hybrids MagH-PEDOT (b, c) obtained with, (b) 50% MagH, 24 h, 90˚C and (c) 10% MagH, 96 h, 90˚C; (B) XRD enlargement in area of the diffraction angle (q˚): 3 - 10.
sence of the characteristic peak of montmorillonite, and also of the amorphous zones, which results in a broad peak, and an partially exfoliation of montmorillonite in the MagH-PEDOT (c) composite.
3.4. Determination of the Composites Morphology
The comparison of the MagH SEM morphology (Figure 4(A)) with the first hybrid MagH-PEDOT (Figure 4(B)) shows a montmorillonite structure more organized in small particles. In the sample MagH-PEDOT hybrid (Figures 4(C) and (D)), the observation of the hybrid at 20 μm (Figure 4(C)), reveals a formation of montmorillonite plate separated, that is a partial exfoliation, this result is in the same way than results obtained by DRX analysis, the same sample observed at 10 μm (Figure 4(D)), shows a rougher surface and a covering of the montmorillonite surface by the PEDOT.
3.5. Characterization by 27Al Solid State NMR
The 27Al NMR data (Figure 5) show that the octahedral and tetrahedral Aluminium peaks are preserved in the MagH-PEDOT (1) composite, compared to the octahedral and tetrahedral peaks of MagH (Figure 6), thus PEDOT does not form a bond with aluminium, or is not in the aluminium environment.
3.6. Thermal Stability of Composites (Thermogravimetric Analysis: TGA)
The thermograms of Figure 7 show that MagH brings a greater stability to the composites, more the product is rich in polymer more it is degraded easily. Thus, the sta-

Figure 4. The scanning electron micrographs images of MagH (A) hybrids MagH-PEDOT (B) obtained with 50% MagH, 24 h, 90˚C; and MagH-PEDOT (C), (D) obtained with 10% MagH, 96 h, 90˚C, with a work distance: 7.0 - 7.4 mm and an amplification: 1.5 - 6.0 k.
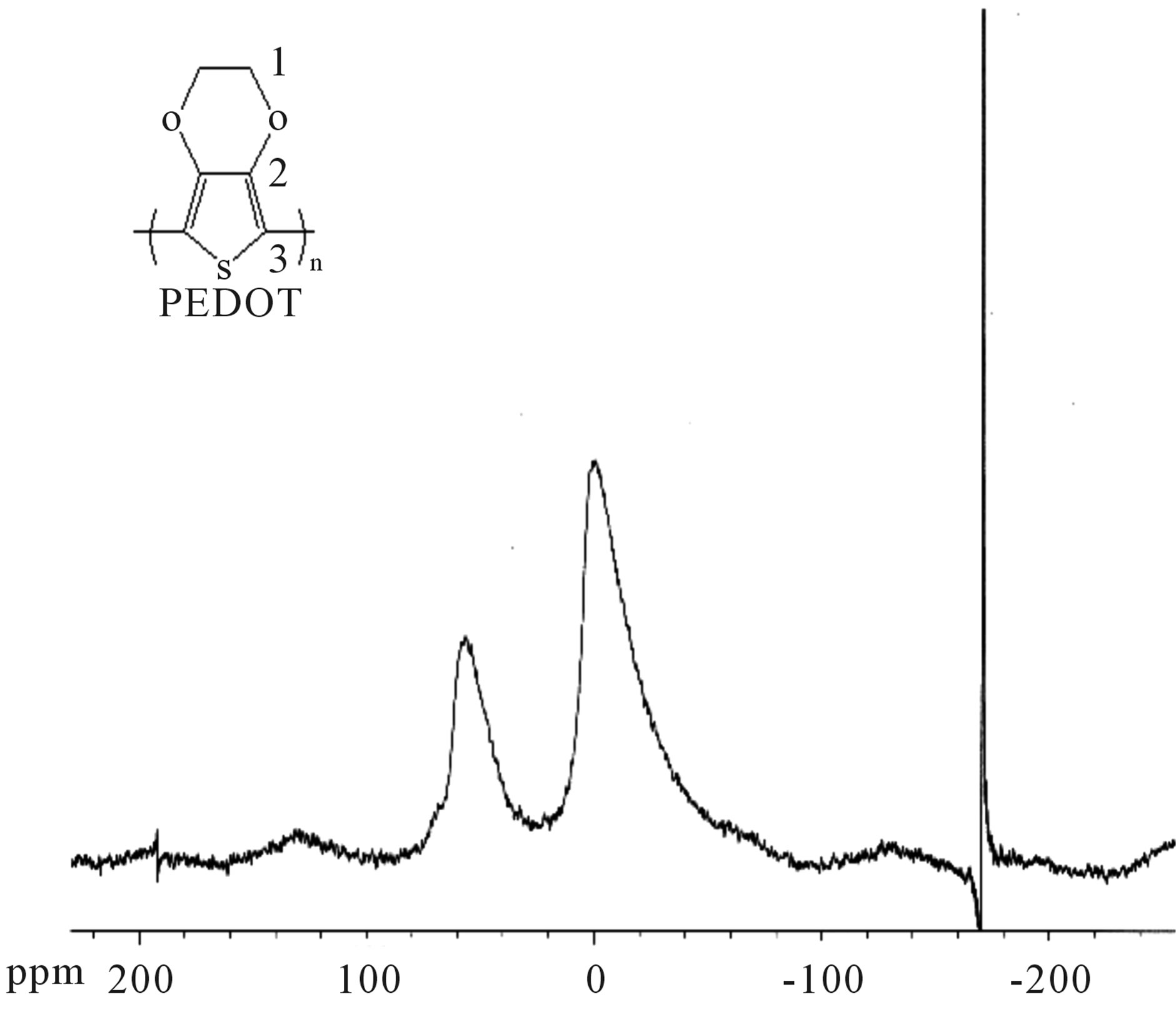
Figure 5. Spectrum 27Al solid state NMR of hybrid MagHPEDOT (1): obtained with 50% MagH, 24 h, 90˚C.
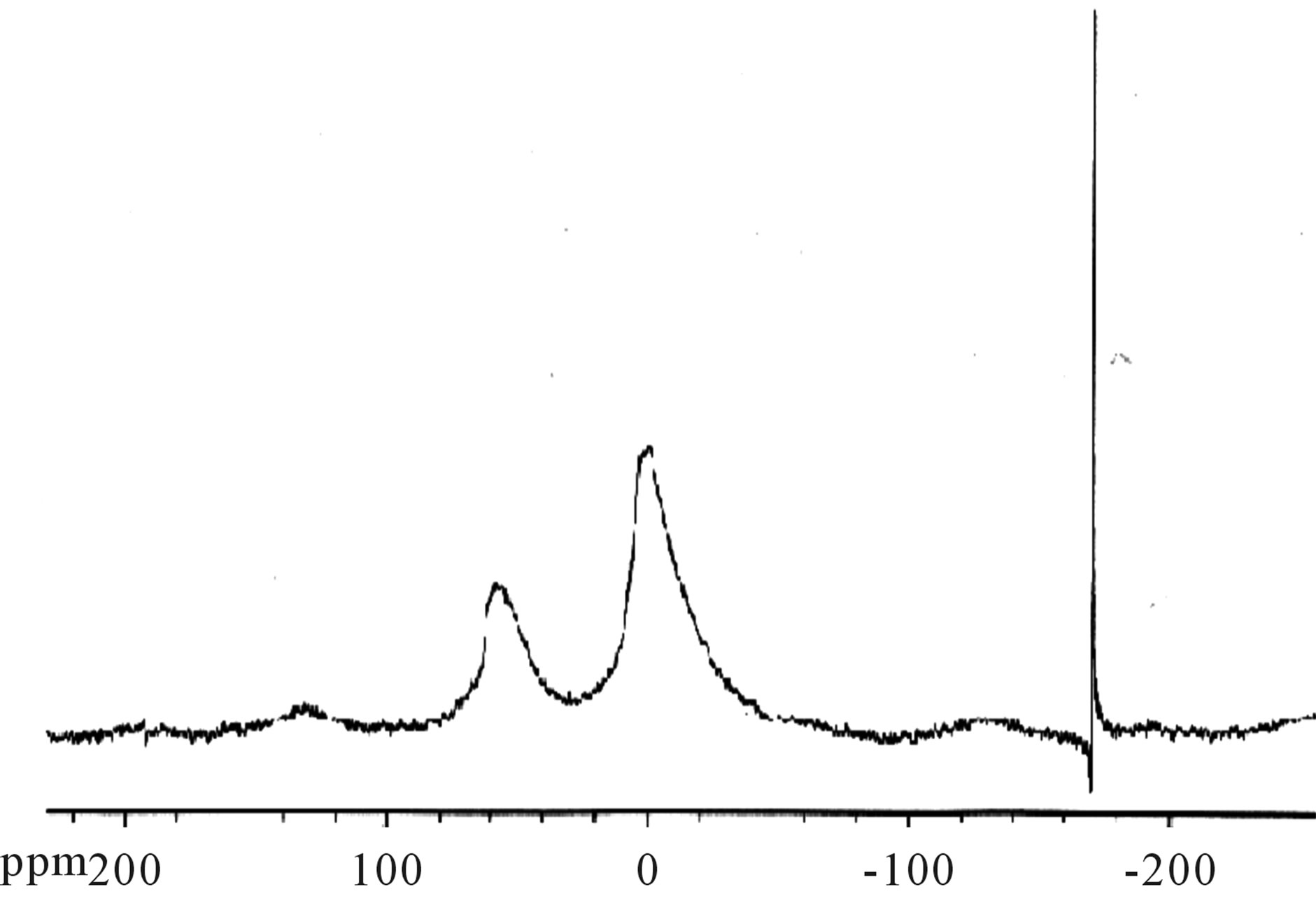
Figure 6. Spectrum 27Al solid state NMR of MagH.
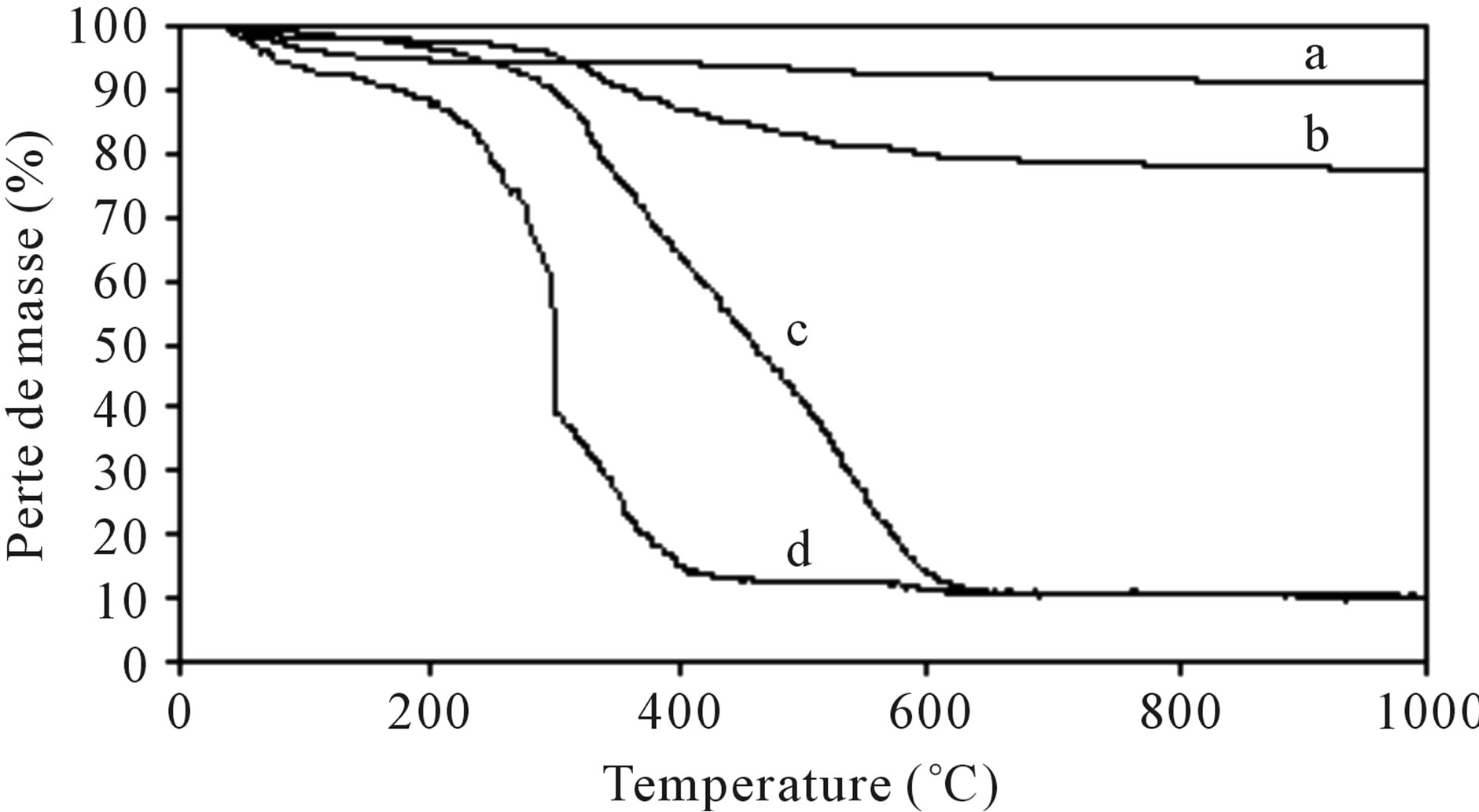
Figure 7. TGA curves of: (a) (MagH); (b, c) MagH-PEDOT; (b) obtained with 50% MagH, 24 h, 90˚C; (c) obtained with 10% MagH, 96 h, 90˚C; (d) PEDOT.
bility order obtained for all materials is as follows: MagHPEDOT (b) > MagH-PEDOT (c) > PEDOT. Similar results were observed for the montmorillonite/polythiophene composites (MMT-PTP) [27]. The percentage of the PEDOT grafted, determined from thermograms, is 19% in the MagH-PEDOT composite (Figure 7(b)) whereas it is 89% in the MagH-PEDOT composite (Figure 7(c)).
4. Conclusion
The in situ polymerization of 3,4-ethylenedioxythiophene (EDOT) by MagH, led to the synthesis of the MagH-PEDOT hybrids. The DRX study of MagH-PEDOT hybrids shows an intercalation of PEDOT between the MagH layers and a partial exfoliation in certain hybrids rich in PEDOT. The morphology study by SEM of hybrids confirmed an organization of certain particles, and in other cases a separation in plates made up of montmorillonite layers. This confirms partial exfoliation of montmorillonite in the hybrids and formation of the composite. The 27Al solid state NMR shows that the polymer is not in the environment of aluminium. The thermal analysis of hybrids and montmorillonite shows a high thermal stability of composites and that it is possible to quantify intercalated polymer.
5. Acknowledgements
Authors are grateful to Mr. Yannig Nedellec, engineer CNRS of the Molecular aggregates and organic Materials laboratory (LAMMI) in Montpellier (French) and Mr. A Addou, engineer of the Chemistry Laboratory of Polymers (LCP) for IR, NMR, XRD and all analysis.
REFERENCES
- N. Hari-Singh, “Handbook of Organic Conductive Molecules and Polymers,” John Wiley & Sons, Ltd., New York, 1997.
- G. Tourrillan, “Polythiophene and Its Derivatives,” In: T. A. Skotheim, Ed., Handbook of Conducting Polymers, Marcel Dekker, New York, 1986, p. 293.
- A. Kossmehl, “Semi-Conducting and Conducting Polymers with Aromatic and Heteroaromatic Units,” In: T. A. Skotheim, Ed., Handbook of Conducting Polymers, Marcel Dekker, New York, 1986, p. 351.
- G. Schopf and G. Kossmehl, “Polythiophenes—Electrically Conducting Polymers,” Springer-Verlag, Berlin, 1997.
- D. Kumar and R. C. Sharma, “Advances in Conductive Polymers,” European Polymer Journal, Vol. 34, No. 8, 1998, pp. 1053-1060. doi:10.1016/S0014-3057(97)00204-8
- L. Groenendaal, F. Jonas, D. Freitag, H. Pielartzik and J. R. Reynolds, “Poly(3,4-Ethylenedioxythiophene) and Its Derivatives: Past, Present, and Future,” Advanced Materials, Vol. 12, No. 7, 2000, pp. 481-494. doi:10.1002/(SICI)1521-4095(200004)12:7<481::AID-ADMA481>3.0.CO;2-C
- H. Yamato, M. Ohwa and W. Wernet, “Stability of Polypyrrole and Poly(3,4-Ethylenedioxythiophene) for Biosensor Application,” Journal of Electroanalytical Chemistry, Vol. 397, No. 1-2, 1995, pp.163-170.
- I. Winter, C. Reese, J. Hormes, G. Heywang and F. Jonas, “The Thermal Ageing of Poly(3,4-Ethylenedioxythiophene). An Investigation by X-Ray Absorption and XRay Photoelectron Spectroscopy,” Chemical Physics, Vol. 194, No. 1, 1995, pp. 207-213. doi:10.1016/0301-0104(95)00026-K
- C. Kvarnstrom, H. Neugebauer, S. Blomquiste, H. J. Ahomen, J. Kankare and A. Ivaska, “In Situ SpectroElectrochemical Characterization of Poly(3,4-Ethylenedioxythiophene),” Electrochimica Acta, Vol. 44, No. 16, 1999, pp. 2739-2750. doi:10.1016/S0013-4686(98)00405-8
- A. N. Aleshin, R. Hiebooms and A. J. Heeger, “Metallic Conductivity of Highly Doped Poly(3,4-Ethylenedioxythiophene),” Synthetic Metals, Vol. 101, No. 1-3, 1999, pp. 369-370. doi:10.1016/S0379-6779(98)00758-9
- F. Jonas and L. Schrader, “Conductive Modifications of Polymers with Poly-Pyrroles and Polythiophenes,” Synthetic Metals, Vol. 41, No. 3, 1991, pp. 831-836.
- F. Jonas and J. T. Morrisson, “3,4-Polyethylenedioxy Thiophene (PEDT): Conductive Coatings Technical Applications and Properties,” Synthetic Metals, Vol. 85, No. 1-3, 1997, pp. 1397-1398.
- Q. Pei, G. Zucarello, M. Ahlskog and O. Inganäs, “Electrochromic and Highly Stable Poly(3,4-Ethylenedioxythiophene) Switches between Opaque Blue-Black and Transparent Sky Blue,” Polymer, Vol. 35, No. 7, 1994, pp. 1347-1351. doi:10.1016/0032-3861(94)90332-8
- A. Kumar, D. M. Welsh, M. C. Morvant, F. Piroux, K. A. Abboud and J. R. Reynolds, “Conducting Poly(3,4-Alkylenedioxythiophene) Derivatives as Fast Electrochromics with High-Contrast Ratios,” Chemistry of Materials, Vol. 10, No. 3, 1998, pp. 896-902. doi:10.1021/cm9706614
- J. C. Carlberg and O. Inganäs, “Poly(3,4-Ethylenedioxythiophene) as Electrode Material in Electrochemical Capacitors,” Journal of the Electrochemical Society, Vol. 144, No. 4, 1997, pp. L61-L64. doi:10.1149/1.1837553
- N. Rozlosnik, “New Directions in Medical Biosensors Employing Poly(3,4-Ethylenedioxy Thiophene) DerivativeBased Electrodes,” Analytical and Bioanalytical Chemistry, Vol. 395, No. 3, 2009, pp. 637-645. doi:10.1007/s00216-009-2981-8
- R. D. McCullough, P. C. Ewbank and R. S. Loewe, “SelfAssembly and Disassembly of Regioregular, Water Soluble Polythiophenes: Chemoselective Ionchromatic Sensing in Water,” Journal of the American Chemical Society, Vol. 119, No. 3, 1997, pp. 633-634. doi:10.1021/ja963713j
- F. Le Floch, H. A. Ho, P. Harding-Lepage, M. Bedard, R. Neagu-Plesu and M. Leclerc, “Ferrocene-Functionalized Cationic Polythiophene for the Label-Free Electrochemical Detection of DNA,” Advanced Materials, Vol. 17, No. 10, 2005, pp. 1251-1254. doi:10.1002/adma.200401474
- C. J. Brabec, N. S. Sariciftci and J. C. Hummelen, “Plastic Solar Cells,” Advanced Functional Materials, Vol. 11, No. 1, 2001, pp. 15-26. doi:10.1002/1616-3028(200102)11:1<15::AID-ADFM15>3.0.CO;2-A
- K. M. Coakley and M. D. McGehee, “Conjugated Polymer Photovoltaic Cells,” Chemistry of Materials, Vol. 16, No. 23, 2004, pp. 4533-4542. doi:10.1021/cm049654n
- L. Akcelrud, “Electroluminescent Polymers,” Progress in Polymer Science, Vol. 28, No. 6, 2003, pp. 875-962. doi:10.1016/S0079-6700(02)00140-5
- I. F. Perepichka, D. F. Perepichka, H. Meng and F. Wudl, “Light-Emitting Polythiophenes,” Advanced Materials, Vol. 17, No. 19, 2005, pp. 2281-2305. doi:10.1002/adma.200500461
- F. Jonas, W. Krafft and B. Muys, “Poly(3, 4-Ethylenedioxythiophene): Conductive Coatings, Technical Applications and Properties,” Macromolecular Symposia, Vol. 100, No. 1, 1995, pp. 169-173. doi:10.1002/masy.19951000128
- B. Sankaran and J. R. Reynolds, “High-Contrast Electrochromic Polymers from Alkyl-Derivatized Poly(3,4-Ethylenedioxy Thiophenes),” Macromolecules, Vol. 30, No. 9, 1997, pp. 2582-2588. doi:10.1021/ma961607w
- W. C. Jeong, G. H. Moon, Y. Sook, G. O. Seong and S. I. Seung, “Poly(3,4-Ethylenedioxythiophene) Nanoparticles Prepared in Aqueous DBSA Solutions,” Synthetic Metals, Vol. 141, No. 3, 2004, pp. 293-299.
- S. K. Lim, J. W. Kim, I. Chim, Y. K. Kwon and H. J. Choi, “Preparation and Interaction Characteristics of Organically Modified Montmorillonite Nanocomposite with Miscible Polymer Blend of Poly(Ethylene Oxide) and Poly(Methyl Methacrylate),” Chemistry of Materials, Vol. 14, No. 5, 2002, pp. 1989-1994. doi:10.1021/cm010498j
- N. Boutaleb, A. Benyoucef, H-J. Salavagione, M. Belbachir and E. Morallón, “Electrochemical Behaviour of Conducting Polymers Obtained into Clay-Catalyst Layers. An in Situ Raman Spectroscopy Study,” European Polymer Journal, Vol. 42, No. 4, 2006, pp. 733-739. doi:10.1016/j.eurpolymj.2005.10.012
- J. W. Kim, S. G. Kim, H. J. Choi, M. S. Suh, M. J. Shin and M. J. Jhon, “Synthesis and Electrorheological Characterization of Polyaniline and Na+-Montmorillonite Clay Nanocomposite,” International Journal of Modern Physics B, Vol. 15, No. 06n07, 2001, pp. 657-664.
- J.W. Kim, F. Liw, H. J. Choi, S. H. Hong and J. Joo, “Intercalated Polypyrrole/Na+-Montmorillonite Nano-Composite via an Inverted Emulsion Pathway Method,” Polymer, Vol. 44, No. 1, 2003, pp. 289-293. doi:10.1016/S0032-3861(02)00749-8
- M. Biswas and S. S. Ray, “Preparation and Evaluation of Composites from Montmorillonite and Some Heterocyclic Polymers. 1: Poly(N-Vinylcarbazole)-Montmorillonite Nanocomposite System,” Polymer, Vol. 39, No. 25, 1998, pp. 6423-6428. doi:10.1016/S0032-3861(97)10366-4
- S. S. Ray and M. Biswas, “Preparation and Evaluation of Composites from Montmorillonite and Some Heterocyclic Polymers: 3. a Water Dispersible Nanocomposite from Pyrrole-Montmorillonite Polymerization System,” Materials Research Bulletin, Vol. 34, No. 8, 1999, pp. 1187- 1194.
- L. Sadok, A. Pilar and R. H. Eduardo, “Influence of Iron in the Formation of Conductive Polypyrrole-Clay Nanocomposites,” Applied Clay Science, Vol. 28, No. 1-4, 2005, pp. 183-198.
- N. Ballav and M. Biswas, “A Conducting Nanocomposite via Intercalative Polymerisation of Thiophene in Montmorillonite Clay,” Synthetic Metals, Vol. 142, No. 1-3, 2004, pp. 309-315. doi:10.1016/j.synthmet.2003.08.004
- M. Belbachir and A. Bensaoula, “Composition and Method for Catalysis Using Bentonites,” US Patent No. 7094823, 2006.
- A. Harrane, R. Meghabar and M. Belbachir, “A Protons Exchanged Montmorillonite Clay as an Efficient Catalyst for the Reaction of Isobutylene Polymerization,” International Journal of Molecular Science, Vol. 3, No. 7, 2002, pp. 790-800. doi:10.3390/i3070790
- R. Meghabar, A. Megherbi and M. Belbachir, “MaghniteH+, an Ecocatalyst for Cationic Polymerization of N-Vinyl- 2-Pyrrolidone,” Polymer, Vol. 44, No. 15, 2003, pp. 4097- 4100. doi:10.1016/S0032-3861(03)00400-2
- M. Chabani, A. Yahiaoui, A. Hachemaoui and M. Belbachir, “New Approach for the Polymerization of 2-Chloroethyl Vinyl Ether Using a Maghnite Clay as Eco-Catalyst,” Journal of Applied Polymer Science, Vol. 122, No. 3, 2011, pp. 1800-1806. doi:10.1002/app.34242
- A. Yahioui, M. Belbachir and A. Hachmaoui, “An Acid Exchanged Montmorillonite Clay-Catalyzed Synthesis of Polyepichlorhydrin,” International Journal of Molecular Science, Vol. 4, No. 10, 2003, pp. 548-561. doi:10.3390/i4100548
- A. Harrane, K. Oussadi, M. E. Belaoudj, R. Meghabar and M. Belbachir, “Cationic Ring Opening Polymerization of Glycolide Catalysed by a Montmorillonite Clay Catalyst,” Journal of Polymer Research, Vol. 12, No. 5, 2005, pp. 361-365.
- A. Harrane, R. Meghabar and M. Belbachir, “Polymerization of Epsilon-Caprolactone Using a Montmorillonite Clay as Catalyst,” Designed Monomers and Polymers, Vol. 8, No. 1, 2005, pp. 11-24. doi:10.1163/1568555053084203
- A. Harrane, R. Meghabar and M. Belbachir, “In Situ Polymerization of Caprolactone Catalysed by MaghniteTOA to Produce Poly(-Caprolactone)/Montmorillonite Nanocomposites,” Designed Monomers and Polymers, Vol. 9, No. 2, 2006, pp. 181-191. doi:10.1163/156855506776382673
- K. Oussadi, V. Montembault, M. Belbachir and L. Fontaine, “Ring-Opening Bulk Polymerization of Fiveand Six-Membered Cyclic Phosphonates Using Maghnite, a Nontoxic Proton Exchanged Montmorillonite Clay,” Journal of Applied Polymer Science, Vol. 122, No. 2, 2011, pp. 891-897. doi:10.1002/app.34193
- H. Meng, D. F. Perpichka and F. Wudl, “Facile SolidState Synthesis of Highly Conducting Poly(Ethylenedioxythiophene),” Angewandte Chemie International Edition, Vol. 42, No. 6, 2003, pp. 658-661. doi:10.1002/anie.200390181
NOTES
*Corresponding author.

