 Vol.2, No.8, 473-478 (2013) Case Reports in Clinical Medicine http://dx.doi.org/10.4236/crcm.2013.28125 Surgical strategy in case with co-existence of malignant oligodendroglioma and arteriovenous malformation: A case report Hirohito Yano1*, Noriyuki Nakayama1, Naoyuki Ohe1, Toshinori Takagi1, Jun Shinoda2, Toru Iwama1 1Department of Neurosurgery, Gifu University Graduate School of Medicine, Gifu, Japan; *Corresponding Author: hirohito@gifu-u.ac.jp 2Chubu Medical Center for Prolonged Traumatic Brain Dysfunction, Department of Neurosurgery, Kizawa Memorial Hospital, Minokamo, Japan Received 26 September 2013; revised 20 October 2013; accepted 3 November 2013 Copyright © 2013 Hirohito Yano et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT A brain tumor associated with an arteriovenous malformation (AVM) is very rare. A 42-year-old female presented with two separate lesions in her right frontal lobe on MRI. An angiogram di- agnosed one of the lesions as an AVM. The second lesion appeared to be a tumor. Tumor removal was difficult due to bleeding from the nearby AVM, necessitating removal of the AVM and allowing complete excision of the tumor. Histopathological analysis revealed the tumor was an anaplastic o ligodendroglioma. There was no recurrence of the tumor 5 year after comple- tion of therapy. We discuss the operative strat- egy in case of synchronous diseases and pro- vide a review of the literature. Keywords: Oligodendroglioma; Arteriovenous Malformation; Co-Existence 1. INTRODUCTION The association of a brain tumor and an arteriovenous malformation (AVM) is rare. While there have been a number of reports, few of these have reported the details of the operative strategy, particularly the approaches used based on the preoperative information of both lesions. We describe our chosen surgical strategy in a case with two such synchronous lesions and provide a review of the literature. 2. CASE REPORT A 42-year-old previously healthy female presented with complaints of headache and vomiting. Her level of consciousness was normal and she had no neurologic deficits. Magnetic resonance images (MRI) on admission revealed a 7 cm in diameter mass lesion that showed low signal intensity on the T1 weighted images (WI) and high signal intensity on the T2 WI in the right frontal lobe. The lesion demonstrated heterogeneous enhance- ment by gadolinium-diethylenetriamine penta-acetic acid (Gd-DTPA) adjacent to the Rolandic vein (Figure 1(A)). The T2 WI also demonstrated a second 3 cm in diameter lesion with multiple flow void signs in the right inferior frontal gyrus (Figure 1(B)). An angiogram showed an arteriovenous malformation (AVM) that was predominantly fed by the middle cerebral arteries and drained through the superficial frontal ascending vein and the Rolandic vein (Figures 1(C) and (D)). Spetzler-Martin Grade was 1 point. The angiogram also demonstrated an avascular region in the area of the for- mer lesion. A plain computed tomography scan showed partial calcification, and a dynamic study of the me- thionine positron emission tomography (PET) scan pre- sented a reduction of accumulation in the longitudinal course in the former lesion. The results of dynamic PET study indicated oligodendroglial tumor according to our study previously reported [1]. Comprehensively, these radiological findings led to a diagnosis of malignant oli- godendroglial tumor associated with AVM. The patient’s headache was likely caused by the large tumor, and so we planed to resect the oligodendroglioma first, followed by the AVM. For the operation, we performed a large osteoplastic frontotemporal craniotomy beyond the mid-line including both lesions, with the goal being to remove the oligodendroglioma and AVM consecutively. During this procedure, severe bleeding occurred from the Copyright © 2013 SciRes. OPEN ACCESS 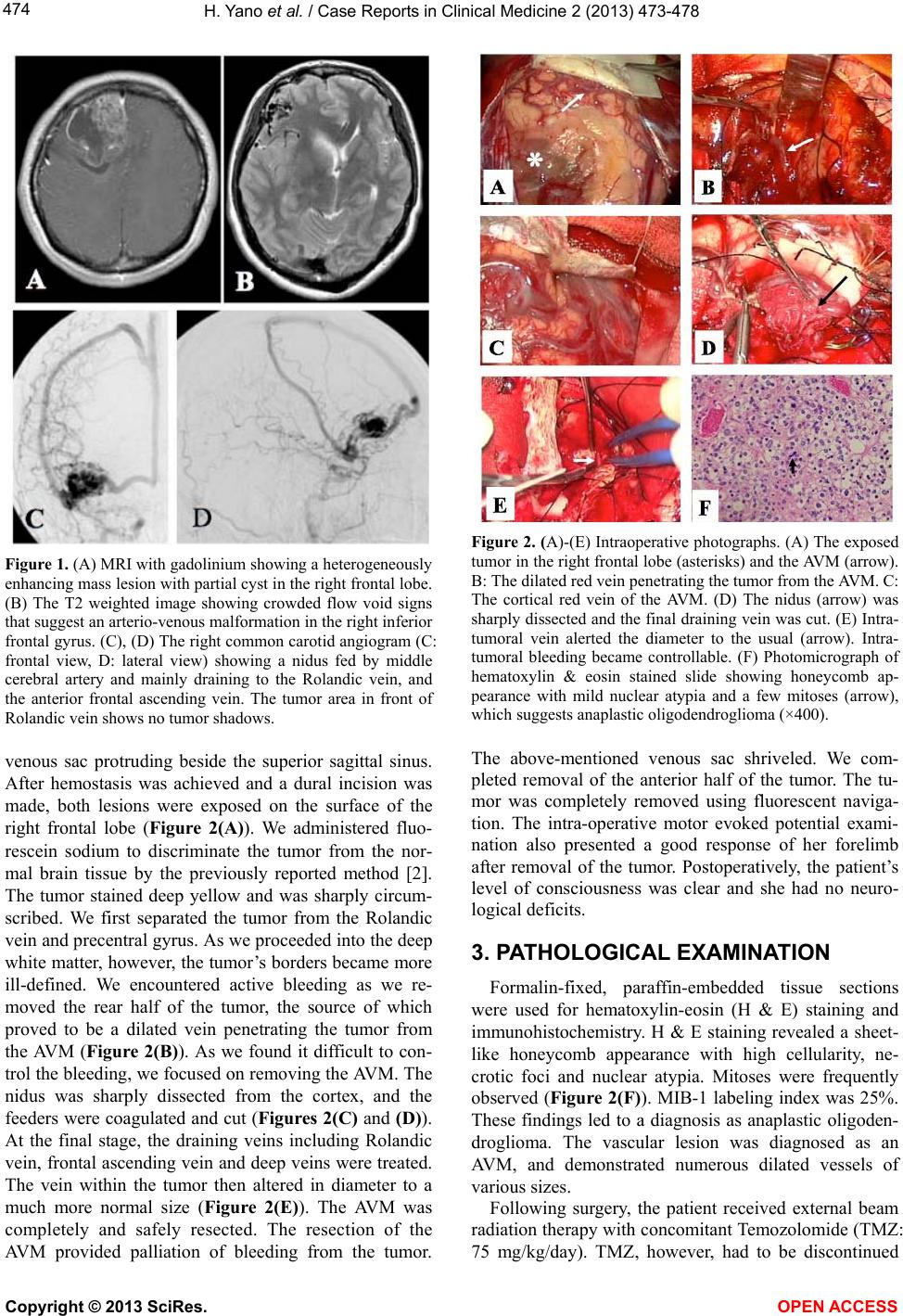 H. Yano et al. / Case R eports in Clinical Medicine 2 (2013) 473-478 474 Figure 1. (A) MRI with gadolinium showing a heterogeneously enhancing mass lesion with partial cyst in the right frontal lobe. (B) The T2 weighted image showing crowded flow void signs that suggest an arterio-venous malformation in the right inferior frontal gyrus. (C), (D) The right common carotid angiogram (C: frontal view, D: lateral view) showing a nidus fed by middle cerebral artery and mainly draining to the Rolandic vein, and the anterior frontal ascending vein. The tumor area in front of Rolandic vein shows no tumor shadows. venous sac protruding beside the superior sagittal sinus. After hemostasis was achieved and a dural incision was made, both lesions were exposed on the surface of the right frontal lobe (Figure 2(A)). We administered fluo- rescein sodium to discriminate the tumor from the nor- mal brain tissue by the previously reported method [2]. The tumor stained deep yellow and was sharply circum- scribed. We first separated the tumor from the Rolandic vein and precentral gyrus. As we proceeded into the deep white matter, however, the tumor’s borders became more ill-defined. We encountered active bleeding as we re- moved the rear half of the tumor, the source of which proved to be a dilated vein penetrating the tumor from the AVM (Figure 2(B)). As we found it difficult to con- trol the bleeding, we focused on removing the AVM. The nidus was sharply dissected from the cortex, and the feeders were coagulated and cut (Figures 2(C) and (D)). At the final stage, the draining veins including Rolandic vein, frontal ascending vein and deep veins were treated. The vein within the tumor then altered in diameter to a much more normal size (Figure 2(E)). The AVM was completely and safely resected. The resection of the AVM provided palliation of bleeding from the tumor. Figure 2. (A)-(E) Intraoperative photographs. (A) The exposed tumor in the right frontal lobe (asterisks) and the AVM (arrow). B: The dilated red vein penetrating the tumor from the AVM. C: The cortical red vein of the AVM. (D) The nidus (arrow) was sharply dissected and the final draining vein was cut. (E) Intra- tumoral vein alerted the diameter to the usual (arrow). Intra- tumoral bleeding became controllable. (F) Photomicrograph of hematoxylin & eosin stained slide showing honeycomb ap- pearance with mild nuclear atypia and a few mitoses (arrow), which suggests anaplastic oligodendroglioma (×400). The above-mentioned venous sac shriveled. We com- pleted removal of the anterior half of the tumor. The tu- mor was completely removed using fluorescent naviga- tion. The intra-operative motor evoked potential exami- nation also presented a good response of her forelimb after removal of the tumor. Postoperatively, the patient’s level of consciousness was clear and she had no neuro- logical deficits. 3. PATHOLOGICAL EXAMINATION Formalin-fixed, paraffin-embedded tissue sections were used for hematoxylin-eosin (H & E) staining and immunohistochemistry. H & E staining revealed a sheet- like honeycomb appearance with high cellularity, ne- crotic foci and nuclear atypia. Mitoses were frequently observed (Figure 2(F)). MIB-1 labeling index was 25%. These findings led to a diagnosis as anaplastic oligoden- droglioma. The vascular lesion was diagnosed as an AVM, and demonstrated numerous dilated vessels of various sizes. Following surgery, the patient received external beam radiation therapy with concomitant Temozolomide (TMZ: 75 mg/kg/day). TMZ, however, had to be discontinued Copyright © 2013 SciRes. OPEN ACCESS  H. Yano et al. / Case R eports in Clinical Medicine 2 (2013) 473-478 Copyright © 2013 SciRes. OPEN ACCESS 475 due to pancytopenia a few weeks later. Sixty gray of ra- diation therapy produced a satisfactory result. Both le- sions completely disappeared and there was no recur- rence of the tumor 5 year after completion of therapy. 4. DISCUSSION There have been 74 reported cases of brain tumors as- sociated with AVM to the best of our knowledge (Table 1) [3-40]. The rate of this rare association was reported to be 0.1% [19]. There is a male predominance (male: 47 cases, female: 27 cases). The ages ranged from 0 to 70 years of age; the median age was 28.5. The age of onset was unlikely to be related to the association of brain tu- mor and AVM, and seemed more likely to depend on the kind of brain tumor involved. There are several hypothe- ses about the etiology and timing of the development of the two lesions in terms of which occurs first and whether one lesion causes the other [26]. Environmental factors and viruses have also been speculated as triggers [26]. Concerning the histology of the brain tumors involved in these cases, there were 12 cases of pilocytic astrocy- toma [27,40], 10 of astrocytoma [3,6,8,15,29,30,33,35, 36], 6 of malignant astrocytoma [13,18,25,34,36], 20 of oligodendroglioma [4,9,10,20,24,27,38], 3 of glioblas- toma [7,19,36], 8 of meningioma [10,11,16,17,21,22,26, 32], 5 of acoustic tumors [12], and 3 of pleomorphic xan- thoastrocytoma [28,31,39]. Other tumors found in asso- ciation with AVM include hemangioblastoma [5], he- mangiopericytoma [22], ganglioneuroma, [23] subepen- dymal giant astrocytomas [29], and craniopharyngioma [14]. In the remaining 2 cases, histology was simply de- scribed as glioma [27,37]. In the present case, the histol- ogy was that of an anaplastic oligodendroglioma, which is the type of tumor most frequently associated with AVM. Concerning the positional relation between the tumor and the AVM, the two lesions were considered “separate” in 18 cases, “intermixed” in 39 cases and “adjacent” in 17 cases (Table 1). In our case, it appeared that the tumor had occurred in contiguity with a congenital AVM in the nearby region of the same lobe. Accordingly, the rela- tionship was judged as “separate”. There have been only a handful of cases in which both the AVM and the brain tumor were preoperatively diagnosed using angiography and MRI [29,31-34]. In some reported cases with inter- mixed AVM in the brain tumor, vascular components were pathologically diagnosed after the surgery. Those were diagnosed as “angioglioma”. In such a case, exten- sive bleeding during surgery would be assumed; however, the reports make little mention of the operative approach. In the present case, we preoperatively diagnosed both lesions, and decided to remove the tumor first, consider- ing it to be the etiology of the patient’s headaches be- cause we thought the AVM was asymptomatic. As both lesions were located in the right frontal lobe, we could Table 1. Summary of the positional relationship between brain tumor and AVM. Tumor histology Intermixed Adjacent Separate References Pilocytic astrocytoma 11 1 0 [27,40] Astrocytoma 2 5 3 [3,6,8,15,29,30,33,35,36] Malignant astrocytoma 2 2 2 [13,18,25,34,36] Glioblastoma 1 0 2 [7,19,36] Oligodendroglioma 14 2 4 [4,9,10,20,24,27,38] Meningioma 1 4 3 [10,11,16,17,21,22,26,32] Acoustic tumor 5 0 0 [12] Hemangioblastoma 1 0 0 [5] Hemangiopericytoma 0 1 0 [22] Craniopharyngioma 0 0 1 [14] Ganglioneuroma 0 1 0 [23] PXA 1 0 2 [28,31,39] SEGA* 1 0 0 [29] Glioma 0 1 1 [27,37] Total 39 17 18 *SEGA: subependymal giant astrocytoma.  H. Yano et al. / Case R eports in Clinical Medicine 2 (2013) 473-478 476 fortunately observe them in the same bone window at the operation. The tumor was sharply dissected from the Rolandic vein at the beginning of the operation; however, the tumor’s boundaries were less clear in the deep white matter. We had difficulty controlling bleeding from a dilated vein passing through the tumor from the AVM to the superior sagittal sinus. The vein was one of the draining veins of the AVM. Although the preoperative angiogram had not revealed them, we should have taken them into consideration from the T2 WI that showed some flow void signs passing through the tumor. We proceeded to remove the AVM with a general procedure. Fortunately, we could safely remove the nidus from the superficial frontal cortex. An intraoperative angiogram showed no residual nidus. It followed that we could dramatically diminish the bleeding and completely re- move the tumor. There have been 5 case reports with the separate type [11,20,21,24,28], in which both of the lesions were re- moved. Two cases had a good clinical course as the pre- sent case [24,28]. As a whole, in 51 cases described the cli- nical results, 23 cases were good recovery, 18 were mod- erate disability and 10 cases were dead. Accordingly, the entity does not necessary, which have a good prognosis. In conclusion, in a case of brain tumor nearby associ- ated with AVM, a large craniotomy including both le- sions should be planned to remove the nidus, allowing one to control bleeding from the AVM even if it was preoperatively asymptomatic. The procedures will make a good clinical course. REFERENCES [1] Aki, T., Nakayama, N., Yonezawa, S., Takenaka, S., Miwa, K., Asano, Y., Shinoda, J., Yano, H. and Iwama, T. (2012) Evaluation of brain tumors using dynamic 11C-methion- ine-PET. Journal of Neuro-Oncology, 109, 15-122. http://dx.doi.org/10.1007/s11060-012-0873-9 [2] Shinoda, J., Yano, H., Yoshimura, S., Okumura, A., Kaku, Y., Iwama, T. and Sakai, N. (2003) Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium. Technical note. Journal of Neurosur- gery, 99, 597-603. http://dx.doi.org/10.3171/jns.2003.99.3.0597 [3] White, R.J., Kernohan, J.W. and Wood, M.W. (1958). A study of fifty intracranial vascular tumors found inciden- tally at necropsy. Journal of Neuropathology & Experi- mental Neurology, 17, 392-398. http://dx.doi.org/10.1097/00005072-195804000-00022 [4] Fine, R.D., Paterson, A. and Gaylor, J.B. (1960) Recur- rent attacks of subarachnoid haemorrhage in presence of a cerebral angioma and an intraventricular oligodendro- glioma. Scottish Medical Journal, 5, 342-345. [5] Raynor, R.B. and Kingman Jr., A.F. (1965) Hemangio- blastoma and vascular malformations as one lesion. Ar- chives of Neurology, 12, 39-48. http://dx.doi.org/10.1001/archneur.1965.00460250043006 [6] Welker, E.R. and Seidel, K. (1966) Kombination eines angioma arteriovenosum aneurysmaticum mit einem as- trocytoma. Deutsche Zeitschrift fur Ne rvenheilkunde, 189, 231-239. [7] Warren, G.C. (1969). Intracranial arteriovenous malfor- mation, pulmonary arteriovenous fistula, and malignant glioma in the same patient. Case report. Journal of Neu- ro surgery , 30, 618-621. http://dx.doi.org/10.3171/jns.1969.30.5.0618 [8] Heffner Jr., R.R., Porro, R.S. and Deck, M.D. (1971) Benign astrocytoma associated with arteriovenous mal- formation. Case report. Journal of Neurosurgery, 35, 229- 233. http://dx.doi.org/10.3171/jns.1971.35.2.0229 [9] Crowell, R.M., DeGirolami, U. and Sweet, W.H. (1975) Arteriovenous malformation and oligodendroglioma. Case report. Journal of Neurosurgery, 43, 108-111. http://dx.doi.org/10.3171/jns.1975.43.1.0108 [10] Fine, R.D. and Gonski, A. (1976) Cerebral tumour with intracranial angioma. Medical Journal of Australia, 1, 227-230. [11] Fukawa, O. and Tanaka, T. (1977) The association of intracranial meningioma with arteriovenous malformation and aneurysm. Neurological Surgery, 5, 175-180 (Japa- nese). [12] Kasantikul, V. and Netsky, M.G. (1979) Combined neuri- lemmoma and angioma. Tumor of ectomesenchyme and a source of bleeding. Journal of Neurosurgery, 50, 81-87. http://dx.doi.org/10.3171/jns.1979.50.1.0081 [13] Zuccarello, M., Giordano, R., Scanarini, M. and Mingrino, S. (1979) Malignant astrocytoma associated with arterio- venous malformation. Case report. Acta Neurochirurgica (Wien ), 50, 305-309. http://dx.doi.org/10.1007/BF01808529 [14] Mori, K., Handa, H., Murata, T., Ishikawa, M., Takeuchi, J. and Osaka, K. (1980). Craniopharyngiomas with un- usual topography and associated with vascular pathology. Acta Neurochirurgica (Wi en ), 53, 53-68. http://dx.doi.org/10.1007/BF02074521 [15] Ho, K.L. and Wolfe, D.E. (1981) Concurrence of multiple sclerosis and primary intracranial neoplasms. Cancer, 47, 2913-2919. http://dx.doi.org/10.1002/1097-0142(19810615)47:12<29 13::AID-CNCR2820471229>3.0.CO;2-1 [16] Kasantikul, V. and Brown, W.J. (1981) Meningioangio- matosis in the absence of von Recklinghausen’s disease. Surgical Neurology, 15, 71-75. http://dx.doi.org/10.1016/S0090-3019(81)80095-X [17] Kasantikul, V. and Brown, W.J. (1984) Lipomatous men- ingioma associated with cerebral vascular malformation. Journal of Surgical Oncology, 26, 35-39. http://dx.doi.org/10.1002/jso.2930260109 [18] Shah, D., Naheedy, M.H. and O’Hara, R. (1985). Intrac- ranial vascular malformation and astrocytoma. IMJ Illi- nois Medical Journal, 168, 388-390. [19] Licata, C., Pasqualin, A., Freschini, A., Barone, G. and Da Pian, R. (1986) Management of associated primary cere- bral neoplasms and vascular malformations: 2. Intracra- Copyright © 2013 SciRes. OPEN ACCESS  H. Yano et al. / Case R eports in Clinical Medicine 2 (2013) 473-478 477 nial arterio-venous malformations. Acta Neurochirurgica (Wien ), 83, 38-46. http://dx.doi.org/10.1007/BF01420506 [20] Martinez-Lage, J.F., Poza, M., Esteban, J.A. and Sola, J. (1986) Subarachnoid hemorrhage in the presence of a cerebral arteriovenous malformation and an intraven- tricular oligodendroglioma: Case report. Neurosurgery, 19, 125-128. http://dx.doi.org/10.1227/00006123-198607000-00022 [21] Ohno, M., Nagai, H., Misawa, I. and Sugiyama, N. (1986) A meningioma associated with AVM: A case report. No Shinkei Geka, 14, 215-219 (Japanese). [22] Bitoh, S., Hasegawa, H., Kato, A., Tamura, K., Mabuchi, E. and Kobayashi, Y. (1987) Meningeal neoplasms asso- ciated with cerebral vascular malformations. Surgical Neurology, 27, 469-475. [23] Chovanes, G.I. and Truex Jr., R.C. (1987) Association of a ganglioneuroma with an arteriovenous malformation: Case report. Neurosurgery, 21, 241-243. http://dx.doi.org/10.1227/00006123-198708000-00022 [24] Nazek, M., Mandybur, T.I. and Kashiwagi, S. (1988) Oli- godendroglial proliferative abnormality associated with arteriovenous malformation: Report of three cases with review of the literature. Neurosurgery, 23, 781-785. http://dx.doi.org/10.1227/00006123-198812000-00022 [25] Goodkin, R., Zaias, B. and Michelsen, W.J. (1990) Arte- riovenous malformation and glioma: Coexistent or se- quential? Case report. Journal of Neurosurgery, 72, 798- 805. http://dx.doi.org/10.3171/jns.1990.72.5.0798 [26] Soria, E., Fine, E. and Hajdu, I. (1990) Association of intracranial meningioma with arteriovenous malformation. Surgical Neurology, 34, 111-117. http://dx.doi.org/10.1016/0090-3019(90)90106-Y [27] Lombardi, D., Scheithauer, B.W., Piepgras, D, Meyer, F.B. and Forbes, G.S. (1991) Angioglioma and the arterioven- ous malformation-glioma association. Journal of Neuro- surgery, 75, 589-566. http://dx.doi.org/10.3171/jns.1991.75.4.0589 [28] Malcolm, G.P., Symon, L, Tan, L.C. and Pires, M. (1991). Astrocytoma and associated arteriovenous malformation. Surgical Neurology, 36, 59-62. http://dx.doi.org/10.1016/0090-3019(91)90135-V [29] Ecklund, J., Schut, L. and Rorke, L. (1993) Associated vascular malformations and neoplasms in children. Pedi- atric Neurosurgery, 19, 196-201. http://dx.doi.org/10.1159/000120730 [30] Kroh, H., Bidziński, J. and Majchrowski, A. (1994) Su- pratentorial astrocytoma associated with arteriovenous malformation. Case report. Folia Neuropathologica, 32, 167-169. [31] Lee, T.T., Landy, H.J. and Bruce, J.H. (1996) Arterio- venous malformation associated with pleomorphic xan- thoastrocytoma. Acta Neurochirurgica (Wien), 138, 590- 591. http://dx.doi.org/10.1007/BF01411181 [32] Castillo, M., Thompson, J.E. and Mukherji, S.K. (1998) Association of an intracranial arteriovenous malformation and a meningioma. Neuroradiology, 40, 574-576. http://dx.doi.org/10.1007/s002340050646 [33] Umezawa, K., Kumabe, T. and Shirane, R. (1999) Coex- istence of intracranial arteriovenous malformation with Turcot’s syndrome: A case report. Surgical Neurology, 52, 397-399. http://dx.doi.org/10.1016/S0090-3019(99)00099-3 [34] Harris, O.A., Chang, S.D. and Harris, B.T. (2000) Ac- quired cerebral arteriovenous malformation induced by an anaplastic astrocytoma: An interesting case. Journal of Neurology Research, 22, 473-477. [35] Suzuki, H., Uenohara, H., Utsunomiya, A., Kurihara, N., Suzuki, S., Tadokoro, M., Iwasaki, Y., Sakurai, Y., Takei, H. and Tezuka, F. (2002) A case of angioglioma com- posed of astrocytoma with a papillary growth pattern: immunohistochemical and ultrastructural studies. Brain Tumor Pathology, 19, 111-116. http://dx.doi.org/10.1007/BF02478937 [36] Borges, L.R., Malheiros, S.M., Pelaez, M.P., Stávale, J.N., Santos, A.J., Carrete Jr., H., Nogueira, R.G., Ferraz, F.A. and Gabbai, A.A. (2003) Arteriovenous malformation- glioma association: Study of four cases. Arquivos de Neuro- Psiquiatria, 61, 426-429 (Portuguese). http://dx.doi.org/10.1590/S0004-282X2003000300018 [37] Ziyal, I.M., Ece, K., Bilginer, B., Tezel, G.G. and Ozcan, O.E. (2004). A glioma with an arteriovenous malforma- tion: An association or a different entity? Acta Neurochi- rurgica (Wi en ), 146, 83-86. http://dx.doi.org/10.1007/s00701-003-0161-8 [38] McKinney, J.S., Steineke, T., Nochlin, D. and Brisman, J.L. (2008) De novo formation of large arteriovenous shunting and a vascular nidus mimicking an arterioven- ous malformation within an anaplastic oligodendroglioma: Treatment with embolization and resection. Journal of Neurosurgery , 109, 1098-1102. http://dx.doi.org/10.3171/JNS.2008.109.12.1098 [39] Pallud, J., Belaid, H., Guillevin, R., Vallee, J.N. and Capelle, L. (2009). Management of associated glioma and arteriovenous malformation—The priority isthe glioma. British Journal of Neurosurgery, 23, 197-198. http://dx.doi.org/10.1080/02688690802688146 [40] Soltanolkotabi, M., Schoeneman, S.E., Dipatri, A.J., Hur- ley, M.C., Ansari, S.A., Rajaram, V., Tomita, T. and Shaibani, A. (2012) Juvenile pilocytic astrocytoma in as- sociation with arteriovenous malformation. Interventional Neuroradiology, 18, 140-147. Copyright © 2013 SciRes. OPEN ACCESS  H. Yano et al. / Case R eports in Clinical Medicine 2 (2013) 473-478 478 LIST OF ABBREVIATIONS AVM: arteriovenous malformation MRI: magnetic resonance image WI: weighted images Gd-DTPA: gadolinium-diethylenetriamine penta-acetic acid PET: positron emission tomography H&E: hematoxylin-eosin; TMZ: temozolomide Copyright © 2013 SciRes. OPEN ACCESS
|