 Pharmacology & Pharmacy, 2013, 4, 584-589 Published Online November 2013 (http://www.scirp.org/journal/pp) http://dx.doi.org/10.4236/pp.2013.48084 Open Access PP Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial Jiangbei Cao, Wenzhu Shi, Weidong Mi, Hong Zhang* Department of Anesthesiology and Operation Center, PLA General Hospital, Beijing, China. Email: cjb2000@sina.com, *mazuimao301@yahoo.com.cn Received September 15th, 2013; revised October 16th, 2013; accepted October 25th, 2013 Copyright © 2013 Jiangbei Cao et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Background: The efficacy and safety of dexmedetomidine during the anesthesia induction of intracranial tumor pa- tients remain unknown. We wondered whether loading infusion of dexmedetomidine 1 g/kg over 10 min to intracra- nial tumor patients was as efficient and safe as to those abdominal disease patients. Methods: Patients aged 18 - 60 years, male or female, ASA I or II, scheduled for intracranial tumor resection (Group N, n = 30) or abdominal operation (Group A, n = 30) were enrolled in this observational trial. Dexmedetomidine was administrated with a loading dosage of 1 g/kg over 10 min following with continuous infusing of 0.5 g/kg/h. Fentanyl, propofol and rocuronium were sequentially administered for anesthesia induction. Heart rate (HR), blood pressure (BP), pulse oxygen saturation (SpO2), bispectral index (BIS) and other adverse effects were recorded from the beginning of loading infusion of dex- medetomidine to the end of endotracheal intubation. Results: Among with loading infusion, HR and BIS value de- creased and were significantly lower at the end of infusion than before infusion (P < 0.01), but BP did not (P > 0.05). One patient of Group N dropped out from this trial because of a serious headache. 14 of 29 patients during dexmede- tomidine loading infusion suffered hypoxemia (SpO2 < 90%) in Group N, which was higher than 6 of 30 of in Group A (P < 0.05). No other side effects were recorded. Conclusion: A loading dosage of 1 g/kg of dexmedetomidine was not suitable for the anesthesia induction of intracranial tumor patients as compared to patients undergoing abdominal opera- tion. Keywords: Dexmedetomidine; Intracranial Tumor; Adverse Effects; Loading Infusion 1. Introduction Dexmedetomidine, a highly selective α2-adrenoreceptor agonist, is widely used as a sedative or an adjuvant agent for anesthesia induction and maintenance [1-6]. Patients sedated with dexmedetomidine could well tolerate the endotracheal intubation. When using as sedative, dex- medetomidine can increase hemodynamic stability, cause little respiratory depression and allow a rapid recovery [1,7,8]. Preliminary data of the perioperative usage of dex- medetomidine in patients undergoing neurosurgery have indicated that preoperative usage of dexmedetomidine may spare opioid requirements and offer more stable hemodynamics during neurosurgery [9-11]. Besides dex- medetomidine’s well-proven sedative effects, an in- creasing body of both in vitro and in vivo evidence indi- cates that dexmedetomidine also exerts a cell-protective effect on nervous tissue under ischemic conditions [12- 15]. In addition, laryngoscopy and endotracheal intubation are apt to result in hypertension, tachycardia and ar- rhythmias, which is hazardous to those patients undergo- ing neurosurgery for brain tumor [16,17]. Therefore, it seems that dexmedetomidine used as an adjunct of anes- thesia induction may be helpful to those patients under- going neurosurgery. Dexmedetomidine loading infusion with the dosage of 1 g/kg over 10 min showed its effi- cacy and safety to several kinds of operation patients [18-20]. However,the efficacy and safety of dexmede- tomidine loading infusion during anesthesia induction to *Corresponding author.  Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial 585 those intracranial tumor patients remain unknown. The objective of this preliminary observational trial was to investigate into the sedative effects, respiration inhibition, hemodynamic changes and adverse effects of dexmede- tomidine with a loading infusion dosage 1 g/kg over 10 min during introduction in neurosurgery patients. 2. Patients and Methods This trial protocol was approved by the Ethics Commit- tee of the General Hospital of PLA, Beijing, China. The written informed consents were obtained from all pa- tients. Sixty patients, male or female, aged 18 - 60 years, with American Society of Anesthesiologists (ASA) physical status I or II, were enrolled from April 2011 to October 2011 in this trial. Among these patients, 30 patients were scheduled to intracranial tumor resection (Group N), and the other 30 patients were scheduled for abdominal op- eration (Group A). The exclusion criteria included: pa- tients with hepatic or renal insufficiency, a history of allergy to α2-adrenoreceptor agonist, a psychiatric or neuromuscular disorder, or predicted difficult airway. All the patients were premeditated with intramuscular atropine of 0.5 mg at 30 min before operation. After en- tering the operation room, patients were inserted intra- venously catheter with a 20- or 22-gauge in a peripheral vein. The patients were first administered with a loading dosage of 1 g/kg of dexmedetomidine (200 g/ml, di- luted with normal saline to 4 g/ml) over 10 min by a computer controlled pump infuser then followed to in- fuse with the rate of 0.5 g/kg/h. When loading infusion was ended, fentanyl 2 g/kg, propofol 2 mg/kg and ro- curonium 0.9 mg/kg were sequentially administered within 1 min. Three minutes later, endotracheal intuba- tion was performed by one of the two appointed attend- ing anesthesiologists. Propofol and isoflurane were used for anesthesia maintenance. Heart rate (HR), blood pressure (systolic and diastolic blood pressure, SBP and DBP), bispectral index (BIS) and pulse oxygen saturation (SpO2) were recorded at the following time points: before dexmedetomidine loading infusion (T1), at the end of loading infusion (T2), before, during and 5 min after endotracheal intubation (T3-5, respectively). Normally, during the period of loading infusion, the patients inhaled room air without extra-oxygen supplying. If a patient’s HR < 45 bpm for >3 min, SBP < 80 mmHg for >3 min, or SpO2 < 90% for >1 min, he (she) will drop out from this trial and then atropine, ephedrine or 100% oxygen via mask will be given [21]. The occurrence of adverse reactions including nausea and dry mouth were also noted. 3. Statistical Analysis Data are presented as means ± SD or proportion. Statis- tical analysis was performed using the SPSS 17.0 soft- ware for Windows (Chicago, IL). Proportion was com- pared using chi-square test. Age, weight and height were compared using independent-samples t tests. HR, SBP, DBP, BIS and SpO2 were analyzed by using analysis of variance for repeated measurements. P < 0.05 was con- sidered to be statistically significant. 4. Result All the surgeries were successfully completed. One pa- tient from Group N was ceased to continue infusing dex- medetomidine because a severe headache occurred dur- ing the loading infusion. There was no significant differ- ence with respect to demographic data between the two groups (Table 1). As shown in Figure 1, the HR of Group N is faster than that of Group A (83 ± 11 bpm vs. 75 ± 10 bpm, P < 0.01) before anesthesia. After loading infusion of dex- medetomidine at dosage of 1 g/kg, HR decreased sig- nificantly in both groups (the average HR of Group N was 62 bpm and Group A was 61 bpm independently, P < 0.01, compared with T1, Figure 1). However, there was no significant change in blood pressure during load- ing infusion or even during endotracheal intubation (Figure 2). Among with the infusion of dexmedetomidine, the pa- tient could be sedated effectively. After loading infusion, it showed that patients were deep sedated (the average BIS value decreased from 94 to 74 in Group N, from 95 to 76 in Group A, P < 0.01, Figure 3). In this trial, we designed that all patients were limited to inhale room air during dexmedetomidine loading infusion. The results showed that at the end of loading infusion SpO2 value decreased from 98% to 95% of Group N and from 99% to 96% respectively (P < 0.01) (Figure 4). Though no patient was dropped off in case of SpO2 lower than 90% for more than 1 minute. However, it happened to some patients of both groups that their SpO2 value were fluctu- ating between 86% - 96%. Among them, 14 of 29 pa- tients in Group N while 6 of 30 patients in Group A whose SpO2 value dropped below 90% at least once dur- ing dexmedetomidine loading infusion (P < 0.05). Table 1. Demographics in the two groups. Characteristic Group N Group A Sex, male/female 15/15 14/16 Age, mean (SD), yr 44 (11) 45 (12) Weight, mean (SD), kg 63 (10) 62 (8) Height, mean (SD), cm 165 (9) 164 (7) Open Access PP 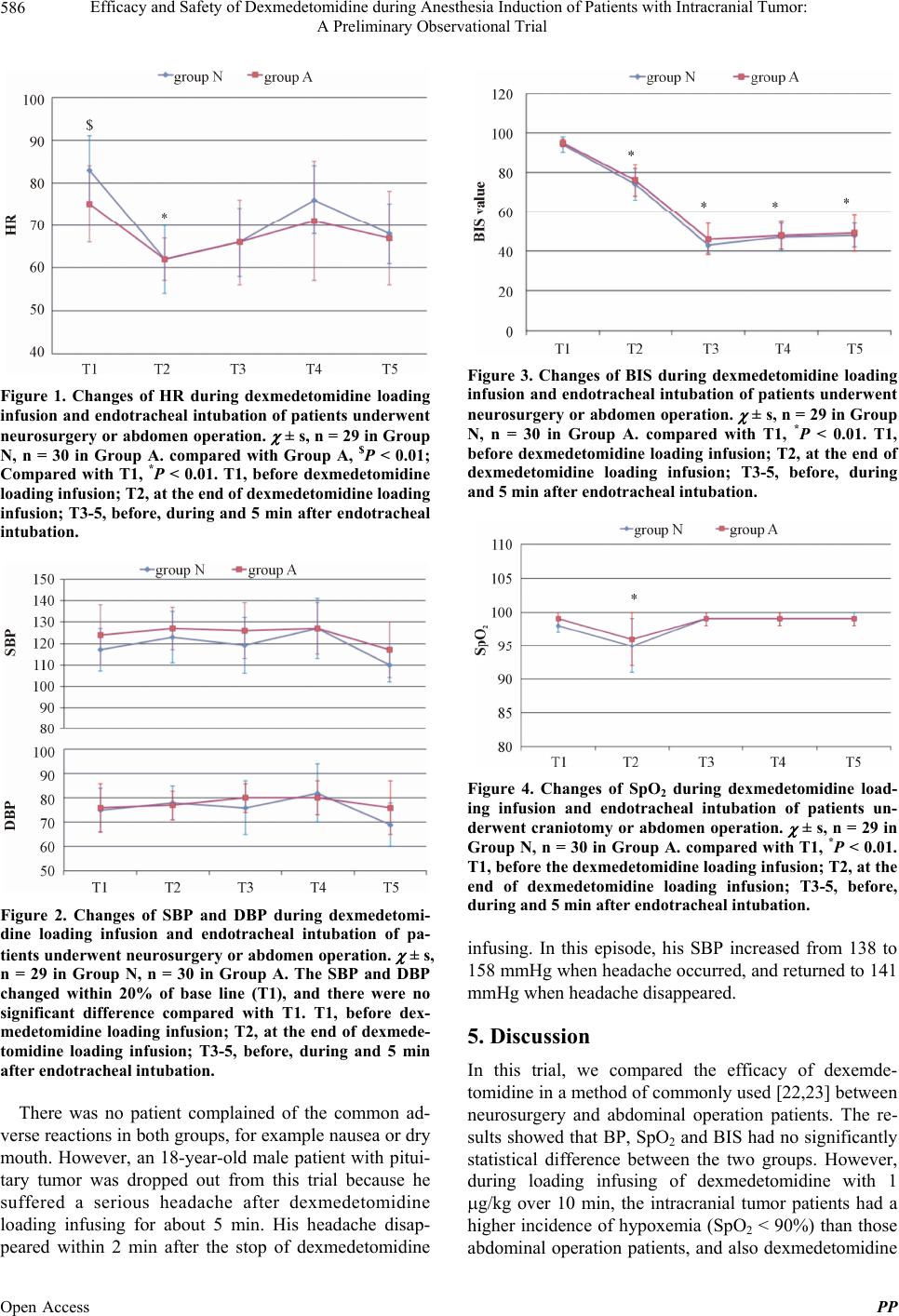 Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial 586 Figure 1. Changes of HR during dexmedetomidine loading infusion and endotracheal intubation of patients underw ent neurosurgery or abdomen operation. ± s, n = 29 in Group N, n = 30 in Group A. compared with Group A, $P < 0.01; Compared with T1, *P < 0.01. T1, before dexmedetomidine loading infusion; T2, at the end of dexmedetomidine loading infusion; T3-5, before, during and 5 min after endotracheal intubation. Figure 2. Changes of SBP and DBP during dexmedetomi- dine loading infusion and endotracheal intubation of pa- tients underwent neur osurgery or abdomen opera tion. ± s, n = 29 in Group N, n = 30 in Group A. The SBP and DBP changed within 20% of base line (T1), and there were no significant difference compared with T1. T1, before dex- medetomidine loading infusion; T2, at the end of dexmede- tomidine loading infusion; T3-5, before, during and 5 min after endotracheal intubation. There was no patient complained of the common ad- verse reactions in both groups, for example nausea or dry mouth. However, an 18-year-old male patient with pitui- tary tumor was dropped out from this trial because he suffered a serious headache after dexmedetomidine loading infusing for about 5 min. His headache disap- peared within 2 min after the stop of dexmedetomidine Figure 3. Changes of BIS during dexmedetomidine loading infusion and endotracheal intubation of patients underw ent neurosurgery or abdomen operation. ± s, n = 29 in Group N, n = 30 in Group A. compared with T1, *P < 0.01. T1, before dexmedetomidine loading infusion; T2, at the end of dexmedetomidine loading infusion; T3-5, before, during and 5 min after endotracheal intubation. Figure 4. Changes of SpO2 during dexmedetomidine load- ing infusion and endotracheal intubation of patients un- derwent craniotomy or abdomen operation. ± s, n = 29 in Group N, n = 30 in Group A. compared with T1, *P < 0.01. T1, before the dexmedetomidine loading infusion; T2, at the end of dexmedetomidine loading infusion; T3-5, before, during and 5 min after endotracheal intubation. infusing. In this episode, his SBP increased from 138 to 158 mmHg when headache occurred, and returned to 141 mmHg when headache disappeared. 5. Discussion In this trial, we compared the efficacy of dexemde- tomidine in a method of commonly used [22,23] between neurosurgery and abdominal operation patients. The re- sults showed that BP, SpO2 and BIS had no significantly statistical difference between the two groups. However, during loading infusing of dexmedetomidine with 1 g/kg over 10 min, the intracranial tumor patients had a higher incidence of hypoxemia (SpO2 < 90%) than those abdominal operation patients, and also dexmedetomidine Open Access PP  Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial 587 might induce serious headache in patients with intracra- nial tumor. Dexmedetomidine as an α2-adrenoceptor agonist is widely used for the sedation of intubated and mechani- cally ventilated patients in the intensive care setting, as well as for the sedation of non-intubated patients before and during surgical procedures [7,24,25]. In addition to its sedative properties, dexmedetomidine causes sym- patholysis and decreases anesthetic requirements, which may decrease opioid use and make it attractive for use as an adjunct to general anesthesia. Moreover, the neuro- protective property of dexmedetomidine has been dem- onstrated in various animal models of cerebral ischemia [9,26]. Drummond and colleagues [27] recently demon- strated a simultaneous decrease of CBF and CMRO2 with dexmedetomidine in healthy volunteers. However, the cerebral blood flow (CBF)/cerebral metabolic rate (CMR) ratio did not decrease and suggest that CMR-CBF cou- pling is preserved during dexmedetomidine administra- tion. Also, researchers found that dexmedetomidine could decrease the adrenergic response to the trauma of surgery and improve perioperative hemodynamic stabil- ity [28]. All these documents show that pretreatment or loading infusion with dexmedetomidine before induction has the advantage to those neurosurgery patients. However, there are few researches on the safety of us- ing dexmedetomidine to intracranial tumor patients and which dosage should be used as an adjunct to anesthesia induction, though it was recommended that dexmede- tomidine 1 g/kg as loading infusion over 10 min was a common clinical dosage [22]. In this study, we designed a trial to investigate the safety and efficiency of dex- medetomidine usage to those intracranial patients who received a loading infusion dosage of 1 g/kg over 10 min as an adjunct to anesthesia induction. Additionally, patients were allowed to inhale room air but not the 100% O2 during loading infusion during this trial to in- vestigate the dexmedetomidine’ effect on patients’ respi- ration function. The primary composite endpoint of this study was de- fined as patients having any of the following during loading infusion period: 1) HR < 45 bpm for >3 minutes, 2) systolic blood pressure (SBP) < 80 mmHg for >3 minutes, 3) or SpO2 < 90% for >1 min, or 4) administra- tion of vasoactive agents (including <500 μg phenyle- phrine by bolus or infusion, any epinephrine) or any at- ropine. It was found that the HR of almost all the patients including intracranial tumor patients and abdominal op- eration patients decreased obviously. The HR of some patients was less than 50 bpm (but haven’t match the deadline of 45 bpm for more than 3 min) during dex- medetomidine loading infusion. In our study, the base HR of intracranial tumor patients was faster significantly than abdominal operation patients. It may be due to dif- ferent treatments of diseases before operation. However, HR was almost the same of both groups at the end of dexmedetomidine loading infusion. In addition, the hemodynamic frustration changed slightly during dex- medetomidine infusion and even during endotracheal intubation. It is of very important for patients to keep normal cen- tral respiratory rhythm during sedation or introduction of anesthesia. Dexmedetomidine, an α2-adrenergic agonist, causes an unique kind of sedation, acting on the subcor- tical areas, which resembles natural sleep without respi- ratory depression [29]. The monitoring of BIS value in our study showed that during dexmedetomidine loading infusion, patients could be sedated and reached a level of deep sleep gradually (the BIS of both group at the end of loading infusion <76, 0 = no cortical activity, or coma; 40 - 60 = unconscious; 70 - 90 = varying levels of con- scious sedation; 100-fully awake). During loading infu- sion, patients could be still breathing almost normally. It was shown that SpO2 could always keep beyond 90% when patients inhaling room air but not 100% oxygen. However, during dexmedetomidine loading infusion, some patients’ SpO2 decreased and fluctuated in a wide range between 86% and 96%. And the results could tell us that the incidence of hypoxemia (the SpO2 was dropped below 90% at least one time) was much higher in those patients with intracranial tumor than those pa- tients undergoing abdominal operation. In other words, our data implied that dexemdetomidine might lead to more serious respiration depression in patients with in- tracranial tumor. The reason why the incidence of hy- poxemia was much higher in intracranial tumor patients than in abdominal patients may be due to the ICP changes of intracranial tumor patients, but still remains unclear. Dexmedetomidine has been used in over one million patients since its approval in 1999 [30]. The most fre- quently reported adverse reactions of dexmetomidine usage were hypotension, nausea, and bradycardia [31-33]. In this study, during dexmedetomidine loading infusion, there was a young man suffered an unpredictable head- ache. The exact reason for headache happening was far to know, but the occurrence of headache warning us to notice this adverse reaction when dexemdetomidine used in clinics, particularly in patients with intracranial tumor. Sometimes, a higher loading infusion doses may be re- quired to ensure adequate sedation. However, a higher dose of dexmedetomidine can have a higher incidence of hypertension, bradycardia, and hypotension [7,34,35]. Patients with intracranial tumor showed less endurance than those patients without intracranial tumor. The main limitation of our study was the small sample Open Access PP  Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial 588 size. The sample size was too small especially when ob- serving adverse reactions. Therefore, further large sam- ple size investigations are needed in the near future to determine the safety of dexemdetomidine in neurosur- gery. Further studies are necessary to establish an opti- mal dosage regimen especially for patients underwent craniotomy, because those patients maybe always endure a high ICP and experience an unexpected adverse reac- tion. Secondly, we had not checked the precise sedation scores of patients and also had not recorded the frequent of respiration. Therefore, further investigations maybe need to answer these questions. 6. Conclusion Compared with those without intracranial tumor patients, dexemdetomidine with a loading dosage 1 g/kg may induce more serious respiration depression and some- times headache in intracranial tumor patients. And fur- ther investigations with a large sample size are needed to determine the safety rang of dexemdetomidine in intrac- ranial tumor patients. 7. Acknowledgements We thank Dr. Jianjun Yang for reviewing this manu- script. REFERENCES [1] R. Siddappa, J. Riggins, S. Kariyanna, P. Calkins and A. T. Rotta, “High-Dose Dexmedetomidine Sedation for Pe- diatric MRI,” Pediatric Anesthesia, Vol. 21, No. 2, 2011, pp. 153-158. http://dx.doi.org/10.1111/j.1460-9592.2010.03502.x [2] S. Strom, “Preoperative Evaluation, Premedication, and Induction of Anesthesia in Infants and Children,” Current Opinion in Anesthesiology, Vol. 25, No. 3, 2012, pp. 321- 325. http://dx.doi.org/10.1097/ACO.0b013e3283530e0d [3] A. Akin, A. Bayram, A. Esmaoglu, Z. Tosun, R. Aksu, R. Altuntas and A. Boyaci, “Dexmedetomidine vs Mida- zolam for Premedication of Pediatric Patients Undergoing Anesthesia,” Pediatric Anesthesia, Vol. 22, No. 9, 2012, pp. 871-876. http://dx.doi.org/10.1111/j.1460-9592.2012.03802.x [4] A. Patel, M. Davidson, M. C. Tran, H. Quraishi, C. Schoenberg, M. Sant, A. Lin and X. Sun, “Dexmede- tomidine Infusion for Analgesia and Prevention of Emer- gence Agitation in Children with Obstructive Sleep Ap- nea Syndrome Undergoing Tonsillectomy and Adenoi- dectomy,” Anesthesia & Analgesia, Vol. 111, No. 4, 2010, pp. 1004-1010. [5] N. Ohtani, K. Kida, K. Shoji, Y. Yasui and E. Masaki, “Recovery Profiles from Dexmedetomidine as a General Anesthetic Adjuvant in Patients Undergoing Lower Ab- dominal Surgery,” Anesthesia & Analgesia, Vol. 107, No. 6, 2008, pp. 1871-1874. http://dx.doi.org/10.1213/ane.0b013e3181887fcc [6] K. Kida, N. Ohtani, K. Shoji, Y. Yasui and E. Masaki, “Postoperative Pain Status after Intraoperative Systemic Dexmedetomidine and Epidural Neostigmine in Patients Undergoing Lower Abdominal Surgery,” European Jour- nal of Anaesthesiology, Vol. 25, No. 11, 2008, pp. 869- 875. http://dx.doi.org/10.1017/S0265021508004493 [7] K. P. Mason, D. Zurakowski, S. Zgleszewski, R. Prescilla, P. J. Fontaine and J. A. Dinardo, “Incidence and Predic- tors of Hypertension during High-Dose Dexmedetomi- dine Sedation for Pediatric MRI,” Pediatric Anesthesia, Vol. 20, No. 6, 2010, pp. 516-523. http://dx.doi.org/10.1111/j.1460-9592.2010.03299.x [8] J. W. Berkenbosch, P. C. Wankum and J. D. Tobias, “Prospective Evaluation of Dexmedetomidine for Nonin- vasive Procedural Sedation in Children,” Pediatric Criti- cal Care Medicine, Vol. 6, No. 4, 2005, pp. 435-439. http://dx.doi.org/10.1097/01.PCC.0000163680.50087.93 [9] K. Sato, T. Kimura, T. Nishikawa, Y. Tobe and Y. Ma- saki, “Neuroprotective Effects of a Combination of Dex- medetomidine and Hypothermia after Incomplete Cere- bral Ischemia in Rats,” Acta Anaesthesiologica Scandi- navica, Vol. 54, No. 3, 2010, pp. 377-382. http://dx.doi.org/10.1111/j.1399-6576.2009.02139.x [10] N. E. Ngwenyama, J. Anderson, D. G. Hoernschemeyer and J. D. Tobias, “Effects of Dexmedetomidine on Pro- pofol and Remifentanil Infusion Rates during Total In- travenous Anesthesia for Spine Surgery in Adolescents,” Pediatric Anesthesia, Vol. 18, No. 2, 2008, pp. 1190- 1195. [11] R. N. Soliman, A. R. Hassan, A. M. Rashwan and A. M. Omar, “Prospective, Randomized Study to Assess the Role of Dexmedetomidine in Patients with Supratentorial Tumors Undergoing Craniotomy under General Anaes- thesia,” Middle East Journal of Anesthesiology, Vol. 21, No. 3, 2011, pp. 325-334. [12] M. Schoeler, P. D. Loetscher, R. Rossaint, A. V. Fahlenk- amp, G. Eberhardt, S. Rex, J. Weis and M. Coburn, “Dex- medetomidine Is Neuroprotective in an in Vitro Model for Traumatic Brain Injury,” BMC Neurology, Vol. 12, 2012, p. 20. http://dx.doi.org/10.1186/1471-2377-12-20 [13] S. Dahmani, D. Rouelle, P. Gressens and J. Mantz, “Cha- racterization of the Postconditioning Effect of Dexmede- tomidine in Mouse Organotypic Hippocampal Slice Cul- tures Exposed to Oxygen and Glucose Deprivation,” An- esthesiology, Vol. 112, No. 2, 2010, pp. 373-383. http://dx.doi.org/10.1097/ALN.0b013e3181ca6982 [14] K. Engelhard, C. Werner, E. Eberspacher, M. Bachl, M. Blobner, E. Hildt, P. Hutzler and E. Kochs, “The Effect of the Alpha 2-Agonist Dexmedetomidine and the n-Methyl- d-Aspartate Antagonist s(+)-Ketamine on the Expression of Apoptosis-Regulating Proteins after Incomplete Cere- bral Ischemia and Reperfusion in Rats,” Anesthesia & Analgesia, Vol. 96, No. 2, 2003, pp. 524-531. [15] R. N. Soliman, A. R. Hassan, A. M. Rashwan and A. M. Omar, “Prospective, Randomized Controlled Study to Assess the Role of Dexmedetomidine in Patients with Su- pratentorial Tumors Undergoing Craniotomy under Gen- eral Anesthesia,” Middle East Journal of Anesthesiology, Open Access PP  Efficacy and Safety of Dexmedetomidine during Anesthesia Induction of Patients with Intracranial Tumor: A Preliminary Observational Trial Open Access PP 589 Vol. 21, No. 1, 2011, pp. 23-33. [16] R. K. Stoelting, “Blood Pressure and Heart Rate Changes during Short-Duration Laryngoscopy for Tracheal Intuba- tion: Influence of Viscous or Intravenous Lidocaine,” Anesthesia & Analgesia, Vol. 57, No. 2, 1978, pp. 197- 199. http://dx.doi.org/10.1213/00000539-197803000-00009 [17] R. A. Allberry and H. F. Drake, “Preoperative Beta- Blockade for Patients Undergoing Craniotomy: A Com- parison between Propranolol and Atenolol,” Canadian Journal of Anaesthesia, Vol. 37, No. 4, 1990, pp. 448-451. http://dx.doi.org/10.1007/BF03005625 [18] W. S. Kang, S. Y. Kim, J. C. Son, J. D. Kim, H. B. Mu- hammad, S. H. Kim, T. G. Yoon and T. Y. Kim, “The Effect of Dexmedetomidine on the Adjuvant Propofol Requirement and Intraoperative Hemodynamics during Remifentanil-Based Anesthesia,” Korean Journal of An- esthesiology, Vol. 62, No. 2, 2012, pp. 113-118. http://dx.doi.org/10.4097/kjae.2012.62.2.113 [19] Z. Ozkose, F. S. Demir, K. Pampal and S. Yardim, “He- modynamic and Anesthetic Advantages of Dexmede- tomidine, an Alpha 2-Agonist, for Surgery in Prone Posi- tion,” Tohoku Journal of Experimental Medicine, Vol. 210, No. 2, 2006, pp. 153-160. http://dx.doi.org/10.1620/tjem.210.153 [20] S. R. Arain and T. J. Ebert, “The Efficacy, Side Effects, and Recovery Characteristics of Dexmedetomidine versus Propofol When Used for Intraoperative Sedation,” Anes- thesia & Analgesia, Vol. 95, No. 2, 2002, pp. 461-466. [21] R. Y. Klinger, W. D. White, B. Hale, A. S. Habib and E. Bennett-Guerrero, “Hemodynamic Impact of Dexmede- tomidine Administration in 15,656 Noncardiac Surgical Cases,” Journal of Clinical Anesthesia, Vol. 24, No. 3, 2012, pp. 212-220. http://dx.doi.org/10.1016/j.jclinane.2011.07.016 [22] M. Shukry and J. A. Miller, “Update on Dexmedetomi- dine: Use in Nonintubated Patients Requiring Sedation for Surgical Procedures,” Therapeutics and Clinical Risk Management, Vol. 6, 2010, pp. 111-121. http://dx.doi.org/10.2147/TCRM.S5374 [23] A. Bekker, M. Sturaitis, M. Bloom, M. Moric, J. Golfinos, E. Parker, R. Babu and A. Pitti, “The Effect of Dexmede- tomidine on Perioperative Hemodynamics in Patients Undergoing Craniotomy,” Anesthesia & Analgesia, Vol. 107, No. 4, 2008, pp. 1340-1347. http://dx.doi.org/10.1213/ane.0b013e3181804298 [24] M. A. Mirski, Lewin Jr., S. Ledroux, C. Thompson, P. Murakami, E. K. Zink and M. Griswold, “Cognitive Im- provement during Continuous Sedation in Critically Ill, Awake and Responsive Patients: The Acute Neurological ICU Sedation Trial (Anist),” Intensive Care Medicine, Vol. 36, No. 9, 2010, pp. 1505-1513. http://dx.doi.org/10.1007/s00134-010-1874-9 [25] A. Bekker and M. K. Sturaitis, “Dexmedetomidine for Neurological Surgery,” Neurosurgery, Vol. 57, No. 1, 2005, pp. 1-10. http://dx.doi.org/10.1227/01.NEU.0000163476.42034.A1 [26] M. Cosar, O. Eser, H. Fidan, O. Sahin, S. Buyukbas, Y. Ela, M. Yagmurca and O. A. Ozen, “The Neuroprotective Effect of Dexmedetomidine in the Hippocampus of Rab- bits after Subarachnoid Hemorrhage,” Surgical Neurology, Vol. 71, No. 1, 2009, pp. 54-59. http://dx.doi.org/10.1016/j.surneu.2007.08.020 [27] J. C. Drummond, A. V. Dao, D. M. Roth, C. R. Cheng, B. I. Atwater, A. Minokadeh, L. C. Pasco and P. M. Patel, “Effect of Dexmedetomidine on Cerebral Blood Flow Velocity, Cerebral Metabolic Rate, and Carbon Dioxide Response in Normal Humans,” Anesthesiology, Vol. 108, No. 2, 2008, pp. 225-232. http://dx.doi.org/10.1097/01.anes.0000299576.00302.4c [28] A. S. Uyar, H. Yagmurdur, Y. Fidan, C. Topkaya and H. Basar, “Dexmedetomidine Attenuates the Hemodynamic and Neuroendocrinal Responses to Skull-Pin Head-Holder Application during Craniotomy,” Journal of Neurosurgi- cal Anesthesiology, Vol. 20, No. 3, 2008, pp. 174-179. http://dx.doi.org/10.1097/ANA.0b013e318177e5eb [29] I. Rozet, “Anesthesia for Functional Neurosurgery: The Role of Dexmedetomidine,” Current Opinion in Anaes- thesiology, Vol. 21, No. 5, 2008, pp. 537-543. http://dx.doi.org/10.1097/ACO.0b013e32830edafd [30] P. O. Talke and M. Maze, “Expecting the Unexpected,” Anesthesia & Analgesia, Vol. 106, No. 6, 2008, pp. 1605- 1606. http://dx.doi.org/10.1213/ane.0b013e31816c3f69 [31] M. Venn, J. Newman and M. Grounds, “A Phase II Study to Evaluate the Efficacy of Dexmedetomidine for Seda- tion in the Medical Intensive Care Unit,” Intensive Care Medicine, Vol. 29, No. 2, 2003, pp. 201-207. [32] J. D. Tobias, J. W. Berkenbosch and P. Russo, “Addi- tional Experience with Dexmedetomidine in Pediatric Pa- tients,” Southern Medical Journal, Vol. 96, No. 9, 2003, pp. 871-875. http://dx.doi.org/10.1097/01.SMJ.0000053557.75799.09 [33] J. D. Tobias and J. W. Berkenbosch, “Sedation during Mechanical Ventilation in Infants and Children: Dex- medetomidine versus Midazolam,” Southern Medical Journal, Vol. 97, No. 5, 2004, pp. 451-455. http://dx.doi.org/10.1097/00007611-200405000-00007 [34] K. P. Mason, D. Zurakowski, S. E. Zgleszewski, C. D. Robson, M. Carrier, P. R. Hickey and J. A. Dinardo, “High Dose Dexmedetomidine as the Sole Sedative for Pediatric MRI,” Pediatric Anesthesia, Vol. 18, No. 5, 2008, pp. 403-411. http://dx.doi.org/10.1111/j.1460-9592.2008.02468.x [35] K. P. Mason, S. Zgleszewski, R. E. Forman, C. Stark and J. A. DiNardo, “An Exaggerated Hypertensive Response to Glycopyrrolate Therapy for Bradycardia Associated with High-Dose Dexmedetomidine,” Anesthesia & Anal- gesia, Vol. 108, No. 3, 2009, pp. 906-908. http://dx.doi.org/10.1213/ane.0b013e3181948a6f
|