 Advances in Bioscience and Biotechnology, 2013, 4, 1-9 ABB http://dx.doi.org/10.4236/abb.2013.411A2001 Published Online November 2013 (http://www.scirp.org/journal/abb/) Synergistic effect of Mucuna pruriens and Withania somnifera in a paraquat induced Parkinsonian mouse model* Jay Prakash1, Satyndra Kumar Yadav1, Shikha Chouhan1, S atya Prakash 2, Surya Pratap Singh1# 1Department of Biochemistry, Faculty of Science, Banaras Hindu University, Varanasi, India 2Biomedical Technology and Cell Therapy Research Laboratory, Department of Biomedical Engineering, Faculty of Medicine, McGill University, Montreal, Canada Email: jaiprakash_biotech@yahoo.co.in, satyndra_yadav@yahoo.co.in, shikhachouhan16@gmail.com, satya.prakash@mcgill.ca, #suryasinghbhu16@gmail.com, #ssingh35@bhu.ac.in Received 2 August 2013; revised 2 September 2013; accepted 21 September 2013 Copyright © 2013 Jay Prakash et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Parkinson’s disease (PD) is a neurodegenerative dis- order characterized by the development of rigidity, resting tremors and postural instability. Recently, the focus of PD’s treatment has shifted towards herbal medicines. Mucuna pruriens (Mp) and Withania som- nifera (Ws) are traditional herbal medicines known to have neuro-protective effects due to the L-DOPA pre- sent in Mp seed powder and withanoloides present in Ws root extract. Here, the synergistic effect of Mp and Ws in Parkinsonian mice induced by chronic exposure to paraquat was evaluated. Co-treatment with Mp and Ws for 9 weeks, significantly decreased the elevated nitrite levels and lipid peroxidation found in Parkinsonian mice. In behavioural tests, Mp and Ws treated mice showed a significant decrease in the time taken to cross a narrow beam, an increase in the time of stay on drum in rotarod test and an im- provement in the hanging time. Furthermore, it was found that the use of Mp and Ws considerably im- proved the tyrosine hydroxylase expression in the substantianigra region of the brain. The results sug- gest that Mp and Ws may provide a platform for fu- ture drug discoveries and novel treatment strategies for PD. Keywords: Withinia somnifera; Mucuna pruriens; Oxidative Stress; Tyrosine Hydroxylase; Parkinson’s Disease; Substantia nigra; Motor Dysfunctions 1. INTRODUCTION Parkinson’s disease (PD) is the second most common neurodegenerative disease ranking next to Alzheimer’s disease [1]. The loss of dopaminergic neurons in the Sub- stantia nigra (SN) pars compacta results in the reduction of the level of dopamine in this region [2]. In modern medicine, Levodopa (L-dopa) is used as a dopamine sup- plement and provides effective treatment against the symptoms of PD [3]. Despite its wide usage, long term administration of L-dopa leads to motor complications called L-dopa induced dyskinesia (LIDS) [4]. Thus, the use of L-dopa as a therapy for PD is now being chal- lenged due to its side effects and extensive research has been opened up for developing new and potent drugs for treating PD. Recently, many epidemiological studies have vali- dated the relationship between PD and environmental factors such as farming [5], drinking water from wells [6], agricultural chemicals, pesticides, and herbicides [7]. Notably, there are a number of pesticides including para- quat (PQ), rotenone and maneb (MB) that can be used to create animal models of PD and to study its mechanism and therapeutic interventions [8-10]. Despite the wide usage of these models, they have limitations to being perfect PD models [11]. As suggested by various studies, the PQ and MB induced PD model is considered to be the best due to the slow progression of the disease [12]. In addition, the generation of free radicals, mitochondrial dysfunctions, microglial activation, increased lipid per- oxidation and nitric oxide levels are well documented in PQ + MB intoxicated mice [13]. *The study was supported financially by Department of Science and Technology (100/(IFD)/1130/2012-2013 SR/CSI/38/2011 (G) DST P- 07-520) New Delhi, India. #Corresponding author. Mucuna pruriens Linn. (Mp) (Fabaceae), commonly known as Kapikacho or Kevach in Hindi, is used as a therapeutic drug in Ayurveda, the traditional medical OPEN ACCESS  J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 2 system of India [14]. It is a climbing legume native to southern China and eastern India [15]. The seed, root and stem of Mp possess valuable medicinal properties [16]. It has been reported to contain analgesic, anti-neoplastic anti-inflammatory, anti-epileptic, anti-microbial and learn- ing and memory enhancing properties [17,18]. Further, some studies, including those conducted in the present laboratory, have demonstrated Mp’s potent neuroprotec- tive properties in PQ-induced Parkinsonian mice [15]. Interestingly, Mp seed extract contains L-DOPA, the dopamine precursor that is used as a therapeutic agent against PD [15,16]. Although the antioxidative properties of Mp are well reported, the exact mechanism of Mp’s antioxidative action remains unknown [19]. Withania somnifera (Ws) is regarded as the wonder shrub of Ayurveda, commonly found on the Indian sub- continent [20]. It is an important indigenous medicinal plant used for the treatment of many diseases including stress, insomnia, anxiety, arthritis and other disorders related to the central nervous system (CNS) such as PD and Alzheimer’s disease [21]. Further, it has a signifi- cant role in the prevention and management of drug ad- diction [22,23]. Using a MPTP-induced PD mouse model, Ws was shown to have antioxidant and free radical scav- enging potential [24]. Further, using a PQ model of PD in mice, our laboratory demonstrated the neuroprotective role of Ws [23]. The objective of the present work is to elucidate the synergistic neuroprotective effects of Ws root extract and Mp seed extract in PQ induced Parkinsonian mice. In the present study, the efficacy of Ws root extract and Mp seed extract in providing protection to dopamirnergic neurons against neurodegenaration caused by oxidative stress in the SN was examined. The neuro-protective activity of Mp and Ws was evaluated through the ex- pression of tyrosine hydroxylase (TH) in the SN of PD mice and also the observation of improvements in motor coordination with narrow beam, hanging and rotarod tests. 2. MATERIAL & METHODS 2.1. Medicinal Plants and Preparation of Extracts Mp seed powder and Ws root powder were purchased from the Ayurveda Pharmacy, Institute of Medical Sci- ence, Banaras Hindu University, Varanasi. To prepare the ethanolic extract of the powdered material, 600 g of each were soaked separately in 1000 mL of ethanol over- night. The extracts were refluxed using a soxhlet appa- ratus and concentrated under reduced pressure. Finally the extracts were stored at 4˚C and suspended in 0.7% carboxy methyl cellulose (CMC, S. D fine chemicals, India) for in vivo assays. 2.2. Animal Treatment Male Swiss albino mice weighing 25 ± 5 g were used in all experiments. Swiss albino mice were obtained from the animal house of the Institute of Medical Science, BHU, Varanasi, India. The study was approved by the Institutional Ethics Committee for use of laboratory ani- mals and all the experimental procedures were performed under the national guidelines on the proper care and use of animals in laboratory research. Animals were main- tained under standard conditions of temperature (22˚C ± 5˚C), humidity (45% - 55%) and light (12:12 h light: dark cycle). The animals were fed with a standard pellet diet and water ad libitum [25]. Animals were randomly divided into three experimen- tal groups (n = 6) as follows: Group I: Control mice. Mice were administered in- traperitoneal (i.p.) injections of saline (0.9%) per day. Group II: Parkinsonian mice. Mice were administered i.p. injections of PQ (10 mg/kg body wt.) twice weekly for 9 weeks. Group III: Treated Mice. In addition to the treatment given to Group II, animals were orally administered al- coholic seed extract of Mp (100 mg/kg) daily. Group IV: Treated mice. In addition to PQ, animals were orally administered alcoholic root extract of Ws (100 mg/kg) daily. Group V: Treated mice. In addition to PQ, animals were orally administered alcoholic seed extract of Mp (50 mg/kg) [26] and alcoholic root extract of Ws (48 mg/ kg) [27] daily. PQ was obtained from Sigma Aldrich (St. Louis, Mo, USA). All the above treatments were carried out for 9 weeks to check disease development and the effect on its treatment. At the end of the experiment, behavioural studies were performed to understand motor skill abnor- malities. 2.3. Neurobehavioral Parameters 2.3.1. Ha ngi ng Test The hanging test was performed as previously described by Mohanasundari et al. [28]. Briefly, mice were placed on a horizontal grid and inverted upside down. The mice were allowed to hang by gripping the grid and the time it took for the mice to fall (hanging time) was recorded for all the treatment groups separately. 2.3.2. Narrow Be am W alki ng Test The narrow beam walking test was performed as previ- ously described by Pisa [29]. In brief, a narrow flat beam was placed at a height of 100 cm from the floor and mice were trained to walk on it. Following training, the mice were tested by recording the time it took to cross the beam. This measure is used to assess the motor coordina- Copyright © 2013 SciRes. OPEN ACCESS  J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 3 tion of the experimental groups. 2.3.3. Rot ar od Test The rotarod test was performed to measure the muscular coordination skills of mice. In this test, the beam re- volves around its longitudinal axis and the mice walk or run forward in synchrony. Mice were trained for 3 con- secutive days before the day of final treatment at a fixed speed for 5 minutes. Mice adjust their posture in re- sponse to a moving speed of 5 rpm and the time it took for the mice to fall from the rotarod was recorded. An average of four experimental readings was calculated for each animal [30]. 2.4. Biochemical Parameters 2.4.1. Lipid Peroxidation Lipid peroxidation in the nigrostraital tissue of the mouse brain was estimated according to the method described previously [31] with slight modifications. Briefly, in or- der to measure the concentration of malondialdehye (MDA) an assay mixture containing 10% tissue homo- genate (0.1 mL) was mixed with 10% SDS solution (0.1 mL) and incubated for 5 minutes at room temperature followed by the addition of 20% acetic acid (0.6 mL) and further incubation for 2 - 5 minutes. Finally 0.8% Thio- barbituric acid TBA (0.6 mL) was added and the reaction mixture was incubated in a boiling water bath for 1 hr. The assay mixture was cooled, centrifuged and absorb- ance of the supernatant was read at 532 nm against con- trol. LPO levels are expressed as nano moles MDA/mg protein. 2.4.2. Nitrite Estimation Nitrite was estimated in the tissue homogenate super- natant as previously described by [32]. Briefly, super- natant of 10% w/v tissue homogenate was incubated with ammonium chloride (0.7 mM) followed by addition of Griess reagent (0.1% N-naphthylethylenediamine and 1% sulfanilamide in 2.5% phosphoricacid). The reaction mixture was incubated for 30 minutes at 37˚C and the absorbance was measured at 540 nm. The nitrite content was calculated using a standard curve for sodium nitrite (10 - 100 μM) in units of μmoles/ml. Following behavioural and biochemical tests further experiments were conducted only with control, PQ and, Mp + Ws and PQ co-treated groups. 2.4.3. Immunoreactivity Immunohistochemical (IHC) staining of tyrosine hy- droxylase (TH)-positive cells in dopaminergic (DAergic) neurons was performed in mice brain sections of control and treated groups using a standard procedure [33]. Bri- efly, perfused mouse brains were post-fixed with para- formaldehyde and cryoprotected in sucrose. Following this, the brain was cut into 20 μm sections using a cryostat. Sections were washed with PBS and incubated in blocking buffer 1 (0.5% H2O2 in methanol and PBS) for 15 minutes, to block endogenous peroxidase activity, followed by incubation in Blocking Buffer 2 (2% normal goat serum, in PBS) for 2 hr and washed again. The sec- tions were then incubated with a primary monoclonal anti-TH antibody (dilution 1:1000, Santa Cruz, USA) at 4˚C for 48 hr and washed again. The sections were incu- bated with a biotinylated secondary antibody (Merck, dilution 1:500) for 2 hr and subsequently treated with a streptavidin peroxidase complex for 30 min. The colour was developed with 3, 3 diaminobenzidine and the sec- tions were permanently mounted with dextrenepthylate xylene (DPX) after dehydration in graded ethanol, as described previously [34]. The mounted sections were examined under bright field microscopy (Nikon, Japan Tokyo, bright field microscope) and images were cap- tured at 10× magnification. Counting of TH-positive cells was done using a standard procedure as described previously by [35]. 2.4.4. Western Blotting Western blot analysis was done as described previously [36]. Briefly, a 10% w/v tissue homogenate was made in lysis buffer (20 mM Tris-HCl, pH 7.4, 2 mM EDTA, 2 mM EGTA, 1 mM PMSF, 30 mM NaF, 30 mM sodium pyrophosphate, 0.1 % SDS, 1% Triton X-100 and pro- tease inhibitor cocktail). Protein content was measured using a standard Bradford Assay [37]. The proteins (80 - 90 g) were separated on a 12% SDS-PAGE and elec- troblotted onto a PVDF membrane. The membrane was incubated with mouse monoclonal antibodies for TH or β-actin (Santa crutz, USA; dilution 1:500) in Tris-buff- ered saline (TBS, pH 7.4) containing 5% non-fat dry milk, overnight at 4˚C. The blot was washed three times with TBS containing 0.2% Tween-20 to remove unbound antibodies. The blot was further incubated with goat anti- mouse IgG peroxidase conjugate (1:2000 dilution) for 2 hr at room temperature. The blot was washed three times with TBS and developed with TMB/H2O2 western blot kits (Bangalore Genei, India). Finally, the developed blots were subjected to densitometric analysis using β-actin as an internal control. 2.5. Statistical Analysis Statistical analysis of the data was performed using one- way analysis of variance (ANOVA) using Graph Pad Instat software. Data were expressed as mean ± standard error mean (SEM) for separate groups. The significance of the data was evaluated by using Tukey’s post hoc analyses and differences were considered statistically Copyright © 2013 SciRes. OPEN ACCESS 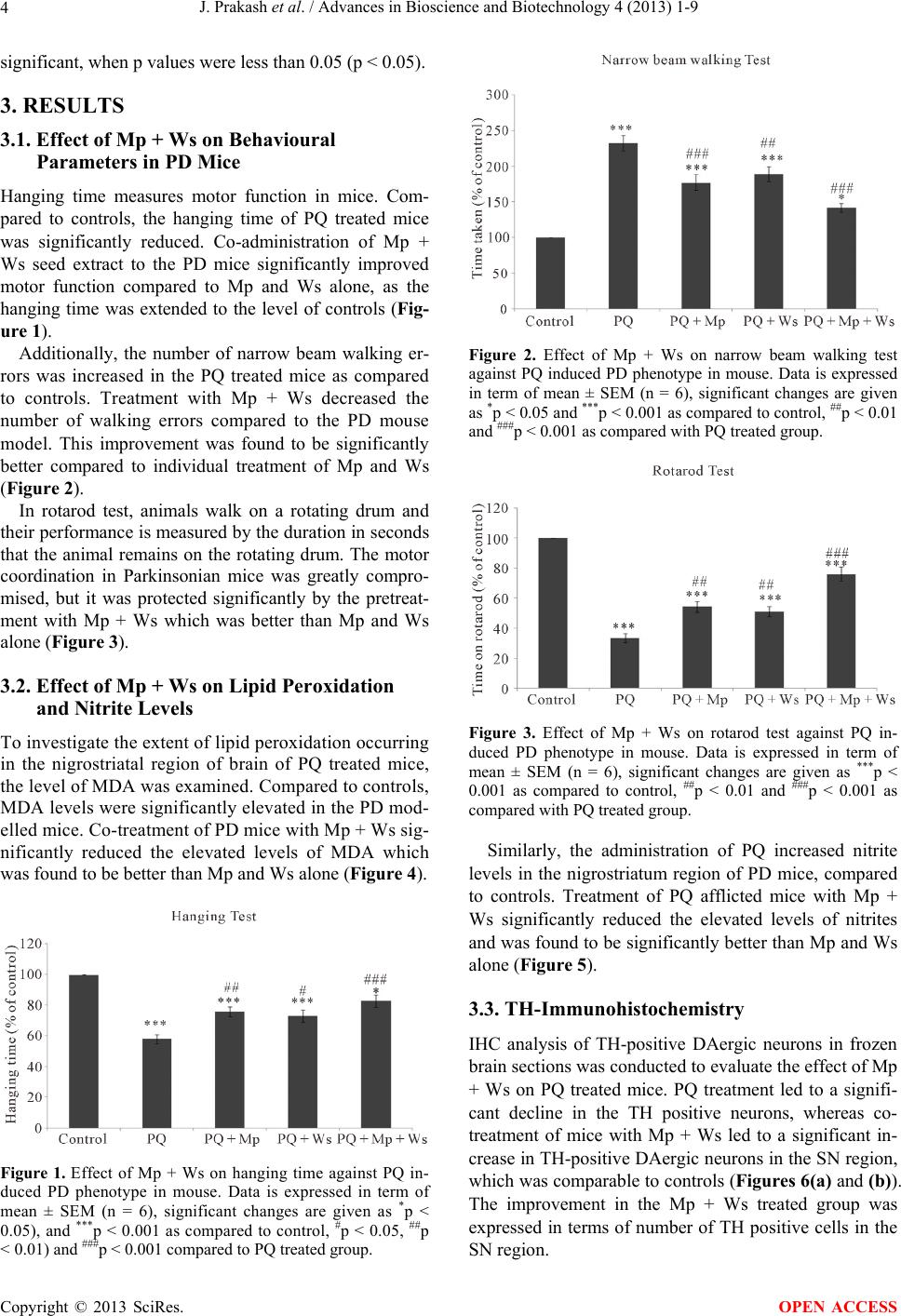 J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 4 significant, when p values were less than 0.05 (p < 0.05). 3. RESULTS 3.1. Effect of Mp + Ws on Behavioural Parameters in PD Mice Hanging time measures motor function in mice. Com- pared to controls, the hanging time of PQ treated mice was significantly reduced. Co-administration of Mp + Ws seed extract to the PD mice significantly improved motor function compared to Mp and Ws alone, as the hanging time was extended to the level of controls (Fig- ure 1). Additionally, the number of narrow beam walking er- rors was increased in the PQ treated mice as compared to controls. Treatment with Mp + Ws decreased the number of walking errors compared to the PD mouse model. This improvement was found to be significantly better compared to individual treatment of Mp and Ws (Figure 2). In rotarod test, animals walk on a rotating drum and their performance is measured by the duration in seconds that the animal remains on the rotating drum. The motor coordination in Parkinsonian mice was greatly compro- mised, but it was protected significantly by the pretreat- ment with Mp + Ws which was better than Mp and Ws alone (Figure 3). 3.2. Effect of Mp + Ws on Lipid Peroxidation and Nitrite Levels To investigate the extent of lipid peroxidation occurring in the nigrostriatal region of brain of PQ treated mice, the level of MDA was examined. Compared to controls, MDA levels were significantly elevated in the PD mod- elled mice. Co-treatment of PD mice with Mp + Ws sig- nificantly reduced the elevated levels of MDA which was found to be better than Mp and Ws alone (Figure 4). Figure 1. Effect of Mp + Ws on hanging time against PQ in- duced PD phenotype in mouse. Data is expressed in term of mean ± SEM (n = 6), significant changes are given as *p < 0.05), and ***p < 0.001 as compared to control, #p < 0.05, ##p < 0.01) and ###p < 0.001 compared to PQ treated group. Figure 2. Effect of Mp + Ws on narrow beam walking test against PQ induced PD phenotype in mouse. Data is expressed in term of mean ± SEM (n = 6), significant changes are given as *p < 0.05 and ***p < 0.001 as compared to control, ##p < 0.01 and ###p < 0.001 as compared with PQ treated group. Figure 3. Effect of Mp + Ws on rotarod test against PQ in- duced PD phenotype in mouse. Data is expressed in term of mean ± SEM (n = 6), significant changes are given as ***p < 0.001 as compared to control, ##p < 0.01 and ###p < 0.001 as compared with PQ treated group. Similarly, the administration of PQ increased nitrite levels in the nigrostriatum region of PD mice, compared to controls. Treatment of PQ afflicted mice with Mp + Ws significantly reduced the elevated levels of nitrites and was found to be significantly better than Mp and Ws alone (Figure 5). 3.3. TH-Immunohistochemistry IHC analysis of TH-positive DAergic neurons in frozen brain sections was conducted to evaluate the effect of Mp + Ws on PQ treated mice. PQ treatment led to a signifi- cant decline in the TH positive neurons, whereas co- treatment of mice with Mp + Ws led to a significant in- crease in TH-positive DAergic neurons in the SN region, which was comparable to controls (Figures 6(a) and (b)). The improvement in the Mp + Ws treated group was expressed in terms of number of TH positive cells in the SN region. Copyright © 2013 SciRes. OPEN ACCESS 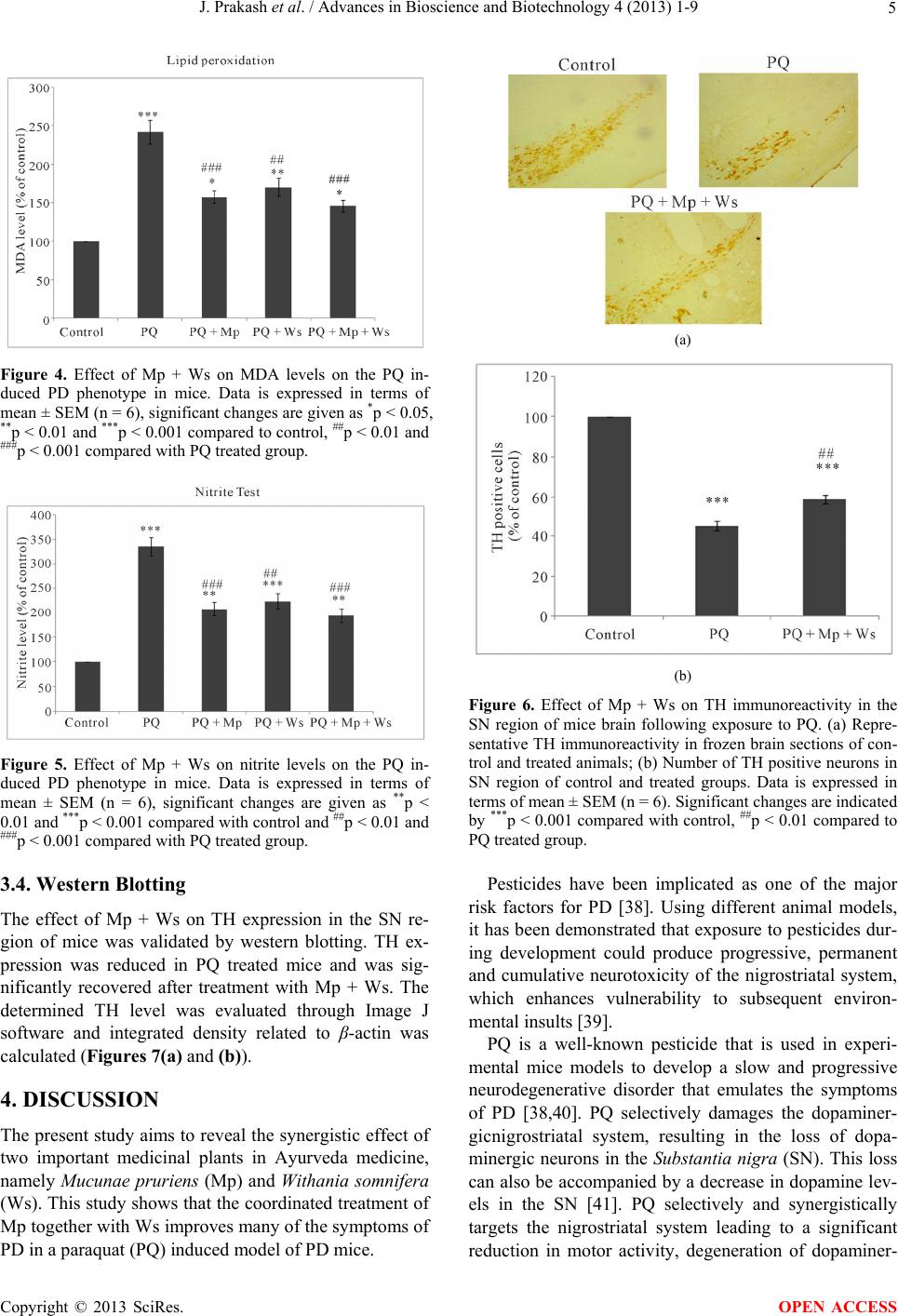 J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 5 Figure 4. Effect of Mp + Ws on MDA levels on the PQ in- duced PD phenotype in mice. Data is expressed in terms of mean ± SEM (n = 6), significant changes are given as *p < 0.05, **p < 0.01 and ***p < 0.001 compared to control, ##p < 0.01 and ###p < 0.001 compared with PQ treated group. Figure 5. Effect of Mp + Ws on nitrite levels on the PQ in- duced PD phenotype in mice. Data is expressed in terms of mean ± SEM (n = 6), significant changes are given as **p < 0.01 and ***p < 0.001 compared with control and ##p < 0.01 and ###p < 0.001 compared with PQ treated group. 3.4. Western Blotting The effect of Mp + Ws on TH expression in the SN re- gion of mice was validated by western blotting. TH ex- pression was reduced in PQ treated mice and was sig- nificantly recovered after treatment with Mp + Ws. The determined TH level was evaluated through Image J software and integrated density related to β-actin was calculated (Figures 7(a) and (b)). 4. DISCUSSION The present study aims to reveal the synergistic effect of two important medicinal plants in Ayurveda medicine, namely Mucunae pruriens (Mp) and Withania somnifera (Ws). This study shows that the coordinated treatment of Mp together with Ws improves many of the symptoms of PD in a paraquat (PQ) induced model of PD mice. (a) (b) Figure 6. Effect of Mp + Ws on TH immunoreactivity in the SN region of mice brain following exposure to PQ. (a) Repre- sentative TH immunoreactivity in frozen brain sections of con- trol and treated animals; (b) Number of TH positive neurons in SN region of control and treated groups. Data is expressed in terms of mean ± SEM (n = 6). Significant changes are indicated by ***p < 0.001 compared with control, ##p < 0.01 compared to PQ treated group. Pesticides have been implicated as one of the major risk factors for PD [38]. Using different animal models, it has been demonstrated that exposure to pesticides dur- ing development could produce progressive, permanent and cumulative neurotoxicity of the nigrostriatal system, which enhances vulnerability to subsequent environ- mental insults [39]. PQ is a well-known pesticide that is used in experi- mental mice models to develop a slow and progressive neurodegenerative disorder that emulates the symptoms of PD [38,40]. PQ selectively damages the dopaminer- gicnigrostriatal system, resulting in the loss of dopa- minergic neurons in the Substantia nigra (SN). This loss can also be accompanied by a decrease in dopamine lev- els in the SN [41]. PQ selectively and synergistically targets the nigrostriatal system leading to a significant reduction in motor activity, degeneration of dopaminer- Copyright © 2013 SciRes. OPEN ACCESS 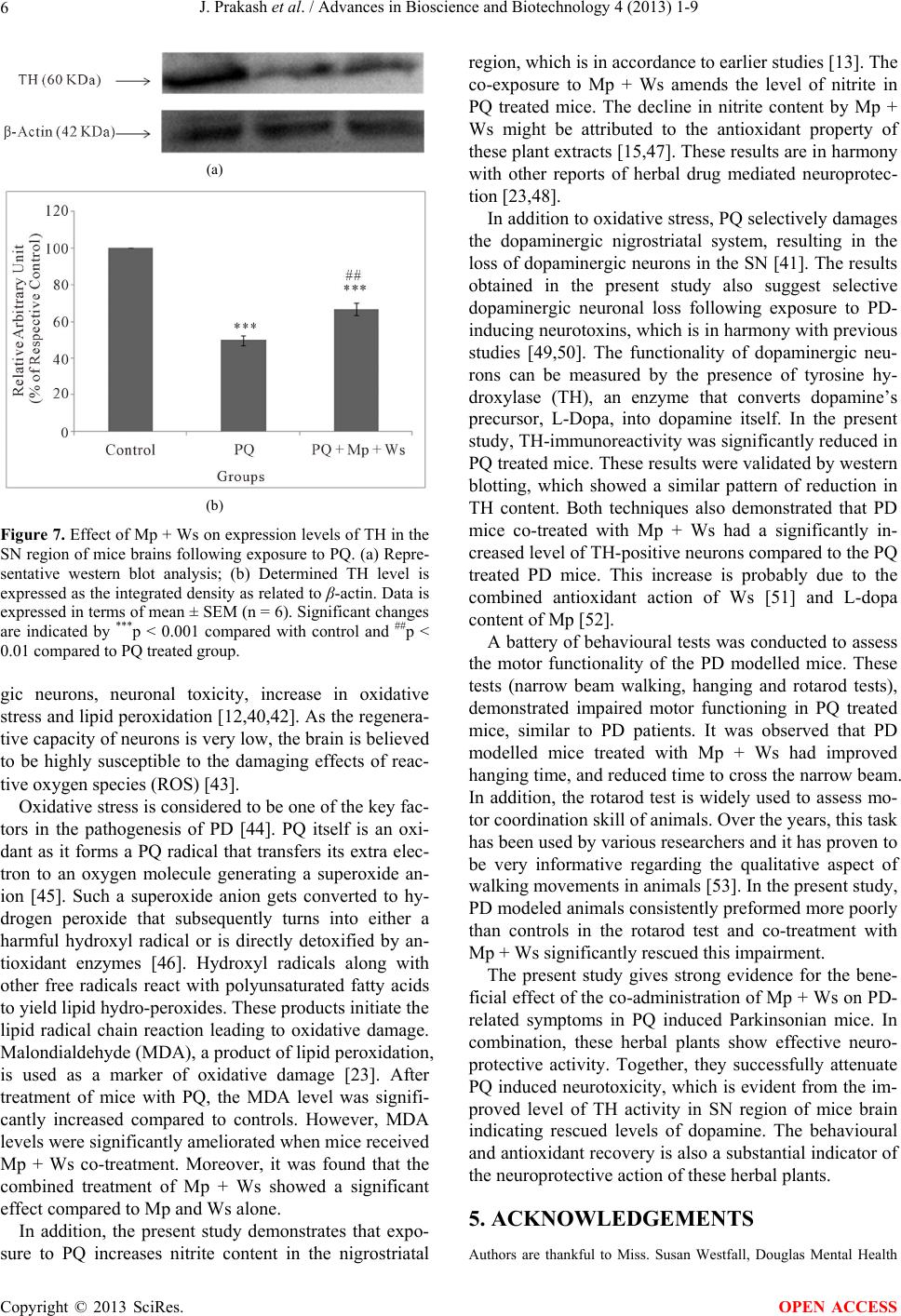 J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 6 (a) (b) Figure 7. Effect of Mp + Ws on expression levels of TH in the SN region of mice brains following exposure to PQ. (a) Repre- sentative western blot analysis; (b) Determined TH level is expressed as the integrated density as related to β-actin. Data is expressed in terms of mean ± SEM (n = 6). Significant changes are indicated by ***p < 0.001 compared with control and ##p < 0.01 compared to PQ treated group. gic neurons, neuronal toxicity, increase in oxidative stress and lipid peroxidation [12,40,42]. As the regenera- tive capacity of neurons is very low, the brain is believed to be highly susceptible to the damaging effects of reac- tive oxygen species (ROS) [43]. Oxidative stress is considered to be one of the key fac- tors in the pathogenesis of PD [44]. PQ itself is an oxi- dant as it forms a PQ radical that transfers its extra elec- tron to an oxygen molecule generating a superoxide an- ion [45]. Such a superoxide anion gets converted to hy- drogen peroxide that subsequently turns into either a harmful hydroxyl radical or is directly detoxified by an- tioxidant enzymes [46]. Hydroxyl radicals along with other free radicals react with polyunsaturated fatty acids to yield lipid hydro-peroxides. These products initiate the lipid radical chain reaction leading to oxidative damage. Malondialdehyde (MDA), a product of lipid peroxidation, is used as a marker of oxidative damage [23]. After treatment of mice with PQ, the MDA level was signifi- cantly increased compared to controls. However, MDA levels were significantly ameliorated when mice received Mp + Ws co-treatment. Moreover, it was found that the combined treatment of Mp + Ws showed a significant effect compared to Mp and Ws alone. In addition, the present study demonstrates that expo- sure to PQ increases nitrite content in the nigrostriatal region, which is in accordance to earlier studies [13]. The co-exposure to Mp + Ws amends the level of nitrite in PQ treated mice. The decline in nitrite content by Mp + Ws might be attributed to the antioxidant property of these plant extracts [15,47]. These results are in harmony with other reports of herbal drug mediated neuroprotec- tion [23,48]. In addition to oxidative stress, PQ selectively damages the dopaminergic nigrostriatal system, resulting in the loss of dopaminergic neurons in the SN [41]. The results obtained in the present study also suggest selective dopaminergic neuronal loss following exposure to PD- inducing neurotoxins, which is in harmony with previous studies [49,50]. The functionality of dopaminergic neu- rons can be measured by the presence of tyrosine hy- droxylase (TH), an enzyme that converts dopamine’s precursor, L-Dopa, into dopamine itself. In the present study, TH-immunoreactivity was significantly reduced in PQ treated mice. These results were validated by western blotting, which showed a similar pattern of reduction in TH content. Both techniques also demonstrated that PD mice co-treated with Mp + Ws had a significantly in- creased level of TH-positive neurons compared to the PQ treated PD mice. This increase is probably due to the combined antioxidant action of Ws [51] and L-dopa content of Mp [52]. A battery of behavioural tests was conducted to assess the motor functionality of the PD modelled mice. These tests (narrow beam walking, hanging and rotarod tests), demonstrated impaired motor functioning in PQ treated mice, similar to PD patients. It was observed that PD modelled mice treated with Mp + Ws had improved hanging time, and reduced time to cross the narrow beam. In addition, the rotarod test is widely used to assess mo- tor coordination skill of animals. Over the years, this task has been used by various researchers and it has proven to be very informative regarding the qualitative aspect of walking movements in animals [53]. In the present study, PD modeled animals consistently preformed more poorly than controls in the rotarod test and co-treatment with Mp + Ws significantly rescued this impairment. The present study gives strong evidence for the bene- ficial effect of the co-administration of Mp + Ws on PD- related symptoms in PQ induced Parkinsonian mice. In combination, these herbal plants show effective neuro- protective activity. Together, they successfully attenuate PQ induced neurotoxicity, which is evident from the im- proved level of TH activity in SN region of mice brain indicating rescued levels of dopamine. The behavioural and antioxidant recovery is also a substantial indicator of the neuroprotective action of these herbal plants. 5. ACKNOWLEDGEMENTS Authors are thankful to Miss. Susan Westfall, Douglas Mental Health Copyright © 2013 SciRes. OPEN ACCESS  J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 7 University Institute, Montreal, QC, Canada and Department of Neu- rology and Neurosurgery, McGill University, Montreal, QC, Canada for her constructive suggestions while writing the paper. Authors wish to acknowledge Dr. T. D. Singh, Associate Professor, Department of Medicinal Chemistry, IMS, BHU, for helping us to prepare ethanolic root extract of Ws in his laboratory. The authors sincerely thank Indian Council of Medical Research (ICMR), New Delhi, India for providing research fellowship to Jay Prakash, Council of Scientific and Industrial Research (CSIR), New Delhi, India for providing research fellowship to Satyndra Kumar Yadav and Department of Science and Technology (DST), New Delhi, India for providing fellowship to Shikha Chouhan. REFERENCES [1] Dauer, W. and Przedborski, S. (2003) Parkinson’s disease: Mechanisms and models. Neuron, 39, 889-909. http://dx.doi.org/10.1016/S0896-6273(03)00568-3 [2] Singh, C., Ahmad, I. and Kumar, A. (2007) Pesticides and metals induced Parkinson’s disease: involvement of free radicals and oxidative stress. Cell and Molecular Bi- ology, 30, 19-28. [3] Maheswari, T., Vijayraja, D., Kundhavai, N.R., Kalaivani, K., Rajasankar, S., Tamilselvama, K. and Manivasagama, T. (2010) Synergistic neuropreventive effect of Withania somnifera root powder and Mucuna pruriens seed powder in parkinsonic mice model. Journal of Herbal Medicine and Toxicology, 4, 63-69. [4] Borah, A. and Mohanakumar, K.P. (2012) L-DOPA in- duced-endogenous 6-hydroxydopamine is the cause of aggravated dopaminergic neurodegeneration in Parkin- son’s disease patients. Medical Hypotheses, 79, 271-273. http://dx.doi.org/10.1016/j.mehy.2012.05.008 [5] Cannon, J.R. and Greenamyre, J.T. (2011) The role of environmental exposures in neurodegeneration and neu- rodegenerative diseases. Toxicological Sciences, 124, 225- 250. http://dx.doi.org/10.1093/toxsci/kfr239 [6] Gatto, N.M., Cockburn, M., Bronstein, M.A.D. and Ritz, B. (2009) Well-water consumption and Parkinson’s dis- ease in rural California. Environmental Health Perspec- tives, 117, 1912-1918. [7] Semchuk, K.M., Love, E.J. and Lee, R.G. (1992) Parkin- son’s disease and exposure to agricultural work and pes- ticide chemicals. Neurology, 42, 1328-1335. http://dx.doi.org/10.1212/WNL.42.7.1328 [8] Duty, S. and Jenner, P. (2011) Animal models of Parkin- son’s disease: A source of novel treatments and clues to the cause of the disease. British Journal of Pharmacology, 164, 1357-1391. http://dx.doi.org/10.1111/j.1476-5381.2011.01426.x [9] Singh, S., Singh, K., Patel, S., Patel, D.K., Singh, C., Nath, C. and Singh, M.P. (2008) Nicotine and caffeine- mediated modulation in the expression of toxicant re- sponsive genes and vesicular monoamine transporter-2 in 1-methyl 4-phenyl-1,2,3,6-tetrahydropyridineinduced Par- kinson’s disease phenotype in mouse. Brain Research, 1207, 193-206. http://dx.doi.org/10.1016/j.brainres.2008.02.023 [10] Yadav, S., Dixit, A., Agrawal, S., Singh, A., Srivastava, G., Singh, A.K., Srivastava, P.K., Prakash, O. and Singh, M.P. (2012) Rodent models and contemporary molecular techniques: Notable feats yet incomplete explanations of Parkinson’s disease pathogenesis. Molecular Neurobiol- ogy, 46, 495-512. http://dx.doi.org/10.1007/s12035-012-8291-8 [11] Tiwari, M.N., Singh, A.K., Ahmad, I., Upadhyay, G., Singh, D., Patel, D.K., Singh, C., Prakash, O. and Singh, M.P. (2010) Effects of cypermethrin on monoamine transporters, xenobiotic metabolizing enzymes and lipid peroxidation in the rat nigrostriatal system. Free Radical Research, 44, 1416-1424. http://dx.doi.org/10.3109/10715762.2010.512041 [12] Patel, S., Singh, V., Kumar, A. Gupta, Y.K. and Singh, M.P. (2006) Status of antioxidant defence system and ex- pression of toxicant responsive genes in striatum of maneb- and paraquat induced Parkinson’s disease pheno- type in mouse: mechanism of neurodegeneration. Brain Research, 1081, 9-18. http://dx.doi.org/10.1016/j.brainres.2006.01.060 [13] Gupta, S.P., Patel, S., Yadav, S., Singh, A.K., Singh, S. and Singh, M.P. (2010) Involvement of nitric oxide in Maneb and paraquat-induced Parkinson’s disease phe- notype in mouse: Is there any link with lipid peroxidation. Neurochemical Research, 35, 1206-1213. http://dx.doi.org/10.1007/s11064-010-0176-5 [14] Ngatchic, J.T., Sokeng, S.D., Njintang, N.Y., Maoun- dombaye, T., Oben, J. and Mbofung, C.M. (2013) Eva- luation of some selected blood parameters and histopa- thology of liver and kidney of rats fed protein-substituted mucuna flour and derived protein rich product. Food and Chemical Toxicology, 57, 46-53. http://dx.doi.org/10.1016/j.fct.2013.02.045 [15] Yadav, S.K., Prakash, J., Chouhan, S. and Singh, S.P. (2013) Mucuna pruriens seed extract reduces oxidative stress in nigrostriatal tissue and improves neurobehav- ioral activity in paraquat-induced Parkinsonian mouse model. Neurochemistry International, 62, 1039-1047. http://dx.doi.org/10.1016/j.neuint.2013.03.015 [16] Suresh, S., Prithiviraj, E., Lakshmi, N.V., Ganesh, M.K., Ganesh, L. and Prakash, S. (2012) Effect of Mucunapru- riens (Linn.) on mitochondrial dysfunction and DNA damage in epididymal sperm of streptozotocin induced diabetic rat. Journal of Ethnopharmacology, 145, 32-41. http://dx.doi.org/10.1016/j.jep.2012.10.030 [17] Adepoju, G.K.A. and Odubena, O.O. (2009) Effect of Mucuna pruriens on some haematological and biochemi- cal parameters. Journal of Medicinal Plants Research, 3, 73-76. [18] Sathiyanarayanan, L. and Arulmozhi, S. (2007) Mucuna- pruriens L. A comprehensive review. Pharmacognosy Re- views, 1, 157-162. [19] Tripathi, Y.B. and Upadhyay, A.K. (2001) Antioxidant property of Mucuna pruriens L. Current Science, 80, 1-11. [20] Widodo, N., Kaur, K., Shrestha, B.G., Takagi, Y., Ishii, T., Wadhwa, R. and Kaul S.C. (2007) Selective killing of cancer cells by leaf extract of Ashwagandha: Identifica- tion of a tumor-inhibitory factor and the first molecular Copyright © 2013 SciRes. OPEN ACCESS  J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 8 insights to its effect. Clinical Cancer Research, 13, 2298- 2306. http://dx.doi.org/10.1158/1078-0432.CCR-06-0948 [21] Kataria, H., Wadhwa, R. and Kaul, S.C. (2013) Withani- asomnifera water extract as a potential candidate for dif- ferentiation based therapy of human neuroblastomas. PLoS One, 8, e55316. http://dx.doi.org/10.1371/journal.pone.0055316 [22] Kulkarni, S.K. and Dhir, A. (2008) Withaniasomnifera: An Indian ginseng. Progress in Neuro-Psychopharma- cology & Biological Psychiatry, 32, 1093-1105. [23] Prakash, J., Yadav, S.K., Chouhan, S. and Singh, S.P. (2013) Neuroprotective role of Withaniasomnifera root extract in maneb-paraquat induced mouse model of Parkinsonism. Neurochemical Research, 38, 972-980. http://dx.doi.org/10.1007/s11064-013-1005-4 [24] Rajasankara, S., Manivasagam, T., Sankar, V., Prakash, S., Muthusamy, R., Krishnamurti, A. and Surendran, S. (2009). Withaniasomnifera root extract improves cate- cholamines and physiological abnormalities seen in a Parkinson’s disease model mouse. Journal of Ethnophar- macology, 125, 369-373. http://dx.doi.org/10.1016/j.jep.2009.08.003 [25] Chouhan, S., Prakash, J., Yadav, S.K., Agrawal, N.K. and Singh, S.P. (2013) Effect of bisphenol A on fertility of male mice. Journal of Scientific Research, 57, 77-84. [26] Bhattacharya, A., Ghosal, S. and Bhattacharya, S.K. (2001) Anti-oxidant effect of Withaniasomniferaglycowithano- lides in chronic footshock stress-induced perturbations of oxidative free radical scavenging enzymes and lipid per- oxidation in rat frontal cortex and striatum. Journal of Ethnopharmacology, 74, 1-6. http://dx.doi.org/10.1016/S0378-8741(00)00309-3 [27] Kasture, S., Pontis, S., Pinna, A., Schintu, N., Spina, L., Longoni, R., Simola N., Ballero M. and Morelli, M. (2009) Assessment of symptomatic and neuroprotective efficacy of Mucuna pruriens seed extract in rodent model of Park- inson’s disease. Neurotoxicity Research, 15, 111-122. http://dx.doi.org/10.1007/s12640-009-9011-7 [28] Mohanasundari, M., Srinivasan, M.S., Sethupathy, S. and Sabesan, M. (2006) Enhanced neuroprotective effect by combination of bromocriptine and Hypericumperforatum extract against MPTP-induced neurotoxicity in mice. Jour- nal of the Neurological Sciences, 249, 140-144. http://dx.doi.org/10.1016/j.jns.2006.06.018 [29] Pisa, M. (1998) Regional specialization of motor func- tions in the rat striatum: implications for the treatment of Parkinsonism. Progress in Neuro-Psychopharmacology & Biological Psychiatry, 12, 217-224. [30] Manna, S., Bhattacharyya, D., Mandal, T.K. and Dey, S. (2006) Neuropharmacological effects of deltamethrin in rats. Journal of Veterinary Science, 7, 133-136. http://dx.doi.org/10.4142/jvs.2006.7.2.133 [31] Ohkawa, H., Ohishi, N. and Yag, K. (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95, 351-358. http://dx.doi.org/10.1016/0003-2697(79)90738-3 [32] Granger, D.L., Taintor, R.R., Boockvar, K.S. and Hibbs, J.B. (1996) Measurement of nitrate and nitrite in biologi- cal samples using nitrate reductase and Greiss reaction. Methods in Enzymology, 268, 142-151. http://dx.doi.org/10.1016/S0076-6879(96)68016-1 [33] Gorbatyuk, O.S., Li, S., Sullivan, L.F., Chen, W., Kon- drikova, G., Manfredsson, F.P., Mandel, R.J. and Mu- zyczka, N. (2008) The phosphorylation state of Ser-129 in human alpha-synuclein determines neurode-generation in a rat model of Parkinson disease. Proceedings of the National Academy of Sciences of the USA, 105, 763-768. http://dx.doi.org/10.1073/pnas.0711053105 [34] Singh, S., Singh, K., Patel, D.K., Singh, C., Nath, C., Singh, V.K., Singh, R.K. and Singh, M.P. (2009) The ex- pression of CYP2D22, an ortholog of human CYP2D6, in mouse striatum and its modulation in 1-methyl 4-phenyl- 1,2,3,6-tetrahydropyridine induced Parkinson’s disease phe- notype and nicotine mediated neuroprotection. Rejuvena- tion Research, 12, 185-197. http://dx.doi.org/10.1089/rej.2009.0850 [35] Mochizuki, H., Hayakawa, H., Migit, M., Shibata, M., Tanaka, R., Suzuki, A., Shimo, N.Y., Urabe, T., Yamada, M., Tamayos, K., Shimada, T., Miura, M. and Mizuno, Y. (2001) An AAV-derived apaf-1 dominant negative in- hibitor prevents MPTP toxicity as antiapoptotic gene therapy for Parkinson’s disease. Proceedings of the Na- tional Academy of Sciences of the USA, 98, 10918-10923. http://dx.doi.org/10.1073/pnas.191107398 [36] Martin, P.Y., Bianchi, M., Roger, F., Niksic, L., and Fé- raille, E. (2002) Arginine vasopressin modulates expres- sion of neuronal NOS in rat renal medulla. American Journal of Physiology, 283, 559-568. [37] Bradford, M.M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utiliz- ing the principle of protein-dye binding. Analytical Bio- chemistry, 72, 248-254. http://dx.doi.org/10.1016/0003-2697(76)90527-3 [38] Mccormack, A.L., Thiruchelvam, M., Manning-Bog, A.B., Thiffault, C., Langston, J.W., Cory-Slechta, D.A., and Di Monte, D.A. (2002) Environmental risk factors and Park- inson’s disease: Selective degeneration of nigral dopami- nergic neurons caused by the herbicide paraquat. Neuro- biology of Disease, 10, 119-127. http://dx.doi.org/10.1006/nbdi.2002.0507 [39] Cory-Slechta, D.A., Thiruchelvam, M., Barlow, B.K. and Richfield, E.K. (2005) Developmental pesticide models of the Parkinson disease phenotype. Environmental Health Perspectives, 113, 1263-1270. http://dx.doi.org/10.1289/ehp.7570 [40] Thiruchelvam, M., Brockel, B.J., Richfield, E.K., Baggs, R.B., and Cory-Slechta, D.A. (2000) Potentiated and pre- ferential effects of combined paraquat and maneb on ni- grostriatal dopamine systems: Environmental risk factors for Parkinson’s disease? Brain Research, 873, 225-234. http://dx.doi.org/10.1016/S0006-8993(00)02496-3 [41] Kumar, A., Ahmad, I., Shukla, S., Singh, B.K., Patel, D.K., Pandey, H.P, and Singh, C. (2010) Effect of zinc and PQ co-exposure on neurodegeneration: Modulation of oxidative stress and expression of metallothioneins, toxicant responsive and transporter genes in rats. Free Radical Research, 44, 950-965. Copyright © 2013 SciRes. OPEN ACCESS  J. Prakash et al. / Advances in Bioscience and Biotechnology 4 (2013) 1-9 Copyright © 2013 SciRes. 9 OPEN ACCESS [42] Patel, S., Singh, K., Singh, S. and Singh, M.P. (2008) Gene expression profiles of mouse striatum in control and maneb+paraquat-induced Parkinson’s disease phenotype: Validation of differentially expressed energy metaboliz- ing transcripts. Molecular Biotechnology, 40, 59-68. http://dx.doi.org/10.1007/s12033-008-9060-9 [43] Uttara, B., Singh, A.V., Zamboni, P. and Mahajan, R.T. (2009) Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant thera- peutic options. Current Neuropharmacology, 7, 65-74. http://dx.doi.org/10.2174/157015909787602823 [44] Farooqui, T. and Farooqui, A.A. (2011) Lipid-mediated oxidative stress and inflammation in the pathogenesis of Parkinson’s disease. Parkinson’s Disease, 2011, 1-9. [45] Kappus, H. (1986) Overview of enzyme systems involved in bioreduction of drugs and in redox cycling. Biochemi- cal Pharmacology, 35, 1-6. http://dx.doi.org/10.1016/0006-2952(86)90544-7 [46] Beckman, J.S., Beckman, T.W., Chen, J., Marshall, P.A., and Freeman, B.A. (1990) Apparent hydroxyl radical pro- duction by peroxynitrite: Implications for endothelial in- jury from nitric oxide and superoxide. Proceedings of the National Academy of Sciences of the USA, 87, 1620-1624. http://dx.doi.org/10.1073/pnas.87.4.1620 [47] Bhatnagar, M., Sharma, D. and Salvi, M. (2009) Neuro- protective effects of Withaniasomniferadunal: A possible mechanism. Neurochemical Research, 34, 1975-1983. http://dx.doi.org/10.1007/s11064-009-9987-7 [48] Yadav, S., Gupta, S.P., Srivastava, G., Srivastava, P.K., and Singh, M.P. (2011) Role of secondary mediators in caffeine-mediated neuroprotection in maneb- and para- quat-induced Parkinson’s disease phenotype in the mouse. Neurochemical Research, 37, 875-884. http://dx.doi.org/10.1007/s11064-011-0682-0 [49] Dixit, A., Srivastava, G., Verma, D., Mishra, M., Singh, P.K., Prakash, O. and Singh, M.P. (2013) Minocycline, levodopa and MnTMPyP induced chamges in the mito- chondrial protein profile of MPTP and maneb and pa- raquat mice models of Parkinson’s disease. Biochimica et Biophysica Acta, 1832, 1227-1240. [50] Somayajulu-Nit, M., Sandhu, J.K., Cohen, J., Sikorska, M., Sridhar, T.S., Matei, A., Borowy-Borowski, H., and Pandey, S. (2009) Paraquatinduces oxidative stress, neu- ronal loss in substantianigra region and parkinsonism in adult rats: Neuroprotection and amelioration of symp- toms by water soluble formulation of coenzyme Q10. BMC Neuroscience, 10, 1-12. [51] Ahmad, M., Saleem, S., Ahmad, A.S., Ansari, M.A., Yousuf, S., Hoda, M.N. and Islam, F. (2005) Neuropro- tective effects of Withaniasomnifera on 6-hydroxydopa- mine induced Parkinsonism in rats. Human & Experimen- tal Toxicology, 24, 137-147. http://dx.doi.org/10.1191/0960327105ht509oa [52] Dhanasekaran, M., Tharakan, B., and Manyam, B.V. (2008) Antiparkinson drug—Mucuna pruriens shows antioxidant and metal chelating activity. Phytotherapy Research, 22, 6-11. http://dx.doi.org/10.1002/ptr.2109 [53] Whishaw, I.Q., Li, K., Whishaw, P.A., Gorny, B. and Metz, G.A. (2008) Use of rotorod as a method for the qualitative analysis of walking in rat. Journal of Visual- ized Experiments, 22, 1030.
|