 Advances in Chemical Engineering and Science, 2013, 3, 20-26 doi:10.4236/aces.2013.34B004 Published Online October 2013 (http://www.scirp.org/journal/aces) Precipitation and Crystallization of Struvite from Synthetic Wastewater under Stoichiometric Conditions Anna Kozik1, Nina Hutnik1, Krzysztof Piotrowski2*, Agata Mazienczuk1, Andrzej Matynia1 1Wroclaw University of Technology, Faculty of Chemistry, Wybrzeze Wyspianskiego, Wroclaw, Poland 2Silesian University of Technology, Department of Chemical & Process Engineering, M. Strzody Gliwice, Poland Email: *krzysztof.piotrowski@polsl.pl Received June, 2013 ABSTRACT Phosphate (V) ions were continuously removed from synthetic wastewater containing inorganic impurities using mag- nesium and ammonium ions. The product was magnesium ammonium phosphate (V) hexahydrate, struvite, MgNH4PO4 × 6H2O. Research ran in stoichiometric conditions in DT MSMPR type crystallizer with internal circulation of suspen- sion. Increase in process environment pH from 9 to 11 resulted in 3-time decrease of mean struvite crystals size (from 40.1 to 12.6 m). Elongation of mean residence time of suspension in a crystallizer up to 3600 s resulted in improve- ment of the product quality. Mean size of struvite crystals enlarged up to 50.2 m. Based on kinetic calculations results (SIG MSMPR model) it was concluded, that linear struvite crystal growth rate varied within 5.04 × 10–9 – 1.69 × 10–8 m/s range, whereas nucleation rate within 1.4 × 107 – 1.7 × 1010 1/(s m3) limits. In solid product, besides struvite, also all impurities present in wastewater were identified analytically as hydroxides, phosphates and other salts. Keywords: Struvite; Precipitation; Continuous Reaction Crystallization; Phosphate(V) Ions; Impurity Ions; Continuous DT MSMPR Crystallizer; Crystal Size Distribution; Kinetics 1. Introduction Inexpensive and easily available, secondary phosphorus sources can be industrial, municipal liquid wastes, liquid manure, urine and other waste solutions containing phosphate (V) ions [1]. Technological idea of recovery from them some useful phosphorus compounds is based on reaction crystallization of sparingly soluble phosphate salts, mainly struvite MgNH4PO4 × 6H2O (MAP) [2, 3]. Controlled reaction crystallization of struvite is not sim- ple process. Its course and final results are strongly af- fected by temperature, concentrations of main reacting substances (phosphate(V), magnesium and ammonium ions), process environment’s pH, reaction crystallization of not only expected product, but also co-precipitating sparingly soluble salts or/and hydroxides of some metals present in wastewaters [1, 4-6]. Process success depends also on the continuous crystallizer construction and its work mode/parameters, including: mean residence time of suspension in working volume of the crystallizer, in- tensity of mixing and circulation of suspension inside the apparatus, inlet places and contact method of the reacting substances, etc. [1, 7-9]. The experimental test results concerning recovery of phosphate(V) ions from solution of similar chemical composition to agricultural, animal breeding or mineral fertilizer industry wastewater are presented. Precipitation of phosphate (V) ions with magnesium and ammonium ions in alkaline aqueous environment, followed by stru- vite mass crystallization was carried out in a laboratory continuous DT MSMPR (Draft Tube, Mixed Suspension Mixed Product Removal) type crystallizer with propeller stirrer. Crystallizer was provided with feed solution of determined chemical composition, prepared earlier in a mixer from chemically pure substances and deionized water. The solution contained, besides phosphate (V) ions, also: magnesium, ammonium, aluminium, calcium, copper, iron, potassium, zinc and nitrate (V) ions. In this environment also sodium and chloride ions were present from dissolved in water crystalline salts from which syn- thetic wastewater was made. Concentration of phosphate (V) ions was assumed to be 1.0 mass %. This concentration is ca. 2 - 5-time higher than reported in real wastewaters. However, one can thus compare the presented results with the data concerning struvite production process from solutions containing phosphate (V) ions only, of concentration 1.0 mass % [10]. The research was carried out at molar ratio of the substrates PO4 3– : Mg2+ : NH4 + as 1 : 1 : 1 in temperature 298 K. Influence of pH (from 9 to 11) and mean residence time of suspension in a crystal- lizer (from 900 to 3600 s) on product crystal size distri- *Corresponding author. Copyright © 2013 SciRes. ACES  A. KOZIK ET AL. 21 butions, mean size and homogeneity within product population were investigated. Linear growth rate of stru- vite crystals and its nucleation rate were estimated. Cal- culations were based on the simplified model of mass crystallization kinetics in MSMPR crystallizer – SIG (Size Independent Growth) model. 2. Material and Methods Photo of experimental plant is shown in Figure 1. It is fully automated. Continuous Bioengineering RALF plus Solo plan. Steering, control and acquisition of measure- ment data streams were carried out with the use of PC computer (driven by BioScadaLab software). Process ran in DT MSMPR type crystallizer of working volume Vw 0.6 dm3 (total volume Vt 1.3 dm3). Crystallizer, made of glass, was equipped with heating/cooling coat providing stable process temperature, as well as with the system delivering compressed air, required for stripping of ab- sorbed CO2 and oxidation of possible organic substances present in struvite continuous reaction crystallization environment. Crystallizer diameter was d 100 mm, its working part height hw 90 mm, total height ht 200 mm. Inside the crystallizer circulation profile (DT, Draft Tube , ddt 52 mm, hdt 50 mm) was installed, inside which four- paddle propeller stirrer of diameter dm 48 mm operated. Mixer speed, process temperature, inlet stream of air, inflows of feed and alkalising solution, as well as out- flow of product crystal suspension from the crystallizer were strictly controlled and adjusted by computer. Synthetic wastewater feeding the crystallizer was aqueous solution of ammonium di hydrogen phosphate (V) NH4H2PO4, magnesium chloride MgCl2×6H2O, chlorides of impurity cations (AlCl3×6H2O, CaCl2×2H2O, CuCl2 × 2H2O, FeCl3×6H2O, KCl and ZnCl2), as well as sodium salt of impurity anion (NaNO3). The mixture was pre- pared in external mixer using crystalline substances (p.a., POCh Gliwice, Poland) and deionized water (Barnstead– NANOpure DIamond). Concentrations of main substrates: phosphate (V) ions (1.0 mass %) [10], magnesium (0.256 mass %) and ammonium (0.190 mass %) resulted from their assumed molar ratio 1 : 1 : 1. Detailed chemi- cal composition of the feed was presented in Table 1. This solution was continuously introduced into circula- tion profile (mixer speed: 4.0 1/s; suspension movement – downward). Between crystallizer body and circulation profile (suspension movement – upward) aqueous solu- tion of sodium hydroxide, of concentration 3 mass % NaOH was dosed in amount providing the assumed, con- trolled pH of continuous struvite reaction crystallization environment. Tests ran in temperature 298 ±0.2 K as- suming pH 9, 10 or 11 (±0.1) and mean residence time of suspension in a crystallizer 900, 1800 or 3600 (±20) s. Compressed air flow was established on the 100 Ndm3/h level (pressure ca. 2.5 bar). After stabilisation in a crys- tallizer the assumed parameter values, process in a steady state ran through another 5. After this time whole crys- tallizer content was transferred on vacuum filter. Product crystals were not washed. Using appropriate analytical methods and procedures there were determined: solid phase concentration in product crystals suspension (MT), chemical composition of mother solution and solid phase (using, among others, plasma emission spectrometer ICP–AES CPU 7000, spectrometer IR PU9712, atomic absorption spectrometer iCE 3000, spectrophotometer UV–Vis Evolution 300), product crystal size distribu- tions (solid particle laser analyser Beckman Coulter LS 13 320) and crystal habit (scanning electron microscope JEOL JSM 5800LV). Accuracy of process data determi- nation in the continuous plant used was estimated to be ca. 10%. Kinetic parameters of the investigated continuous struvite reaction crystallization process were determined based on population density distributions n(L) of product crys- tals [11]. The most simplified kinetic model for continu- ous MSMPR crystallizer – SIG model [12], was used for the calculations. Crystal population density distribution equation resulting from the assumed SIG kinetic model constraints can be presented in the form of Eq. (1): a b Figure 1. Photo of experimental plant for continuous reaction crystallization of struvite: (a) general view, (b) continuous DT MSMPR type crystallizer unit with internal circulation of suspension. Copyright © 2013 SciRes. ACES 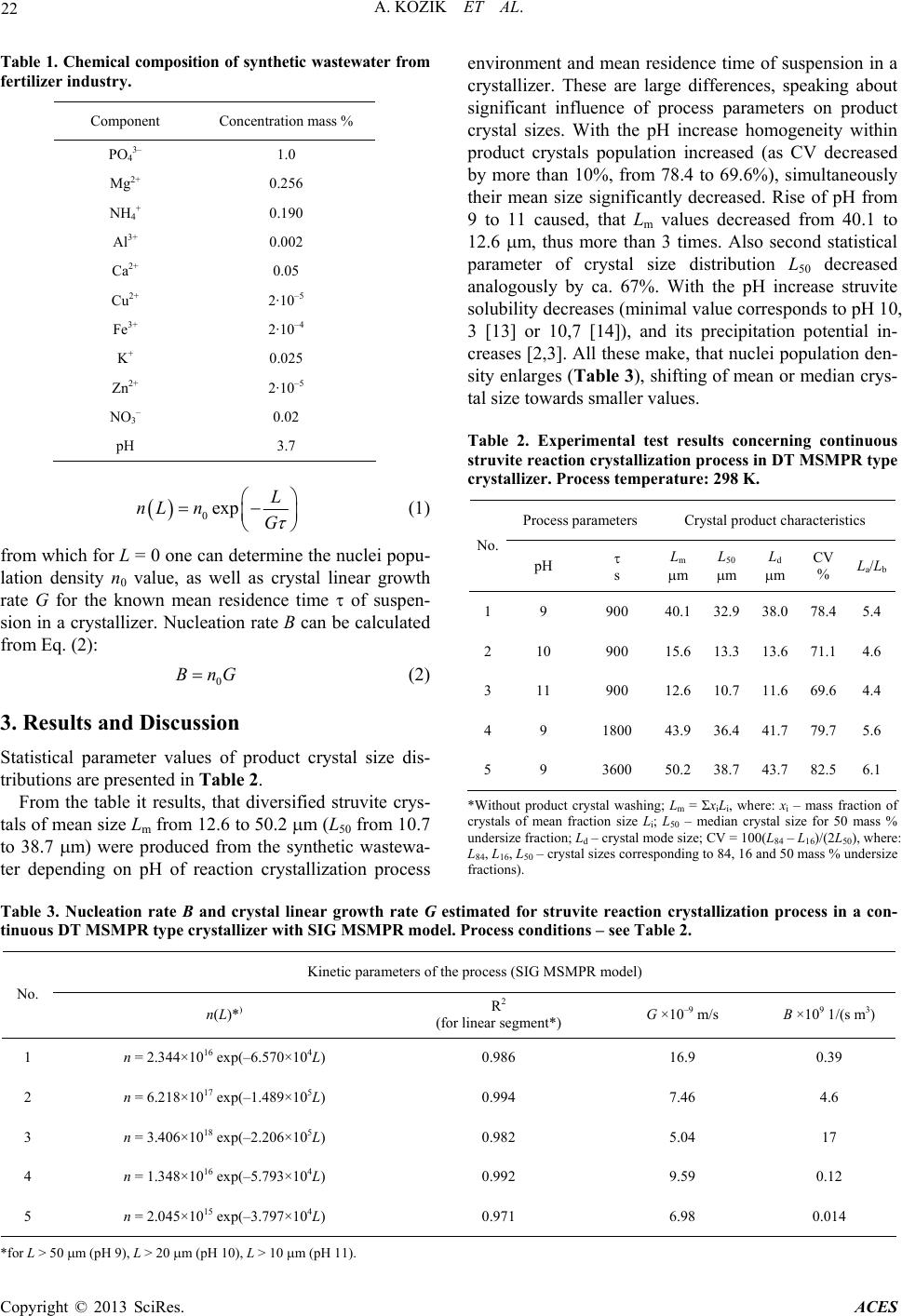 A. KOZIK ET AL. 22 Table 1. Chemical composition of synthetic wastewater from fertilizer industry. Component Concentration mass % PO43– 1.0 Mg2+ 0.256 NH4+ 0.190 Al3+ 0.002 Ca2+ 0.05 Cu2+ 2·10–5 Fe3+ 2·10–4 K+ 0.025 Zn2+ 2·10–5 NO3– 0.02 pH 3.7 0exp L nL nG (1) from which for L = 0 one can determine the nuclei popu- lation density n0 value, as well as crystal linear growth rate G for the known mean residence time of suspen- sion in a crystallizer. Nucleation rate B can be calculated from Eq. (2): 0 BnG (2) 3. Results and Discussion Statistical parameter values of product crystal size dis- tributions are presented in Table 2. From the table it results, that diversified struvite crys- tals of mean size Lm from 12.6 to 50.2 m (L50 from 10.7 to 38.7 m) were produced from the synthetic wastewa- ter depending on pH of reaction crystallization process environment and mean residence time of suspension in a crystallizer. These are large differences, speaking about significant influence of process parameters on product crystal sizes. With the pH increase homogeneity within product crystals population increased (as CV decreased by more than 10%, from 78.4 to 69.6%), simultaneously their mean size significantly decreased. Rise of pH from 9 to 11 caused, that Lm values decreased from 40.1 to 12.6 m, thus more than 3 times. Also second statistical parameter of crystal size distribution L50 decreased analogously by ca. 67%. With the pH increase struvite solubility decreases (minimal value corresponds to pH 10, 3 [13] or 10,7 [14]), and its precipitation potential in- creases [2,3]. All these make, that nuclei population den- sity enlarges (Table 3), shifting of mean or median crys- tal size towards smaller values. Table 2. Experimental test results concerning continuous struvite reaction crystallization process in DT MSMPR type crystallizer. Process temperature: 298 K. Process parameters Crystal product characteristics No. pH s Lm m L50 m Ld m CV % La/Lb 1 9 900 40.1 32.9 38.0 78.45.4 2 10 900 15.6 13.3 13.6 71.14.6 3 11 900 12.6 10.7 11.6 69.64.4 4 9 1800 43.9 36.4 41.7 79.75.6 5 9 3600 50.2 38.7 43.7 82.56.1 *Without product crystal washing; Lm = ΣxiLi, where: xi – mass fraction of crystals of mean fraction size Li; L50 – median crystal size for 50 mass % undersize fraction; Ld – crystal mode size; CV = 100(L84 – L16)/(2L50), where: L84, L16, L50 – crystal sizes corresponding to 84, 16 and 50 mass % undersize fractions). Table 3. Nucleation rate B and crystal linear growth rate G estimated for struvite reaction crystallization process in a con- tinuous DT MSMPR type crystallizer with SIG MSMPR model. Process conditions – see Table 2. Kinetic parameters of the process (SIG MSMPR model) No. n(L)*) R2 (for linear segment*) G ×10–9 m/s B ×109 1/(s m3) 1 n = 2.344×1016 exp(–6.570×104L) 0.986 16.9 0.39 2 n = 6.218×1017 exp(–1.489×105L) 0.994 7.46 4.6 3 n = 3.406×1018 exp(–2.206×105L) 0.982 5.04 17 4 n = 1.348×1016 exp(–5.793×104L) 0.992 9.59 0.12 5 n = 2.045×1015 exp(–3.797×104L) 0.971 6.98 0.014 *for L > 50 m (pH 9), L > 20 m (pH 10), L > 10 m (pH 11). Copyright © 2013 SciRes. ACES 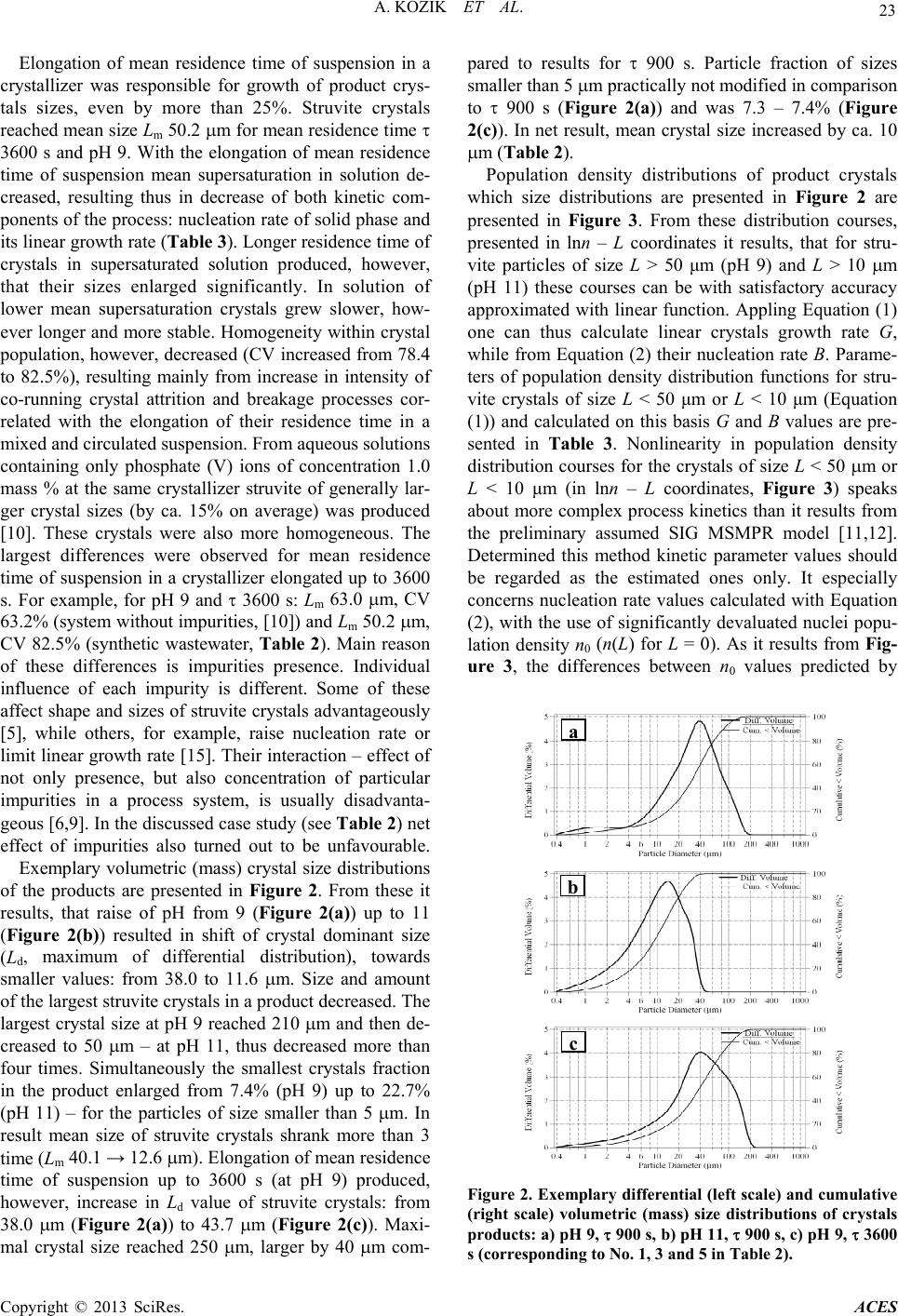 A. KOZIK ET AL. 23 Elongation of mean residence time of suspension in a crystallizer was responsible for growth of product crys- tals sizes, even by more than 25%. Struvite crystals reached mean size Lm 50.2 m for mean residence time 3600 s and pH 9. With the elongation of mean residence time of suspension mean supersaturation in solution de- creased, resulting thus in decrease of both kinetic com- ponents of the process: nucleation rate of solid phase and its linear growth rate (Tabl e 3). Longer residence time of crystals in supersaturated solution produced, however, that their sizes enlarged significantly. In solution of lower mean supersaturation crystals grew slower, how- ever longer and more stable. Homogeneity within crystal population, however, decreased (CV increased from 78.4 to 82.5%), resulting mainly from increase in intensity of co-running crystal attrition and breakage processes cor- related with the elongation of their residence time in a mixed and circulated suspension. From aqueous solutions containing only phosphate (V) ions of concentration 1.0 mass % at the same crystallizer struvite of generally lar- ger crystal sizes (by ca. 15% on average) was produced [10]. These crystals were also more homogeneous. The largest differences were observed for mean residence time of suspension in a crystallizer elongated up to 3600 s. For example, for pH 9 and 3600 s: Lm 63.0 m, CV 63.2% (system without impurities, [10]) and Lm 50.2 m, CV 82.5% (synthetic wastewater, Table 2). Main reason of these differences is impurities presence. Individual influence of each impurity is different. Some of these affect shape and sizes of struvite crystals advantageously [5], while others, for example, raise nucleation rate or limit linear growth rate [15]. Their interaction – effect of not only presence, but also concentration of particular impurities in a process system, is usually disadvanta- geous [6,9]. In the discussed case study (see Table 2) net effect of impurities also turned out to be unfavourable. Exemplary volumetric (mass) crystal size distributions of the products are presented in Figure 2. From these it results, that raise of pH from 9 (Figure 2(a)) up to 11 (Figure 2(b)) resulted in shift of crystal dominant size (Ld, maximum of differential distribution), towards smaller values: from 38.0 to 11.6 m. Size and amount of the largest struvite crystals in a product decreased. The largest crystal size at pH 9 reached 210 m and then de- creased to 50 m – at pH 11, thus decreased more than four times. Simultaneously the smallest crystals fraction in the product enlarged from 7.4% (pH 9) up to 22.7% (pH 11) – for the particles of size smaller than 5 m. In result mean size of struvite crystals shrank more than 3 time (Lm 40.1 → 12.6 m). Elongation of mean residence time of suspension up to 3600 s (at pH 9) produced, however, increase in Ld value of struvite crystals: from 38.0 m (Figure 2(a)) to 43.7 m (Figure 2(c)). Maxi- mal crystal size reached 250 m, larger by 40 m com- pared to results for 900 s. Particle fraction of sizes smaller than 5 m practically not modified in comparison to 900 s (Figure 2(a)) and was 7.3 – 7.4% (Figure 2(c)). In net result, mean crystal size increased by ca. 10 m (Table 2). Population density distributions of product crystals which size distributions are presented in Figure 2 are presented in Figure 3. From these distribution courses, presented in lnn – L coordinates it results, that for stru- vite particles of size L > 50 μm (pH 9) and L > 10 m (pH 11) these courses can be with satisfactory accuracy approximated with linear function. Appling Equation (1) one can thus calculate linear crystals growth rate G, while from Equation (2) their nucleation rate B. Parame- ters of population density distribution functions for stru- vite crystals of size L < 50 μm or L < 10 μm (Equation (1)) and calculated on this basis G and B values are pre- sented in Table 3. Nonlinearity in population density distribution courses for the crystals of size L < 50 m or L < 10 μm (in lnn – L coordinates, Figure 3) speaks about more complex process kinetics than it results from the preliminary assumed SIG MSMPR model [11,12]. Determined this method kinetic parameter values should be regarded as the estimated ones only. It especially concerns nucleation rate values calculated with Equation (2), with the use of significantly devaluated nuclei popu- lation density n0 (n(L) for L = 0). As it results from Fig- ure 3, the differences between n0 values predicted by a b c Figure 2. Exemplary differential (left scale) and cumulative (right scale) volumetric (mass) size distributions of crystals products: a) pH 9, 900 s, b) pH 11, 900 s, c) pH 9, 3600 s (corresponding to No. 1, 3 and 5 in Table 2). Copyright © 2013 SciRes. ACES 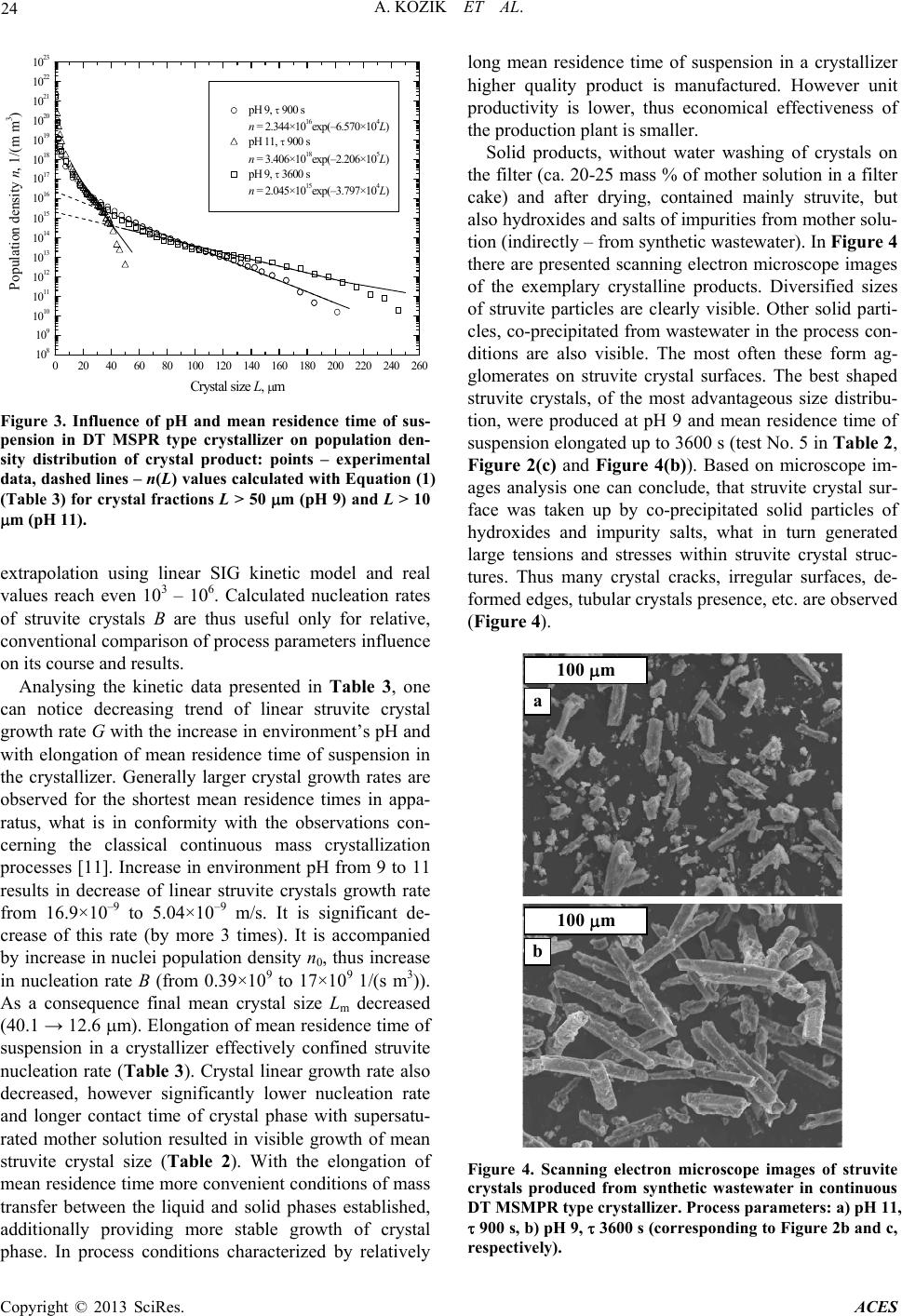 A. KOZIK ET AL. 24 020406080100 120 140 160 180 200 220 240 260 108 109 1010 1011 1012 1013 1014 1015 1016 1017 1018 1019 1020 1021 1022 1023 pH 9, 900 s n = 2.344×1016exp(–6.570×104L) pH 11, 900 s n = 3.406×1018exp(–2.206×105L) pH 9, 3600 s n = 2.045×1015exp(–3.797×104L) Population density n, 1/(m m3) Crystal size L, m Figure 3. Influence of pH and mean residence time of sus- pension in DT MSPR type crystallizer on population den- sity distribution of crystal product: points – experimental data, dashed lines – n(L) values calculated with Equation (1) (Table 3) for crystal fractions L > 50 m (pH 9) and L > 10 m (pH 11). extrapolation using linear SIG kinetic model and real values reach even 103 – 106. Calculated nucleation rates of struvite crystals B are thus useful only for relative, conventional comparison of process parameters influence on its course and results. Analysing the kinetic data presented in Table 3, one can notice decreasing trend of linear struvite crystal growth rate G with the increase in environment’s pH and with elongation of mean residence time of suspension in the crystallizer. Generally larger crystal growth rates are observed for the shortest mean residence times in appa- ratus, what is in conformity with the observations con- cerning the classical continuous mass crystallization processes [11]. Increase in environment pH from 9 to 11 results in decrease of linear struvite crystals growth rate from 16.9×10–9 to 5.04×10–9 m/s. It is significant de- crease of this rate (by more 3 times). It is accompanied by increase in nuclei population density n0, thus increase in nucleation rate B (from 0.39×109 to 17×109 1/(s m3)). As a consequence final mean crystal size Lm decreased (40.1 → 12.6 m). Elongation of mean residence time of suspension in a crystallizer effectively confined struvite nucleation rate (Table 3 ). Crystal linear growth rate also decreased, however significantly lower nucleation rate and longer contact time of crystal phase with supersatu- rated mother solution resulted in visible growth of mean struvite crystal size (Table 2). With the elongation of mean residence time more convenient conditions of mass transfer between the liquid and solid phases established, additionally providing more stable growth of crystal phase. In process conditions characterized by relatively long mean residence time of suspension in a crystallizer higher quality product is manufactured. However unit productivity is lower, thus economical effectiveness of the production plant is smaller. Solid products, without water washing of crystals on the filter (ca. 20-25 mass % of mother solution in a filter cake) and after drying, contained mainly struvite, but also hydroxides and salts of impurities from mother solu- tion (indirectly – from synthetic wastewater). In Figure 4 there are presented scanning electron microscope images of the exemplary crystalline products. Diversified sizes of struvite particles are clearly visible. Other solid parti- cles, co-precipitated from wastewater in the process con- ditions are also visible. The most often these form ag- glomerates on struvite crystal surfaces. The best shaped struvite crystals, of the most advantageous size distribu- tion, were produced at pH 9 and mean residence time of suspension elongated up to 3600 s (test No. 5 in Tabl e 2 , Figure 2(c) and Figure 4(b)). Based on microscope im- ages analysis one can conclude, that struvite crystal sur- face was taken up by co-precipitated solid particles of hydroxides and impurity salts, what in turn generated large tensions and stresses within struvite crystal struc- tures. Thus many crystal cracks, irregular surfaces, de- formed edges, tubular crystals presence, etc. are observed (Figure 4). 100 m b 100 m a Figure 4. Scanning electron microscope images of struvite crystals produced from synthetic wastewater in continuous DT MSMPR type crystallizer. Process parameters: a) pH 11, 900 s, b) pH 9, 3600 s (corresponding to Figure 2b and c, respectively). Copyright © 2013 SciRes. ACES  A. KOZIK ET AL. 25 Table 4. Chemical composition of solid phase and mother solution after filtration of crystal suspension removed from continuous DT MSMPR type crystallizer (see Table 2). Concentration in: Component Mother solution mg/kg Solid phase1 mass % PO43– 15 – 66 37.2 – 40.2 Mg2+ 31 – 66 8.6 – 9.6 NH4+ 70 – 110 6.5 – 7.0 Al 0.05 – 0.5 0.05 – 0.16 Ca 20 – 60 1.8 – 3.4 Cu 0.002 – 0.004 (6 – 11)×10–4 Fe 0.005 – 0.008 0.006 – 0.018 K 63 – 160 0.20 – 0.66 Zn < 0.1 0.09 – 0.18 NO3– 100 – 150 0.20 – 0.29 1after drying, without water washing of crystals on a filter. Based on planimetric measurement results involving 50 crystals of each product, randomly selected from three scanning electron microscope images it was concluded, that average ratio of their length La to their width Lb var- ied from 4.4 to 6.1, depending on the process parameter values (Table 2). Struvite produced in the same crystal- lizer from aqueous solutions of phosphate (V) ions with- out impurities characterized with La/Lb ratio ca. 6 (for [PO4 3–]RM 1.0 mass %, pH 9 and 900 s) [10]. Increase in pH from 9 to 11 caused, that struvite crystals became clearly shorter and thinner (La/Lb 5.4 → 4.4, see Figure 4(a)). Contrary, elongation of mean residence time of suspension up to 3600 s favoured production of longer and wider crystals, and length La increment was clearly larger than width Lb increment. The La/Lb ratio increased thus up to 6.1 (see Figure 4(b)). One can assume that struvite crystal sizes and their shape are the net result of wastewater impurities action and parameters of reaction crystallization process. From the microscope images it also results, that agglomeration within struvite crystals was not significant. It generally speaks advantageously about process conditions established in the crystallizer in respect to nucleation and growth of struvite crystals. Taking, however, under consideration all components of struvite continuous reaction crystallization process in DT MSMPR type crystallizer one can notice, that main factor influencing the process course is supersaturation in mother solution, very strongly dependent (at constant composition of feed solution, constant temperature and constant mixing/circulation intensity) on environment pH and on mean residence time of suspension in the crystal- lizer working volume. In Table 4 the concentration ranges of components in the post processed mother solution and in solid phase (without water washing of crystals on the filter and after their drying) removed from the crystallizer were pre- sented. Crystal product, as it results from Table 4, be- sides main component MgNH4PO4 × 6H2O, contained also all impurities present in synthetic wastewater (hy- droxides, phosphates (V), chlorides, nitrates (V)). From the data analysis it results, that aluminium, calcium, copper, iron and zinc ions practically totally precipitated (compare concentration of these ions in wastewater (Ta- ble 1) and in post processed mother solution (Table 4)). One can also notice, that concentration of phosphate (V) ions in a post processed mother solution varied from 66 mg/kg (pH 9, 900 s) to 15 mg/kg (pH 9, 3600 s). This concentration values decreased regularly with the pH raise and with elongation of mean residence time of struvite crystals suspension in the crystallizer. From the comparison it results, that concentration of phosphate (V) ions can be decreased even 4 times. It is connected with decrease of struvite solubility with the rising of reacting mixture pH while longer contact time of crystals with mother solution in a crystallizer is responsible for more thorough discharge of the generated supersaturation. The values of phosphate (V) ions concentration in mother solution can be regarded small, thus effectiveness of their removal from the feed solution as fully satisfactory. 4. Conclusions The sparingly soluble salt, MgNH4PO4 × 6H2O, struvite was produced from synthetic wastewater, simulating wastewater from mineral fertilizer industry, agricultural industry or liquid manure. Process ran in continuous DT MSMPR type crystallizer. Struvite crystals of mean size Lm from ca. 13 to ca. 50 m were removed from the crystallizer. It was proved, that increase in pH (from 9 to 11) of struvite reaction crystallization process environ- ment produced decrease of crystal mean size by more than 3 times (Lm 40.1 → 12.6 m, 900 s). Contrary, elongation of mean residence time of suspension in a crystallizer from 900 to 3600 s produced significant enlargement of this characteristic size by ca. 25% (Lm 50.2 m at pH 9, 3600 s). Products of moderate crystal homogeneity (CV ca. 76%) were removed from the crystallizer. It is complex, net effect of feed chemical composition, pH, mean residence time of suspension, as well as crystals attrition and breakage on the supersatura- tion level establishing in mother solution. From the population density distribution of product crystals nucleation rate and linear growth rate of struvite were estimated. It was concluded, that linear crystal growth rate varied within 5.04×10–9 – 16.9×10–9 m/s range, while nucleation rate changed within the 0.014×109 – 17×109 1/(s m3) limits depending on process parameter values. With the increase in environment pH Copyright © 2013 SciRes. ACES  A. KOZIK ET AL. Copyright © 2013 SciRes. ACES 26 nucleation rate increased while linear crystal growth rate decreased, what influenced final size of struvite crystals disadvantageously. Elongation of mean residence time of suspension in a crystallizer caused, that both kinetic pa- rameter (B and G) values decreased. On the other hand longer contact time of crystals from the supersaturated mother solution compensated with excess lower crystal growth rate. In result struvite crystals of relatively large sizes were produced. Concentration of phosphate (V) ions decreased from 1.0 mass % in a feed to 15 – 66 mg/kg in a postprocessed mother solution. It can be regarded as a very good result of their removal process from the feed. In solid product, besides main component – struvite, all impurities from wastewater appeared in form of hydroxides, phosphates and other salts. 5. Acknowledgements This work was supported by the National Science Centre of Poland under grant No. NN 209 0959 40 (2011–2014). REFERENCES [1] K. S. Le Corre, E. Valsami–Jones, P. Hobbs and S. A. Parsons, “Phosphorus Recovery from Wastewater by Struvite Crystallization: A Review,” Critical Reviews En- vironmental Science and Technology, Vol. 39, 2009, pp. 433-477. doi:10.1080/10643380701640573 [2] S. Parsons, “Recent Scientific and Technical Develop- ments: Struvite Precipitation,” CEEP Scope Newslett., Vol. 41, 2001, pp. 15-22. [3] J. Doyle and S. A. Parsons, “Struvite Formation, Control and Recovery,” Water Research, Vol. 36, 2002, pp. 3925-3940. doi:10.1016/S0043-1354(02)00126-4 [4] A. Matynia, B. Wierzbowska, N. Hutnik, K. Piotrowski, R. Liszka, T. Ciesielski and A. Mazienczuk, “Method for Struvite Recovery from Wastewater of Mineral Fertilizer Industry,” Przem. Chem., Vol. 89, 2010, pp. 478-485. [5] N. Hutnik, K. Piotrowski, J. Gluzinska and A. Matynia, “Effect of Selected Inorganic Impurities Present in Real Phosphate(V) Solutions on the Quality of Struvite Crys- tals Produced in Continuous Reaction Crystallization Process,” Progr. Environ. Sci. Technol., Vol. 3, 2011, pp. 559-566. [6] N. Hutnik, A. Kozik, A. Mazienczuk, K. Piotrowski, B. Wierzbowska and A. Matynia, “Phosphates(V) Recovery from Phosphorus Mineral Fertilizers Industry Wastewater by Continuous Struvite Reaction Crystallization Process,” Water Research, Vol. 47, 2013. doi:10.1016/j.watres.2013.04.026 [7] J. Koralewska, K. Piotrowski, B. Wierzbowska and A. Matynia, “Kinetics of Reaction–crystallization of Struvite in the Continuous Draft Tube Magma Type Crystallizers – influence of Different Internal Hydrodynamics,” Chi- nese Journal of Chemical Engineering, Vol. 17, 2009, pp. 330-339. doi:10.1016/S1004-9541(08)60212-8 [8] A. Mazienczuk, N. Hutnik, K. Piotrowski, B. Wierz- bowska and A. Matynia, “Continuous Crystallizers with Jet Pump Driven by Recirculated Mother Solution in Pro- duction of Struvite,” Przem. Chem., Vol. 91, 2012, pp. 890-895. [9] A. Kozik, K. Piotrowski, B. Wierzbowska and A. Matynia, “Recovery of Phosphate (V) Ions from Waste- waters of Different Chemical Compositions,” Progr. En- viron. Sci. Technol., Vol. 3, 2011, pp. 550-558. [10] A. Kozik, A. Matynia and K. Piotrowski, “Continuous Reaction Crystallization of Struvite from Diluted Aque- ous Solutions of Phosphate (V) Ions in DT MSMPR Crystallizer,” Proceedia Engineering, Vol. 42, 2012, pp. 313-322. doi:10.1016/j.proeng.2012.07.422 [11] J. W. Mullin, “Crystallization”, Butterworth–Heinemann, Oxford, 1993. [12] A. D. Randolph and M. A. Larson, “Theory of Particulate Processes: Analysis and Techniques of Continuous Crys- tallization”, Academic Press, New York, 1988. [13] K. N. Ohlinger, T. M. Young and E. D. Schroeder, “Pre- dicting Struvite Formation in Digestion,” Water Research, Vol. 32, 1998, pp. 3607-3614. doi:10.1016/S0043-1354(98)00123-7 [14] V. L. Snoeyink and D. Jenkins, “Water Chemistry,” Wiley, New York, 1980. [15] N. Hutnik, K. Piotrowski, A. Kozik and A. Matynia, “Continuous Reaction Crystallization of Struvite from phosphate(V) Solutions Containing Inorganic Impurities –SIG kinetic Approach,” Proceedings of the 38th Inter- national Conference of Slovak Society of Chemical Engi- neering SSCHE, Tatranské Matliare, 23-27 May 2011, pp. 325-0333.
|