Paper Menu >>
Journal Menu >>
 Engineering, 2012, 5, 121-123 doi:10.4236/eng.2012.410B031 Published Online October 2012 (http://www.SciRP.org/journal/eng) Copyright © 2012 SciRes. ENG Leptin Receptor in Ram Epididymal Spermatozoa Mohammad Roostaei-Ali Mehr, Reza Rajabi-Toustan i , Ras ool Motamedi -Mojdehi Department of Anim al Science, Faculty of Agricultural Sciences, University of Guilan, Rasht, Iran Email: roostaei@guilan.ac.ir, rezarajabi.t@gmail.com, r.motamedimojdehi@gmail.com Received 2012 ABSTRACT This experi ment was des i gned to investi gate lep ti n recep to rs ( Ob-R and Ob-Rb) mRNA in ram epididymal spermatozoa by RT-PCR. Ten testes were obtained from abattoir and epididymal spermatozoa recovery was performed. To purify spermatozoa, motile sperm were isolated by the swim-up procedure. Total RNA was isolated from epididymal spermatozoa and placental cotyledon, as a posi- tive control, and then they were purified. Specific bands (98 and 308 bp) for Ob-R and Ob -Rb were detected a fter RT-P CR in bo th epididymal spermatozoa and placental cotyledon. We may conclude that Ob-R and Ob-Rb mRNA are present in ram epididymal spermatozoa and leptin perhaps exerts physiological effects, as already de mon strated in hum a n. Keywords: Epididymal Spermat ozoa; mRNA; Ob-R; Ob-Rb 1. Introduction Leptin is a 16 kDa protein and mainly secreted by white adi- pose tissue [1]. Leptin modulates different systems via neu- roendocrine regulation, including adrenal, thyroid and gonadal axes. Other functions of leptin have been suggested recently in peripheral organs that leptin independently interacts with leptin recepto r in peri pheral ti ssue, as wel l as repr oducti ve organs [2] . Recent o bservation s in female suggest th at leptin is involved in relaying energy status to reproduction [3-7]. Various studies have also pointed out a direct role of leptin in the control of testicular function, but its actual role in the regulatory network controlling male reproduction is still a matter of debate [3,6,8]. To date, at least five Ob-R isoforms (Ob-Ra-e) have been cloned from rodents and exhibit widespread distribution both in central and peripheral tissues [6,9]. Alternative splicing of the Ob-R transcript results in the various products that vary in the length of the cytoplasmic region [10]. Three Ob-R isoforms, the long form (Ob-Rb), the middle form (Ob-Ra) and the short form (Ob-Rc) were cloned and sequenced in bovine and ovine [11,12]. Ob-Rb was repo rted i n most o f the farm ani mal tissues [10,13]. It is known that animal and human models of leptin resis- tance and deficiency show a severe impairment of the repro- duct ive functio n [6]. Leptin recepto rs are presen t in several cel l types in the human, bovine and porcine male and female re- productive system [6,14]. Therefore, it is seemed that leptin could exert a direct effect on the gonads [2]. There is little in- formation about the expression of leptin and leptin receptor in ruminant such as sheep. To fully understand the peripheral effects of leptin as a local factor, it is necessary to study the structure and peripheral tissue distribution of its receptors (Ob-R and Ob-Rb). The aim of this experiment was to determine the expression of Ob-R and Ob-Rb mRNA in ram epididymal spermatozoa, providing evidence for a role of leptin in sperm physiology. 2. Materials and Methods Ten testes were obtained from abattoir. In the laboratory, the caudal epididymis was isolated from testis. Epididymal sper- matozoa were obtained by slicing and suspending of the caudal epididymal tissue in Petri dish containing 5 ml Tris (300 mM tris (hydroxymethyl) aminomethane, 95 mM citric acidmono- hydrate, 28 mM D-(+)-Glucose, 2000 IU/ml penicillin G and 0.4 mg/ml streptomycin, pH 7.0). The samples were agitated and incubated at room temperature for 10 min; liquid phase containing epididymal spermatozoa was collected. A complete semen analysis was performed on each sample: sperm motility was >65% with normal morphology >65% and viability >70 %. Motility, viability, normal morphology and sperm concentra- tion were evaluated by microscopic visual method, one-step eosin -nigrosin staining and hematocytometer method, respec- tively. Recovered epididymal spermatozoa were pooled and centrifuged for 10 min at 700 × g at room temperature; the su- pernatant was removed. To purify spermatozoa, motile sperm were isol ated by the s wim-up procedure; 250 µl of semen were layered under 1 ml of Tris diluent in each of 15 ml centrifuge tubes. After incubation for 1 h at 37 °C in an atmosphere with 5% CO2, 750 µl were removed from the top of each tube. The purity of spermatozoa was checked under a phase contrast mi- crosco pe. Sperm were concen trated b y centrifugation at 800 ×g for 10 min, the top of suspension was removed frozen in liquid nitrogen vapor and held in liquid nitrogen until RNA extrac- tion. Total RNA from placental cotyledon and epididymal sper- matozoa were extracted according to Chomczynski and Saachi [15]. RNA samples were treated by DNAase I at 37 °C for 30 min and then the enzyme was inactivated at 75 °C for 15 min. After that Purity of RNA were determined by Biophotometer (Eppendorf, Germany) (OD 260/280 and 260/230 ratio) and visu a lized b y electroph oresis on a 2% agaro se gel, stai ned with 0.5 μg/ml ethidium bromide. RNA samples with no DNA bands 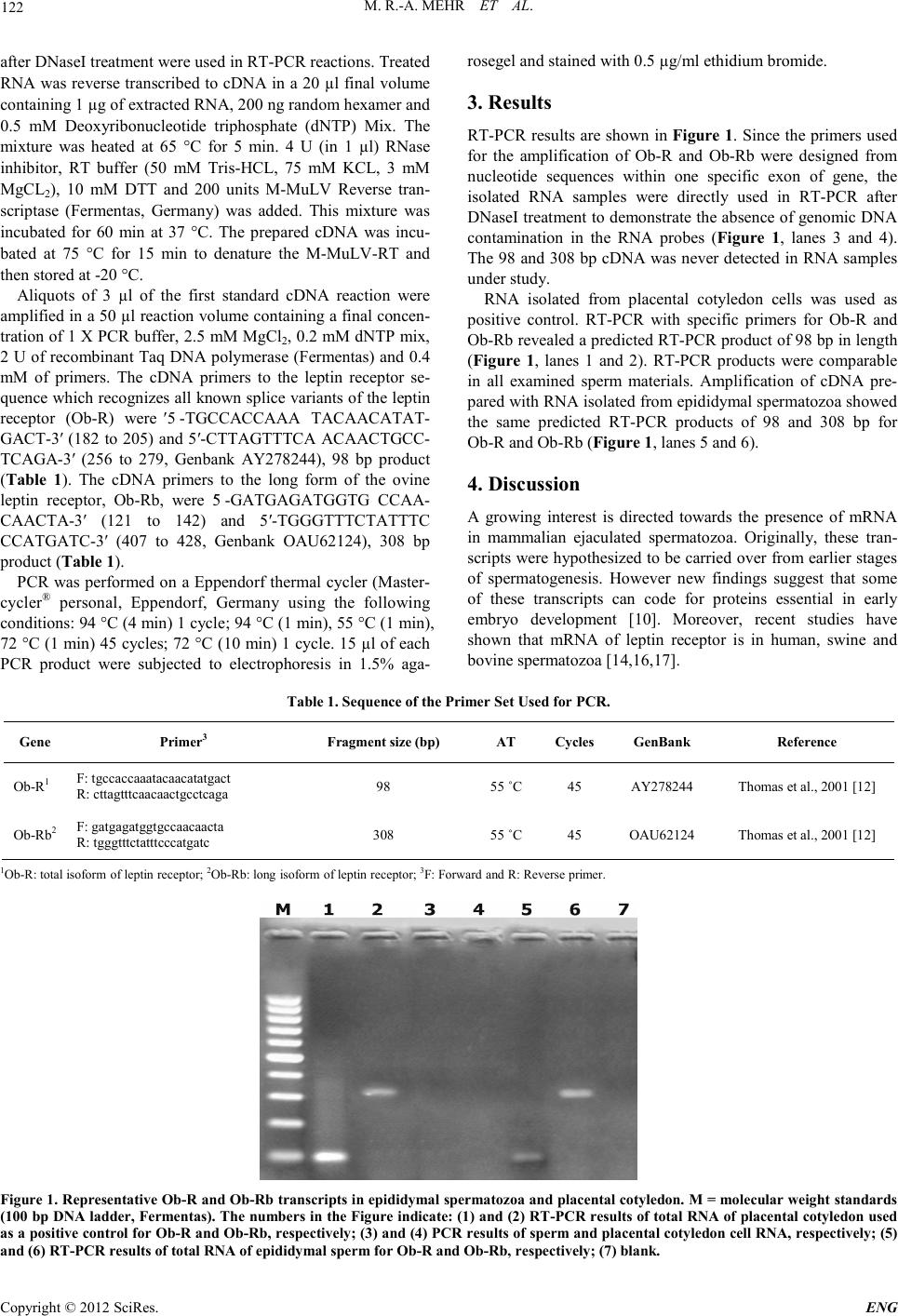 M. R.-A. MEHR ET AL. Copyright © 2012 SciRes. ENG 122 after DNaseI treatment were used in RT-PCR reactions. Treat ed RNA was reverse transcribed to cDNA in a 20 µl final volume containing 1 µg of extracted RNA, 200 ng random hexamer and 0.5 mM Deoxyribonucleotide triphosphate (dNTP) Mix. The mixture was heated at 65 °C for 5 min. 4 U (in 1 µl) RNase inhibitor, RT buffer (50 mM Tris-HCL, 75 mM KCL, 3 mM MgCL2), 10 mM DTT and 200 units M-MuLV Reverse tran- scriptase (Fermentas, Germany) was added. This mixture was incubated for 60 min at 37 °C. The prepared cDNA was incu- bated at 75 °C for 15 min to denature the M-MuLV-RT and then stored at -20 °C. Aliquots of 3 µl of the first standard cDNA reaction were amplified in a 50 µl reaction volume contain ing a final concen- tration of 1 X PCR buffer, 2.5 mM MgCl2, 0. 2 mM dN TP mix, 2 U of recombinant Taq DNA polymerase (Fermentas) and 0.4 mM of primers. The cDNA primers to the leptin receptor se- quen ce which recognizes all known splice variants of the leptin receptor (Ob-R) were 5′-TGCCACCAAA TACAACATAT- GACT-3′ (182 to 205) and 5′-CTTAGTTTCA ACAACTGCC- TCAGA-3′ (256 to 279, Genbank AY278244), 98 bp product (Table 1). The cDNA primers to the long form of the ovine leptin receptor, Ob-Rb, were 5′-GATGAGATGGTG CCAA- CA ACTA-3′ (121 to 142) and 5′-TGGGTTTCTATTTC CCATGATC-3′ (407 to 428, Genbank OAU62124), 308 bp product (Table 1). PCR was performed o n a Eppendorf th ermal cycler (Master- cycler® personal, Eppendorf, Germany using the following conditions: 94 °C (4 min) 1 cycle; 94 °C (1 min), 55 °C (1 min), 72 °C (1 min) 45 cycles; 72 °C (10 min) 1 cycle. 15 µl of each PCR product were subjected to electrophoresis in 1.5% aga- rosegel and stained with 0.5 µg/ml ethidium bromide. 3. Results RT-PCR results are shown in Figure 1. Since t he primers used for the amplification of Ob-R and Ob-Rb were designed from nucleotide sequences within one specific exon of gene, the isolated RNA samples were directly used in RT-PCR after DNaseI treatmen t to d emonst rate the absen ce of genomic DN A contamination in the RNA probes (Figure 1, lanes 3 and 4). The 98 and 308 bp cDNA was never detected in RNA samples under study. RNA isolated from placental cotyledon cells was used as positive control. RT-PCR with specific primers for Ob-R and Ob-Rb r evealed a predi cted RT-PCR product of 98 bp in length (Figure 1, lanes 1 and 2). RT-PCR products were comparable in all examined sperm materials. Amplification of cDNA pre- pared with RNA isolated from epididymal spermat ozo a sh owed the same predicted RT-PCR products of 98 and 308 bp for Ob-R and Ob-R b (Figure 1, lanes 5 and 6). 4. Discussion A growing interest is directed towards the presence of mRNA in mammalian ejaculated spermatozoa. Originally, these tran- scripts were h ypothesi zed to be carri ed over fro m earlier stag es of spermatogenesis. However new findings suggest that some of these transcripts can code for proteins essential in early embryo development [10]. Moreover, recent studies have shown that mRNA of leptin receptor is in human, swine and bovine spermatozoa [14,16,17]. Tabl e 1. Sequence of the Primer Set Used for PCR. Gene Primer3 Fragment si ze (bp) AT Cycles GenBank Reference Ob-R1 F: tgccaccaaatacaacatatgact R: cttagtttcaacaactgcctcaga 98 55 ˚C 45 AY278244 Thomas et al., 2001 [12] Ob-Rb2 F: gatgagatggtgccaacaacta R: tgggtttctatttcccatgatc 308 55 ˚C 45 OAU62124 Thomas et al., 2001 [12] 1Ob-R: total isoform of leptin receptor; 2Ob-Rb: long isoform of leptin receptor; 3F: Forward and R: Reverse prime r. Fig ure 1 . Representative Ob-R a nd O b-Rb transcri pts in epididymal spermatozoa and placental cotyledon. M = molecular weight standards (100 bp DNA ladder, Fermentas). The numbers in the Figure i ndicate: (1) and (2) RT-PCR res ults of total RNA of placen tal cot yledo n used as a positive control for Ob-R and Ob-Rb, respectively; (3) and (4) PCR results of sperm and placental cotyledon cell RNA, respectively; (5) and (6) RT-PCR results of total RNA of epididymal sperm for Ob-R and Ob -Rb, respectively; (7) blank.  M. R.-A. MEHR ET AL. Copyright © 2012 SciRes. E NG 123 However, to our knowledge, this is the first time that the presence of mRNA of Ob-R and Ob-Rb in ram epididymal spermatozoa has been demonstrated. A recent study in pig has suggested that leptin receptor may induce transduction and molecular changes associated with sperm capacitation and sur- vival [18]. These findings suggest that leptin, through leptin receptor, may play an important role in both sperm function and male fertility. Furthermore, as leptin interacts with insulin to regulate glycogen synthesis in mature spermatozoa, it might affect sperm motility, as already demonstrated in humans [8]. The presence of Ob-R mRNA in ram epididymal spermatozoa might suggest that leptin acts through leptin receptor to regulate spermatozoa energy expenditure in this species. At present, we do not have any information on the possible actions of leptin on ram spermatozoa. REFERENCES [1] L. A. Campfield, F. J. Smith, and P. Burn, “The OB protein (leptin) pathway. A link between adipose tissue mass and central neu ra l net work s, ” Hor m. Met ab . R es. , vol . 2 8 , pp . 619 –63 1, Dec . 1996. [2] I. J. Clarke, and B. A. Henry, “Leptin and reproduction,” Rev. Reprod., vol. 4 , pp. 48–55, Jan. 1 99 9 . [3] D. P. Shalev, Y. Soffer, and L. M. Lewin, “Investigation on the motili ty of hu man spermat ozoa in a defined medium in the pres- ence of metabolic inhibitors and of carnitine,” Andrologia, vol. 18, pp. 368–375, Aug. 1986. [4] C. Cunningham, D. K. Clifton, and R. A. Steiner, “Leptin’s actions on t he reproduct ive axis: persp ectives and mechanisms,” Biol. Reprod. , vol. 60, pp. 216–222, Feb. 1999. [5] K. Laud, I. Gourdou, L. Be Iair, D. H. Keisler, and J. Djinane, “Detection and regulation of leptin receptor mRNA in ovine mammary epithelial cells during pregnancy and lactation,” FEBS. Lett., vol. 463, pp. 194–198, Dec. 1999. [6] M. Caprio, E. Fabrini, G. Ricci, S. Basciani, L. Gnessi, M. Arizzi, A. R. Carta, M. U. De Martino, A. M. Isidori, G. V. Frajese, and A. Fabbri, “Ontogenesis of leptin receptor in rat leydig cells,” Biol. Reprod. , vol. 68, pp. 1199–1207, Apr. 2003. [7] S. C. Liefers, R. F. Veerkamp, M. F. W. Te Pas, Y. Chilliard and T. van der Lende. “Genetics and physiology of lept in in peripar- turi ent dairy,” Domest. Ani m. E ndocrin ol., vol. 29, pp. 227–238, Jul. 2 0 0 5. [8] S. Aquila, M. Gentile, E. Middea, S. Catalano, and S. Andò, “Autocrine regulation of insulin secretion in human ejaculated sperma tozoa,” Endocrinology, vol. 146, pp. 552–557, Feb. 2005. [9] L. A. Tartaglia, M. Dembski, X. Weng, N. Deng, J. Culpepper, R. Devos, G. J. Rich ards , L. A. Camp field , F. T. C lark, J . Deeds , C. Muir, S. Sanker, A. Moriarty, K. J. Moore, J. S. Smutko, G. G. Mays, E. A. Wool, C. A. Monroe, and R. I. Tepper, “Identifica- tion and express ion c lonin g of a leptin recep t or, OB-R,” C ell, vol. 83, pp. 1263–1271, Dec. 1995. [10] M. Haniu, T. Arakawa, E. J. Bures, Y. Young, J. O. Hui, M. F. Rohde, A. A. Welcher, and T. Horan, “Human leptin receptor. Determin ation of disulfid e struct ure and N-glycosylation sites of the extracellular domain,” J. Biol. Chem., vol. 273, pp. 28691–28699, Oct. 1998. [11] H. Kawachi, S. H. Yang, A. Hamano, T. Matsui, S. B. Smith, and H. Yano, “Molecu lar cloning an d expression of bovine (B os taurus) leptin receptor isoform mRNAs,” Comp. Biochem. Phy- siol., vol. 148, pp. 167–173, Oct. 2007. [12] L. Thomas, J. M. Wallace, R. P. Aitken, J. G. Mercer, P. Tray- hurn, and N. Hoggard, “Circulating leptin during ovine pregnan- cy in relation to maternal nutrition, body composition and preg- nancy outcome,” J. Endocrinol., vol. 169, pp. 465–476, Jun. 2001. [13] L. A. Tartaglia, “The leptin receptor,” J. Biol. Chem., vol. 272, pp. 6093–6099, Mar. 1997. [14] G. Nikbakht, M. Roostaei Ali Mehr, A. Baghbanzadeh, P. Tajik, C. Tamanini, and M. Emam, “Leptin receptor mRNA in bull ejaculated spermatozoa,” Reprod. Dom. Anim., vol. 45, pp. 237–242, Apr. 2010. [15] P. Chomczynski, and N. Sacchi, “Single step method of RNA isolation by acid guanidinium hiocyanate-phenol-choloroform extraction,” Anal. Biochem., vol. 16 2, pp. 156–159 , Apr. 1987. [16] T. Jope, A. Lammert, J. Kratzsch, U. Paasch, and H. J. Glander, “Leptin and leptin receptor in human seminal plasma and in hu- man spermatozoa,” Int. J. Androl, vol. 26, pp. 335–341, Dec. 2003. [17] M . De Ambrogi, M. Spinaci, G Galeati, and C. Tamanini, “Lep- tin receptor in boar spermatozoa,” Int. J. Androl, vol. 30, pp. 458–461, Oct. 2007. [18] S. Aquila, V. Rago, C. Guido, S. Zupo, I. Casaburi, and A. Car- pin o, “Lep ti n and lep t in rec ep t or in p ig sp er ma t ozoa : evid en c e of their involvement in sperm capacitation and survival,” Repro- ducti on, vol. 136, pp. 23–32. Epub. |

