I. DEREVICH, D. GALDINA
6
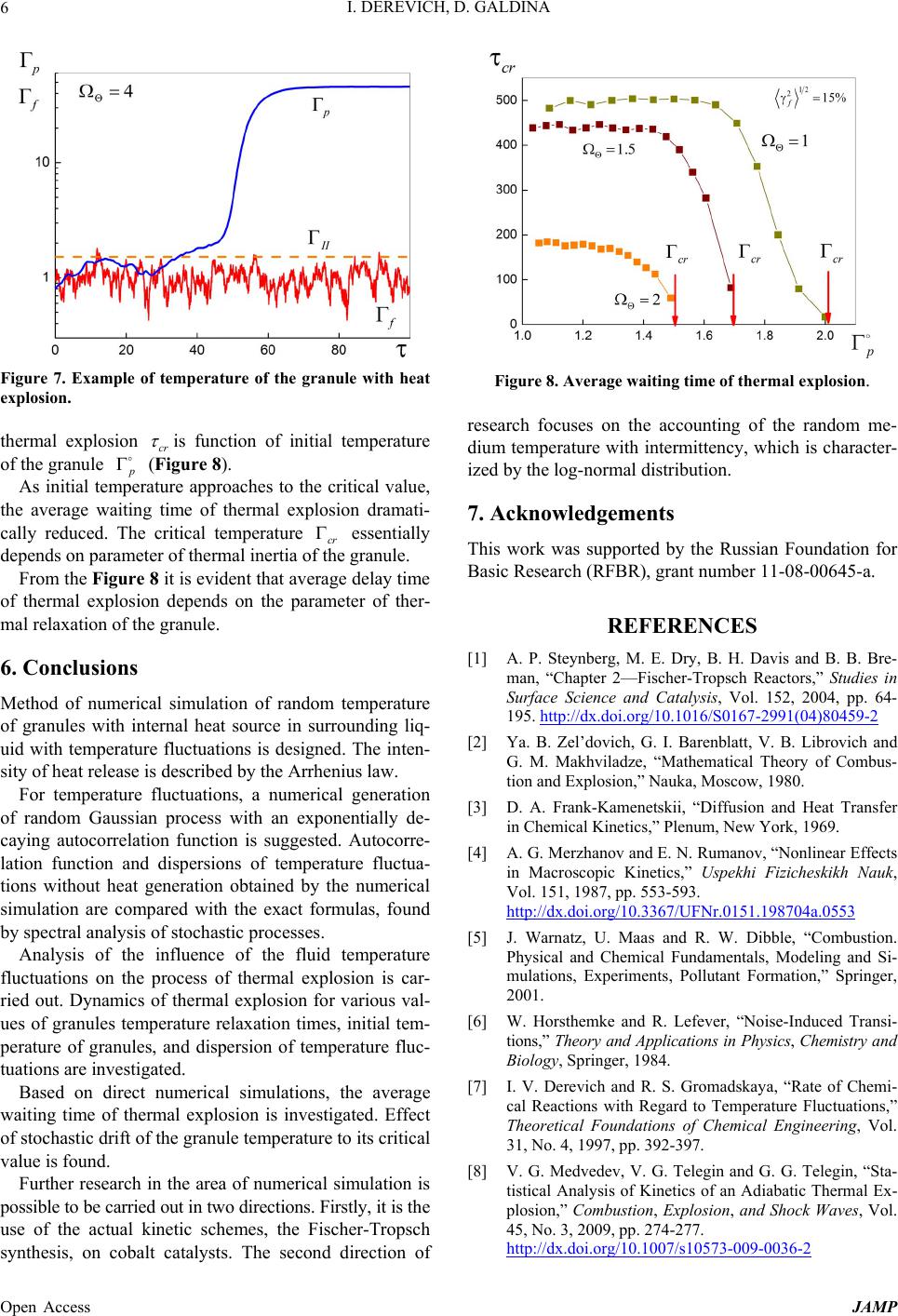
Figure 7. Example of temperature of the granule with heat
explosion.
thermal explosion cr
is function of initial temperature
of the granule
mp (Fi
eratur
gure 8).
As initial tee approaches to the critical value,
the average waiting time of thermal explosion dramati-
cally reduced. The critical temperature essentially
depends on parameter of thermal inertiagranule.
From the Figure 8 it is ev ident that averag e delay time
of thermal explosion depends on the parameter of ther-
mal relaxation of the granule.
6. Conclusions
Method of numerical simulation of random temperatue
of granules with internal heat source in surrounding l-
release is described by the Arrhenius law.
flu
ss
explosion for various val-
erature relaxation times, initial te
and dispersion of temperature fluc-
al
va
cr
of the
r
iq
uid with temperature fluctuations is designed. The inten-
ity of heatsFor temperature ctuations, a numerical generation
of random Gauian process with an exponentially de-
caying autocorrelation function is suggested. Autocorre-
lation function and dispersions of temperature fluctua-
tions without heat generation obtained by the numerical
simulation are compared with the exact formulas, found
by spectral analysis of stochastic processes.
Analysis of the influence of the fluid temperature
fluctuations on the process of thermal explosion is car-
ried out. Dynamics of thermal
ues of granules temp
perature of granules, m-
tuations are investigated.
Based on direct numerical simulations, the average
waiting time of thermal explosion is investigated. Effect
of stochastic drift of the granule temperature to its critic
lue is found.
Further research in the area of numerical simulation is
possible to be carried out in two directions. Firstly, it is the
use of the actual kinetic schemes, the Fischer-Tropsch
synthesis, on cobalt catalysts. The second direction of
Figure 8. Average waiting time of thermal explosion.
research focuses on the accounting of the random me-
dium temperature with intermittenc y, which is character-
ized by the log-normal distribution.
7. Acknowledgements
This work was supported by the Russian Foundation for
Basic Research (RFBR), grant number 11-08-00645-a.
REFERENCES
[1] A. P. Steynberg, M. E. Dry, B. H. Davis and B. B. Bre-
man, “Chapter 2—Fischer-Tropsch Reactors,” Studies in
Surface Science and Catalysis, Vol. 152, 2004, pp. 64-
195. http://dx.doi.org/10.1016/S0167-2991(04)80459-2
[2] nd
[4] A. G. Merzhanov and E. N. Rumanov, “Nonlinear Effects
Uspekhi Fizicheskikh Nauk,
3.
Ya. B. Zel’dovich, G. I. Barenblatt, V. B. Librovich a
G. M. Makhviladze, “Mathematical Theory of Combus-
tion and Explosion,” Nauka, Moscow, 1980.
[3] D. A. Frank-Kamenetskii, “Diffusion and Heat Transfer
in Chemical Kinetics,” Plenum, New York, 1969.
in Macroscopic Kinetics,”
Vol. 151, 1987, pp. 553-59
http://dx.doi.org/10.3367/UFNr.0151.198704a.0553
[5] J. Warnatz, U. Maas and R. W. Dibble, “Combustion.
Physical and Chemical Fundamentals, Modeling and Si-
mulations, Exmation,” Springer,
2001.
i-
al Ex-
periments, Pollutant For
[6] W. Horsthemke and R. Lefever, “Noise-Induced Transi-
tions,” Theory and Applications in Physics, Chemistry and
Biology, Springer, 1984.
[7] I. V. Derevich and R. S. Gromadskaya, “Rate of Chem
cal Reactions with Regard to Temperature Fluctuations,”
Theoretical Foundations of Chemical Engineering, Vol.
31, No. 4, 1997, pp. 392-397.
[8] V. G. Medvedev, V. G. Telegin and G. G. Telegin, “Sta-
tistical Analysis of Kinetics of an Adiabatic Therm
plosion,” Combustion, Explosion, and Shock Waves, Vol.
45, No. 3, 2009, pp. 274-277.
http://dx.doi.org/10.1007/s10573-009-0036-2
Open Access JAMP