 International Journal of Analytical Mass Spectrometry and Chromatography, 2013, 1, 72-80 http://dx.doi.org/10.4236/ijamsc.2013.11009 Published Online September 2013 (http://www.scirp.org/journal/ijamsc) Determination of Lanthanides, Thorium, Uranium and Plutonium in Irradiated (Th, Pu)O2 by Liquid Chromatography Using α-Hydroxyiso Butyric Acid (α-HIBA) Pranaw Kumar, P. G. Jaison, Vijay M. Telmore, Sumana Paul, Suresh K. Aggarwal* Fuel Chemistry Division, Bhabha Atomic Research Centre, Mumbai, India Email: *skaggr2002@rediffmail.com Received July 27, 2013; revised August 28, 2013; accepted September 27, 2013 Academic Editor: Prof. N. Sivaraman, HBNI, India and Head, SCSS, Chemistry Group, Indira Gandhi Centre for Atomic Research, Kalpakkam-603102, INDIA Copyright © 2013 Pranaw Kumar et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT An HPLC method is presented for the separation and determination of lanthanides (Lns), thorium (Th), uranium (U) and plutonium (Pu) from irradiated (Th, Pu)O2. Individual separation of Lns, Th, U and Pu is a challenging task because of 1) lanthanides having similar physical and chemical properties, 2) presence of complex matrix like irradiated fuel and 3) the co-existence of multiple oxidation states of Pu. Different procedures were developed for separation of individual lanthanides and actinides. The individual lanthanides were separated on a dynamically modified reversed phase (RP) column using n-octane sulfonic acid as an ion interaction reagent and employing dual gradient (pH and concentration) of α-hydroxyisobutyric acid (HIBA). In order to improve the precision on the determination of Lns, terbium (Tb) was used as an internal standard. The method was validated employing simulated high level liquid waste. Concentrations of lanthanides viz. lanthanum (La) and neodymium (Nd) in the dissolver solution were determined based on their peak areas. Th, U and Pu were separated on a RP column using mobile phase containing HIBA and methanol. Since Pu is prone to exist in multiple oxidation states, all the oxidation states were converted into Pu (IV) using H2O2 in 3 M HNO3. Under the optimized conditions, Pu(IV) eluted first followed by Th and U. The concentrations of Th, U and Pu were determined by standard addition method and were found to be 1.10 ± 0.02 mg/g, 5.3 ± 0.3 µg/g and 27 ± 1 µg/g, respec- tively, in the dissolver solution of irradiated fuel. These values were in good agreement with the concentration of Th determined by biamperometry and those of U and Pu by isotope dilution thermal ionization mass spectrometry. Keywords: HPLC; Lanthanides; Th; U; Pu; -HIBA; Irradiated (Th, Pu)O2 1. Introduction The Indian nuclear program envisages the effective utili- zation of thorium (Th) as a fertile material for the sus- tained production of electricity in the country [1]. Unlike the natural uranium (U) based fuel which contains 235U as the fissile material, thoria based fuel initially requires the addition of fissile materials like 233U, 235U and 239Pu from outside. Mixture of (Th, 233U) O2 and (Th, 239Pu) O2 was proposed as fuel for advanced heavy water reactor (AHWR) to make system self-sustaining in 233U [2,3]. In order to assess the performance of (Th, Pu) O2 MOX fuel, prior to its introduction in commercial reactors, (Th, 4% Pu)O2 fuel clusters were irradiated in the Pressurized Water Loop (PWL) of CIRUS reactor, BARC. The im- plementation of mixed oxide based fuel cycle requires development of methodologies for evaluating the per- formance of the fuel in the reactor. Burn-up is an impor- tant parameter for nuclear fuel development, fuel man- agement and fuel performance analysis [4,5]. Burn-up is defined as the atom percent fission of heavy element (mass ≥ 225) during its life time in the reactor. Radio- chemical and chemical analysis methods are generally used for burn-up determination of irradiated nuclear fuels [6]. The destructive method involved dissolution of fuels followed by individual separation and determination of * Corresponding author. C opyright © 2013 SciRes. IJAMSC  P. KUMAR ET AL. 73 fission products and heavy elements. Isotope dilution-thermal ionization mass spectrometry (ID-TIMS) is an estab- lished method for the determination of burn-up [5,7,8]. Development of new methodologies for the separation and determination of Lns, Th, U and Pu is essential for burn-up determination of thorium based nuclear fuel [9]. Different methods based on solvent extraction, ion ex- change, precipitation, liquid chromatography, etc. have been reported for the separation of Lns, Th, U and Pu in various matrices [10,11]. Among these methods, high performance liquid chromatography (HPLC) is a fast and highly efficient technique and has been applied for the separation of lanthanides and actinides in nuclear fuel samples and geological samples [12-16]. Due to the fast separation and less amount of the sample handling in HPLC, it minimizes the exposure to highly radioactive samples and is, therefore, attractive for the separation of fission products. Cassidy et al. studied the separation of lanthanides fission products using a dynamic ion ex- change column from different fuel samples like UO2 and (Th, U)O2 fuels [17,18]. However, in presence of bulk of actinides, the individual separation of lanthanides was not reported. Lanthanides were separated in a group by conventional ion exchange method and subsequently injected into HPLC after removal of Th and U. This off- line separation requires multiple monitoring elements and there are chances of loss of analytes. The reported method gave good reproducibility among different runs for the same aliquot but results were inconsistent for dif- ferent aliquots of same fuel. Sivaraman et al. carried out extensive studies on the separation of lanthanide fission products for fast breeder test reactor fuels [19-21]. They determined the stability constant of actinide-HIBA com- plexes under different chromatographic conditions. How- ever, (Th, Pu)O2 is a unique fuel tested for AHWR re- actor and liquid chromatographic method for separation of Lns from this matrix is not reported in literature. Various RP based methods have been reported for the separation of Th, U and Pu using hydroxyl carboxylic acid like HIBA and mandelic acid [22-24]. In most of the reported method, the lanthanides, U and Th are present in comparable amounts. Studies were carried out previously in our laboratory using ion interaction reagent (IIR) on the reversed phase (RP) column for the separation of lanthanides from Th and U [25]. Under the optimized chromatographic conditions using dual gradient of pH and concentration of mobile phase, lanthanides were se- parated out from bulk of Th and U from simulated ir- radiated thoria sample. However, the separation and de- termination of Lns, Th, U and Pu from irradiated (Th, Pu)O2 fuel employing HPLC have not been reported so far. Present paper deals with the individual separation of Lns, Th, U and Pu in an irradiated (Th, Pu)O2 fuel. Han- dling of (Th, Pu)O2 fuel samples is more challenging because of the radiation dose associated with the dissol- ver solution, presence of large amount of Th which is vulnerable to hydrolysis and multiple oxidation states of Pu under the chromatographic conditions [26]. Since the chromatographic behavior of lanthanides is different than tetravalent and hexavalent actinides, two different sepa- ration procedures are developed. The challenges of the present studies are: 1) individual separation of lanthanide fission products in presence bulk of Th and interference of trivalent actinides in Lns separation, 2) difficulties in the quantification of Pu because of its multiple oxidation states, and 3) individual separation of Th(IV), Pu(IV) and U(VI). 2. Experimental 2.1. Instrumentation The HPLC system consisted of an L-2130 (Elite La- Chrom, Hitachi) low-pressure quaternary gradient pump and an L-2450 (Elite LaChrom) diode array detector. C18 monolith RP column (100 mm × 4.6 mm, Chromolith, Merck) and C18 particulate RP column (150 mm × 4.6 mm, 5 µm, Supelcosil) were used as the stationary pha- ses. Solutions were injected into the column using a Rheodyne injector (Model 9725i) with a 100 µL loop. The eluted species were monitored after post-column reac- tion with a metallochromic reagent, which was added us- ing a reciprocating pump (Eldex Laboratories Inc.) into a low dead volume-mixing tee (Valco). The signal from the detector was processed by EZChrom software package. 2.2. Reagents All solutions were prepared using deionised water from Milli-Q system (Millipore) and were filtered through a 0.45 µm membrane filter (Millipore) prior to using. α- HIBA (Lancaster) was used as an eluent. Sodium n-oc- tane sulphonate monohydrate (Fluka) and tetrabutylam- monium iodide (Sigma-Aldrich) were used as the ion interaction reagents (IIR). ICP-standards of the lantha- nides, U and Th (Inorganic Venture) were used after ap- propriate dilutions with Milli-Q water and mobile phase. HNO3 and NH4OH (Merck) were used for adjusting the pH of the mobile phase. H2O2 (Merck) in combination with HNO3 was used as a redox reagent to bring Pu into the IV oxidation state. For ion exchange separation, Dowex 1 × 8, 200 - 400 mesh size (Sigma-Aldrich) and Bio-Rad AG 1 × 2, 200 - 400 mesh size (Bio-Rad) were used as stationary phases. Arsenazo (III) (Fluka) was used as the post-column metallochromic reagent (PCR). The arsenazo complexes of Lns, Th, U and Pu were monitored at 653 nm. Irradiated (Th, Pu)O2 sample re- ceived from Post-Irradiated Examination Division (PIED), BARC was used for the method development. NIST- Copyright © 2013 SciRes. IJAMSC  P. KUMAR ET AL. 74 SRM-950a U3O8, K4Pu(SO 4)4 and enriched isotope of 142Nd were used as spikes for ID-TIMS analysis of U, Pu and Nd, respectively. 2.3. Procedure Appropriate quantities of α-HIBA and sodium n-octane sulfonate were dissolved in water and made to solutions with concentrations 0.5 M and 0.1 M, respectively. α- HIBA was adjusted to the desired pH using high-purity NH4OH and HNO3. Mobile phase flow-rate of 1.0 mL min−1 was used. The PCR solution [1.5 × 10−4 M Ar- senazo (III) and 0.01 M urea in 0.1 M HNO3] was deliv- ered at a flow rate of 0.3 mLmin−1.The lanthanides, Th, U and Pu solution of appropriate concentrations were prepared after dilution with mobile phase. Concentration of Pu was determined by biamperometry titration [27]. Dissolution of irradiated fuel sample was carried out in a facility housed in a shielded glove box. Aliquots from the irradiated fuel dissolver solution were transferred to a shielded fume-hood for further experiments. For the determination of Lns, about 0.5 g of dissolved sample solution was taken and 2.5 ppm of Tb was added as an internal standard. This solution was directly in- jected into HPLC through 100 µL injection port. The mobile phase containing α-HIBA of pH 6.5 was changed from 0.05 M to 0.15 M in 30 min; whereas α-HIBA of pH 3.5 was changed from 0.15 M to 0.3 M in a time in- terval of 30 to 40 min. A C18 RP column (250 mm × 4.6 mm) was used for the individual separation of Lns. For validation of method using ID-TIMS, all the aliquots were subjected to necessary chemical treatments to en- sure depolymerisation and proper isotopic homogeniza- tion. The spiked and unspiked aliquots were used for separation by anion exchange using Dowex 1 × 8, 200 - 400 mesh resin in 9 M HCl medium [8]. The effluent containing Th and fission product fraction was collected. Pu and U fractions were sequentially eluted from the column using 0.1 M hydroxylamine hydrochloride in 5M HCl and 0.5 M HNO3, respectively. The fraction con- taining Th and fission products was subjected to a second stage anion-exchange separation using Dowex 1 × 8200 - 400 mesh resin in 7 M HNO3 medium. The non-retained fission product fraction was collected and subjected to a third stage separation using Bio-Rad AG 1 × 2200 - 400 in a mixture containing HNO3 and MeOH to separate Nd fraction [8]. 3. Results and Discussion 3.1. Separation and Determination of Lns from Irradiated (Th, Pu)O2 3.1.1. Chromatographic Behavior of Lanthanides, Th, U and Pu In solution, under the chromatographic conditions, lan- thanides exist in III oxidation state whereas Th and U exist in IV and VI states, respectively. The situation in the cases of Pu is different. In view of the closeness of redox potential values, Pu can exist in different oxidation state viz. III, IV and VI simultaneously. Retention behav- iors of Lns and Pu(III) is different from Th(IV), U (VI), Pu(VI) and Pu(IV) when α-HIBA was used as an eluent on RP column [21].With α-HIBA, Lns and Pu(III) form mainly cationic complexes which are different from those formed by Th, U, Pu(VI) and Pu(IV) [25]. Thus Lns-HIBA complexes can be separated on dynamically modified RP column using an IIR which is sorbed unto the column converting hydrophobic surface into the charged surface, for ion exchange separation of lantha- nides [28]. However, Th, U and Pu(IV) get sorbed onto the RP column by hydrophobic interaction, when α- HIBA is used as a complexing reagent. In presence of large amounts of IIR, the actinides were found to elute in between the lanthanides which makes lanthanides deter- mination difficult. Hence, at a given concentration of IIR, the surface of RP column remains partly ionic and hy- drophobic in nature. The separation of lanthanides from Th, U and Pu was performed using dual gradient (pH and Concentration) of α-HIBA. At higher pH, tetravalent and hexavalent actinides are retained better than lanthanides on dynamically modified reversed phase column leading to the elution and separation of lanthanides prior to acti- nides. Due to the complex nature of irradiated (Th, Pu) O2 fuel matrix, two different separation procedures were developed as presented in Scheme 1. In the first stage, Lns were separated and determined on dynamically modified column whereas the separation and determina- tion of Th, U and Pu was achieved using RP column. 3.1.2. Determination of Lanthanides in Irradiated (Th, Pu) O2 Sample by HPLC A C18 reversed-phase column, dynamically modified with n-octane sulphonate, was used as the stationary phase for separation of lanthanides from irradiated (Th, Pu)O2 fuel. It was reported that with increase in the con- centration of IIR, the retention of Lns increases whereas the retention of Th and U decreases [28]. Th being the matrix element in the present case, the concentration of IIR selected was 5 mM to separate Lns without affecting the Th holding capacity. Effect of pH showed that at lower pH, higher fraction of HIBA remains in the uni- nonized form, resulting in faster elution of U and Th whereas, at pH ≥ 5.0, Th and U showed stronger reten- tion than lanthanides and eluted after Lns. With the in- crease in concentration of HIBA at pH 4.0, Lns, Th and U showed decrease in retention. Dual gradient (pH and concentration) of HIBA was used for the separation of Lns, Th, U and Pu. Pu was found to elute as multiple peaks in chromatographic run due to the presence of its Copyright © 2013 SciRes. IJAMSC 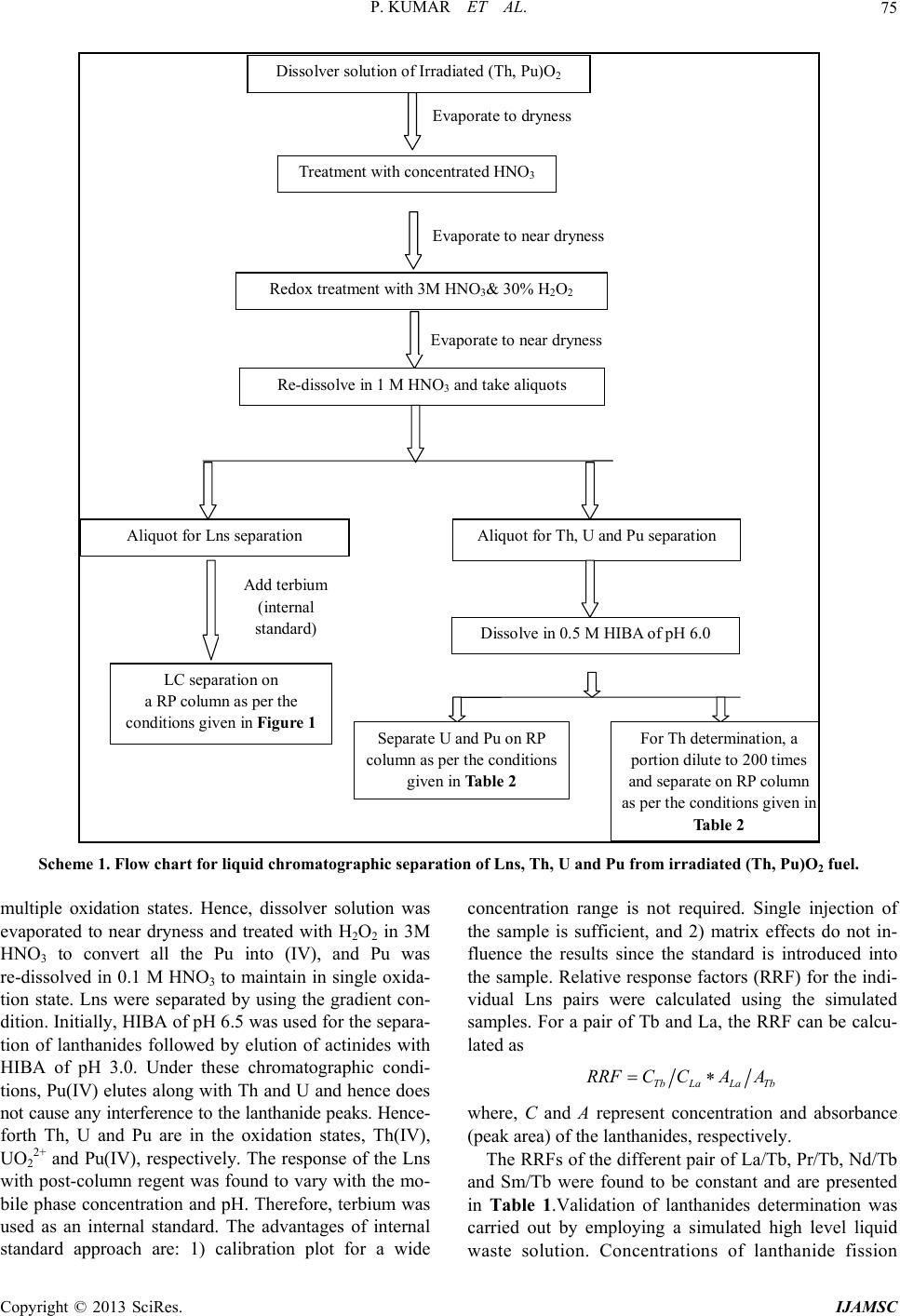 P. KUMAR ET AL. Copyright © 2013 SciRes. IJAMSC 75 Dissolver solution of Irradiated (Th, Pu)O 2 Evaporate to dryness Redox treatment with 3M HNO 3 & 30% H 2 O 2 Treatment with concentrated HNO 3 Evaporate to near dryness Eva orate to near dr ness Re-dissolve in 1 M HNO 3 and take aliquots Aliquot for Lns separation LC separation on a RP column as per the conditions given in Figure 1 Aliquot for Th, U and Pu separation Dissolve in 0.5 M HIBA of pH 6.0 Separate U and Pu on RP column as per the conditions given in Table 2 For Th determination, a portion dilute to 200 times and separate on RP column as per the conditions given in Table 2 Add terbium (internal sta ndard ) Scheme 1. Flow chart for liquid chromatographic separa tion of Lns, Th, U and Pu from irradiated (Th, Pu)O2 fuel. multiple oxidation states. Hence, dissolver solution was evaporated to near dryness and treated with H2O2 in 3M HNO3 to convert all the Pu into (IV), and Pu was re-dissolved in 0.1 M HNO3 to maintain in single oxida- tion state. Lns were separated by using the gradient con- dition. Initially, HIBA of pH 6.5 was used for the separa- tion of lanthanides followed by elution of actinides with HIBA of pH 3.0. Under these chromatographic condi- tions, Pu(IV) elutes along with Th and U and hence does not cause any interference to the lanthanide peaks. Hence- forth Th, U and Pu are in the oxidation states, Th(IV), UO2 2+ and Pu(IV), respectively. The response of the Lns with post-column regent was found to vary with the mo- bile phase concentration and pH. Therefore, terbium was used as an internal standard. The advantages of internal standard approach are: 1) calibration plot for a wide concentration range is not required. Single injection of the sample is sufficient, and 2) matrix effects do not in- fluence the results since the standard is introduced into the sample. Relative response factors (RRF) for the indi- vidual Lns pairs were calculated using the simulated samples. For a pair of Tb and La, the RRF can be calcu- lated as TbLaLa Tb RRF CCAA where, C and A represent concentration and absorbance (peak area) of the lanthanides, respectively. The RRFs of the different pair of La/Tb, Pr/Tb, Nd/Tb and Sm/Tb were found to be constant and are presented in Table 1.Validation of lanthanides determination was carried out by employing a simulated high level liquid waste solution. Concentrations of lanthanide fission 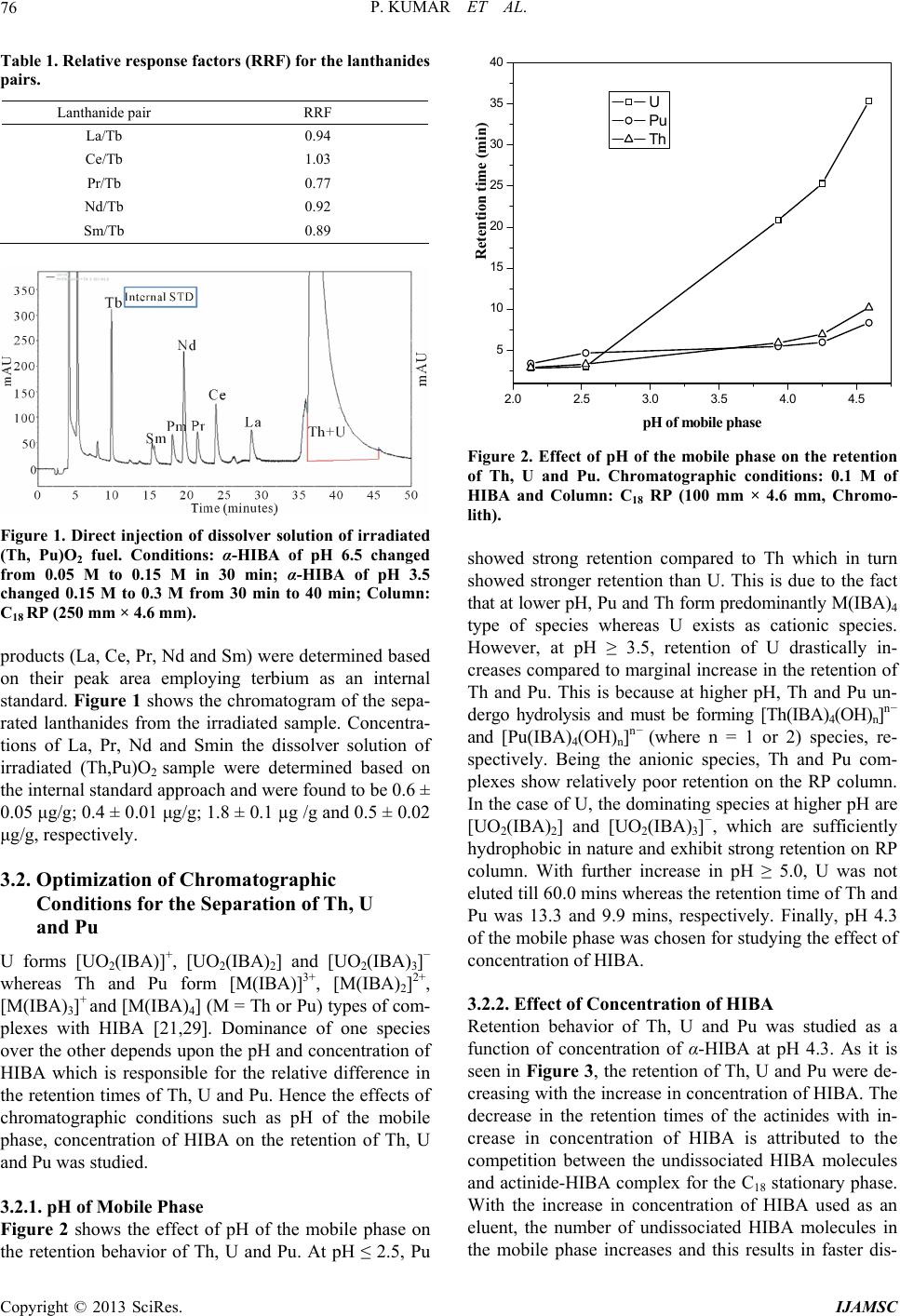 P. KUMAR ET AL. 76 Table 1. Relative response factors (RRF) for the lanthanides pairs. Lanthanide pair RRF La/Tb 0.94 Ce/Tb 1.03 Pr/Tb 0.77 Nd/Tb 0.92 Sm/Tb 0.89 Figure 1. Direct injection of dissolver solution of irradiated (Th, Pu)O2 fuel. Conditions: α-HIBA of pH 6.5 changed from 0.05 M to 0.15 M in 30 min; α-HIBA of pH 3.5 changed 0.15 M to 0.3 M from 30 min to 40 min; Column: C18 RP (250 mm × 4.6 mm). products (La, Ce, Pr, Nd and Sm) were determined based on their peak area employing terbium as an internal standard. Figure 1 shows the chromatogram of the sepa- rated lanthanides from the irradiated sample. Concentra- tions of La, Pr, Nd and Smin the dissolver solution of irradiated (Th,Pu)O2 sample were determined based on the internal standard approach and were found to be 0.6 ± 0.05 μg/g; 0.4 ± 0.01 μg/g; 1.8 ± 0.1 µg /g and 0.5 ± 0.02 μg/g, respectively. 3.2. Optimization of Chromatographic Conditions for the Separation of Th, U and Pu U forms [UO2(IBA)]+, [UO2(IBA)2] and [UO2(IBA)3]− whereas Th and Pu form [M(IBA)]3+, [M(IBA)2]2+, [M(IBA)3]+ and [M(IBA)4] (M = Th or Pu) types of com- plexes with HIBA [21,29]. Dominance of one species over the other depends upon the pH and concentration of HIBA which is responsible for the relative difference in the retention times of Th, U and Pu. Hence the effects of chromatographic conditions such as pH of the mobile phase, concentration of HIBA on the retention of Th, U and Pu was studied. 3.2.1. p H of Mobil e Phase Figure 2 shows the effect of pH of the mobile phase on the retention behavior of Th, U and Pu. At pH ≤ 2.5, Pu 2.0 2.5 3.0 3.5 4.0 4.5 5 10 15 20 25 30 35 40 Retention time (min) pH of mob ile phase U Pu Th Figure 2. Effect of pH of the mobile phase on the retention of Th, U and Pu. Chromatographic conditions: 0.1 M of HIBA and Column: C18 RP (100 mm × 4.6 mm, Chromo- lith). showed strong retention compared to Th which in turn showed stronger retention than U. This is due to the fact that at lower pH, Pu and Th form predominantly M(IBA)4 type of species whereas U exists as cationic species. However, at pH ≥ 3.5, retention of U drastically in- creases compared to marginal increase in the retention of Th and Pu. This is because at higher pH, Th and Pu un- dergo hydrolysis and must be forming [Th(IBA)4(OH)n]n− and [Pu(IBA)4(OH)n]n− (where n = 1 or 2) species, re- spectively. Being the anionic species, Th and Pu com- plexes show relatively poor retention on the RP column. In the case of U, the dominating species at higher pH are [UO2(IBA)2] and [UO2(IBA)3]−, which are sufficiently hydrophobic in nature and exhibit strong retention on RP column. With further increase in pH ≥ 5.0, U was not eluted till 60.0 mins whereas the retention time of Th and Pu was 13.3 and 9.9 mins, respectively. Finally, pH 4.3 of the mobile phase was chosen for studying the effect of concentration of HIBA. 3.2.2. Effect of Concentration of HIBA Retention behavior of Th, U and Pu was studied as a function of concentration of α-HIBA at pH 4.3. As it is seen in Figure 3, the retention of Th, U and Pu were de- creasing with the increase in concentration of HIBA. The decrease in the retention times of the actinides with in- crease in concentration of HIBA is attributed to the competition between the undissociated HIBA molecules and actinide-HIBA complex for the C18 stationary phase. With the increase in concentration of HIBA used as an eluent, the number of undissociated HIBA molecules in the mobile phase increases and this results in faster dis- Copyright © 2013 SciRes. IJAMSC 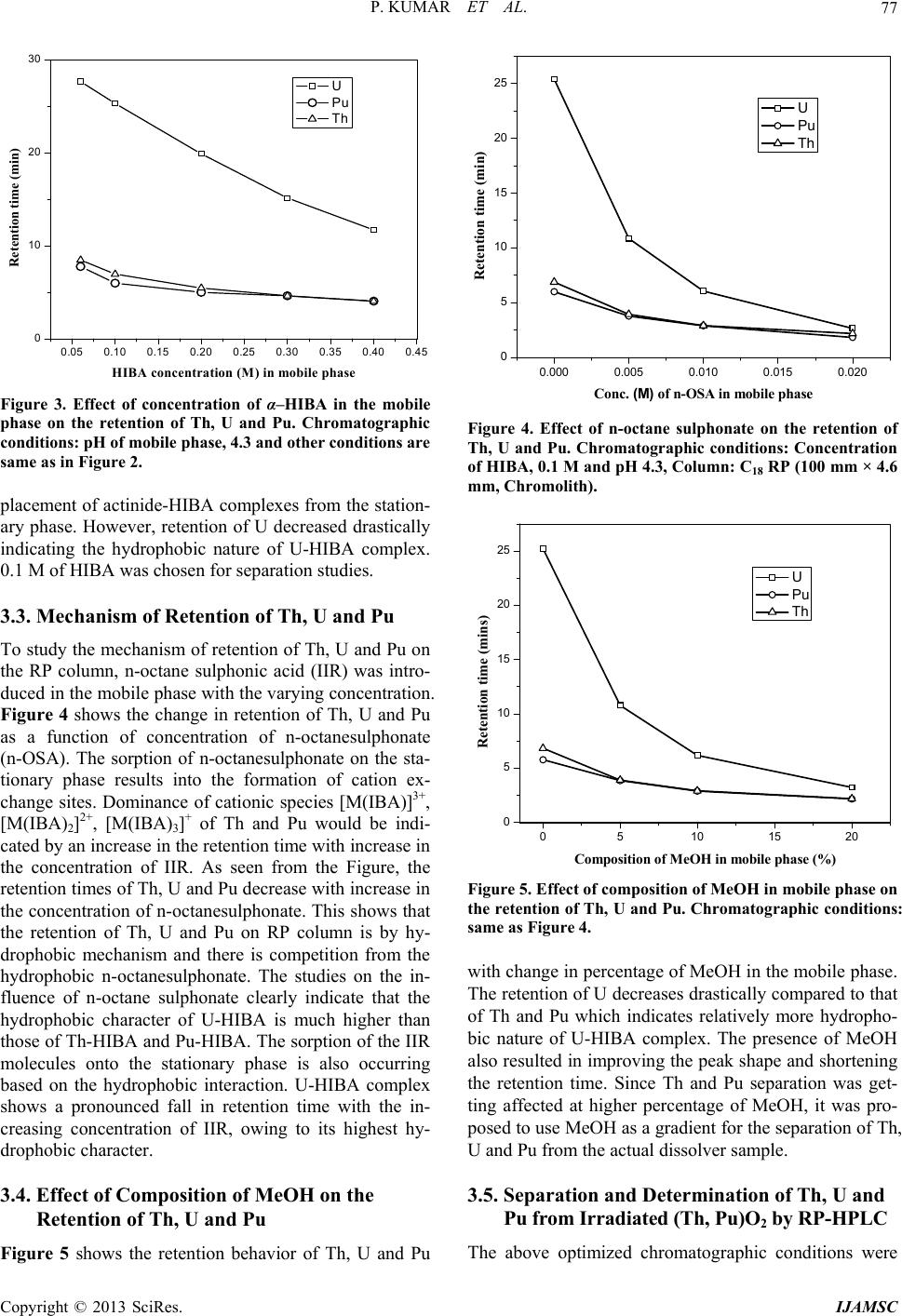 P. KUMAR ET AL. 77 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0 10 20 30 U Pu Th Retention time (min) HIBA concentration (M) in mobile phase Figure 3. Effect of concentration of α–HIBA in the mobile phase on the retention of Th, U and Pu. Chromatographic conditions: pH of mobile phase, 4.3 and other conditions are same as in Figure 2. placement of actinide-HIBA complexes from the station- ary phase. However, retention of U decreased drastically indicating the hydrophobic nature of U-HIBA complex. 0.1 M of HIBA was chosen for separation studies. 3.3. Mechanism of Retention of Th, U and Pu To study the mechanism of retention of Th, U and Pu on the RP column, n-octane sulphonic acid (IIR) was intro- duced in the mobile phase with the varying concentration. Figure 4 shows the change in retention of Th, U and Pu as a function of concentration of n-octanesulphonate (n-OSA). The sorption of n-octanesulphonate on the sta- tionary phase results into the formation of cation ex- change sites. Dominance of cationic species [M(IBA)]3+, [M(IBA)2]2+, [M(IBA)3]+ of Th and Pu would be indi- cated by an increase in the retention time with increase in the concentration of IIR. As seen from the Figure, the retention times of Th, U and Pu decrease with increase in the concentration of n-octanesulphonate. This shows that the retention of Th, U and Pu on RP column is by hy- drophobic mechanism and there is competition from the hydrophobic n-octanesulphonate. The studies on the in- fluence of n-octane sulphonate clearly indicate that the hydrophobic character of U-HIBA is much higher than those of Th-HIBA and Pu-HIBA. The sorption of the IIR molecules onto the stationary phase is also occurring based on the hydrophobic interaction. U-HIBA complex shows a pronounced fall in retention time with the in- creasing concentration of IIR, owing to its highest hy- drophobic character. 3.4. Effect of Composition of MeOH on the Retention of Th, U and Pu 0.000 0.005 0.010 0.015 0.020 0 5 10 15 20 25 Retention time (min) Conc . (M) of n-OSA in mobile phase U Pu Th Figure 4. Effect of n-octane sulphonate on the retention of Th, U and Pu. Chromatographic conditions: Concentration of HIBA, 0.1 M and pH 4.3, Column: C18 RP (100 mm × 4.6 mm, Chromolith). 0510 15 20 0 5 10 15 20 25 Retention ti me ( min s ) Composition of MeOH in mobile phase (%) U Pu Th Figure 5. Effect of composition of MeOH in mobile phase on ith change in percentage of MeOH in the mobile phase. 3.5. Separation and Determination of Th, U and The the retention of Th, U and Pu. Chromatographic conditions: same as Figure 4. w The retention of U decreases drastically compared to that of Th and Pu which indicates relatively more hydropho- bic nature of U-HIBA complex. The presence of MeOH also resulted in improving the peak shape and shortening the retention time. Since Th and Pu separation was get- ting affected at higher percentage of MeOH, it was pro- posed to use MeOH as a gradient for the separation of Th, U and Pu from the actual dissolver sample. Pu from Irradiated (T h, Pu)O 2 by RP-HPLC above optimized chromatographic conditions were Figure 5 shows the retention behavior of Th, U and Pu Copyright © 2013 SciRes. IJAMSC  P. KUMAR ET AL. 78 Table 2. Gradient condition for separation of Th, U and Pu. Time HIBA (pH = 6.5) HIBA (pH=2.0) % MeOH (mins) (M) (M) (v/v) 0.00 0.125 0.005 7 10.00 0.125 0.05 7 20.00 0.125 0.05 30 30.00 0.125 0.05 30 sed for the sequential elution of Pu, Th and U. In order 3.6. Validation of HPLC Method ass spectrometric u to improve the resolution between Th and Pu, gradient condition, as given in Table 2 was employed. To improve the resolution as well as Th holding capacity, a larger stationary phase of dimension 250 mm × 4.6 mm was used. Aliquots of irradiated sample were evaporated to near dryness and were taken in the mobile phases con- taining pH 6.5 of 0.13 Mα-HIBA, and were injected through 100 µL injection port. As shown in Figure 6, lanthanides and other trivalent species are eluted at the solvent front. Th, U and Pu are retained on the column because of the formation of hydrophobic species using α-HIBA as an eluent. Under the optimized conditions, the elution pattern was Pu (IV) followed by Th and U. This is due to the fact that at higher pH for Th and Pu(IV), M(HIBA)4 type of species are most dominating whereas for U, [UO2(IBA)3]− is the major species. How- ever, the lower retention time of Pu and Th compared to U can be explained on the basis of strong hydrolysis na- ture of Th and Pu (IV) in the chromatographic conditions resulting in the formation of [M(IBA)4(OH)2]2−. The concentrations of Pu, Th and U were determined by standard addition method and were found to be 27 ± 1 µg/g, 1.10 ± 0.02 mg/g, and 5.3 ± 0.3 µg/g, respectively, in the dissolver fuel solution. Isotope dilution-thermal ionization m (ID-TIMS) methodology was employed for the concen- tration determination of U, Pu and Nd in the dissolver fuel samples. Chemical separation of U, Pu and Nd frac- tions in the dissolver solution was carried out to elimi- nate the potential isobaric interferences during the mass spectrometric analysis as well as to get good ion yield in TIMS. Nd, U and Pu fractions obtained from unspiked and spiked aliquots were used to determine isotopic composition and concentration by ID-TIMS, respectively. Concentrations of U, Pu and Nd in the sample were found to be 5.3 ± 0.3 % g/g, 26.8 ± 0.2% g/g and 1.8 ± 0.2% g/g, respectively. Concentration of Th in the dis- solver solution, determined biampero metrically using EDTA as a titrant, was found to be 1.03 ± 0.2% mg/g [30]. The concentrations of Nd, Th, U and Pu obtained were in good agreement with the concentrations deter- mined by HPLC. Figure 6. Direct injection of dissolver solution of irradiated 4. Conclusion employing α-HIBA was developed for 5. Acknowledgements . Ramakumar, Director, REFERENCES [1] L. M. GanatayManohar and K. C. tharaman, V. Shivakumar and D. Saha, “Utilisa- .042 (Th, Pu)O2 fuel. Conditions: α-HIBA of pH 6.5, 0.125 M; α-HIBA of pH 2.0 from 0.005 M to 0.05 M in 10.0 mins; MeOH 7.0% till 10 mins, then 7% to 30% till 20 mins; Column: C18 RP (250 mm × 4.6 mm). An HPLC method the separation of Lns, Th, U and Pu in the irradiated fuel samples. Dynamically modified RP column was used for the separation of Lns employing dual gradient condition. The method was successfully applied for determining the concentrations of Lns. Determination of Th, U and Pu was carried out on RP column. Pu was maintained in single oxidation state using redox treatment with H2O2 and HNO3. Under the optimized conditions, the concen- tration of Pu was also determined which was in good agreement with the concentration obtained by ID-TIMS. Authors are thankful to Dr. K. L Radiochemistry and Isotope Group, BARC for the sup- port and encouragement and to Dr. Mrs. D. Alamelu, Head, Mass Spectrometry Section for her interest in HPLC work. Authors are thankful to Mr. A. R. Parab, Mass Spectrometry Section for providing mass spectro- metric analysis and staff members of FRD, BARC for carrying out the dissolution of irradiated fuel samples. et, B. N. Jagatap, K. G. Sahoo, “New Technologies for Thorium Fuel Cycle,” Proceeding of the INSAC-2000, Mumbai, 1-2 June 2000, p. 123. [2] K. Anan tion of Thoriumin Reactors,” Journal of Nuclear Materi- als, Vol. 383, No. 1-2, 2008, pp. 119-121. http://dx.doi.org/10.1016/j.jnucmat.2008.08 lopment des.2005.09.026 [3] R. K. Sinha and A. Kakodkar, “Design and Deve of the AHWR—The Indian Thoriumfuelled Innovative Nuclear Reactor,” Nuclear Engineering and Design, Vol. 236, No. 7-8, 2006, pp. 683-700. http://dx.doi.org/10.1016/j.nuceng Copyright © 2013 SciRes. IJAMSC  P. KUMAR ET AL. 79 [4] S. K. Aggarwal, P. G. Jaison, V. M. Telmore, P. S. Kho- ami, V. D. Kavi- n dade, R. V. Shah, R. Govindan, V. L. Sant and P. M. Shah, “Determination of Burn-Up of Irradiated PHWR Fuel Samples from KAPS-1 by Mass Spectrometry,” BARC Report No. BARC/2007/E/020, 2007. [5] B. Saha, R. Bagyalakshmi, G. Periasw mandan, S. A. Chitambar, H. C. Jain and C. K. Mathews, “Determination of Nuclear Fuel Burn-Up Using Mass Spectrometric Techniques,” BARC Report-891, 1977. [6] M. Gysemans, A. Dobney, L. Adriaensen and L. Sanne, “Destructive Radiochemical Burn-Up Determination at SCK-CEN Using Isotopes of Cs, Ce and Nd as Fission Product Monitors,” 2006. http://www.sckcen.be/HOTLAB/events/proceedings/2006 /HOTLAB%202006/Full%20Papers/13%20Gysemans.pdf [7] S. Koyama, M. Osaka, T. Sekine, K. Morozumi, T. Na- mekawa and M. Itoh, “Measurement of Burnup in FBR MOX Fuel Irradiated to High Burn Up,” Journal of Nu- clear Science and Technology, Vol. 40, 2003, pp. 998- 1013. http://dx.doi.org/10.3327/jnst.40.998 [8] K. L. Ramakumar, S. K. Aggarwal, V. D. Kavimandan, V. A. Raman, P. S. Khodade and H. C. Jain, “Separation and Purification of Magnesium, Lead, and Neodymium from Dissolver Solution of Irradiated Fuel,” Separation Sci- ence and Technology, Vol. 15, No. 7, 1980, pp. 1471- 1481. http://dx.doi.org/10.1080/01496398008056098 [9] I. Burcik and V. Mikulaj, “Separation of Thorium, Ura- nium and Plutonium by Neutral and Basic Organic Ex- tractants,” Journal of Radioanalytical and Nuclear Chem- istry, Vol. 150, No. 2, 1991, pp. 247-253. http://dx.doi.org/10.1007/BF02035309 [10] N. Dacheux and J. Aupias, “Determination of Uranium, Thorium, Plutonium, Americium and Curium Ultratraces by Photon Electron Rejecting α-Liquid Scintillation,” Analytical Chemistry, Vol. 69, No. 13, 1997, pp. 2275- 2282. http://dx.doi.org/10.1021/ac961209r [11] P. G. Jaison, V. M. Telmore, P. Kumar and K. Aggarwal, “Reversed-Phase Liquid Chromatography Using Mande- lic Acid as an Eluent for the Determination of Uranium in Presence of Large Amounts of Thorium,” Journal of Chromatography A, Vol. 1216, No. 9, 2009, pp. 1383- 1389. http://dx.doi.org/10.1016/j.chroma.2008.12.076 [12] N. R. Larsen, “High-Pressure Liquid Chromatography of Irradiated Nuclear Fuel: Separation of Neodymium for Burn-Up Determination,” Journal of Radioanalytical Che- mistry, Vol. 52, No. 1, 1979, pp. 85-91. http://dx.doi.org/10.1007/BF02517702 [13] P. E. Jackson, J. Carnevale, H. Fuping and P. R. Haddad, “Determination of Thorium and Uranium in Mineral Sands by Ion Chromatography,” Journal of Chromatography A, Vol. 671, No. 1-2, 1994, pp. 181-191. http://dx.doi.org/10.1016/0021-9673(94)80237-8 [14] S. Rollin, Z. Kopajtic, B. Wernli and B. Magyar, “Deter- mination of Lanthanides and Actinides in Uranium Mate- rials by High-Performance Liquid Chromatography with Inductively Coupled Plasma Mass Spectrometric Detec- tion,” Journal of Chromatography A, Vol. 739, No. 1-2, 1996, pp. 139-149. http://dx.doi.org/10.1016/0021-9673(96)00037-4 [15] M. R. Buchmeiser, G. Seeber and R. Tessadri, “Quantifi- cation of Lanthanides in Rocks Using Succinic Acid-De- rivatized Sorbents for On-Line SPE-RP-Ion-Pair HPLC,” Analytical Chemistry, Vol. 72, No. 11, 2000, pp. 2595- 2602. http://dx.doi.org/10.1021/ac991217i [16] G. Seeber, P. Brunner, M. R. Buchmeiser and G. K. Bonn, “Poly(7-oxanorborn-2-ene-5,6-dicarboxylate)-Coated Sil- ica Prepared by Ring-Opening Metathesis Polymerization for the Selective Enrichment of Radioactive Lantha- nides,” Journal of Chromatography A, Vol. 848, No. 1-2, 1999, pp. 193-202. http://dx.doi.org/10.1016/S0021-9673(99)00399-4 [17] R. M. Cassidy, S. Elchuk, N. L. Elliot, L. W. Green, C. H. Knight and B. M. Recoskie, “Dynamic Ion Exchange Chromatography for the Determination of Number of Fis- sions in Uranium Dioxide Fuels,” Analytical Chemistry, Vol. 58, No. 6, 1986, pp. 1181-1186. http://dx.doi.org/10.1021/ac00297a045 [18] C. H. Knight, R. M. Cassidy, B. M. Recoskie and L. W. Green, “Dynamic Ion Exchange Chromatography for De- termination of Number of Fissions in Thorium-Uranium Dioxide Fuels,” Analytical Chemistry, Vol. 56, No. 3, 1984, pp. 474-478. http://dx.doi.org/10.1021/ac00267a041 [19] N. Sivaraman, R. Kumar, S. Subramaniam and P. R. Va- sudeva Rao, “Separation of Lanthanides Using Ion-Inter- action Chromatography with HDEHP Coated Columns,” Journal of Radioanalytical and Nuclear Chemistry, Vol. 252, No. 3, 2002, pp. 491-495. http://dx.doi.org/10.1023/A:1015894418606 [20] N. Sivaraman, S. Subramaniam, T. G. Srinivasan and P. R. Vasudeva Rao, “Burn-Up Measurements on Nuclear Reactor Fuels Using High Performance Liquid Chroma- tography,” Journal of Radioanalytical and Nuclear Che- mistry, Vol. 253, No. 1, 2002, pp. 35-40. http://dx.doi.org/10.1023/A:1015800114488 [21] A. Datta, N. Sivaraman, K. S. Viswanathan, S. Ghosh, T. G. Srinivasan and P. R. Vasudeva Rao, “Correlation of Retention of Lanthanide and Actinide Complexes with Stability Constants and Their Speciation,” Radiochimica Acta, Vol. 101, No. 2, 2013, pp. 81-92. http://dx.doi.org/10.1524/ract.2013.2005 [22] H. Fuping, P. R. Haddad, P. E. Jackson and J. Carnevale, “Studies on the Retention Behaviour of α-Hydroxyiso- butyric Acid Complexes of Thorium(IV) and Uranyl Ion in Reversed-Phase High-Performance Liquid Chromato- graphy,” Journal of Chromatography A, Vol. 640, No. 1-2, 1993, pp. 187-194. http://dx.doi.org/10.1016/0021-9673(93)80181-7 [23] H. Fuping, B. Paull and P. R. Haddad, “Retention Beha- viour of Thorium(IV) and Uranyl on a Reversed-Phase Column with Glycolate and Mandelate as Eluents,” Jour- nal of Chromatography A, Vol. 739, No. 1-2, 1996, pp. 151-161. http://dx.doi.org/10.1016/0021-9673(96)81462-2 [24] S. Elchuk, K. I. Burns, R. M. Cassidy and C. A. Lucy, “Reversed-Phase Separation of Transition Metals, Lan- thanides and Actinides by Elution with Mandelic Acid,” Journal of Chromatography A, Vol. 558, No. 1, 1991, pp. Copyright © 2013 SciRes. IJAMSC  P. KUMAR ET AL. Copyright © 2013 SciRes. IJAMSC 80 oi.org/10.1016/0021-9673(91)80125-Z 197-207. http://dx.d ect De- .2006.04.037 [25] P. G. Jaison, N. M. Raut and S. K. Aggarwal, “Dir termination of Lanthanides in Simulated Irradiated Thoria Fuels Using Reversed-Phase High-Performance Liquid Chromatography,” Journal of Chromatography A, Vol. 1122, No. 1-2, 2006, pp. 47-53. http://dx.doi.org/10.1016/j.chroma R. Vasu-[26] A. Datta, N. Sivaraman, T. G. Srinivasan and P. deva Rao, “Liquid Chromatographic Behaviour of Acti- nides and Lanthanides on Monolith Supports,” Radio- chimica Acta, Vol. 99, 2011, pp. 275-283. http://dx.doi.org/10.1524/ract.2011.1816 [27] P. R. Nair, V. Akhileswaran, P. Venkataramana, M son and S. K. Aggarwal, “Separation oma.2004.08.054 . Xa- vier, S. Vaidyanathan and P. R. Natarajan, BARC Report, BARC/I-882, 1986. [28] N. M. Raut, P. G. Jai and Determination of Lanthanides, Thorium and Uranium Using a Dual Gradient in Reversed-Phase Liquid Chro- matography,” Journal of Chromatography A, Vol. 1052, No. 1-2, 2004, pp. 131-136. http://dx.doi.org/10.1016/j.chr varaman, .010 [29] Ch. Siva KesavaRaju, M. S. Subramanian, N. Si T. G. Srinivasan and P. R. Vasudeva Rao, “Retention Studies on Uranium, Thorium and Lanthanides with Am- ide Modified Reverse Phase Support and Its Applica- tions,” Journal of Chromatography A, Vol. 1156, No. 1-2, 2007, pp. 340-347. http://dx.doi.org/10.1016/j.chroma.2007.01 avier and [30] K. Jayachandran, J. S. Gamare, P. R. Nair, M. X S. K. Aggarwal, “A Novel Biamperometric Methodology for Thorium Determination by EDTA Complexometric Titration,” Radiochimica Acta, Vol. 100, 2012, pp. 311- 314. http://dx.doi.org/10.1524/ract.2012.1920
|