Paper Menu >>
Journal Menu >>
 Journal of Biomaterials and Nanobiotechnology, 2011, 2, 65-70 doi:10.4236/jbnb.2011.21009 Published Online January 2011 (http://www.SciRP.org/journal/jbnb) Copyright © 2011 SciRes. JBNB 65 Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Betcheva Rositza, Yordanov Dancho*, Yotova Lubov Department of Textile Chemistry, University of Chemical Technology and Metallurgy, Sofia, Bulgaria. Email: danchoyordanov@gmail.com Received September 10th, 2010; revised September 24th, 2010; accepted September 30th, 2010. ABSTRACT Scouring of raw wool is a chemical treatmen t that needs a high amount of detergents, a lkalis and water. Effluents pro- duced by this treatment are extremely polluted with chemicals and impurities washed out from the fibers. It is well known that the ultrasound washing can remove effectively different substances from the textile surfaces even without surfactants due to the cavitations occurring at certain parameters of the ultrasound field. On the other side water treatments of woo l comb in ed with mecha n ical ag ita tio n p rovo k ed felting which can impa ir the quality of wool materials. Felting itself depends not only on the parameters of water treatments but also on the structure of wool cuticle. Partial hydrolysis of the cuticle with some proteases can decrease considerably the wool felting. The aim of this work is to study the possibility of applying the ultrasound at the process of raw wool scouring and the influence of proteases on the felting properties of wool at these conditions. It has been found out that ultrasound environment applied does not impair the specific activity of enzyme auxiliaries used and leads to increasing of their effect on the surface of wool fi- bers. Thus the scouring process studied could be used for developing of a technology producing lower amount and less polluted effluents. Keywords: Enzyme, Felting, Scouring, Ultrasound, Wool 1. Introduction The tendency of wool to felt and shrink is mainly due to its scaly structure. Wool shrinkage occurs when the fi- bers are subjected to mechanical treatments combined with higher temperatures and use of different detergents. It is well known that the scouring of raw wool fibers is a textile chemical processing that needs a lot of detergents and water due to the high amount of impurities [1]. On the other side there are many data that ultrasound (US) could destroy and remove the pollutants on textile sur- faces even without any additional auxiliaries in the washing bath [2] Logically it can be expected that US will increase wool fiber felting occurring at the condi- tions of scouring. The chlorine-Hercosett is the most widespread process used to modify the surface of wool fibers in order to provide resistance to felting and shrinkage [3,17,18]. Currently a wide range of enzymatic treatments are being studied as an alternative of chemical treatments [4,20].The corresponding technologies of tex- tile processing usually combine the enzyme application with an intensive mass transfer (for example by stirring) [1,14,16]. There is no available data if US scouring of raw wool fibers is equally effective to the classical de- tergent washing and if it can cause higher wool felting and shrinkage. It is of a practical interest if the applica- tion of protease auxiliaries originally formulated for use as detergent additives can influence the process of US wool scouring. Bearing in mind the above mentioned the aim of this work is to study the possibility of applying the ultrasound at the process of raw wool scouring and the influence of proteases on the felting properties of wool at these condi- tions. 2. Experimental 2.1. Fibers and Chemicals Raw wool treated were Bulgarian merino fibers, quality 64 (22-25μm). The auxiliaries used were the enzyme containing Bioprot Multi and Bioprot Gentle (Biocon S.A.). 2.2. Enzyme Activity and Protein Concentration The activity of proteases in Bioprot Multi and Bioprot Gentle was measured by incubating 1ml of these prod-  Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Copyright © 2011 SciRes. JBNB 66 ucts with 1ml of 2% casein solution and 2ml 0,1 M bo- rate buffer, for 20 min at temperature 37˚C. After incu- bating the reaction was stopped by adding 4 ml 6% tri- cloroacetic acid solution, and then the precipitate was removed by centrifugation at 5000rpm, at room tem- perature for 10 min. The absorbance due to the amino acids produced was analysed at 280 nm. Temperature and pH dependences of enzyme activity were investigated as follows: - at different pH-values and 37˚C, - at pH 7.8, and different temperatures. The concentration of protein was determined by Lowry method [5], using bovine serum albumin as a standard. 2.3. Characterization of Wool Fibers Wool fibers (treated with enzyme and combined en- zyme/ultrasonic) were characterized by their felting properties and by the weight lost after treatment. The felting ability of wool fibers was evaluated by Blankenburg method. Diameters of wool spheres formed during the agitation test were measured and the changes of felting ability were calculated from them. The weight lost of pre-treatment wool fiber was calcu- lated in the following way. Wool fabrics were condi- tioned at 100˚C for 2 h, desiccated and weighed until constant weight was reached. 2.4. Determination of Tyrosine Tyrosine is a mino acid produced from the reaction of protein with enzyme [5]. Its concentration was measured in the following way. A sample of 6ml solution was taken from the enzyme reactor and added to 5ml of 0.11M tricloroacetic acid solution. The precipitate was removed by filtration and centrifugation. Then, 2ml of filtrate were mixed with 5ml of 0.05M Na2CO3 solution and 1ml of two-fold diluted Folin’s reagent. After mixing, the colour was allowed to develop for 30 min at 37˚C. The light absorbance related proportionally to the con- centration of amino acid produced was analyzed at 660 nm, based on dl-tyrosine as standard. 2.5. Experimental Design The experimental equipment designed for this study is shown on Figure 1. The enzymatic treatment (with and without US) of wool fibers was performed in a glass bot- tle with total volume of 300ml. Wool fiber samples of 3 g each were treated with enzymes in phosphate buffer solution (pH 7.8, 0.01M) in a water bath at 37˚C for 5 hours and 80 rpm. In the present study, the cavitations experiment (US irradiation treatment) were carried out by using an ultra- sonic bath form VWR-China with capacity 3,7L, HF-45 Figure 1. 3D plan view of the experimental equipment. kHz and 117V. The actual intensity of US was calculated according to the method introduced by Lorimer et al. (1991) [6]. The field intensity in the glass bottle of ultra- sonic vessel used for our study was 95 W/cm2. 3. Results and Discussion The proteolytic activity of auxiliaries used was calculated in order to give a more precise qualitative evaluation of the influence of US treatment. Proteolytic activity was defined according to Equation 1, pH7.8 and temperature of 37˚C on casein as substrate. Equation: 280nm U /mg(A1000)/(ta) (1) Where: - A is the adsorption of reaction media at 280nm, - a is the quantity enzyme in test chub, - t is the incubation time. The results of these measurements are given in Table 1. They show that the US applied generally increases the enzyme activity most probably by intensifying the mass transfer of reaction products. It is well know that the agi- tation increases the rate of enzymatic hydrolysis and the US in our case play the same role [7,14]. It is well known that every enzyme product has a spe- cific pH and temperature of application for having the highest effectiveness of application. That is why our first task in the presented study was to evaluate the activity of enzyme auxiliaries used. The influence of pH on this parameter can be seen at Figure 2. The results obtained showed that the application of US did not change the position of the maximum of pH de- pendence. For Bioprot Multy these maximums are in pH Table 1. Enzyme activity of the Bioprot Multi and Bioprot Gentle at pH 7.8 and 37˚C on casein as substrate. Enzyme Bioprot Multi, U /mg Bioprot Gentle,U/mg With ultrasound 89672 87570 Without ultrasound 81159 81125 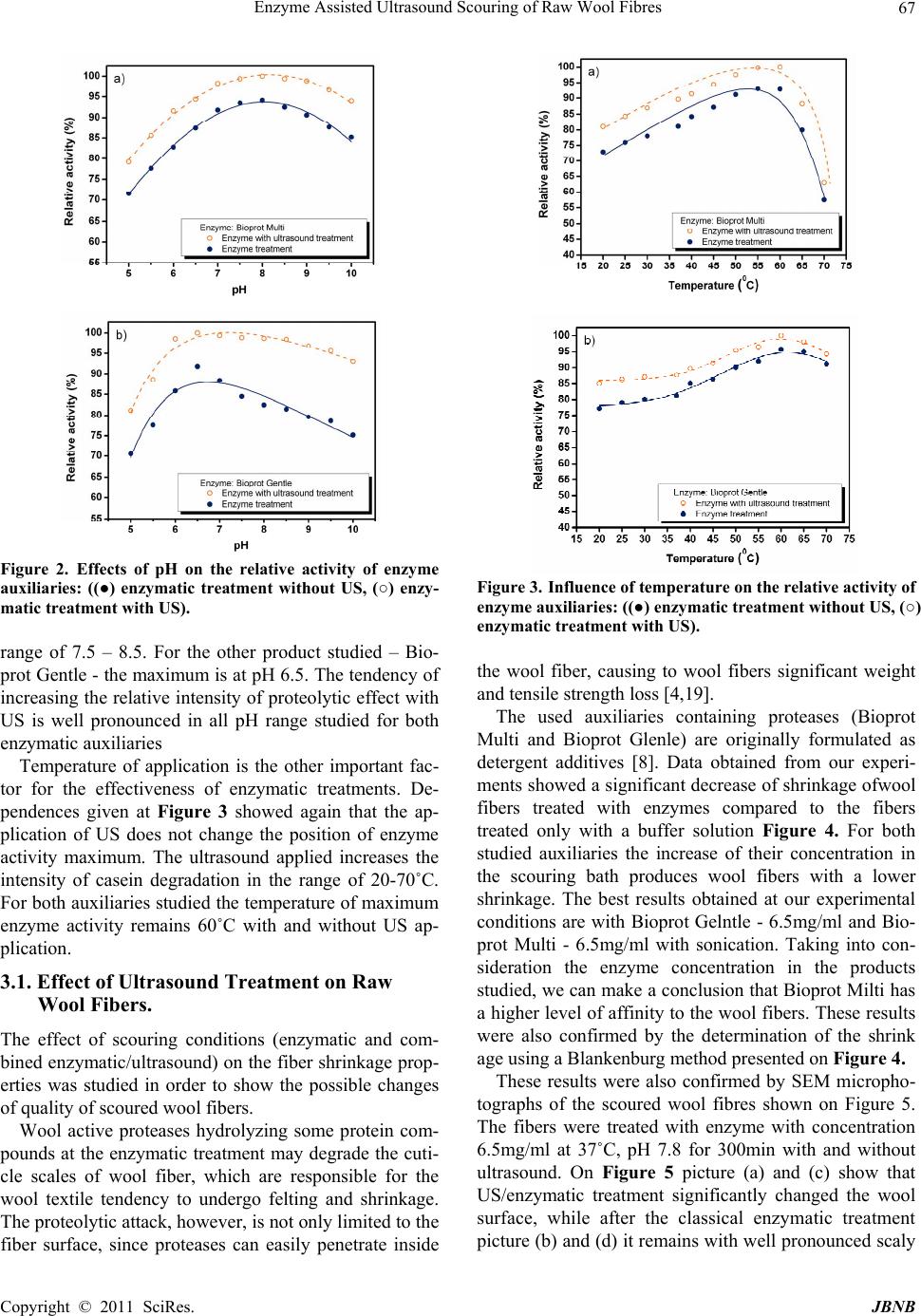 Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Copyright © 2011 SciRes. JBNB 67 Figure 2. Effects of pH on the relative activity of enzyme auxiliaries: ((●) enzymatic treatment without US, (○) enzy- matic treatment with US). range of 7.5 – 8.5. For the other product studied – Bio- prot Gentlе - the maximum is at pH 6.5. The tendency of increasing the relative intensity of proteolytic effect with US is well pronounced in all pH range studied for both enzymatic auxiliaries Temperature of application is the other important fac- tor for the effectiveness of enzymatic treatments. De- pendences given at Figure 3 showed again that the ap- plication of US does not change the position of enzyme activity maximum. The ultrasound applied increases the intensity of casein degradation in the range of 20-70˚C. For both auxiliaries studied the temperature of maximum enzyme activity remains 60˚C with and without US ap- plication. 3.1. Effect of Ultrasound Treatment on Raw Wool Fibers. The effect of scouring conditions (enzymatic and com- bined enzymatic/ultrasound) on the fiber shrinkage prop- erties was studied in order to show the possible changes of quality of scoured wool fibers. Wool active proteases hydrolyzing some protein com- pounds at the enzymatic treatment may degrade the cuti- cle scales of wool fiber, which are responsible for the wool textile tendency to undergo felting and shrinkage. The proteolytic attack, however, is not only limited to the fiber surface, since proteases can easily penetrate inside Figure 3. Influence of temperature on the relative activity of enzyme auxiliaries: ((●) enzymatic treatment without US, (○) enzymatic treatment with US). the wool fiber, causing to wool fibers significant weight and tensile strength loss [4,19]. The used auxiliaries containing proteases (Bioprot Multi and Bioprot Glenle) are originally formulated as detergent additives [8]. Data obtained from our experi- ments showed a significant decrease of shrinkage ofwool fibers treated with enzymes compared to the fibers treated only with a buffer solution Figure 4. For both studied auxiliaries the increase of their concentration in the scouring bath produces wool fibers with a lower shrinkage. The best results obtained at our experimental conditions are with Bioprot Gelntle - 6.5mg/ml and Bio- prot Multi - 6.5mg/ml with sonication. Taking into con- sideration the enzyme concentration in the products studied, we can make a conclusion that Bioprot Milti has a higher level of affinity to the wool fibers. These results were also confirmed by the determination of the shrink age using a Blankenburg method presented on Figure 4. These results were also confirmed by SEM micropho- tographs of the scoured wool fibres shown on Figure 5. The fibers were treated with enzyme with concentration 6.5mg/ml at 37˚C, pH 7.8 for 300min with and without ultrasound. On Figure 5 picture (a) and (c) show that US/enzymatic treatment significantly changed the wool surface, while after the classical enzymatic treatment picture (b) and (d) it remains with well pronounced scaly 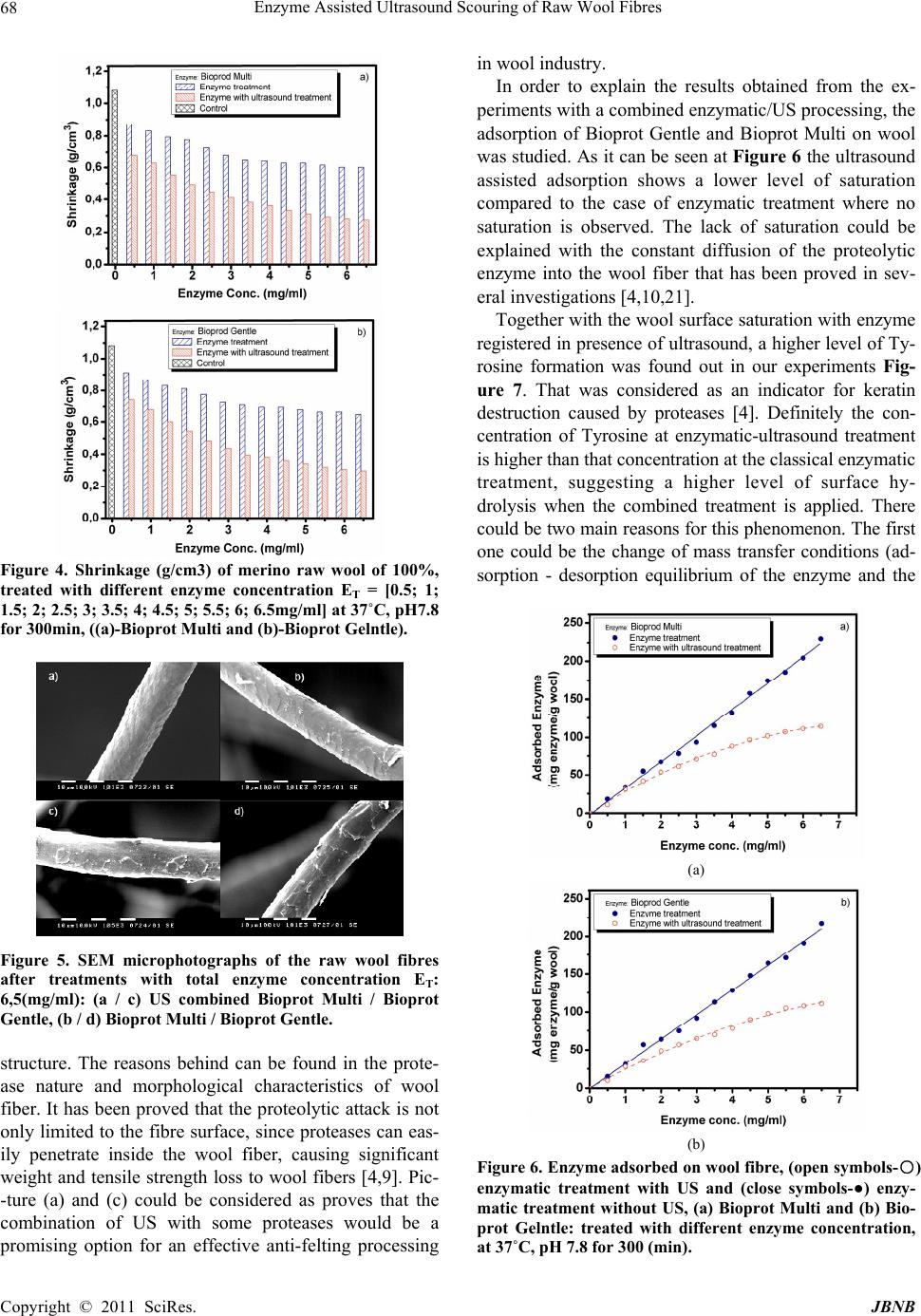 Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Copyright © 2011 SciRes. JBNB 68 Figure 4. Shrinkage (g/cm3) of merino raw wool of 100%, treated with different enzyme concentration ET = [0.5; 1; 1.5; 2; 2.5; 3; 3.5; 4; 4.5; 5; 5.5; 6; 6.5mg/ml] at 37˚C, pH7.8 for 300min, ((a)-Bioprot Multi and (b)-Bioprot Gelntle). Figure 5. SEM microphotographs of the raw wool fibres after treatments with total enzyme concentration ET: 6,5(mg/ml): (a / c) US combined Bioprot Multi / Bioprot Gentle, (b / d) Bioprot Multi / Bioprot Gentle. structure. The reasons behind can be found in the prote- ase nature and morphological characteristics of wool fiber. It has been proved that the proteolytic attack is not only limited to the fibre surface, since proteases can eas- ily penetrate inside the wool fiber, causing significant weight and tensile strength loss to wool fibers [4,9]. Pic- -ture (a) and (c) could be considered as proves that the combination of US with some proteases would be a promising option for an effective anti-felting processing in wool industry. In order to explain the results obtained from the ex- periments with a combined enzymatic/US processing, the adsorption of Bioprot Gentle and Bioprot Multi on wool was studied. As it can be seen at Figure 6 the ultrasound assisted adsorption shows a lower level of saturation compared to the case of enzymatic treatment where no saturation is observed. The lack of saturation could be explained with the constant diffusion of the proteolytic enzyme into the wool fiber that has been proved in sev- eral investigations [4,10,21]. Together with the wool surface saturation with enzyme registered in presence of ultrasound, a higher level of Ty- rosine formation was found out in our experiments Fig- ure 7. That was considered as an indicator for keratin destruction caused by proteases [4]. Definitely the con- centration of Tyrosine at enzymatic-ultrasound treatment is higher than that concentration at the classical enzymatic treatment, suggesting a higher level of surface hy- drolysis when the combined treatment is applied. There could be two main reasons for this phenomenon. The first one could be the change of mass transfer conditions (ad- sorption - desorption equilibrium of the enzyme and the (a) (b) Figure 6. Enzyme adsorbed on wool fibre, (open symbols-○) enzymatic treatment with US and (close symbols-●) enzy- matic treatment without US, (a) Bioprot Multi and (b) Bio- prot Gelntle: treated with different enzyme concentration, at 37˚C, pH 7.8 for 300 (min). 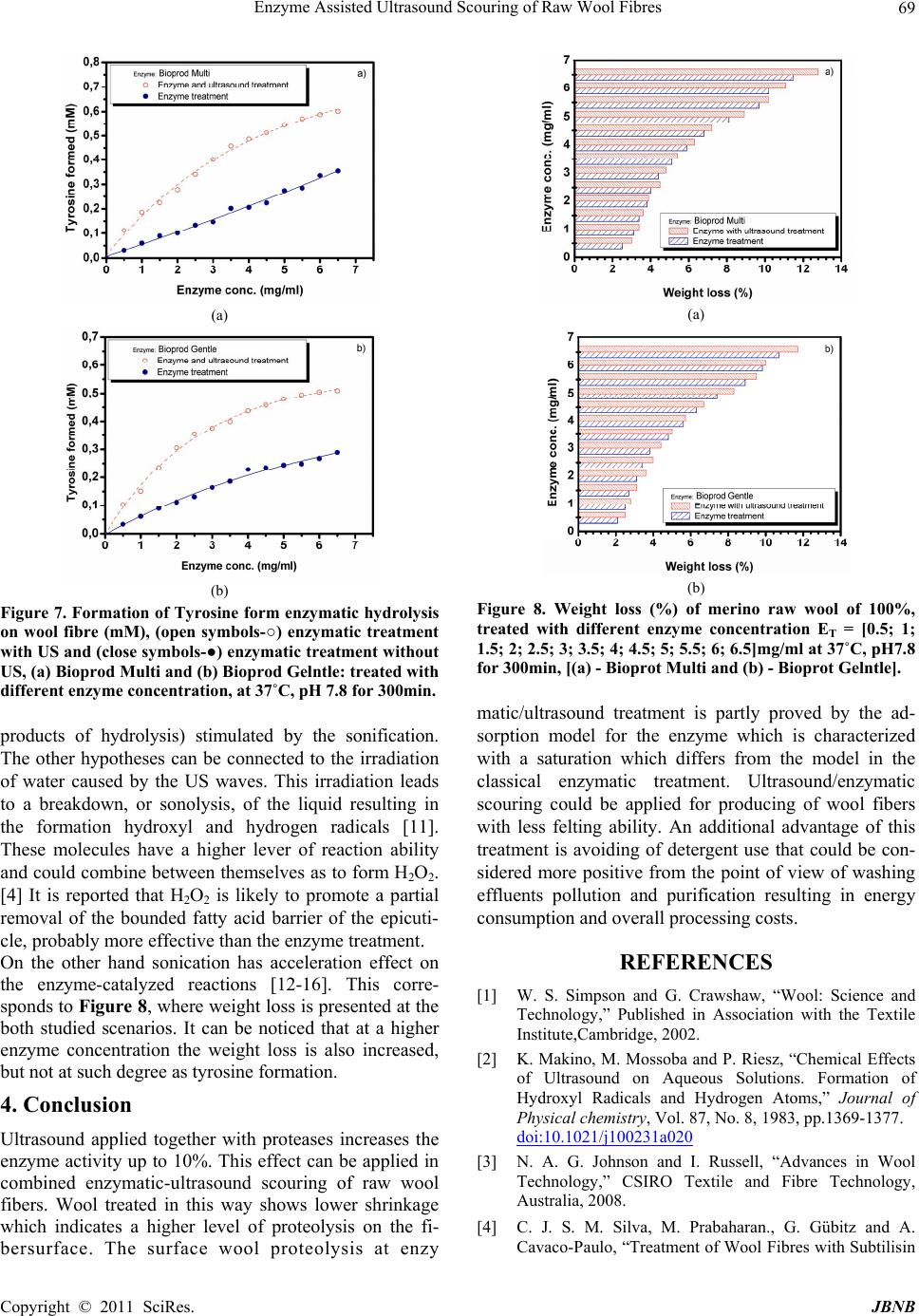 Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Copyright © 2011 SciRes. JBNB 69 (a) (b) Figure 7. Formation of Tyrosine form enzymatic hydrolysis on wool fibre (mM), (open symbols-○) enzymatic treatment with US and (close symbols-●) enzymatic treatment without US, (a) Bioprod Multi and (b) Bioprod Gelntle: treated with different enzyme concentration, at 37˚C, pH 7.8 for 300min. products of hydrolysis) stimulated by the sonification. The other hypotheses can be connected to the irradiation of water caused by the US waves. This irradiation leads to a breakdown, or sonolysis, of the liquid resulting in the formation hydroxyl and hydrogen radicals [11]. These molecules have a higher lever of reaction ability and could combine between themselves as to form H2O2. [4] It is reported that H2O2 is likely to promote a partial removal of the bounded fatty acid barrier of the epicuti- cle, probably more effective than the enzyme treatment. On the other hand sonication has acceleration effect on the enzyme-catalyzed reactions [12-16]. This corre- sponds to Figure 8, where weight loss is presented at the both studied scenarios. It can be noticed that at a higher enzyme concentration the weight loss is also increased, but not at such degree as tyrosine formation. 4. Conclusion Ultrasound applied together with proteases increases the enzyme activity up to 10%. This effect can be applied in combined enzymatic-ultrasound scouring of raw wool fibers. Wool treated in this way shows lower shrinkage which indicates a higher level of proteolysis on the fi- bersurface. The surface wool proteolysis at enzy (a) (b) Figure 8. Weight loss (%) of merino raw wool of 100%, treated with different enzyme concentration ET = [0.5; 1; 1.5; 2; 2.5; 3; 3.5; 4; 4.5; 5; 5.5; 6; 6.5]mg/ml at 37˚C, pH7.8 for 300min, [(a) - Bioprot Multi and (b) - Bioprot Gelntle]. matic/ultrasound treatment is partly proved by the ad- sorption model for the enzyme which is characterized with a saturation which differs from the model in the classical enzymatic treatment. Ultrasound/enzymatic scouring could be applied for producing of wool fibers with less felting ability. An additional advantage of this treatment is avoiding of detergent use that could be con- sidered more positive from the point of view of washing effluents pollution and purification resulting in energy consumption and overall processing costs. REFERENCES [1] W. S. Simpson and G. Crawshaw, “Wool: Science and Technology,” Published in Association with the Textile Institute,Cambridge, 2002. [2] K. Makino, M. Mossoba and P. Riesz, “Chemical Effects of Ultrasound on Aqueous Solutions. Formation of Hydroxyl Radicals and Hydrogen Atoms,” Journal of Physical chemistry, Vol. 87, No. 8, 1983, pp.1369-1377. doi:10.1021/j100231a020 [3] N. A. G. Johnson and I. Russell, “Advances in Wool Technology,” CSIRO Textile and Fibre Technology, Australia, 2008. [4] C. J. S. M. Silva, M. Prabaharan., G. Gübitz and A. Cavaco-Paulo, “Treatment of Wool Fibres with Subtilisin  Enzyme Assisted Ultrasound Scouring of Raw Wool Fibres Copyright © 2011 SciRes. JBNB 70 and Subtilisin-PEG,” Enzyme and Microbial Technology, Vol. 36, No. 7, 2005, pp. 917-922. [5] O. H. Lowry, W. J. Rosenberg, A. L. Farr and R. J. Randell, “Quantitation of Protein Using Folin Ciocalteau Reagent,” Journal of Biological Chemistry, 951, 193, pp. 265-75. [6] J. P. Lorimer, T. Mason and K. Fiddy, “Enhancement of Chemical Reactivity by Power Ultrasound: An Alternative Interpretation of the Hot Spot,” Ultrasonics, Vol. 29, No. 4, 1991, pp. 338-343. doi:10.1016/0041-624X(91)90032-4 [7] Y. Ishimori, I. Karube and S. Suzuki, “Acceleration of Immobilized Chymotrypsin Activity with Ultrasonic Irradiation,” Journal of Molecular Catalysis, Vol. 12, No. 2, 1981, pp. 253-259. doi:10.1016/0304-5102(81)80012-0 [8] N. Hmidet, N. El-Hadj Ali, A. Haddar, S. Kanoun, S.-K. Alya and M. Nasri, “Alkaline Proteases and Thermostable Amylase Co-Produced by Bacillus Licheniformis NH1: Characterization and Potential Application as Detergent Additive,” Biochemical Engineering Journal, Vol. 47, No. 1-3, 2009, pp. 71-79. doi:10.1016/j.bej.2009.07.005 [9] C. Hurren, P. Cookson and X. Wang, “The Effects of Ultrasonic Agitation in Laundering on the Properties of Wool Fabrics,” Ultrason Sonochem. Vol. 15, No. 6, 2008, 1069-1074. doi:10.1016/j.ultsonch.2008.04.002 [10] X.-W. Yu, W.-J. Guan, Y.-Q. Li, T.-J. Guo and J.-D. Zhou, “A Biological Treatment Technique for Wool Textile,” Brazilian Archives of Biology and Technology, Vol. 48, No. 5, 2005, pp. 675-680. [11] C. Little, M. El-Sharif and M. J. Hepher. “The Effect of Solution Level on Calorific and Dosimetric Results in a 70 Khz Tower Type Sonochemical Reactor,” Ultrasonics Sonochemistry, Vol. 14, No. 3, 2007, pp. 375-379. doi:10.1016/j.ultsonch.2006.07.009 [12] Y. Yan, L. Feng, C. Zhang, H. Zhu and Q. Zhou, “Effect of Ultrasonic Specific Energy on Waste Activated Sludge Solubilization and Enzyme Activity,” African Journal of Biotechnology, Vol. 9, No. 12, March 2010, pp. 1776-1782. [13] P. C. Sangave and A. B. Pandit, “Ultrasound and Enzyme Assisted Biodegradation of Distillery Wastewater,” Journal of Environmental Management, Vol. 80, No. 1, 2006, pp. 36-46. doi:10.1016/j.jenvman.2005.08.010 [14] V. G. Yachmenev, N. R. Bertoniere and E. J. Blanchard. “Intensification of the Bio-processing of Cotton Textiles by Combined Enzyme/Ultrasound Treatment,” Jurnal of Chemical Technology and Biotechnology, Vol. 77, No. 5, 2002, pp. 559-567. doi:10.1002/jctb.579 [15] Y.-M. Xiao, Q. Wu, Y. Cai and X.-F. Lin, “Ultrasound- Accelerated Enzymatic Synthesis of Sugar Esters in Nonaqueous Solvents,” Carbohydrate Research, Vol. 340, No. 13, 2005, pp. 2097-2103. [16] C. Basto, T. Tzanov and A. Cavaco-Paulo, “Combined Ultrasound-Laccase Assisted Bleaching of Cotton,” Ultrasonics Sonochemistry, Vol. 14, No. 3, 2007, pp. 350-354. doi:10.1016/j.ultsonch.2006.07.006 [17] H. Nolte, D. Bishop and H. Hocker, “Effects of Proteolytic And Lipolytic Enzymes on Untreated and Shrink Resist Treated Wool,” Journal of the Textile Institute, Vol. 87, No. 1, 1996, pp. 212-226. doi:10.1080/00405009608659069 [18] J. Cortez, P. L. R. Bonner and M. Griffin, “Application of Transglutaminases in the Modification of Wool Textiles,” Enzyme Microbial Technol, Vol. 34, No. 1, 2004, pp.64-72. doi:10.1016/j.enzmictec.2003.08.004 [19] K. Sawada and M. Ueda, “Enzyme Processing of Textiles in Reverse Micellar Solution,” Journal of Biotechnology, Vol. 89, No. 2-3, 2001, pp.263-269. doi:10.1016/S0168-1656(01)00310-8 [20] I. Bearpark, F. Marriott and J. Park, “A Practical Introduction to the Dyeing and Finishing of Wool Fabrics,” Society of Dyers and Colourists, Bradford, 1986. [21] C. J. S. M. Silva, Q. Z., J. Shen and A. Cavaco-Paulo, “Immobilization of Proteases with a Water Soluble- Insoluble Reversible Polymer for Treatment of Wool,” Enzyme and Microbial Technology, Vol. 39, No. 4, 2006, pp. 634-640. doi:10.1016/j.enzmictec.2005.11.016 |

