 World Journal of AIDS, 2013, 3, 221-230 http://dx.doi.org/10.4236/wja.2013.33030 Published Online September 2013 (http://www.scirp.org/journal/wja) 221 The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection Martínez Pedro Ariel1*, Kourí Vivian1*, Blanco Orestes1, Capó Virginia1, Abad Yoandra1, Alemán Yoan1, Verdasquera Denis1, Jiménez Narciso1, Caballero Iraida2, Fleites Gilberto1, Ugarte Yaumara1, Calderón Odalys1, Álvarez Alina1, Ulrich Hengge3 1Institute of Tropical Medicine “Pedro Kourí” (IPK), Autopista Novia del Mediodía, Habana, Cuba; 2“Hermanos Ameijeiras” Hospi- tal, Havana, Cuba; 3Haut Zentrum (Skin Center), Dusseldorf, Germany. Email: arielmr@ipk.sld.cu, arimaroz@infomed.sld.cu Received April 16th, 2013; revised May 16th, 2013; accepted June 16th, 2013 Copyright © 2013 Martínez Pedro Ariel et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Despite the progress has been reached with Human herpesvirus 8 (HHV-8) research, there are gaps in the knowledge of viral induced oncogenesis. The aim of the present study was to identify possible associations between HHV-8 subtypes, HHV-8 loads and clinical manifestations of HIV infected patients diagnosed with different malignancies associated with HHV-8 infection. Forty six HIV-1 infected individuals diagnosed with different HHV-8 associated diseases were studied [37 epidemic Kaposi’s sarcoma (KS), 3 pleural effusion lymphoma (PEL); 5 peripheral lymphadenopathies (PL); 1 Hodgkin’s lymphoma (HL); 1 non Hodgkin’s lymphoma (NHL)]. HHV-8 loads were determined by quantitative real time PCR (qRT-PCR) whilst HHV-8 subtypes were determined by open-reading frame (ORF)-K1 gen genotyping. HHV-8 subtypes B, A, C, A5 and E were exhibited by 31.8%, 23.4%, 19.1%, 17% and 8.5% of the studied patients, respectively. The median HHV-8 viral load did not differ between subtypes (p > 0.05) but HHV-8 viral loads were sig- nificantly higher in PEL than in epidemic KS lesion or lymph nodes (p = 0.04). Subtype B was detected in 60% of pa- tients with B cell lymphoma (NHL, PEL and HL) whereas subtype E was only detected in patients with epidemic KS diagnosis. Our data suggest that HHV-8 DNA quantification instead of subtype identification could be used as a surro- gate marker for monitoring its infection, not only in epidemic KS patients but also in HIV infected individuals with lymphoproliferative disorders. Keywords: Human Herpesvirus 8 or Kaposi’s Sarcoma-Associated Herpesvirus; Real Time PCR; Subtypes; Lymphoproliferative Disorders; Cuban; HIV/AIDS 1. Introduction HHV-8 also referred as Kaposi’s Sarcoma-Associated Herpesvirus is the first known human Rhadinovirus which belongs to the Gammaherpesvirinae subfamily, Herpes- viridae family. It is implicated as the causative agent of all clinical forms of Kaposi’s Sarcoma (classical, epide- mic, endemic and iatrogenic), PEL and multicentric Cas- tleman’s disease (MCD) [1] but its role in other lympho- proliferative disorders has not been clearly established. Although the HHV-8 genome is highly conserved along most of the unique coding region (approx. 145 kbp), sev- eral genomic regions display remarkable sequence vari- ability making them useful markers of strain diversity and potential epidemiologic patterns of HHV-8 spread. Five major subtypes are recognized by K1 genotyping: A, B, C, D and E. The K1 protein is highly variable in its cysteine-rich N-terminal ectodomain and its amino acid sequence has been shown to vary by up to 40%, with changes concentrated in two hypervariable regions (VR1 and VR2) [2]. The influence of particular subtypes on disease pro- gression remains largely unknown. Therefore, it is im- portent to identify biological markers related with HHV- *These authors have equally contributed. Copyright © 2013 SciRes. WJA  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 222 8-associated diseases outcome. Some authors have alert- ed that the risk of KS remains substantially increased in HIV infected subjects and further decreases have not been observed [3]; being KS recently diagnosed in patients with controlled HIV infection and CD4 counts over 200 T cells [4]. Epidemic KS incidence has not been signifi- cantly reduced in Cuba although locally produced anti- retroviral drugs started being introduced as a form of treatment since 2001. Previous studies have shown that, with the exception of subtype D, the majority of HHV-8 subtypes were circulating among Cuban HIV population [5,6]. However, a change in HHV-8 subtype distribution was recently identified with an increase in subtype B detection [7]. Thus, we decided to identify how viral fac- tors (K1 subtype and viral load) may impact on the onco- genesis induced by HHV-8 among HIV infected indivi- duals diagnosed with different malignancies. 2. Materials and Methods 2.1. Patients and Samples The study was approved by local and national ethics com- mittees. All participants provided their written informed consent to participate in the study. Forty six HIV-1 in- fected individual diagnosed with HHV-8 associated dis- eases between 2005 and 2011 inclusive were included and different samples were collected (Table 1). One pa- tient contributed with two samples since KS in the lymph node was diagnosed six month after the initial diagnosis of PEL in the pericardium. The diagnosis of vasoprolife- rative lesion of the lymph node and the AIDS-related complex lymphadenitis were based on the histopatholo- gical diagnosis as described elsewhere [8]. Clinical, im- munological and epidemiological data from each patient were obtained throughout a retrospective review of indi- vidual’s clinical records and are depicted in Table 2. 2.2. Sampling and DNA Extraction DNA was extracted from frozen tissues samples by QIAamp® DNA Mini Kit (QIAGEN, Germany), accord- ing to the protocol for DNA purification from tissues de- scribed by the manufacturer. In contrast, 200 µL of the effusion fluid from PEL patients and the sample of saliva obtained from the patient diagnosed with Hodgkin lym- phoma were used to purify DNA using the same kit but following the protocol for blood or body fluids. Genomic DNA (gDNA) concentration was determined by spectro- photometer (GeneQuant II, Pharmacia Biotech, USA) and adjusted to 100 ng (10 µL). 2.3. qRT-PCR for HHV-8 DNA Quantification An “in-house” Taqman methodology based qRT-PCR was used for HHV-8 quantification in clinical samples. The protocol conditions, primers and probe were de- scribed by Watzinger et al. [9] with minor modifications adapted for the LightCycler 1.5 [10]. The HHV-8 loads were expressed as copies/100 ng of DNA. Human glo- bin was amplified by Real Time PCR using primers, probes and the protocol previously described elsewhere [11]. 2.4. DNA Sequencing of HHV-8 ORF K1 A fragment of ORF-K1 gen was amplified by nested PCR from the assayed samples following the protocol published elsewhere [2]. PCR products from nested PCR were purified using MiniEluteTM Purification Kit (QIAGEN, Germany) following the manufacturer’s pro- tocol and their final concentration were evaluated through- out horizontal electrophoresis in 2% agarose gel stained with ethidium bromide (0.5 μg/mL) and visualized through a UV transilluminator. The Kit Dye labeled dideoxy Terminator Cycle Se- quencing from Beckman Coulter (USA) was used for nu- cleotide sequence analysis, following the manufacturer recommendations. One hundred fentomoles of PCR pro- ducts were added to a mixture containing 1 µl (5 pmol) of either forward or reverse primer (LGH2090 and LGH2508), 8 µl of sequence reaction mixture (DTCS Quick Star Master Mix) and water up to a final volume of 20 µl. The sequencing reaction was conducted by 50 cycles of two minutes at 96˚C for 20 seconds, 50˚C for 20 seconds and finally 60˚C for 4 minutes. The obtained fragments were purified following the manufacturer in- structions. Finally, the purified products underwent elec- trophoresis on a Beckman Coulter CEQ8800 sequencer. 2.5. Sequence Analysis Initial evaluation of each sequence was performed using the nucleotide search engine BLAST at NCBI (USA), to confirm that the amplified product was K1. Then, nucle- otide sequences were manually edited with both forward and reverse primers using MEGA version 4 [12]. To de- termine the K1 subtype of each subject’s strain(s), nu- cleotide sequences were aligned by Muscle in Jalview version 2.4 [13] along with the following reference strains obtained from Genetic sequence database (Gen-Bank) at the National Center for Biotechnology Information (NCBI) [AF133038 (A1); AF130305 (A2); U86667 (A3); AF133039 (A4); AF178823 (A5); AF133040 (B1); AF130259 (B2); AF133041 (C1); AF133042 (C3); AF133043 (D1) AF133044 (D2); AF220292 (E)]. Maximum likelihood (ML) trees were estimated using PAUP 4.0 beta under the best-fit substitution model cal- culated by Modeltest 3.7 [14] using the Akaike informa- tion criterion. New HHV-8 nucleotide sequences and the Copyright © 2013 SciRes. WJA  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection Copyright © 2013 SciRes. WJA 223 Table 1. HHV-8 subtypes identified by ORF-K1 genotyping from HIV-1 infected individuals in which HHV-8 associated dis- eases were diagnosed between 2005 and 2011 inclusive. Specimen ID Age Gender KSHV subtype Diagnosis materialHistopathological diagnosis Accession number Cub-B06-576 22 Female A5 Tissue Kaposi’s sarcoma FJ986113 Cub-81/07 31 Male A5 Tissue Kaposi’s sarcoma FJ986114 Cub-209/06 35 Male A Tissue Kaposi’s sarcoma FJ986115 Cub-551/06 46 Male A5 Tissue Kaposi’s sarcoma FJ986116 Cub-2033/05 29 Male A5 Tissue Kaposi’s sarcoma FJ986117 Cub-758/06 36 Male A5 Tissue Kaposi’s sarcoma FJ986118 Cub-310/07 44 Male A Tissue Kaposi’s sarcoma FJ986119 Cub-B06-603 22 Male A Tissue Kaposi’s sarcoma FJ986120 Cub-473/07 35 Male A Tissue Kaposi’s sarcoma FJ986121 Cub-1064/06 30 Male A Tissue Kaposi’s sarcoma FJ986122 Cub-2032/05 45 Male B Tissue Kaposi’s sarcoma FJ986123 Cub-553/06 41 Male B Tissue Kaposi’s sarcoma FJ986124 Cub-737/06 46 Male B Tissue Kaposi’s sarcoma FJ986126 Cub-557/06 41 Male B Tissue Kaposi’s sarcoma FJ986128 Cub-1373/07 42 Male B Tissue Kaposi’s sarcoma FJ986129 Cub-626/06 39 Male B Tissue Kaposi’s sarcoma FJ986131 Cub-1109/06 37 Male B Tissue Kaposi’s sarcoma FJ986132 Cub-1835/06 36 Male B Tissue Kaposi’s sarcoma FJ986133 Cub-427/07 41 Male B PEL pleural Kaposi’s sarcoma FJ986134 Cub-9/07 41 Male E Tissue Kaposi’s sarcoma FJ986135 Cub-1105/06 50 Male E Tissue Kaposi’s sarcoma FJ986136 Cub-127/07 29 Male E Tissue Kaposi’s sarcoma FJ986137 Cub-286/07 43 Male C Tissue Kaposi’s sarcoma FJ986138 Cub-296/09 a 25 Male C PEL pericardium PEL FJ986139 Cub-134/06 29 Male C Tissue Kaposi’s sarcoma FJ986140 Cub-234/06 57 Male C Tissue Kaposi’s sarcoma FJ986141 Cub-1106/06 36 Male A Tissue Kaposi’s sarcoma FJ986142 Cub-556I/06 30 Male A Tissue Kaposi’s sarcoma FJ986143 Cub-375/07 35 Male C Tissue Kaposi’s sarcoma FJ986144 1T/2009 32 Male A5 Lymph node AIDS-related complex lymphadenitis GU475457 17T/2009 34 Male B Lymph node KS in the lymph node GU475458 32T/2009 25 Male A Lymph node Vasoproliferative lesion of the lymph node GU475459 50T/2009a 25 Male C Lymph node KS in the lymph node GU475460 Cub-58LN/2009 43 Male B Lymph node non Hodgkin’s lymphoma JF979530 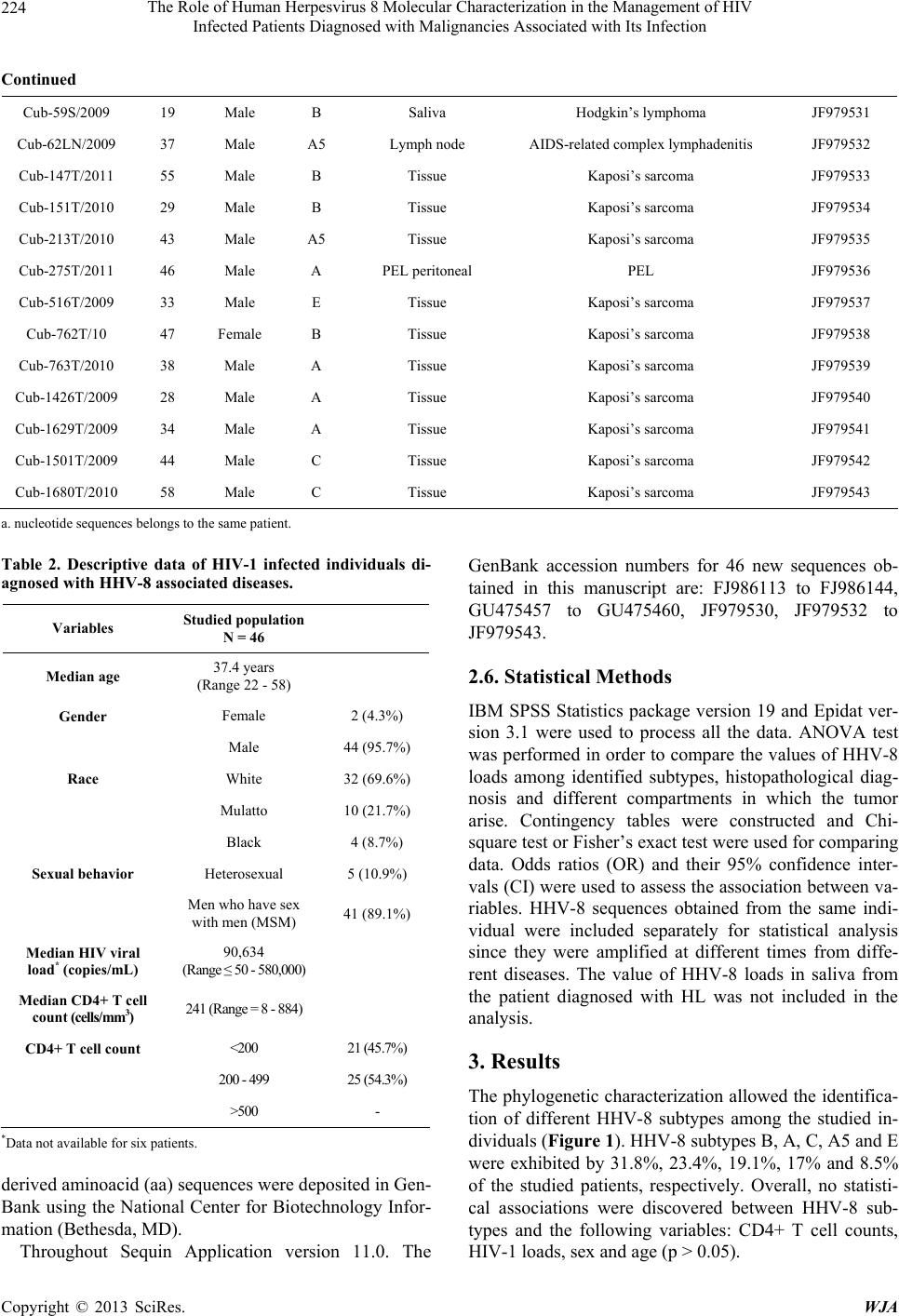 The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 224 Continued Cub-59S/2009 19 Male B Saliva Hodgkin’s lymphoma JF979531 Cub-62LN/2009 37 Male A5 Lymph node AIDS-related complex lymphadenitis JF979532 Cub-147T/2011 55 Male B Tissue Kaposi’s sarcoma JF979533 Cub-151T/2010 29 Male B Tissue Kaposi’s sarcoma JF979534 Cub-213T/2010 43 Male A5 Tissue Kaposi’s sarcoma JF979535 Cub-275T/2011 46 Male A PEL peritoneal PEL JF979536 Cub-516T/2009 33 Male E Tissue Kaposi’s sarcoma JF979537 Cub-762T/10 47 Female B Tissue Kaposi’s sarcoma JF979538 Cub-763T/2010 38 Male A Tissue Kaposi’s sarcoma JF979539 Cub-1426T/2009 28 Male A Tissue Kaposi’s sarcoma JF979540 Cub-1629T/2009 34 Male A Tissue Kaposi’s sarcoma JF979541 Cub-1501T/2009 44 Male C Tissue Kaposi’s sarcoma JF979542 Cub-1680T/2010 58 Male C Tissue Kaposi’s sarcoma JF979543 a. nucleotide sequences belongs to the same patient. Table 2. Descriptive data of HIV-1 infected individuals di- agnosed with HHV-8 associated diseases. Variables Studied population N = 46 Median age 37.4 years (Range 22 - 58) Gender Female 2 (4.3%) Male 44 (95.7%) Race White 32 (69.6%) Mulatto 10 (21.7%) Black 4 (8.7%) Sexual behavior Heterosexual 5 (10.9%) Men who have sex with men (MSM) 41 (89.1%) Median HIV viral load* (copies/mL) 90,634 (Range ≤ 50 - 580,000) Median CD4+ T cell count ( c e l l s / m m 3) 241 (Range = 8 - 884) CD4+ T cell count <200 21 (45.7%) 200 - 499 25 (54.3%) >500 - *Data not available for six patients. derived aminoacid (aa) sequences were deposited in Gen- Bank using the National Center for Biotechnology Infor- mation (Bethesda, MD). Throughout Sequin Application version 11.0. The GenBank accession numbers for 46 new sequences ob- tained in this manuscript are: FJ986113 to FJ986144, GU475457 to GU475460, JF979530, JF979532 to JF979543. 2.6. Statistical Methods IBM SPSS Statistics package version 19 and Epidat ver- sion 3.1 were used to process all the data. ANOVA test was performed in order to compare the values of HHV-8 loads among identified subtypes, histopathological diag- nosis and different compartments in which the tumor arise. Contingency tables were constructed and Chi- square test or Fisher’s exact test were used for comparing data. Odds ratios (OR) and their 95% confidence inter- vals (CI) were used to assess the association between va- riables. HHV-8 sequences obtained from the same indi- vidual were included separately for statistical analysis since they were amplified at different times from diffe- rent diseases. The value of HHV-8 loads in saliva from the patient diagnosed with HL was not included in the analysis. 3. Results The phylogenetic characterization allowed the identifica- tion of different HHV-8 subtypes among the studied in- dividuals (Figure 1). HHV-8 subtypes B, A, C, A5 and E were exhibited by 31.8%, 23.4%, 19.1%, 17% and 8.5% of the studied patients, respectively. Overall, no statisti- cal associations were discovered between HHV-8 sub- types and the following variables: CD4+ T cell counts, HIV-1 loads, sex and age (p > 0.05). Copyright © 2013 SciRes. WJA  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 225 Figure 1. Unrooted maximum likelihood tree generated by PAUP 4.0 beta under the best-fit substitution model calculated by Modeltest 3.7 using the Akaike information criterion with a fragment of the K1 gene of HHV-8. Branch lengths are drawn to scale, with the bar indicating 0.03-nt re placement per site. Numbers on each node indicate the p value by which the cluster is supported. References strains were labeled with circles (). There were no significant differences in subtypes fre- quencies among HHV-8 associated diseases (p > 0.05) (Table 3). No statistical differences were found among K1 subtypes when the median elapsed time between HIV diagnosis and HHV-8 associated diseases appearance were compared (p = 0.444). Nevertheless, individuals in- fected with subtype E and A5 seem to progress slower to HHV-8 associated diseases than individuals infected by subtypes A, B or C. No differences were detected be- tween the median copy number of each identified HHV-8 subtypes irrespective the histhopathological diagnosis (p > 0.05) (Figure 2). However, the median HHV-8 viral load was significantly higher in PEL than in epidemic KS lesion or lymph nodes (p = 0.04) (Figure 3). 3.1. Epidemic KS Patients diagnosed with epidemic KS showed a wide range of subtypes although strains belonged to subtype E were only identified in the tissue of these patients (Table 3). Nodular stage of epidemic KS were identified in 12/37 (patients 32.4%) whilst 8/37 (21.6%) and 2/37 (5.4%) were considered to be in macular and patch stage; Copyright © 2013 SciRes. WJA 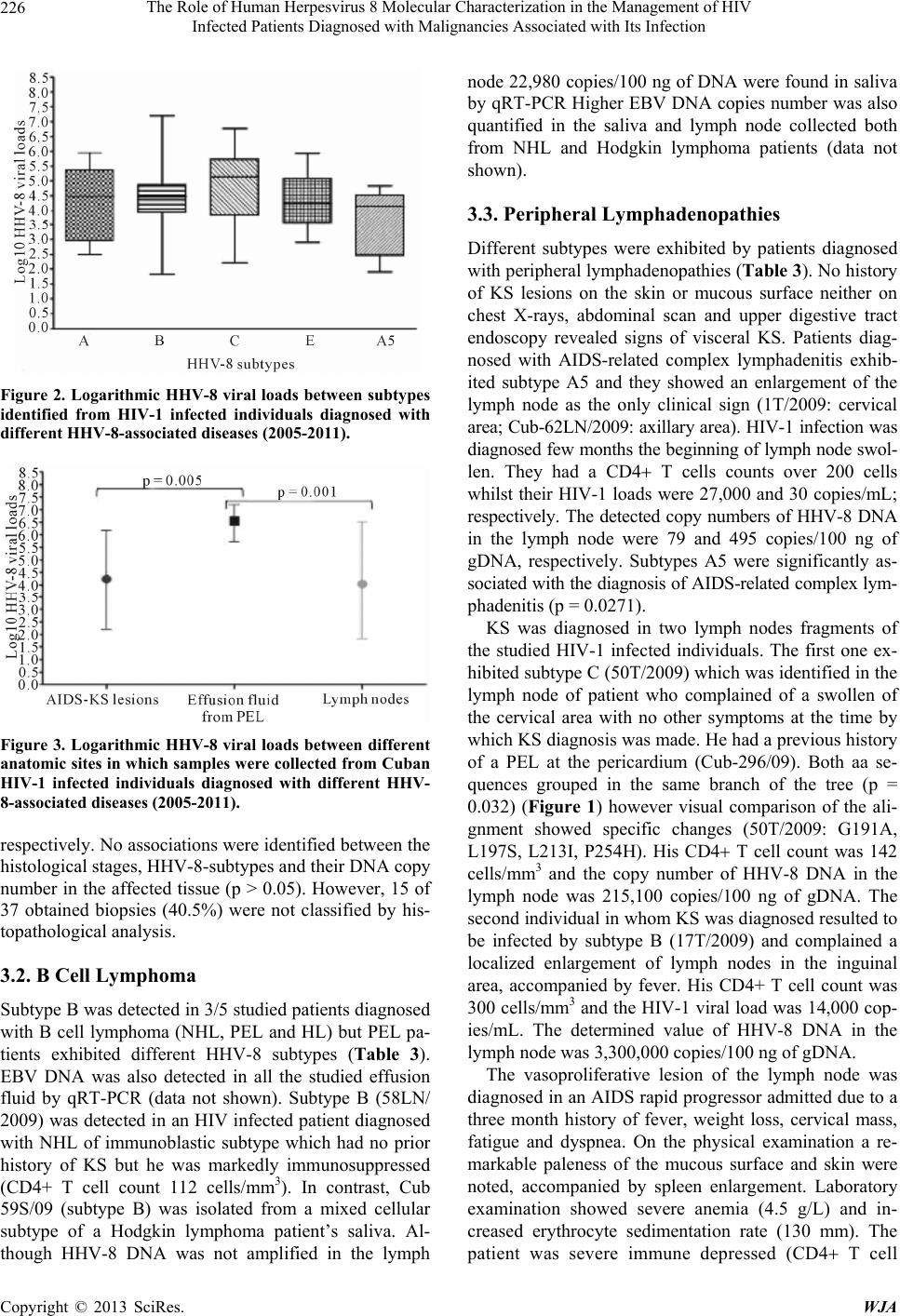 The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 226 Figure 2. Logarithmic HHV-8 viral loads between subtypes identified from HIV-1 infected individuals diagnosed with different HHV-8-associated diseases (2005-2011). Figure 3. Logarithmic HHV-8 viral loads between different anatomic sites in which samples were collected from Cuban HIV-1 infected individuals diagnosed with different HHV- 8-associated diseases (2005-2011). respectively. No associations were identified between the histological stages, HHV-8-subtypes and their DNA copy number in the affected tissue (p > 0.05). However, 15 of 37 obtained biopsies (40.5%) were not classified by his- topathological analysis. 3.2. B Cell Lymphoma Subtype B was detected in 3/5 studied patients diagnosed with B cell lymphoma (NHL, PEL and HL) but PEL pa- tients exhibited different HHV-8 subtypes (Table 3). EBV DNA was also detected in all the studied effusion fluid by qRT-PCR (data not shown). Subtype B (58LN/ 2009) was detected in an HIV infected patient diagnosed with NHL of immunoblastic subtype which had no prior history of KS but he was markedly immunosuppressed (CD4+ T cell count 112 cells/mm3). In contrast, Cub 59S/09 (subtype B) was isolated from a mixed cellular subtype of a Hodgkin lymphoma patient’s saliva. Al- though HHV-8 DNA was not amplified in the lymph node 22,980 copies/100 ng of DNA were found in saliva by qRT-PCR Higher EBV DNA copies number was also quantified in the saliva and lymph node collected both from NHL and Hodgkin lymphoma patients (data not shown). 3.3. Peripheral Lymphadenopathies Different subtypes were exhibited by patients diagnosed with peripheral lymphadenopathies (Table 3). No history of KS lesions on the skin or mucous surface neither on chest X-rays, abdominal scan and upper digestive tract endoscopy revealed signs of visceral KS. Patients diag- nosed with AIDS-related complex lymphadenitis exhib- ited subtype A5 and they showed an enlargement of the lymph node as the only clinical sign (1T/2009: cervical area; Cub-62LN/2009: axillary area). HIV-1 infection was diagnosed few months the beginning of lymph node swol- len. They had a CD4 T cells counts over 200 cells whilst their HIV-1 loads were 27,000 and 30 copies/mL; respectively. The detected copy numbers of HHV-8 DNA in the lymph node were 79 and 495 copies/100 ng of gDNA, respectively. Subtypes A5 were significantly as- sociated with the diagnosis of AIDS-related complex lym- phadenitis (p = 0.0271). KS was diagnosed in two lymph nodes fragments of the studied HIV-1 infected individuals. The first one ex- hibited subtype C (50T/2009) which was identified in the lymph node of patient who complained of a swollen of the cervical area with no other symptoms at the time by which KS diagnosis was made. He had a previous history of a PEL at the pericardium (Cub-296/09). Both aa se- quences grouped in the same branch of the tree (p = 0.032) (Figure 1) however visual comparison of the ali- gnment showed specific changes (50T/2009: G191A, L197S, L213I, P254H). His CD4 T cell count was 142 cells/mm3 and the copy number of HHV-8 DNA in the lymph node was 215,100 copies/100 ng of gDNA. The second individual in whom KS was diagnosed resulted to be infected by subtype B (17T/2009) and complained a localized enlargement of lymph nodes in the inguinal area, accompanied by fever. His CD4+ T cell count was 300 cells/mm3 and the HIV-1 viral load was 14,000 cop- ies/mL. The determined value of HHV-8 DNA in the lymph node was 3,300,000 copies/100 ng of gDNA. The vasoproliferative lesion of the lymph node was diagnosed in an AIDS rapid progressor admitted due to a three month history of fever, weight loss, cervical mass, fatigue and dyspnea. On the physical examination a re- markable paleness of the mucous surface and skin were noted, accompanied by spleen enlargement. Laboratory examination showed severe anemia (4.5 g/L) and in- creased erythrocyte sedimentation rate (130 mm). The patient was severe immune depressed (CD4 T cell Copyright © 2013 SciRes. WJA 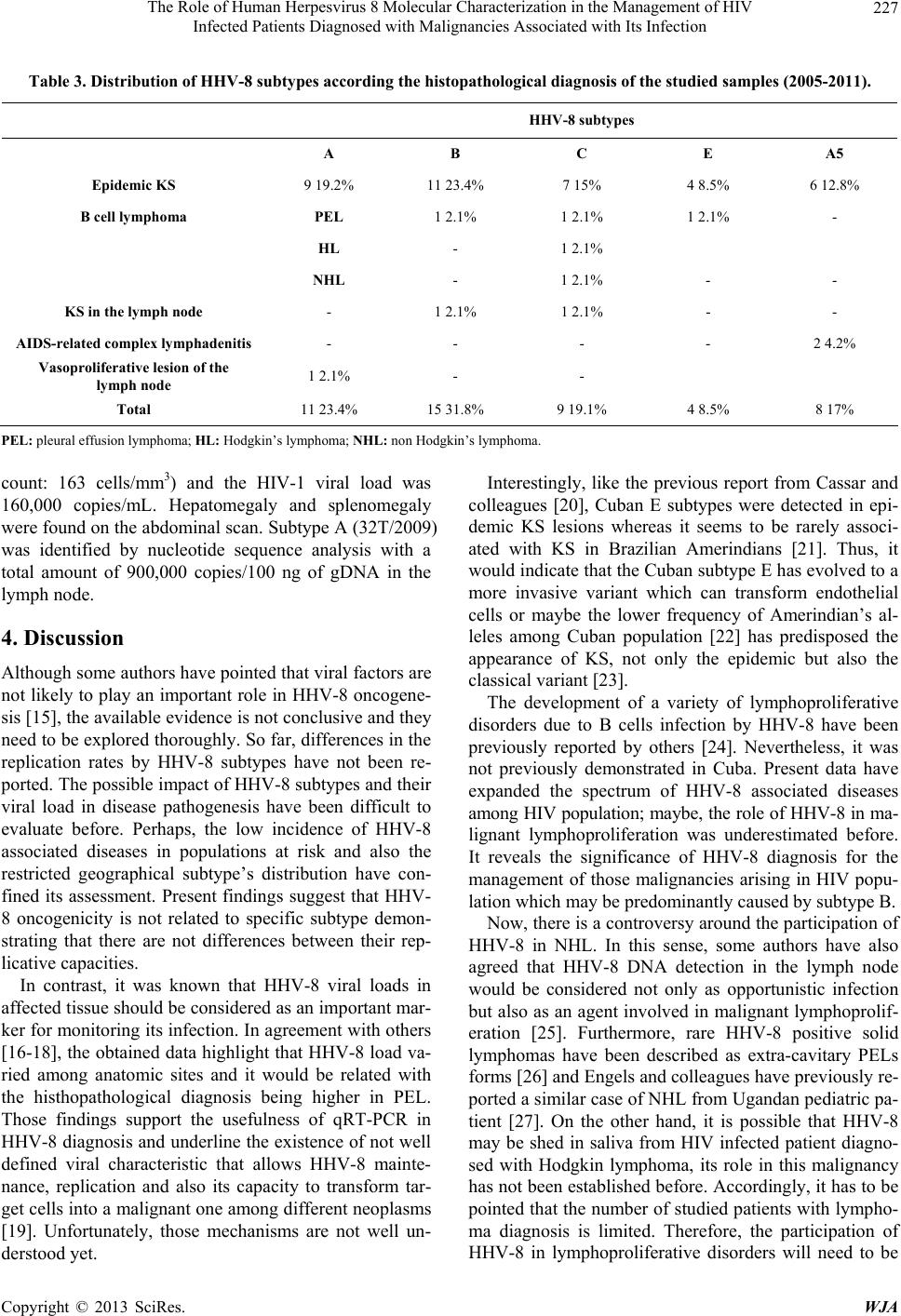 The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection Copyright © 2013 SciRes. WJA 227 Table 3. Distribution of HHV-8 subtypes according the histopathological diagnosis of the studied samples (2005-2011). HHV-8 subtyp es A B C E A5 Epidemic KS 9 19.2% 11 23.4% 7 15% 4 8.5% 6 12.8% B cell lymphoma PEL 1 2.1% 1 2.1% 1 2.1% - HL - 1 2.1% NHL - 1 2.1% - - KS in the lymph node - 1 2.1% 1 2.1% - - AIDS-related complex lymphadenitis - - - - 2 4.2% Vasoproliferative lesion of the lymph node 1 2.1% - - Total 11 23.4% 15 31.8% 9 19.1% 4 8.5% 8 17% PEL: pleural effusion lymphoma; HL: Hodgkin’s lymphoma; NHL: non Hodgkin’s lymphoma. count: 163 cells/mm3) and the HIV-1 viral load was 160,000 copies/mL. Hepatomegaly and splenomegaly were found on the abdominal scan. Subtype A (32T/2009) was identified by nucleotide sequence analysis with a total amount of 900,000 copies/100 ng of gDNA in the lymph node. 4. Discussion Although some authors have pointed that viral factors are not likely to play an important role in HHV-8 oncogene- sis [15], the available evidence is not conclusive and they need to be explored thoroughly. So far, differences in the replication rates by HHV-8 subtypes have not been re- ported. The possible impact of HHV-8 subtypes and their viral load in disease pathogenesis have been difficult to evaluate before. Perhaps, the low incidence of HHV-8 associated diseases in populations at risk and also the restricted geographical subtype’s distribution have con- fined its assessment. Present findings suggest that HHV- 8 oncogenicity is not related to specific subtype demon- strating that there are not differences between their rep- licative capacities. In contrast, it was known that HHV-8 viral loads in affected tissue should be considered as an important mar- ker for monitoring its infection. In agreement with others [16-18], the obtained data highlight that HHV-8 load va- ried among anatomic sites and it would be related with the histhopathological diagnosis being higher in PEL. Those findings support the usefulness of qRT-PCR in HHV-8 diagnosis and underline the existence of not well defined viral characteristic that allows HHV-8 mainte- nance, replication and also its capacity to transform tar- get cells into a malignant one among different neoplasms [19]. Unfortunately, those mechanisms are not well un- derstood yet. Interestingly, like the previous report from Cassar and colleagues [20], Cuban E subtypes were detected in epi- demic KS lesions whereas it seems to be rarely associ- ated with KS in Brazilian Amerindians [21]. Thus, it would indicate that the Cuban subtype E has evolved to a more invasive variant which can transform endothelial cells or maybe the lower frequency of Amerindian’s al- leles among Cuban population [22] has predisposed the appearance of KS, not only the epidemic but also the classical variant [23]. The development of a variety of lymphoproliferative disorders due to B cells infection by HHV-8 have been previously reported by others [24]. Nevertheless, it was not previously demonstrated in Cuba. Present data have expanded the spectrum of HHV-8 associated diseases among HIV population; maybe, the role of HHV-8 in ma- lignant lymphoproliferation was underestimated before. It reveals the significance of HHV-8 diagnosis for the management of those malignancies arising in HIV popu- lation which may be predominantly caused by subtype B. Now, there is a controversy around the participation of HHV-8 in NHL. In this sense, some authors have also agreed that HHV-8 DNA detection in the lymph node would be considered not only as opportunistic infection but also as an agent involved in malignant lymphoprolif- eration [25]. Furthermore, rare HHV-8 positive solid lymphomas have been described as extra-cavitary PELs forms [26] and Engels and colleagues have previously re- ported a similar case of NHL from Ugandan pediatric pa- tient [27]. On the other hand, it is possible that HHV-8 may be shed in saliva from HIV infected patient diagno- sed with Hodgkin lymphoma, its role in this malignancy has not been established before. Accordingly, it has to be pointed that the number of studied patients with lympho- ma diagnosis is limited. Therefore, the participation of HHV-8 in lymphoproliferative disorders will need to be  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 228 established in the future. The studied PEL cases were the first to be diagnosed since the beginning of Cuban AIDS epidemic in 1986. At present, it is revealed that PEL can be caused by different HHV-8 subtypes. In addition, the K1 amino acid changes that were identified in the strain isolated from the same individual are in agreement with the results obtained by Lacoste and colleagues [28]. However, others have ob- tained contrasting results [29] suggesting that K1 varia- tion did not occur over the lifetime of a single infected host. Even though ORF-K1 mutation rates were identi- fied to be similar to genes in other human pathogens [e.g. gen env from HIV-1 [2], there are neither evidences of error-prone replication mechanism that would permit HHV-8 positive selection not specialized mechanisms to rapidly generate a high level of diversity [30]. The causes of this extreme variability are not well understood but host immune pressure seems to be one of the possible ex- planations. However, the effects of Taq polymerase error prone during PCR amplification would not be definitely excluded. The clinical characteristic of HHV-8 primary infection has not been well defined. The first report was done by Oksenhendler and colleagues in an HIV subject [31] and it has been latter described in renal and bone marrow transplant recipients [32,33] as in children from Egypt and Africa [34,35]. Conversely, its occurrence has been difficult to prove since previous serological status is dif- ficult to establish. HHV-8 DNA detection at lymph node fragments obtained from Cuban patients diagnosed with AIDS-related complex lymphadenopathy and vasoprolif- erative lesion of the lymph node would be indicative of its primary infection, since neither signs nor symptoms of KS or other HHV-8 associated diseases were identified. Unfortunately, the seroconversion to HHV-8 was impos- sible to identify since no serum samples collected before the onset of symptoms were available. Thus, it would be impossible to arrive to this diagnosis in the Cuban stud- ied patients. However, a closer follow up was recommen- ded to clinicians since those individuals were recogniz- ed at risk of developing malignancies associated with this Gammaherpesvirus. The association between subtype A5 with AIDS-related complex lymphadenitis as a form of HHV-8 primary infection will need to be elucidated in the future since a small number of patients were included. Overall, our findings suggest that although different HHV-8 subtypes circulate among Cuban HIV-1 popula- tion, it is not necessary to determine the infecting subtype for their clinical management. In contrast, HHV-8 viral load could be used as a surrogate marker for monitoring its infection, not only in epidemic KS patients but also in those diagnosed with different lymphoproliferative dis- orders. Moreover, it was elucidated that the spectrum of HHV-8 associated diseases has expanded among Cuban HIV population irrespective the K1 subtypes. Others host factors will need to be explored in the future if HHV-8 transmission would need to be limited among Cuban HIV population. 5. Acknowledgements The corresponding author is grateful for the training re- ceived at the 16th International Bioinformatics Work- shop on Virus Evolution and Molecular Epidemiology in Rockville, USA, September 2010 (http://www.rega.ku- leuven.be/cev/workshop/); and for the travel grant sup- ported by Vlaamse Interuniversitaire Raad (VLIR) (ZEIN2008PR358). Likewise, he would like to thank the professors working on the Computational Molecular Evo- lution course that he attended 10 - 21 April 2011 run by Wellcome Trust Advanced Courses. We are also indebt- ed to Dr. Mariana Varela from the Department of Vete- rinary Medicine, University of Cambridge, Cambridge, UK for the support in data analysis and also to Dr. Maria Nascimento, PhD, from the London School of Hygiene and Tropical Medicine for her suggestions in the manu- script writing. REFERENCES [1] D. Ganem, “Kaposi’s Sarcoma-Associated Herpesvirus,” In: D. M. Knipe and P. M. Howley, Eds., Fields Virology, 5th Edition, Lippincott Williams & Wilkins, 2007, p. 2847-2888. [2] J. C. Zong, D. M. Ciufo, D. J. Alcendor, X. Wan, J. Ni- cholas, P. J. Browning, et al., “High-Level Variability in the ORF-K1 Membrane Protein Gene at the Left End of the Kaposi’s Sarcoma-Associated Herpesvirus Genome Defines Four Major Virus Subtypes and Multiple Vari- ants or Clades in Different Human Populations,” Journal of Virology, Vol. 73, No. 5, 1999, pp. 4156-4170. [3] S. Franceschi, L. D. Maso, M. Rickenbach, J. Polesel, B. Hirschel, M. Cavassini, et al., “Kaposi Sarcoma Incidence in the Swiss HIV Cohort Study before and after Highly Active Antiretroviral Therapy,” British Journal of Cancer, Vol. 99, No. 5, 2008, pp. 800-804. doi:10.1038/sj.bjc.6604520 [4] T. Maurer, M. Ponte and K. Leslie, “HIV-Associated Ka- posi’s Sarcoma with a High CD4 Count and a Low Viral Load,” New England Journal of Medicine, Vol. 357, No. 13, 2007, pp. 1352-1353. doi:10.1056/NEJMc070508 [5] V. Kouri, A. Marini, R. Doroudi, S. Nambiar, M. E. Ro- driguez, V. Capo, et al., “Molecular Epidemiology of Ka- posi’s Sarcoma Herpesvirus (KSHV) in Cuban and Ger- man Patients with Kaposi’s Sarcoma (KS) and Asympto- matic Sexual Contacts,” Virology, Vol. 337, No. 2, 2005, pp. 297-303. doi:10.1016/j.virol.2005.04.033 [6] V. Kouri, X. Liang, M. E. Rodriguez, V. Capo, S. Resik, J. Barrios, et al., “Molecular Epidemiology and KSHV Copyright © 2013 SciRes. WJA  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection 229 K1 Subtypes in a Cuban AIDS-Kaposi’s Sarcoma Popu- lation,” Aids, Vol. 19, No. 9, 2005, pp. 984-987. doi:10.1097/01.aids.0000171416.07034.d6 [7] V. Kouri, P. A. Martinez, V. Capo, O. Blanco, M. E. Ro- driguez, N. Jimenez, et al., “Kaposi’s Sarcoma and Hu- man Herpesvirus 8 in Cuba: Evidence of Subtype B Ex- pansion,” Virology, Vol. 432, No. 2, 2012, pp. 361-369. doi:10.1016/j.virol.2012.06.014 [8] R. Kumar, A. Abbas, A. DeLancey and E. Malone, “Dis- eases of the Immune System,” In: R. Kumar, A. Abbas, A. DeLancey and E. Malone, Eds., Robbins and Cotran Pa- thologic Basis of Disease, 8 Edition, Elsevier Inc., 2010, pp. 183-257. [9] F. Watzinger, M. Suda, S. Preuner, R. Baumgartinger, K. Ebner, L. Baskova, et al., “Real-Time Quantitative PCR Assays for Detection and Monitoring of Pathogenic Hu- man Viruses in Immunosuppressed Pediatric Patients,” Journal of Clinical Microbiology, Vol. 42, No. 11, 2004, pp. 5189-5198. doi:10.1128/JCM.42.11.5189-5198.2004 [10] V. Kouri, P. A. Martinez, O. Blanco, V. Capo, M. E. Ro- driguez, C. Dovigny Mdel, et al., “Simultaneous Quanti- fication of Human Herpesvirus 8 DNA by Real Time PCR in Different Tissues of HIV Infected Cuban Patients with Kaposi’s Sarcoma,” Herpesviridae, Vol. 1, No. 1, 2010, p. 3. doi:10.1186/2042-4280-1-3 [11] M. Schmitz, C. Scheungraber, J. Herrmann, K. Teller, M. Gajda, I. B. Runnebaum, et al., “Quantitative Multiplex PCR Assay for the Detection of the Seven Clinically Most Relevant High-Risk HPV Types,” Journal of Clinical Vi- rology, Vol. 44, No. 4, 2009, pp. 302-307. doi:10.1016/j.jcv.2009.01.006 [12] K. Tamura, J. Dudley, M. Nei and S. Kumar, “MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) Soft- ware Version 4.0,” Molecular Biology and Evolution, Vol. 24, 2007, pp. 1596-1599. doi:10.1093/molbev/msm092 [13] M. Clamp, J. Cuff, S. M. Searle and G. J. Barton, “The Jal- view Java Alignment Editor,” Bioinformatics, Vol. 20, 2004, pp. 426-427. doi:10.1093/bioinformatics/btg430 [14] D. Posada and K. A. Crandall, “MODELTEST: Testing the Model of DNA Substitution,” Bioinformatics, Vol. 14, 1998, pp. 817-818. doi:10.1093/bioinformatics/14.9.817 [15] N. H. Dukers and G. Rezza, “Human Herpesvirus 8 Epi- demiology: What We Do and Do Not Know,” Aids, Vol. 17, No. 12, 2003, pp. 1717-1730. doi:10.1097/00002030-200308150-00001 [16] A. G. Marcelin, J. Motol, A. Guihot, E. Caumes, J. P. Vi- ard, E. Dussaix, et al., “Relationship between the Quan- tity of Kaposi Sarcoma-Associated Herpesvirus (KSHV) in Peripheral Blood and Effusion Fluid Samples and KSHV-Associated Disease,” Journal of Infectious Dis- ease, Vol. 196, No. 8, 2007, pp. 1163-1166. doi:10.1086/521625 [17] Y. Asahi-Ozaki, Y. Sato, T. Kanno, T. Sata and H. Kata- no, “Quantitative Analysis of Kaposi Sarcoma-Associated Herpesvirus (KSHV) in KSHV-Associated Diseases,” Journal of Infectious Disease, Vol. 193, No. 6, 2006, pp. 773-782. doi:10.1086/500560 [18] C. Casper, “Defining a Role for Antiviral Drugs in the Treatment of Persons with HHV-8 Infection,” Herpes, Vol. 13, No. 2, 2006, pp. 42-47. [19] E. Kadyrova, V. Lacoste, R. Duprez, K. Pozharissky, V. Molochkov, M. Huerre, et al., “Molecular Epidemiology of Kaposi’s Sarcoma-Associated Herpesvirus/Human Her- pesvirus 8 Strains from Russian Patients with Classic, Posttransplant, and AIDS-Associated Kaposi’s Sarcoma,” Journal of Medical Virology, Vol. 71, No. 4, 2003, pp. 548-556. doi:10.1002/jmv.10530 [20] O. Cassar, M. L. Blondot, S. Mohanna, G. Jouvion, F. Bra- vo, V. Maco, et al., “Human Herpesvirus 8 Genotype E in Patients with Kaposi Sarcoma, Peru,” Emerging Infec- tious Diseases, Vol. 16, No. 9, 2010, pp. 1459-1462. doi:10.3201/eid1609.100381 [21] R. J. Biggar, D. Whitby, V. Marshall, A. C. Linhares and F. Black, “Human Herpesvirus 8 in Brazilian Amerindi- ans: A Hyper-Endemic Population with a New Subtype,” Journal of Infectious Diseases, Vol. 181, No. 5, 2000, pp. 1562-1568. doi:10.1086/315456 [22] R. Alegre, J. Moscoso, J. Martinez-Laso, M. Martin-Villa, J. Suarez, A. Moreno, et al., “HLA Genes in Cubans and the Detection of Amerindian Alleles,” Molecular Immu- nology, Vol. 44, No. 9, 2007, pp. 2426-2435. doi:10.1016/j.molimm.2006.10.017 [23] V. Kouri, P. A. Martinez, B. Acosta, M. E. Rodriguez, O. Blanco, V. Capo, et al., “First report of Kaposi’s Sarco- ma-Associated Herpesvirus DNA Sequences from Cuban Kaposi’s Sarcoma Patients without HIV Infection,” Aids, Vol. 21, No. 15, 2007, pp. 2113-2115. doi:10.1097/QAD.0b013e3282f01cc6 [24] E. Cesarman, “Gammaherpesvirus and Lymphoprolifera- tive Disorders in Immunocompromised Patients,” Cancer Letters, Vol. 305, No. 2, 2011, pp. 163-174. doi:10.1016/j.canlet.2011.03.003 [25] A. D’Antonio, M. Addesso, D. Memoli, P. Liguori, R. Cuomo, A. Boscaino, et al., “Lymph Node-Based Disease and HHV-8/KSHV Infection in HIV Seronegative Pa- tients: Report of Three New Cases of a Heterogeneous Group of Diseases,” International Journal of Hematology, Vol. 93, No. 6, 2011, pp. 795-801. doi:10.1007/s12185-011-0849-0 [26] A. Chadburn, E. Hyjek, S. Mathew, E. Cesarman, J. Said and D. M. Knowles, “KSHV-Positive Solid Lymphomas Represent an Extra-Cavitary Variant of Primary Effusion Lymphoma,” The American Journal of Surgical Pathol- ogy, Vol. 28, No. 11, 2004, pp. 1401-1416. doi:10.1097/01.pas.0000138177.10829.5c [27] E. A. Engels, S. M. Mbulaiteye, E. Othieno, M. Gomez, S. Mathew, E. Cesarman, et al., “Kaposi Sarcoma-Associ- ated Herpesvirus in Non-Hodgkin Lymphoma and Reac- tive Lymphadenopathy in Uganda,” Human Pathology, Vol. 38, No. 2, 2007, pp. 308-314. doi:10.1016/j.humpath.2006.08.009 [28] V. Lacoste, E. Kadyrova, I. Chistiakova, V. Gurtsevitch, J. G. Judde and A. Gessain, “Molecular Characterization of Kaposi’s Sarcoma-Associated Herpesvirus/Human Her- pesvirus-8 Strains from Russia,” Journal of General Vi- rology, Vol. 81, No. P5, 2000, pp. 1217-1222. Copyright © 2013 SciRes. WJA  The Role of Human Herpesvirus 8 Molecular Characterization in the Management of HIV Infected Patients Diagnosed with Malignancies Associated with Its Infection Copyright © 2013 SciRes. WJA 230 [29] J. Zong, D. M. Ciufo, R. Viscidi, L. Alagiozoglou, S. Ty- ring, P. Rady, et al., “Genotypic Analysis at Multiple Lo- ci across Kaposi’s Sarcoma Herpesvirus (KSHV) DNA Molecules: Clustering Patterns, Novel Variants and Chi- merism,” Journal of Clinical Virology, Vol. 23, No. 3, 2002, pp. 119-148. doi:10.1016/S1386-6532(01)00205-0 [30] L. A. Dourmishev, A. L. Dourmishev, D. Palmeri, R. A. Schwartz and D. M. Lukac, “Molecular Genetics of Ka- posi’s Sarcoma-Associated Herpesvirus (Human Herpes- virus-8) Epidemiology and Pathogenesis,” Microbiology and Molecular Biology Reviews, Vol. 67, No. 2, 2003, pp. 175-212. doi:10.1128/MMBR.67.2.175-212.2003 [31] E. Oksenhendler, D. Cazals-Hatem, T. F. Schulz, V. Ba- rateau, L. Grollet, J. Sheldon, et al., “Transient Angioly- mphoid Hyperplasia and Kaposi’s Sarcoma after Primary Infection with Human Herpesvirus 8 in a Patient with Human Immunodeficiency Virus Infection,” New Eng- land Journal of Medicine, Vol. 338, No. 22, 1998, pp. 1585-1590. doi:10.1056/NEJM199805283382204 [32] M. Luppi, P. Barozzi, V. Rasini, G. Riva, A. Re, G. Rossi, et al., “Severe Pancytopenia and Hemophagocytosis after HHV-8 Primary Infection in a Renal Transplant Patient Successfully Treated with Foscarnet,” Transplantation, Vol. 74, No. 1, 2002, pp. 131-132. doi:10.1097/00007890-200207150-00023 [33] O. Thaunat, M. F. Mamzer-Bruneel, F. Agbalika, F. Va- lensi, M. Venditto, C. Lebbe, et al., “Severe Human Her- pesvirus-8 Primary Infection in a Renal Transplant Pa- tient Successfully Treated with Anti-CD20 Monoclonal Antibody,” Blood, Vol. 107, No. 7, 2006, pp. 3009-3010. doi:10.1182/blood-2005-08-3213 [34] M. Andreoni, L. Sarmati, E. Nicastri, G. El Sawaf, M. El Zalabani, I. Uccella, et al., “Primary Human Herpesvirus 8 Infection in Immunocompetent Children,” Jama, Vol. 287, No. 10, 2002, pp. 1295-300. doi:10.1001/jama.287.10.1295 [35] F. C. Kasolo, J. Spinks, H. Bima, M. Bates and U. A. Gom- pels, “Diverse Genotypes of Kaposi’s Sarcoma Associ- ated Herpesvirus (KSHV) Identified in Infant Blood In- fections in African Childhood-KS and HIV/AIDS En- demic Region,” Journal of Medical Virology, Vol. 79, No. 10, 2007, pp. 1555-1556. doi:10.1002/jmv.20952
|