 American Journal of Plant Sciences, 2013, 4, 1863-1871 http://dx.doi.org/10.4236/ajps.2013.49229 Published Online September 2013 (http://www.scirp.org/journal/ajps) Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies* Ana Carla Feio1#, Ana Cristina Andrade de Aguiar-Dias2, Raimunda Conceição de Vilhena Potiguara3† 1Doctoral Student in Botany, Universidade Federal de Viçosa—UFV, Viçosa, Brazil; 2Instituto de Ciências Biológicas, Universidade Federal do Pará—UFPA, Belém, Brazil; 3Museu Paraense Emílio Goeldi—MPEG, Coordenação de Botânica, Belém, Brazil. Email: #anacarlafeio@gmail.com Received June 17th, 2013; revised July 17th, 2013; accepted August 20th, 2013 Copyright © 2013 Ana Carla Feio et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT This study describes the anatomy of sterile leaves of Elaphoglossum discolor (Kuhn) C. Chr., E. flaccidum (Fée) T. Moore and E. laminarioides (Bory ex Fée) T. Moore, the most representative species of the genus in the Ecological Park of Gunma in Pará State. It reports the main diagnostic characters and provides new systematic data fo r the group. In addition, it locates the production and accumulation sites of bioactive compounds to determine possible adaptive strategies of these species in the Amazon rainforest environment. Diagnostic structural features include stoma typology, central veins and margin forms, type of mesophyll, an d the presence of schlerenchymatous sheaths in the cortex, among others. Among the bioactive compounds related to defense adaptation are phenolic compounds, which occur in all three species, and alkaloids and mucilage, which are exclusive to E. laminarioides. Of the three species studied, E. lami- narioides has features that make it the best suited to the rainforest environment. Keywords: Fern; Ombrophilous Forest; Epiphytes; Comparative Anatomy; Secondary Metabolites 1. Introduction Because many of the features of Elaphoglossum Schott ex Smith do not allow for the diagnosis of sterile indi- viduals, it is one of the fern genera with the greatest need for further studies [1,2]. In fact, most infrageneric classi- fications are based on morphological characters such as plant size, blade form and size, and scale color and type [2-6]. However, most of these characters are only rele- vant when plants are in their reproductive stage [3]. Elaphoglossum grows in primary formations and is sensitive to environmental changes. Some species may be resistant for some time in human modified habitats, but different light and moisture conditions have consid- erable influence on their morphology [7]. Since the 1970s, these species have been mentioned in floristic studies in the Amazon [8-15]. Their constant presence in inventories is due to different adaptation strategies developed during their evolution, especially in the Amazon rainforest environment. Such strategies in- clude various phytochemical defenses, high resistance to diseases in moist environments, high tolerance to acute nutrient imbalance in substrates, plasticity to adapt to different ecological opportunities, mycotrophic interact- tions [16] and the epiphytic h abitat, in which Elaphoglos- sum prevails [17]. The scarcity of studies on fern anatomy hinders the understanding of their structural organization. Among the main anatomical studies carried out on this genus are [18], who analyzed rhizomes and leaves of Jamaican spe- cies; [19] who described the leaf anatomy of Elaphoglos- sum in Tucumán, Argentina and [20,21], who addressed leaf architecture of Argentinean species by focusing on their induments. None of these studies examined Brazil- ian species, indicating the lack of the knowledge about their anatomy and possible relationships to the environ- ments where they grow. The aim of this study is to characterize the anatomy of sterile leaves of E. discolor, E. flaccidum and E. lami- narioides, listing their main diagnostic characters and providing new data on the systematics of this group, in addition to identifying pro duction and accumulation sites *Part of the dissertation of the first author. #Corresponding author. †In memoria . Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1864 of bioactive compounds, in order to determine possible adaptation strategies of these species in the Amazon rain- forest environment. 2. Material and Methods 2.1. Plant Materials Botanical material was collected in the Ecological Park of Gunma (PEG) located in Santa Bárbara do Pará (01˚13'25''S , 48˚17'40''W), Pará State, Brazil. The area is covered by lowland Amazon forest with a maximum altitude of 100 m above sea level. Adult individuals with totally unwound leaf blad es were collected. The vouchers of the fertile material deposited in the herb ariu m MG are: E. discolor (MG 195547; MG 193856), E. flaccidum (MG 195545; MG 193858), and E. laminarioides (MG 195548; MG 195551; MG 195552 ; MG 193852). 2.2. Field Observations Elaphoglossum discolor and E. flaccidum were always found and collected in shady areas within the forest, while E. laminarioides was found at the forest edges, more exposed to the sunlight. Although the individuals of the latter presented many leaves that seemed to have been chewed, no visitors were observed during our ten excursions to the field over two years. 2.3. Light Microscopy and Histochemical Tests Leaves were fixed in FAA70 for 24 h [22], and in neutral buffered formalin [23] and formalin-ferrous sulphate [22] for 48 h. Leaf samples (n = 7) were dehydrated in a terti- ary butanol series [22] and embedded in histological par- affin. Leaves were cut into sections with a Leica RM 2245 microtome and stained in safranin and Astra blue [24]. The following histochemical tests were applied by hand to sections of the central vein: Sudan black B [25], Nile blue sulphate [26], NADI reagent [27], copper ace- tate/rubeanic acid [28], PAS reaction (Periodic Acid Schiff) [29], ruthenium red [30], tannic acid/ferric chloride [31], Lugol’s solution [22], ferric chloride [22], fluoroglucinol [22], vanillin hydrochloric [32], Dragendorff’s [33] and Wagner’s reagents [34]. Control sections without treat- ment were also analyzed and photographed to compare their color to that of the material fixed in FAA70 and NBF. Photomicrographs were taken with a Zeiss Axiolab microscope equipped with a Canon digital camera. 2.4. Scanning Electron Microscopy (SEM) The samples fixed in FAA70 were dehydrated in an etha- nol series [22], critical point dried with CO2 [35] and me- tallized with gold for SEM analyses. Images were cap- tured with an LEO 145 0 VP electron microscope. Frontal views of epidermis were observed in a scanning electron microscope because chemical and mechanical dissocia- tion techniques were unsuccessful. 3. Results 3.1. Leaf Blade In frontal view, the epidermis of each of the three species presented sinuous, thin anticlinal walls on both faces (Figures 1(a) and (b)), with subtle cuticular striations (Figure 1(c)), as well as fungal hyphae along the surface. All species were hypostomatic (Figures 1(a) and (b)). Stomata are on the same level as the other epidermal cells (Figures 3(a) and (e)). In E. discolor and E. flac- cidum, stomata were exclusively polocytic (Figures 1(d) and (e)), while E. laminarioides presented anomocytic (Figure 1(g)) and copolocytic stomata (Figure 1(h)) as- sociated to polocytic (Figure 1(f)) stomata. Cross sec- tions of the epidermis of each species showed that it con- sists of one layer of cells covered with a thin cuticle (Figures 2(b), (f) and (j)). However, in E. laminarioides, the outer periclinal walls are prominent on both faces (Figure 2(j)). In cross sections, the midrib differed according to spe- cies: in E. discolor, both the adaxial and abaxial faces (Figure 2(a)) are similarly concave; in E. flaccidum both faces were concave, but the adaxial one was more pro- nounced (Figure 2(e)) and in E. laminarioides, the ad- axial face is flat (Figure 2(i)). The midrib also revealed a pattern of the structural organization of the studied spe- cies: schlerenchymatous cells subjacent to the epidermis of both faces formed an arch comprised of 2 - 6 layers (Figures 2(b), (f) and (j)) with the rest filled with paren- chymatous tissue. The midrib enclosed two large and one small vascular bundles, each of which was surrounded by a cortical schlerenchymatous sheath (Figures 2(c), (g) and (k)), which was thicker in E. laminarioi des . Adjacent to this sheath was the endodermis, followed by a 2-lay- ered pericycle (Figures 2(d) and (h)). Vascular bundles were bicollateral, with curved xylem at their tips. Mesophyll was homogenous (Figures 3(a), (e) and (i)), with 4 - 6 strata of branched cells in E. flaccidum (Fig- ures 3(e) and (f)) and lob ed cells in the oth er two species (Figures 3(a), (b), (i) and (j)). In addition, the intracel- lular spaces were conspicuous with scarce collateral bun- dles. Although these species had revolute leaf margins, (Figures 3(c), (g) and (k)), they differed according to taxa: in E. discolor, they were less revolute and the epi- dermis had lignified walls (Figure 3(c)); in E. flaccidum, they were rostriform (Figure 3(g)) and in E. laminario- ides they were more revolute, with slightly truncated ex- tremities (Figure 3(k)). Copyright © 2013 SciRes. AJPS 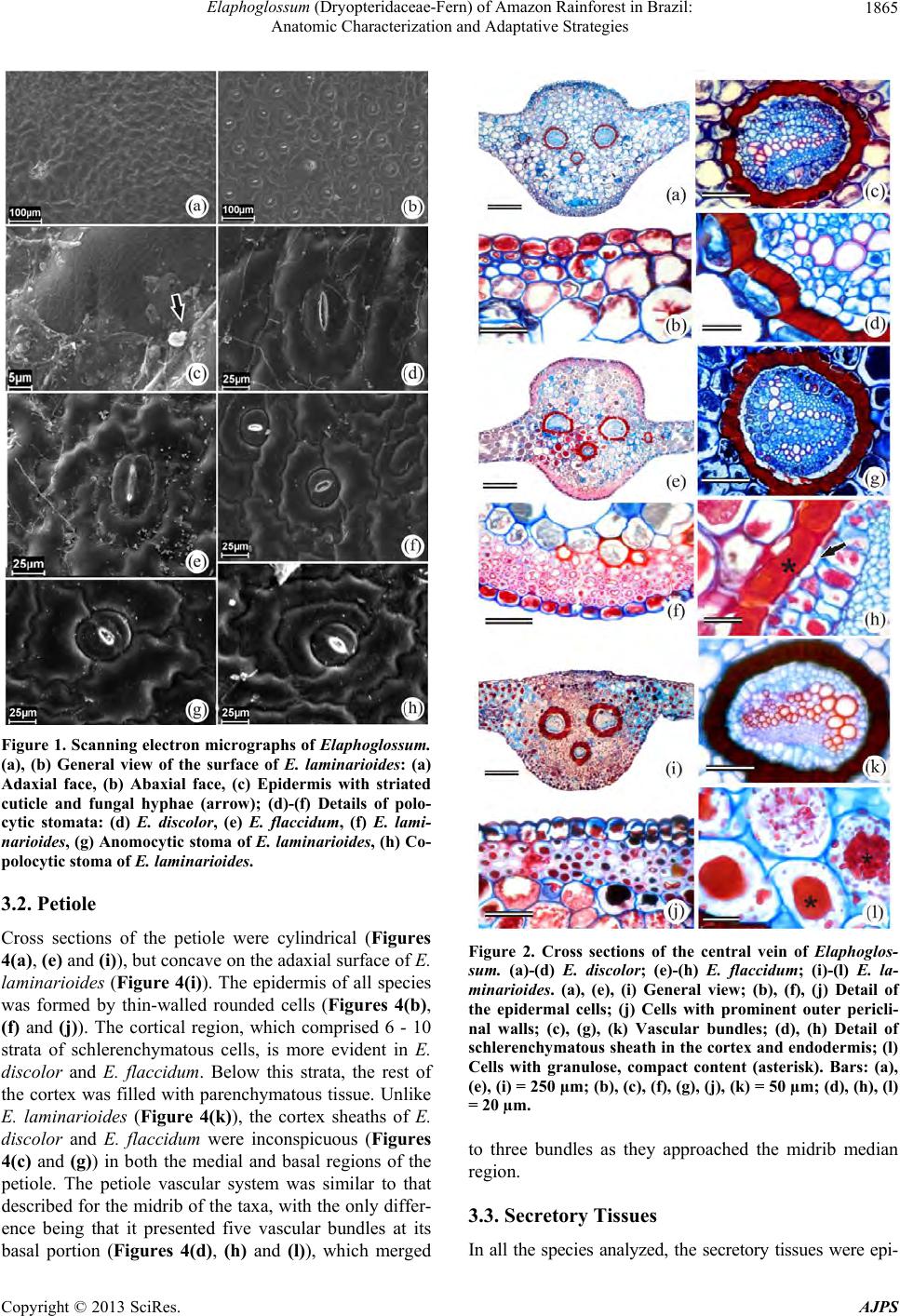 Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1865 Figure 1. Scanning electron mic rographs of Elaphoglossum. (a), (b) General view of the surface of E. laminarioides: (a) Adaxial face, (b) Abaxial face, (c) Epidermis with striated cuticle and fungal hyphae (arrow); (d)-(f) Details of polo- cytic stomata: (d) E. discolor, (e) E. flaccidum, (f) E. lami- narioides, (g) Anomocytic stoma of E. laminarioides, (h) Co- polocytic stoma of E. laminarioides. 3.2. Petiole Cross sections of the petiole were cylindrical (Figures 4(a), (e) and (i)), but concave on the adaxial surface of E. laminarioides (Figure 4(i)). The epidermis of all species was formed by thin-walled rounded cells (Figures 4(b), (f) and (j)). The cortical region, which comprised 6 - 10 strata of schlerenchymatous cells, is more evident in E. discolor and E. flaccidum. Below this strata, the rest of the cortex was filled with parenchymatous tissue. Unlike E. laminarioides (Figure 4(k)), the cortex sheaths of E. discolor and E. flaccidum were inconspicuous (Figures 4(c) and (g)) in both the medial and basal regions of the petiole. The petiole vascular system was similar to that described for the midrib of the taxa, with the only differ- ence being that it presented five vascular bundles at its basal portion (Figures 4(d), (h) and (l)), which merged Figure 2. Cross sections of the central vein of Elaphoglos- sum. (a)-(d) E. discolor; (e)-(h) E. flaccidum; (i)-(l) E. la- minarioides. (a), (e), (i) General view; (b), (f), (j) Detail of the epidermal cells; (j) Cells with prominent outer pericli- nal walls; (c), (g), (k) Vascular bundles; (d), (h) Detail of schlerenchymatous sheath in the cortex and endodermis; (l) Cells with granulose, compact content (asterisk). Bars: (a), (e), (i) = 250 µm; (b), (c), (f), (g), (j), (k) = 50 µm; (d), (h), (l) = 20 µm. to three bundles as they approached the midrib median region. 3.3. Secretory Tissues In all the species analyzed, the secretory tissues were epi- Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1866 Figure 3. Sections of the leaf blade of Elaphoglossum. (a)-(d) E. discolor; (e)-(h) E. flaccidum; (i)-(l) E. laminarioides. (a), (e), (i) Cross sections of the blade showing homogeneous mesophyll; (b), (f), (j) Longitudinal sections of the meso- phyll; (b), (j) Mesophyll with lobed cells; (f) Mesophyll with branched cells; (c), (g), (k) General view of the revolute margins; (d), (h), (l) Detail of the cells with content; (l) Cell with dense content (asterisk) and adjacent starch grains (arrow). Bars: (a), (e), (i) = 50 µm; (b), (d), (f), (h), (j), (l) = 20 µm; (c), (g), (k) = 200 µm. Figure 4. Cross sections of the petiole of Elaphoglossum. (a)-(d) E. discolor; (e)-(h) E. flaccidum; (i)-(l) E. lamina- rioides; (a), (e), (i) General view; (b), (f), (j) Detail of the epidermis and adjacent schlerenchy matous cells; (c), (g), (k) Detail of the vascular bundles in the median region; (d), (h), (l) Vascular bundles in the basal region. Bars: (a), (e), (i) = 250 µm; (b)-(d), (f)-(h), (j)-(l) = 50 µm. dermis (Figures 2(b), (f), (j), 3(e) and (i)), parenchyma (Figures 3(d), (h) and (l)) (including that of the xylem and phloem), and pericycle. No exudate was released into the environment. Independent of the individuals or parts, the exudate had a compact or granulose aspect, but it may appear differently in neighboring cells (Figure 2(l)), or even in the same cell (Figure 3(l)). In the mate- rial fixed in either FAA or NBF, exudate color varied from yellow to light brown (Figures 5(a) and (b)). Exu- date was more abundant on leaf blades and more con- spicuous in E. laminarioides. The results of the histoche- mical tests are shown in Table 1 and Figures 5(a)-(t). Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies Copyright © 2013 SciRes. AJPS 1867 4. Discussion The species analyzed showed significant variation in their structural characters, especially leaf blades and his- tochemical features. Based on our results, E. laminari- oides was the most different in terms of structural varia- tion and the highest number of bioactive compounds. Some of the leaf structural features studied, such as sin- uous epidermis and hypostomatic leaf blades, concur with the descriptions by [18] for E. latifolium, E. musco- sum, E. pallidum and E. villosum and by [20,21] for E. crassipes (Hieron.) Diels, E. gayanum (Fée) T. Moore, E. lindbergii (Mett. ex Kuhn) Rosenst., E. lorentzii (Hieron.) H. Christ, E. pachydermum (Fée) T. Moore, E. pilosel- loides (C. Presl) T. Moore and E. yungense De la Sota. While polocytic stomata predominated in all species, they were associated to copolocytic and anomocytic sto- mata in Elaphoglossum laminarioides. Although polo- cytic and copolocytic stomata are among the five types that can be found in Elaphoglossum [36], anomocytic stomata are not described for this genus. [37,38] did not correlate this stoma type with those found in E. laminari- oides, but Sen and De (1992) asserted that polocytic sto- mata are never associated to anomocytic ones. Neverthe- less, latter studies carried out by [20,21] identified ano- mocytic stomata in Argentinean species of Elaphoglos- sum. This variation in stomata typology is relevant since it can be used as an infrageneric diagnostic feature. As only E. flaccidum presented branched cells and ro- striform margins, mesophyll and leaf margins are also useful infrageneric features. [39] stated that homogene- ous mesophyll with branched cells occurs only in Pteri- daceae species that grow in shady habitats. Since this is the case of E. flaccidum, the present study corroborates their assertion. One of the most common features mentioned in ana- tomical studies on ferns is their lignified schlerenchyma- tous sheath in the cortex [40-46], which only encloses the petiole vascu lar bundles in E. laminarioides. It is consid- ered a specialized character found in specimens of most of the derived families, such as Dryopteridaceae [45]. Although no visitors were observed in loco, many leaves were chewed and pathogens such as fungal hy- phae were observed on leaf epidermis. This is likely due to the presence of lipids, which are considered nutrition- ally important for different classes of visitors and patho- gens [47]. The starch grains observed on the leaf blade of the studied species are believed to be transitorily stocked in the chloroplast during the day and degraded at night to maintain plant metabolism [48]. Although we know that epiphytes are more vulnerable to water stress [49], E. laminarioides has always been collected in areas more exposed to the sun. The muci- Table 1. Results of histochemical tests on secreting of Elaphoglossum. Species Metabolites Tests E. discolorE. flaccidum E. laminarioides Total lipids Sudan black B − − + Acids and neutral lipids Nile blue sulphate − − ++ Lipids Free fatty acids Cooper acetate/rubeanic acid − − + Terpenoids Essential oils and oil-resin NADI reagent − − − FFS + + ++ Total phenolic compounds Ferric chloride + + + Tannins Vanillin hydrochloric − − − Phenolic compounds Lignins Fluoroglucinol − − − Wagner’s reagent − − ++ Alkaloids Dragendorff’s reagent − − ++ Starch Lugol’s solution ++ ++ ++ Mucilage Ruthenium red − − − Total polysaccharides PAS reaction − − ++ Polysaccharides Mucilage Tannic acid/ferric chloride − − + Notes: ++ = high positive; + = positive; − = negative.  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1868 Figure 5. Histochemical characterization of the secretion in leaf blade cross sections of E. laminarioides. (a), (b) Sections without treatment, note yellow to light brown color; (c), (d) Detection of lipoic acid with Nile blue, note the secretion in the cell (asterisk); (e), (f). Positive result for fatty acids with copper acetate/rubeanic acid. (g)-(j) Positive reaction for phenolic compounds: (g), (h) Detection with FFS, note the secretion in the cell (asterisk), (i), (j) Detection with ferrous chloride; (k), (l) Positive result for alkaloids: (k) Detection with Wagner’s reagent, (l) Detection with Dragendorff’s reagent; (m), (n) positive reaction for starch grains; (o)-(r) Detection of polysaccharides with PAS reaction, note the secretion (asterisk) and starch grains (arrow) in the cell; (s), (t) Positive reaction for mucilage with tannic acid/ferric chloride. Bars: (a), (c), (e), (g), (i), (k), (m), (o), (s) = 150 µm; (b), (f), (j), (l), (q), (t) = 50 µm; (d), (h), (n), (p), (r) = 20 µm. laginous compounds found exclusively in this species thus serve to retain water and protect the plant against herbivory [50], since some chewed leaves were observed. When [51] studied fungus-insect-plant interactions, they observed that insects feed on the fungal hyphae present on the plants (mycophagy), which could explain the oc- currence of both chewed leaves and fungal hyphae on leaf surfaces. Phenolic compounds are known components of defen- sive adaptations. According to [52], they present pharma- cological and antinutritional properties that inhibit lipid oxidation and the proliferation of fungi. [53 ] asserted that Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1869 they are involved in adaptation processes as a defense against radiation and pathogenic aggressions. Likewise, alkaloids, found only in E. laminarioides, are toxic and act as a defense against herbivores and pa- rasites, in addition to playing an allelopathic role [54]. Although no Elaphoglossum monospecific formations were observed in the field (which could lead us to induce some kind of dominance), the potential allelopathic ac- tivity of many fern genera has been proved, e.g. by [55] for Pteridium Gled. ex Scop., and [56] for Dicranopteris Bernh., Gleichenia Sm. and Sticherus C. Pr esl. Thus, the structural data reported constitute new char- acters of the representatives of Elaphoglossum in vege- tative stage. The bioactive compounds identified in situ also provide important unpublished data, adding infor- mation on the species and suggesting the development of adaptive strategies. This could be observed especially on E. laminarioides, which in addition to the compounds of communal occurrence on the species studied, was the only species presenting alkaloids and mucilage. These compounds possess peculiar properties related to herbi- vore and pathogen avoidance, demonstrating that E. la- minarioides is possibly better adapted to its environment, producing secondary metabolites that are more varied and directly related to adaptive success. 5. Acknowledgements The authors thank Dr. Alejandra Vasco, New York Bo- tanical Garden (NYBG), for information on the systema- tics studies in the group and the National Coun cil for Sci- entific and Technological Development (CNPq) for the Master scholarship of the first author (CNPq 132809/ 2009-5). We also thank Nicholas J. Walker and Anna Luiza Ilkiu Borges Benkendorff for revising the English of this manuscript. REFERENCES [1] R. E. G. Pichi Sermolli, “Adumbratio Florae Aetiopicae: l5. Elaphoglossaceae,” Webbia, Vol. 23, No. 1, 1968, pp. 209-246. doi:10.1080/00837792.1968.10669885 [2] J. T. Mickel and L. Atehortúa, “Subdivision of the Genus Elaphoglossum,” American Fern Journal, Vol. 70, No. 2, 1980, pp. 47-68. doi:10.2307/1546218 [3] J. T. Mickel and A. R. Smith, “The Pteridophytes of Mexico,” Memories of the New York Botanical Garden, Vol. 88, 2004, p. 282. [4] A. Vasco, R. C. Moran and G. Rouhan, “Circumscription and Phylogeny of the Elaphoglossum ciliatum Group (E. sect. Lepidoglossa, Dryopteridaceae) Based on cpDNA Sequences,” Taxon, Vol. 58, No. 3, 2009, pp. 825-834. [5] A. F. Rojas-Alvarado, “New Taxa, New Records and Redefined Concepts in the Elaphoglossum sect. Elaph- oglossum subsec. Pachyglossa (Lomariopsidaceae) from Mexico and Central America,” Revista de Biología Tro- pical, Vol. 51, No. 1, 2003, pp. 1-32. [6] A. F. Rojas-Alvarado, “A New Species in Elaphoglossum sect. Elaphoglossum subsect. Pachyglossa (Dryopteri- daceae) from Costa Rica and Panama,” Novon, Vol. 20, 2010, pp. 334-337. doi:10.3417/2008122 [7] A. C. Brade, “O Gênero Elaphoglossum (Pteridophyta) no Brasil,” 2003. http://www.saude.unisinos.br/pteridos [8] R. M. Tryon and D. S. Conant, “The Ferns of Brasilian Amazonia,” Acta Amazononica, Vol. 5, No. 1, 1975, pp. 23-24. [9] S. T. Rodrigues, S. S. Almeida, L. H. C. Andrade, I. C. L. Barros and M. E. Van den Berg, “Composição Florística e Abundância de Pteridófitas em três Ambientes da Bacia do rio Guamá, Belém, Pará, Brasil,” Acta Amazonica, Vol. 34, No. 1, 2004, pp. 35-42. doi:10.1590/S0044-59672004000100005 [10] J. Prado, “Flora da Reserva Ducke, Amazonas, Brasil: Pteridophyta—Lomariopsidaceae,” Rodriguésia, Vol. 56, No. 86, 2005, pp. 59-66. [11] J. M. Costa, M. G. C. Souza and M. R. Pietrobom, “Levantamento Florístico das Pteridófitas (Lycophyta e Monilophyta) do Parque Ambiental de Belém (Belém, Pará, Brasil),” Revista de Biologia Neotropical, Vol. 3, No. 1, 2006, pp. 4-12. [12] J. M. Costa and M. R. Pietrobom, “Pteridófitas (Lyco- phyta e Monilophyta) da Ilha de Mosqueiro, Município de Belém, Estado do Pará, Brasil,” Boletim do Museu Pa- raense Emílio Goeldi Ciências Naturais, Vol. 2, No. 3, 2007, pp. 45-56. [13] S. Maciel, M. G. C. Souza and M. R. Pietrobom, “Licó- fitas e Monilófitas do Bosque Rodrigues Alves Jardim Botânico da Amazônia, Município de Belém, Estado do Pará, Brasil,” Boletim do Museu Paraense Emílio Goeldi Ciências Naturais, Vol. 2, No. 2, 2007, pp. 69-83. [14] M. R. P. Silva and S. M. Rosário, “Licófitas e Monilófitas (Pteridophyta) da Floresta Nacional de Caxiuanã, Estado do Pará, Brasil: Chave Para as Famílias e as Espécies de Aspleniaceae e Blechnaceae,” Boletim do Museu Para- ense Emílio Goeldi Ciências Naturais, Vol. 3, No. 2, 2008, pp. 151-163. [15] J. M. Costa and M. R. Pietrobom, “Samambaias e Licó- fitas do Parque Ecológico do Gunma, Município de Santa Bárbara do Pará, Estado do Pará, Brasil,” Rodriguésia, Vol. 61, No. 2, 2010, pp. 223-232. [16] C. N. Page, “Ecological Strategies in Fern Evolution: A Neopteridological Overview,” Review of Palaeobotany and Palynology, Vol. 119, No. 1-2, 2002, pp. 1-33. doi:10.1016/S0034-6667(01)00127-0 [17] H. Tuomisto and A. D. Poulsen, “Pteridophyte Diversity and Species Composition in Four Amazonian Rain For- ests,” Journal of Vegetable Science, Vol. 11, 2000, pp. 383-396. doi:10.2307/3236631 [18] P. R. Bell, “Studies in the Genus Elaphoglossum. III. Anatomy of the Rhizome and Frond,” Annals of Botany, Vol. 14, 1951, pp. 347-357. [19] M. E. Guantay and M. H. Terán, “Estudio Morfoana- Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies 1870 tómico de las Especies del Género Elaphoglossum Schott de Tucumán,” Lilloa, Vol. 38, 1995, pp. 73-84. [20] M. C. Lavalle and M. Rodríguez, “Arquitectura Foliar y Otros Caracteres Vegetativos en Especies Argentinas de Elaphoglossum Schott ex Sm. (Lomariopsidaceae— Pteridophyta),” Boletín de la Sociedad Argentina de Bo- tánica, Vol. 42, 2007, p. 107. [21] M. C. Lavalle and M. Rodríguez, “Indumento y Modelos Epidérmicos en Especies Argentinas de Elaphoglossum (Dryopteridaceae—Pteridophyta),” Boletín de la Socie- dad Argentina de Botánica, Vol. 45, No. 3-4, 2010, pp. 245-256. [22] D. A. Johansen, “Plant Microtechnique,” 2nd Edition, Mc- Graw Hill, New York, 1940. [23] R. D. Lillie, “Histopathologic Technic and Pratical Histo- chemistry,” 3rd Edition, McGraw Hill, New York, 1965. [24] D. Gerlach, “Botanische Mikrotechnik, Eine Einfuhrung,” Georg Thieme, Stuttgart, 1969. [25] A. G. E. Pearse, “Histochemistry Theoretical and Apl- lies,” Vol. 2, C. Livingstone, Edinburgh, 1985. [26] A. J. Cain, “The Use of Nile Blue in the Examination of Lipids,” The Quarterly Journal of Microscopical Science, Vol. 33, 1947, pp. 383-392. [27] R. David and J. P. Carde, “Coloration Differentielle des Inclusions Lipidique et Terpeniques des Pseudophylles du Pin maritime au Moyen du Reactif Nadi,” Comptes Ren- dus de l’Académie des Sciences, Paris, Vol. 258, 1964, pp. 1338-1340. [28] P. Ganter and G. Jollés, “Histologie Normale et Patholo- gique,” Vol. 1-2, Gauthier-Villars, Paris, 1969, 1970. [29] W. A. Jensen, “Botanical Histochemistry: Principles and Practice,” W. H. Freeman and Co., San Francisco, 1962. [30] M. Gregory and P. Baas, “A Survey of Mucilage Cells in Vegetative Organs of the Dicotyledons,” Israel Journal of Botany, Vol. 38, 1989, pp. 125-174. [31] P. Pizzolato and R. D. Lillie, “Mayer’s Tannic Acid-Fer- ric Chloride Stain for Mucins,” Journal of Histochemistry & Cytochemistry, Vol. 21, 1973, pp. 56-64. doi:10.1177/21.1.56 [32] M. E. Mace and C. R. Howell, “Histochemistry and Iden- tification of Condensed Tannin Precursors in Roots of Cotton Seedlings,” Canadian Journal of Botany, Vol. 52, 1974, pp. 2423-2426. doi:10.1139/b74-314 [33] A. B. Svedsen and R. Verpoorte, “Chromatography of Alkaloids,” Elsevier Scientific Publish Company, New York, 1983. [34] M. Furr and P. G. Mahlberg, “Histochemical Analyses of Laticifers and Glandular Trichomes in Cannabis sativa,” Journal of Natural Products, Vol. 44, No. 2, 1981, pp. 153-159. doi:10.1021/np50014a002 [35] J. J. Bozzola and L. D. Russel, “Eletron Microscopy: Principles and Techniques for Biologists,” Jones and Bar- lett Publishers, New York, 1991. [36] U. Sen and B. De, “Structure and Ontogeny of Stomata in Ferns,” Blumea, Vol. 37, No. 1, 1992, pp. 239-261. [37] E. Fryns-Clae ssens and W. van Cotthem, “A New Classi- fication of the Ontogenetic Types of Stomata,” Botanical Review, Vol. 39, No. 1, 1973, pp. 71-131. doi:10.1007/BF02860071 [38] W. R. J. Van Cotthem, “Stomatal Types and Systematic,” In: J. A. Crabbe and B. A. Thomas, Eds., The Phylogeny and Classification of the Ferns, Academic, Nueva York, 1973, pp. 59-71. [39] D. Graçano, A. A. Azevedo and J. Prado, “Anatomia Foliar das Espécies de Pteridaceae do Parque Estadual do Rio Doce (PERD)-MG,” Revista Brasileira de Botanica, Vol. 24, No. 3, 2001, pp. 333-347. doi:10.1590/S0100-84042001000300012 [40] Y. Ogura, “Comparative Anatomy of Vegetative Organs of the Pteridophytes,” 2nd Edition, Gebrüder Bornt Rae- ger, Berlin, 1972. [41] L. Atehortúa, “Contribuciones al Conocimiento de los He- lechos del Gênero Elaphoglossum (Elaphoglossaceae),” Actualidades Biológicas, Vol. 13, No. 50, 1985, pp. 83- 93. [42] R. Bercu, “Anatomical Aspects of Asplenium adiantum- nigrum L,” Journal of Biological Research, Vol. 2, 2004, pp. 57-61. [43] V. Hernández, T. Terrazas and G. Angeles, “Anatomía de seis Espécies de Helechos del Gênero Dryopteris (Dryo- pteridaceae) de México,” Revista de Biología Tropical, Vol. 54, No. 4, 2006, pp. 1157-1169. [44] A. Bagniewska-Zadworna and E. Zenkteler, “Ultra- structure of Endodermis and Stele Cells of Dehydrated Polypodium vulgare L. Rhizomes,” Acta Biologica Cra- coviensia Series Botanica, Vol. 48, No. 2, 2006, pp. 73- 81. [45] V. Hernández-Hernández, T. Terrazas and K. Mehltreter, “Anatomía Vegetativa de Ctenitis melanosticta (Dry- opteridaceae, Pteridophyta),” Boletín de la Sociedad Botánica de México, Vol. 80, 2007, pp. 7-17. [46] I. B. Suffredini, E. M. Bacchi and J. E. Kraus, “Estudo Farmacognóstico Do Caule e Raízes de Microgramma squamulosa (Kaulf.) Sota (Polypodiaceae),” Revista Bra- sileira de Farmacognosia, Vol. 18, No. 2, 2008, pp. 279- 286. doi:10.1590/S0102-695X2008000200023 [47] H. G. Baker and I. Baker, “The Predictive Value of Nec- tar Chemistry to the Recognition of Pollinator Type,” Is- rael Journal of Botany, Vol. 39, No. 1-2, 1990, pp. 157- 166. [48] P. L. Keeling and K. L. Myers, “Biochemistry and Ge- netics of Starch Synthesis,” Annual Review of Food Sci- ence Technology, Vol. 1, 2010, pp. 271-303. doi:10.1146/annurev.food.102308.124214 [49] D. H. Benzing, “Vascular Epiphytes: General Biology and Related Biota,” University Press, Cambridge, 1990. doi:10.1017/CBO9780511525438 [50] A. Fahn, “Secretory Tissues in Plants,” Academic Press, London, 1979. [51] C. Gehring and A. Bennett, “Mycorrhizal Fungal-Plant- Insect Interactions: The Importance of a Community Ap- proach,” Environmental Entomology, Vol. 38, No. 1, Copyright © 2013 SciRes. AJPS  Elaphoglossum (Dryopteridaceae-Fern) of Amazon Rainforest in Brazil: Anatomic Characterization and Adaptative Strategies Copyright © 2013 SciRes. AJPS 1871 2009, pp. 93-102. doi:10.1603/022.038.0111 [52] S. E. Soares, “Ácidos Fenólicos como Antioxidantes,” Revista de Nutrição, Vol. 15, No. 1, 2002, pp. 71-81. doi:10.1590/S1415-52732002000100008 [53] A. Farah and C. M. Donangelo, “Phenolic Compounds in Coffee,” Brazilian Journal of Plant Physiology, Vol. 18, No. 1, 2006, pp. 23-36. doi:10.1590/S1677-04202006000100003 [54] T. Robinson, “Metabolism and Function of Alkaloids in Plants,” Science, Vol. 184, No. 4135, 1974, pp. 430-435. doi:10.1126/science.184.4135.430 [55] R. E. Stewart, “Allelopathic Potential of Western Bra- ken,” Journal of Chemical Ecology, Vol, 1, No. 2, 1975, pp. 161-169. doi:10.1007/BF00987865 [56] G. L. G. Soares and T. R. Vieira, “Inibição da Germi- nação e do Crescimento Radicular de Alface (cv. ‘Grand Rapids’) por Extratos Aquosos de Cinco Espécies de Gleicheniaceae,” Floresta e Ambiente, Vol. 7, 2000, pp. 180-184.
|