 International Journal of Otolaryngology and Head & Neck Surgery, 2013, 2, 151-155 http://dx.doi.org/10.4236/ijohns.2013.25033 Published Online September 2013 (http://www.scirp.org/journal/ijohns) Results of Montgomery T-Tube in Primary Treatment of Laryngotracheal Stenosis Hale Aslan, Sedat Öztürkcan, Erdem Eren, Mehmet Sinan Başoğlu, Murat Songu, Erkan Kulduk, Ahmet Erdem Kılavuz, Hüseyin Katılmış Department of Otorhinolaryngology and Head and Neck Surgery, İzmir Katip Çelebi Universty Atatürk Research and Training Hospital, İzmir, Turkey Email: drhaleaslan@gmail.com Received July 2, 2013; revised July 31, 2013; accepted August 8, 2013 Copyright © 2013 Hale Aslan et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Introduction: For tracheal stenosis, tracheal resection and anastomosis is widely considered the treatment of choice. However, this surgical approach is not feasible when the glottis and subglottis are involved or in patients with a poor general condition and tracheal stents are a plausible means of providing a permanent or temporary airway opening. Objectives: Evaluate the features and the results of patients with Montgomery T-tube in tracheal stenosis. Methods: Fifteen patients with Myer-Cotton grades 2-3 circular cicatricial tracheal stenosis who received a Montgomery T-tube between 2002-2011 were analyzed in terms of age, gender, etiology, duration of intubation, location and size of the stenotic segment on computed tomography(CT), follow-up time with the T-tube, the complications that occurred after T-tube removed and additional tracheal surgery. Conclusion: A T-tube can be applied in tracheal stenosis at the first treatment before attempting surgery. The patients should be closely followed-up due to the possibility of re-stenosis and other complications. Keywords: Tracheal Stenosis; Montgomery T-Tube; Complications 1. Introduction Tracheal stenosis occurs most commonly following in- tubation trauma, tracheotomy, or neck injury. Additional etiologies include benign and malignant neoplasms and inflammatory or systemic autoimmune diseases [1]. The subglottic area is most sensitive to intubation trauma due to its flaccid areolar tissue that readily becomes edema- tous, in contrast to the circular cricoid cartilage, which prevents the expansion of the edema to the surrounding tissue [2]. Although numerous studies describe various treatment modalities, there is no standard approach to laringora- cheal stenosis. Endoscopic treatment includes laryngeal microsurgery, laser-assisted excision, traditional dilation and endoscopic stent insertion, while external surgical treatment comprises a wide range of techniques such as tracheal resection and anastomosis or laryngotracheal re- construction. In order to maintain laryngotracheal pa- tency, a Montgomery Safe T-tube may be used as a sin- gle dilation treatment or in association with endoscopic and/or open-neck surgery [3]. 2. Materials and Methods We retrospectively analyzed 15 patients with Myer-Cot- ton grades 2-3 circular cicatricial tracheal stenosis be- tween 2002-2012 at the Department of Otorhinolaryn- gology in Faculty of Medicine of İzmir Katip Çelebi Uni- versty Atatürk Training and Research Hospital. Most sur- geries were performed by experienced surgeons. These patients evaluated according to age, gender, etiology, du- ration of intubation, location and size of the stenotic seg- ment on computed tomography (CT), follow-up time with T-tube, the complications that occured after T-tube removed and additional tracheal surgery. This study ap- proved by ethics committee Faculty of medicine of İzmir Katip Çelebi Universty Atatürk Training and Research Hospital. We assessed the location, length and anterior posterior diameter of the stenosis by multiplanar recon- structive computed tomography and observed directly or indirectly with a flexible or rigid laryngoscope before surgery. These patients underwent tracheotomy and a Montgomery T-tube was applied either during or after tracheotomy as the first treatment choice. The T-tubes C opyright © 2013 SciRes. IJOHNS 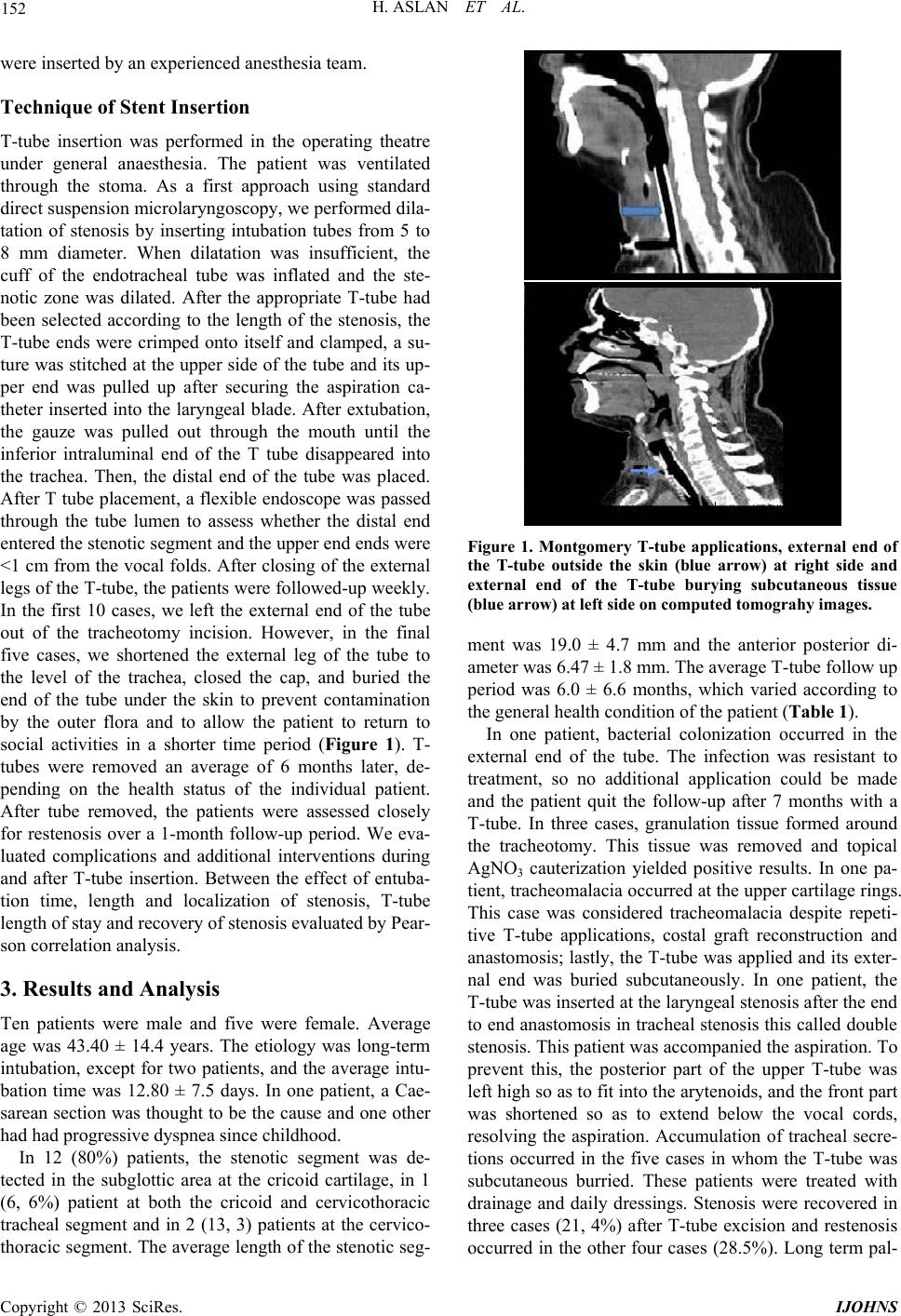 H. ASLAN ET AL. 152 were inserted by an experienced anesthesia team. Technique of Stent Insertion T-tube insertion was performed in the operating theatre under general anaesthesia. The patient was ventilated through the stoma. As a first approach using standard direct suspension microlaryngoscopy, we performed dila- tation of stenosis by inserting intubation tubes from 5 to 8 mm diameter. When dilatation was insufficient, the cuff of the endotracheal tube was inflated and the ste- notic zone was dilated. After the appropriate T-tube had been selected according to the length of the stenosis, the T-tube ends were crimped onto itself and clamped, a su- ture was stitched at the upper side of the tube and its up- per end was pulled up after securing the aspiration ca- theter inserted into the laryngeal blade. After extubation, the gauze was pulled out through the mouth until the inferior intraluminal end of the T tube disappeared into the trachea. Then, the distal end of the tube was placed. After T tube placement, a flexible endoscope was passed through the tube lumen to assess whether the distal end entered the stenotic segment and the upper end ends were <1 cm from the vocal folds. After closing of the external legs of the T-tube, the patients were followed-up weekly. In the first 10 cases, we left the external end of the tube out of the tracheotomy incision. However, in the final five cases, we shortened the external leg of the tube to the level of the trachea, closed the cap, and buried the end of the tube under the skin to prevent contamination by the outer flora and to allow the patient to return to social activities in a shorter time period (Figure 1). T- tubes were removed an average of 6 months later, de- pending on the health status of the individual patient. After tube removed, the patients were assessed closely for restenosis over a 1-month follow-up period. We eva- luated complications and additional interventions during and after T-tube insertion. Between the effect of entuba- tion time, length and localization of stenosis, T-tube length of stay and recovery of stenosis evaluated by Pear- son correlation analysis. 3. Results and Analysis Ten patients were male and five were female. Average age was 43.40 ± 14.4 years. The etiology was long-term intubation, except for two patients, and the average intu- bation time was 12.80 ± 7.5 days. In one patient, a Cae- sarean section was thought to be the cause and one other had had progressive dyspnea since childhood. In 12 (80%) patients, the stenotic segment was de- tected in the subglottic area at the cricoid cartilage, in 1 (6, 6%) patient at both the cricoid and cervicothoracic tracheal segment and in 2 (13, 3) patients at the cervico- thoracic segment. The average length of the stenotic seg- Figure 1. Montgomery T-tube applications, external end of the T-tube outside the skin (blue arrow) at right side and external end of the T-tube burying subcutaneous tissue (blue arrow) at left side on computed tomograhy images. ment was 19.0 ± 4.7 mm and the anterior posterior di- ameter was 6.47 ± 1.8 mm. The average T-tube follow up period was 6.0 ± 6.6 months, which varied according to the general health condition of the patient (Table 1). In one patient, bacterial colonization occurred in the external end of the tube. The infection was resistant to treatment, so no additional application could be made and the patient quit the follow-up after 7 months with a T-tube. In three cases, granulation tissue formed around the tracheotomy. This tissue was removed and topical AgNO3 cauterization yielded positive results. In one pa- tient, tracheomalacia occurred at the upper cartilage rings. This case was considered tracheomalacia despite repeti- tive T-tube applications, costal graft reconstruction and anastomosis; lastly, the T-tube was applied and its exter- nal end was buried subcutaneously. In one patient, the T-tube was inserted at the laryngeal stenosis after the end to end anastomosis in tracheal stenosis this called double stenosis. This patient was accompanied the aspiration. To prevent this, the posterior part of the upper T-tube was left high so as to fit into the arytenoids, and the front part was shortened so as to extend below the vocal cords, resolving the aspiration. Accumulation of tracheal secre- tions occurred in the five cases in whom the T-tube was subcutaneous burried. These patients were treated with drainage and daily dressings. Stenosis were recovered in three cases (21, 4%) after T-tube excision and restenosis occurred in the other four cases (28.5%). Long term pal- Copyright © 2013 SciRes. IJOHNS  H. ASLAN ET AL. 153 Table 1. Definitive statistical analysis. n Minimum Maximum Mean Std. Deviation Age 15 17 70 43.40 14,451 Intubation Time (day) 15 0 27 12.80 7,571 Stenosis length (mm) 15 10 30 19.00 4,706 Ant-post diameter (mm) 15 3 8 6.47 1,846 T-tube duration (month) 15 2 27 6.00 6,665 liative stenting in two cases (14.2%): in one due to tra- cheomalacia and in one due to the patient becoming bed- ridden. Five cases (35,7%) who had the T-tube applied in the past 7 months are still available for follow-up (Table 2). Among three recovered patients, there was no statisti- cally significance between intubation duration, stay with T tube duration and stenosis lenght according to Pearson correlation analysis (p: 0.341, p: 0.143, p: 0.354) (Table 3). Patients with restenosis necessitating additional sur- gery. Three cases applied stenotic segment resection and end to end anastomosis, one case required reconstruction with a costal graft due to front wall collapse. These cases had sufficient respiration and phonation in daily activity. Between three recovered patients and four restenosis patients, there was no statistically significance between intubation duration, stay with T tube duration and steno- sis lenght according to chi-square test (p: 0.603, p: 0.225, p: 0.311) (Table 4). 4. Discussion Predisposing factors for tracheal stenosis related to the endotracheal tube include a wider tube diameter, longer intubation time, continuous movement of the tube in the tracheal lumen, and difficult intubation. Other etiologies include traumatic and nonsterile aspiration, emergency cricothyrotomy, high-level tracheotomies and mechanical ventilation [1]. Some studies claim that tracheal damage begins 17 hours in adults and 1 week in infants following intubation [2]. One of our patients developed tracheal stenosis after just 1 hour of general anesthesia for a Cae- sarian section despite other patients being intubated for 7 - 27 days. Stenosis usually occurs in the cricoid cartilage or in the first tracheal ring due to overinflation of the tube cuff, leading to necrosis. Tracheal stenoses arise frequently on or directly below the vocal cords on the superior side and in the first tracheal ring or directly below it on the infe- rior side [4]. Another cause of tracheal stenosis is trache- otomy. Tracheotomy-induced stenosis is classified as sto- mal, suprastomal, intermediate or distal [5]. In 12 (80%) Table 2. Outcomes of 14 patients who received a T-tube, excluding 1 who quit due to bacterial colonization. Stenosis status Number of patients % Recovery of stenosis 3 21.4 Restenosis 4 28.5 Still with T-tube 5 35.7 Permanent T-tube 2 14.2 Table 3. Effect of intubation time, length of stenosis and with T-tube duration on recovery of stenosis evaluated by Pearson correlation analysis. Patients with recovery of stenosis Intubation duration (day) Length of stenosis (mm) WithT-tube duration (day) 1 14 20 60 2 10 15 60 3 20 20 420 p value p: 0.341 p: 0.143 p: 0.354 Table 4. Comparison of between three recovered patients and four restenosis patients according to intubation dura- tion, stay with T tube duration and stenosis length evalu- ated by chi-square test. Patients of recovery stenosis (n = 3) Patients of Restenosis (n = 4)p value Intubation duration (day)14, 10, 20 21, 7, 9, 14 0.603 Lenght of stenosis (mm)20, 15, 20 20, 20, 22, 30 0.311 Stay with T-tube duration (day)60, 60, 420 120, 60, 180, 60 0.225 of our cases, the stenosis occurred in the subglot- tic area at the cricoid ring level, at the T1-T2 level right below the tracheotomy line in 2 (13, 3%) cases, and 1 (6, 6%) case had stenosis in both the cricoid ring and below the tracheotomy. Myer and Cotton divided tracheal stenoses into four categories based on the degree of obstruction: Grade 1 corresponds to <50%; Grade 2, 50% - 70%; Grade 3, 70% - 99%; and Grade 4 is total obstruction [6]. A 50% reduction in the airway causes exertion dyspnea and a 75% reduction causes dyspnea at rest. Typically in adults, a decrease of the anterio-posterior diameter to 8 mm causes exertion dyspnea and to 5 mm causes dyspnea at rest [7]. Our cases were grades 2 and 3 according to the Cotton classification, with an average anterio-posterior diameter of 6.47 ± 1.846 mm. All of the patients had dyspnea even while at rest. In reality, tracheal resection and anastomosis is con- sidered the treatment of choice for tracheal stenosis [8]. However, when the glottis and/or the subglottis are also Copyright © 2013 SciRes. IJOHNS  H. ASLAN ET AL. 154 involved this surgical approach may not be applicable; moreover, it may not be feasible due to the extent of the stenosis, underlying disease and general health of the patient. Currently, many procedures are available and consist essentially of splitting followed by reconstruction using cartilage or bone. Endoscopic treatment includes laryngeal microsurgery, laser-assisted excision, tradi- tional dilation and endoscopic stent insertion [9]. In cases of longer stenoses, cardiac or additional air- way problems exist and stenosis involved glottis/subglot- tis tracheal stents are a plausible means of providing a permanent or temporary airway opening [10]. Various endotracheal tube types like Montgomery T-tube, Aboul- ker, Dumon and Cotton Lorenz exist. Metallic and sili- cone endotracheal tubes can be placed endoscopically without tracheotomy but have a disadvantage of obstruc- tion of the endoluminal stents in patients with disabled cough reflex and laryngotracheal stenosis. Difficulties in application of expandable metallic stents make these stents not suitable for short term relief of air- way obstruction and migration of the stent in the airway is a disadvantage of the sillicon Dumon stents [11]. The silicon rubber T-tube developed by Montgomery in 1965 has proven widely beneficial in tracheal surgery. The T-tube has three legs of different diameters and lengths. Tracheotomy is needed to insert the T-tube, which can be performed concurrently with surgery, or later to allow completion of stomal epithelialization. The external opening can be left open for ventilation and cleaning but can also be closed for voice and ventilation. Caretta et al. conducted a 158-case study which found that the Montgomery T-tube could be used primarily or complementary to surgical treatment and can be an effec- tive alternative when other stents are unsuccessful [12]. Thus, we use primarily the Montgomery T-tube in our clinic when stenting is desired. The T-tube is generally well-tolerated. It can be left in situ for years but sometimes must be changed due to in- fection, granulation, etc. Complications include infection or granulation at the end contacting the skin, granulation tissue under the cords at the upper side, and bacterial colonization or crust formation in the lumen. There is also a risk of tracheomalacia in areas in contact with the tube edges [13]. The tube became colonized with resis- tant bacteria in one patient, and we found granulation tissue around the tracheotomy site in three patients, and tracheomalacia in one. The subcutaneous burying procedure, described by Keszler in 1987, allows the patient to return to his/her social life earlier [14]. Cooper performed this procedure in 47 patients in 1989 and reported satisfying results [15]. We experienced no problems except collection of tra- cheal secretions in first cases in which we applied this procedure. We treated these patients with drainage and frequent dressing and rotated sternotyroid muscle fleb on tracheotomy site in later patients. In our burying proce- dure we have not observed any crusts or infections in the tube lumen because we have prevented outside contami- nation towards the lumen of the tube. T-tube excision time varies according to the surgeon’s decision, severity of the lesion, and problems relating to control and adaptation of the patient. Gaissert et al. ap- plied T-tubes to 16 patients and left them in place for 20 months [16]. Morshed et al. left T-tubes in place for 3 - 60 months [17]. Our T-tube duration was 2 - 27 months (mean, 6.0 ± 6.665 months), depending on the clinical condition of the patient. Liu et al. reported regression in tracheal stenosis after T-tube removed in 28 of 53 patients (52%) [18]. Ma- niglia applied the T-tube in 53 adult patients with tra- cheal or subglottic stenosis or both, and reported achieve- ment of a sufficient airway in 85% [19]. Five of our cases (33, 3%) were decanullated after T tube removed and three patients (20%) were decanullated after addi- tional surgeries too. This rate is lower than those reported in the literature, possibly due to ongoing follow-up with three cases and that some cases required a long-term stending T-tube due to their clinical condition. 5. Conclusion For tracheal stenosis, tracheal resection and anastomosis is widely considered the treatment of choice. However, this surgical approach is not feasible when the glottis and subglottis are involved or in patients with poor general condition. We prefer the T-tube first treatment in patients with grades 2-3 circular cicatricial tracheal stenosis in the Myer-Cotton classification. We follow our patients for an average of 6 - 9 months and make additional applications in following sessions when necessary. Given the com- plexity of tracheal surgery, we feel that noninvasive T- tube application should be initially attempted in tracheal stenosis. The subcutaneus procedure is with more cases and longer follow-ups, and we think this technique would be more efficient, since the absence of the outer part of T-tube enables more infection and granulatiın tissue-free environment; also better cosmetic and social outcomes for the patients. After discharging, we also do a monthly follow up. None of our patients had a life threatening complication. REFERENCES [1] J. J. Ballenger and J. B. Snow, “Havayolu Kontrolü ve Laringotrakeal Stenoz. Otolaringoloji’de,” Nobel Tıp Kitabevi, İstanbul, 2000, pp. 466-494. [2] C. W. Cummings, J. M. Frederickson, L. A. Harker, C. J. Krause, M. A. Richardson and D. E. Schuller, “Glottik and Subglottic Stenosis,” In: R. T. Cotton and G. H. Copyright © 2013 SciRes. IJOHNS  H. ASLAN ET AL. Copyright © 2013 SciRes. IJOHNS 155 Zalzal, Eds., Otolaryngology Head and Neck Surgery, Mosby, Baltimore, 1998, pp. 303-321. [3] H. C. Herrington, S. M. Weber and P. E. Andersen, “Modern Management of Laryngotracheal Stenosis,” La- ryngoscope, Vol. 116, No. 9, 2006, pp. 1553-1557. doi:10.1097/01.mlg.0000228006.21941.12 [4] S. Ercan and M. Yüksel, “Trakea Cerrahisi,” In: M. Yüksel, G. Kalaycı, Eds., Göğüs Cerrahisi, Bilmedya Grup, İstanbul, 2001, pp. 727-746. [5] R. Meyer and I. Flemming, “Recontructive Surgery of the Trachea,” Thieme, New York, 1982, p. 25. [6] C. M. Meyer III, D. M. O’Connor and R. T. Cotton, “Proposed Grading System for Subglottic Stenosis Based on Endotracheal Tube Sizes,” Annals of Otology, Rhino- logy, and Laryngology, Vol. 103, No. 4, 1994, pp. 319- 323. [7] B. E. Mostafa and A. El Halafawi, “Clinical Facts,” In: B. E. Mostafa, C. Chaouch-Mberek and A. El Halafawi, Eds., Tracheal Stenosis: Diagnosis and Treatment, 2012, pp. 17-32 [8] P. Marques, L. Leal, J. Spatley, et al., “Tracheal Resec- tion with Primary Anastomosis: 10 Years Experience,” American Journal of Otolaryngology, Vol 6, No. 30, 2009, pp. 415-418. doi:10.1016/j.amjoto.2008.08.008 [9] M. Mandour, M. Remacle, P. Van de Heyning, et al., “Chronic Subglottic and Tracheal Stenosis: Endoscopic Management vs. Surgical Reconstruction,” European Ar- chives of Otorhinolaryngology, Vol. 7, No. 260, 2003, pp. 374-380. doi:10.1007/s00405-002-0578-3 [10] A. Gallo, A. Paglıuca, A. Greco, S. Martellucci, A. Mascelli, M. Fusconi and M. De Vıncentis, “Laryngo- tracheal Stenosis Treated with Multiple Surgeries: Ex- perience, Results and Prognostic Factors in 70 Patients,” Acta Otorhinolaryngologica Italıca, Vol. 3, No. 32, 2012, pp. 182-188. [11] J. Strausz and C. T. Bolliger, “Interventional Pulmonol- ogy,” Monograph, 2010, pp. 190-202. [12] A. Carretta, M. Casiraghi, et al., “Montgomery T-Tube Placement in the Treatment of Benign Tracheal Lesions,” Cardiothorac Surgery, Vol. 2, No. 36, 2009, pp. 352-356. doi:10.1016/j.ejcts.2009.02.049 [13] W. W. Montgomery, “T-Tube Tracheal Stent,” Archives of Otolaryngology, No. 82, 1965, pp. 320-321. doi:10.1001/archotol.1965.00760010322023 [14] P. Keszler, “Tracheal T Tube: For İndwelling İntubation as an Alternative Management Method,” In: H. C. Grillo and H. Eschapasse, Eds., International Trends in General Throracic Surgery, Saunders, Philadelphia, 1987, pp. 133-137. [15] J. D. Cooper, F. G. Pearson, G. A. Patterson, et al., “Use of Silicone Stents in the Management of Airway Pro- blems,” Annals of Thoracic Surgery, Vol. 3,No. 47, 1989, pp. 371-378. doi:10.1016/0003-4975(89)90375-5 [16] H. A. Gaissert, H. C. Grillo, D. J. Mathisen and J. C. Wain, “Temporary and Permanent Restoration of Airway Continuity with the Tracheal Tube,” Journal of Thoracic and Cardiovascular Surgery, Vol. 2, No. 107, 1994, pp. 600-606. [17] K. Morshed, M. Szymanski and W. Golabek, “The Use of Tracheostomy T-Tube in the Treatment of Tracheal Stenosis,” Polish Otolaryngology, Vol. 3, No. 59, 2005, pp. 361-364. [18] H. C. Liu, K. S. Lee, C. J. Huang, C. R. Cheng, W. H. Hsu and M. H. Huang, “Silicone T-Tube for Complex Laryngotracheal Problems,” European Journal of Cardio- thoracular Surgery, Vol. 2, No. 21, 2002, pp. 326-330. doi:10.1016/S1010-7940(01)01098-3 [19] A. J. Maniglia, “Tracheal Stenosis: Conservative Surgery as a Primary Mode of Management,” Otolaryngologic Clinics of North America, Vol. 4, No. 12, 1979, pp. 877- 892.
|