Paper Menu >>
Journal Menu >>
 Vol.2, No.12, 1456-1459 (2010) Health doi:10.4236/health.2010.212216 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ Effects of fullerene nanowhiskers on cytotoxicity and gene expression Junko Okuda-Shimazaki1, Shinichi Nudejima2, Saiko Takaku1, Koki Kanehira3, Shuji Sonezaki3 Akiyohshi Taniguchi1* 1Advanced Medical Materials Group, Biomaterials Center, National Institute for Materials Science, Namiki, Tsukuba, Ibaraki, Japan; 2Advanced Nanomaterials Laboratory, National Institute for Materials Science, Namiki, Tsukuba, Ibaraki, Japan; 3TOTO Ltd. Research Institute, Nakashima, Kokurakita, Kitakyushu, Japan; *Corresponding Author: taniguchi.akiyoshi@nims.go.jp Received 23 June 2010; revised 10 July 2010; accepted 14 July 2010 ABSTRACT With recent developments in nanoscience and nanotechnology, carbon-based nanomaterials, such as carbon nanotub es or Fullerene nano whiskers (FNWs), are attracting a great deal of interest. However, nanomaterials have novel properties that may cause safety problems in biological systems. Although the toxic effects of multiwall carbon nanotubes (MWCNTs) in vitro and in vivo have been well described, the effect of FNWs on cells remains unclear. In the present study, we analyzed the gene expression and cytotoxicity of FNWs-treated cells. The results of cell viabil- ity and gene expression analysis indicate that FNWs has a relatively smaller ability to induce cellular gene expression as compared with MWCNTs or titania nanoparticles. Our results suggest that FNWs have weak cytotoxic effects as compared to the effects of MWCNTs and tita- nia nanoparticles. Keywords: Nanomaterials; Cytotoxicity; Gene Expression; Fullerene Nanowhisker 1. INTRODUCTION Nanomaterials are currently being investigated and have potential applications in various fields. With recent developments in nanoscience and nanotechnology, car- bon-based nanomaterials, such as carbon nanotubes (CNTs) and Fullerene nanowhiskers (FNWs), are at- tracting a great deal of interest. Because of their size, chemical composition, surface structure, shape, and ag- gregation properties, nanomaterials are expected to have novel physicochemical properties. The small size of na- nomaterials allows them to penetrate into the body [1]. These novel properties raise safety concerns for the use of nanomaterials in biological systems. Recent studies suggest that nanomaterials affect biological behavior and have the potential to be toxic [2-6]. The toxic effects of multiwall carbon nanotubes (MWCNTs) in vitro and in vivo have been well described [5,7-11]. FNWs also have the potential to be used as carbonaceous engineered na- nomaterials in the future. However, the effect of FNWs on cells remains unclear. In the present study, to understand the biological ac- tivity of FNWs, we analyzed the gene expression and cytotoxicity of FNWs-treated cells. Our results suggest that FNWs have a very weak effect on cytotoxic activity and the induction of gene expression as compared to the effects of MWCNTs and titania nanoparticles. 2. MATERIALS AND METHODS 2.1. Nanomaterials FNWs, which were formed by the liquid-liquid inter- facial precipitation method [12-14], were a kind gift from Dr K. Miyazawa (National Institute for Materials Science). MWCNTs (MITSUI MWNT-7; Lot. 061220- 02) were a kind gift from Dr. S. Tsuruoka (Mitsui & Co.,Ltd.). Small aggregated titania nanoparticles were prepared by the centrifuge method [6]. 2.2. Cell Cultures A human acute monocytic leukemia cell line, THP-1 [14,15], was cultured in RPMI 1640 medium supple- mented with 10% fetal bovine serum, 100 U/mL penicil- lin, and 100 μg/mL streptomycin under 5% CO2 with 100% humidity at 37°C. For the exposure experiments, THP-1 cells were treated with 200 nM phorbol 12- my- ristate 13-acetate (PMA) for 48 h. To expose the cells, PMA-treated THP-1 cells that had been seeded 24 h prior were exposed to titania particles for 24 h. 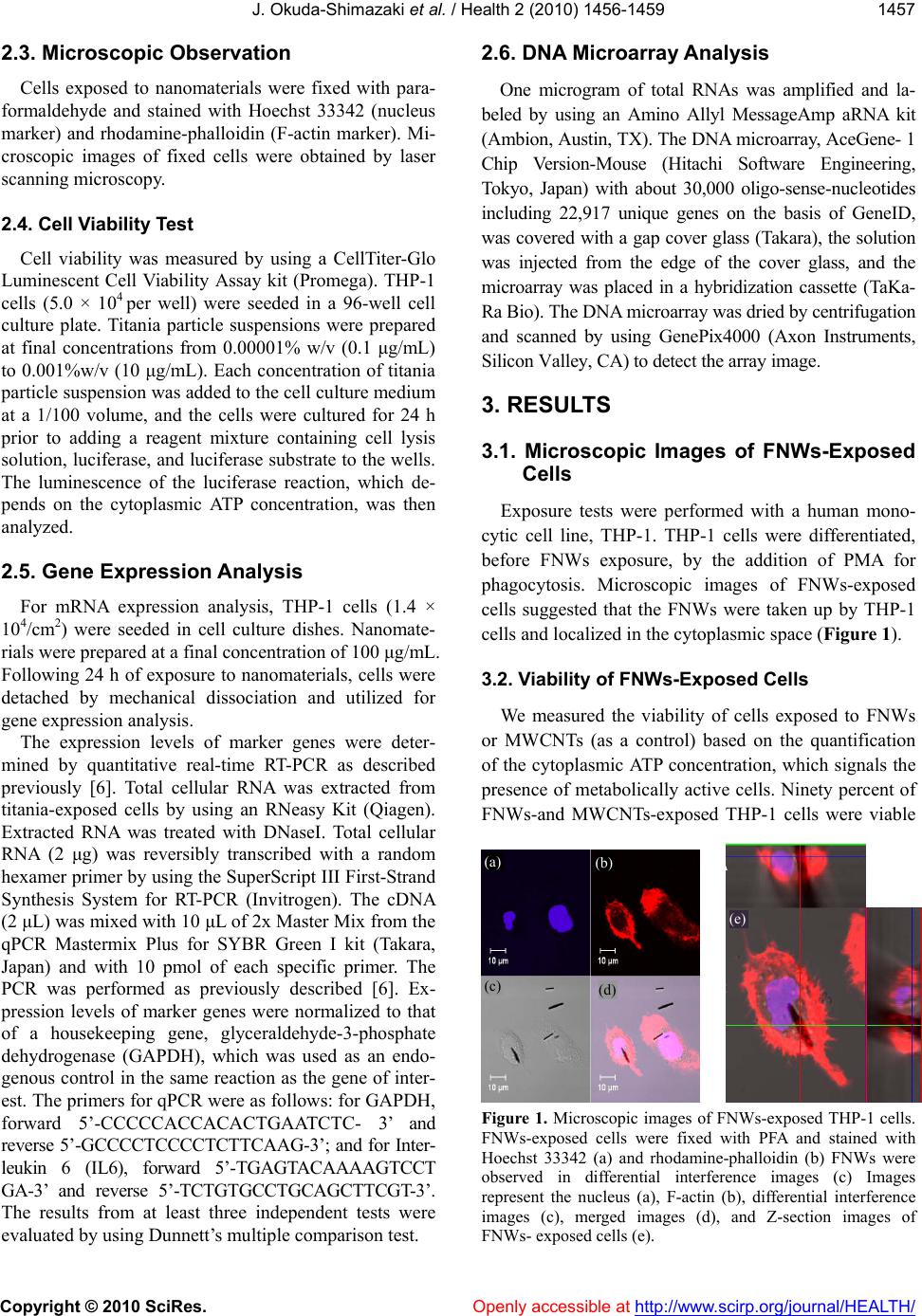 J. Okuda-Shimazaki et al. / Health 2 (2010) 1456-1459 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1457 2.3. Microscopic Observation Cells exposed to nanomaterials were fixed with para- formaldehyde and stained with Hoechst 33342 (nucleus marker) and rhodamine-phalloidin (F-actin marker). Mi- croscopic images of fixed cells were obtained by laser scanning microscopy. 2.4. Cell Viability Test Cell viability was measured by using a CellTiter-Glo Luminescent Cell Viability Assay kit (Promega). THP-1 cells (5.0 × 104 per well) were seeded in a 96-well cell culture plate. Titania particle suspensions were prepared at final concentrations from 0.00001% w/v (0.1 μg/mL) to 0.001%w/v (10 μg/mL). Each concentration of titania particle suspension was added to the cell culture medium at a 1/100 volume, and the cells were cultured for 24 h prior to adding a reagent mixture containing cell lysis solution, luciferase, and luciferase substrate to the wells. The luminescence of the luciferase reaction, which de- pends on the cytoplasmic ATP concentration, was then analyzed. 2.5. Gene Expression Analysis For mRNA expression analysis, THP-1 cells (1.4 × 104/cm2) were seeded in cell culture dishes. Nanomate- rials were prepared at a final concentration of 100 μg/mL. Following 24 h of exposure to nanomaterials, cells were detached by mechanical dissociation and utilized for gene expression analysis. The expression levels of marker genes were deter- mined by quantitative real-time RT-PCR as described previously [6]. Total cellular RNA was extracted from titania-exposed cells by using an RNeasy Kit (Qiagen). Extracted RNA was treated with DNaseI. Total cellular RNA (2 μg) was reversibly transcribed with a random hexamer primer by using the SuperScript III First-Strand Synthesis System for RT-PCR (Invitrogen). The cDNA (2 μL) was mixed with 10 μL of 2x Master Mix from the qPCR Mastermix Plus for SYBR Green I kit (Takara, Japan) and with 10 pmol of each specific primer. The PCR was performed as previously described [6]. Ex- pression levels of marker genes were normalized to that of a housekeeping gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH), which was used as an endo- genous control in the same reaction as the gene of inter- est. The primers for qPCR were as follows: for GAPDH, forward 5’-CCCCCACCACACTGAATCTC- 3’ and reverse 5’-GCCCCTCCCCTCTTCAAG-3’; and for Inter- leukin 6 (IL6), forward 5’-TGAGTACAAAAGTCCT GA-3’ and reverse 5’-TCTGTGCCTGCAGCTTCGT-3’. The results from at least three independent tests were evaluated by using Dunnett’s multiple comparison test. 2.6. DNA Microarray Analysis One microgram of total RNAs was amplified and la- beled by using an Amino Allyl MessageAmp aRNA kit (Ambion, Austin, TX). The DNA microarray, AceGene- 1 Chip Version-Mouse (Hitachi Software Engineering, Tokyo, Japan) with about 30,000 oligo-sense-nucleotides including 22,917 unique genes on the basis of GeneID, was covered with a gap cover glass (Takara), the solution was injected from the edge of the cover glass, and the microarray was placed in a hybridization cassette (TaKa- Ra Bio). The DNA microarray was dried by centrifugation and scanned by using GenePix4000 (Axon Instruments, Silicon Valley, CA) to detect the array image. 3. RESULTS 3.1. Microscopic Images of FNWs-Exposed Cells Exposure tests were performed with a human mono- cytic cell line, THP-1. THP-1 cells were differentiated, before FNWs exposure, by the addition of PMA for phagocytosis. Microscopic images of FNWs-exposed cells suggested that the FNWs were taken up by THP-1 cells and localized in the cytoplasmic space (Figure 1). 3.2. Viability of FNWs-Exposed Cells We measured the viability of cells exposed to FNWs or MWCNTs (as a control) based on the quantification of the cytoplasmic ATP concentration, which signals the presence of metabolically active cells. Ninety percent of FNWs-and MWCNTs-exposed THP-1 cells were viable AB CD A C E Figure 1. Microscopic images of FNWs-exposed THP-1 cells. FNWs-exposed cells were fixed with PFA and stained with Hoechst 33342 (a) and rhodamine-phalloidin (b) FNWs were observed in differential interference images (c) Images represent the nucleus (a), F-actin (b), differential interference images (c), merged images (d), and Z-section images of FNWs- exposed cells (e). (a) (b) (c) (d) (e) 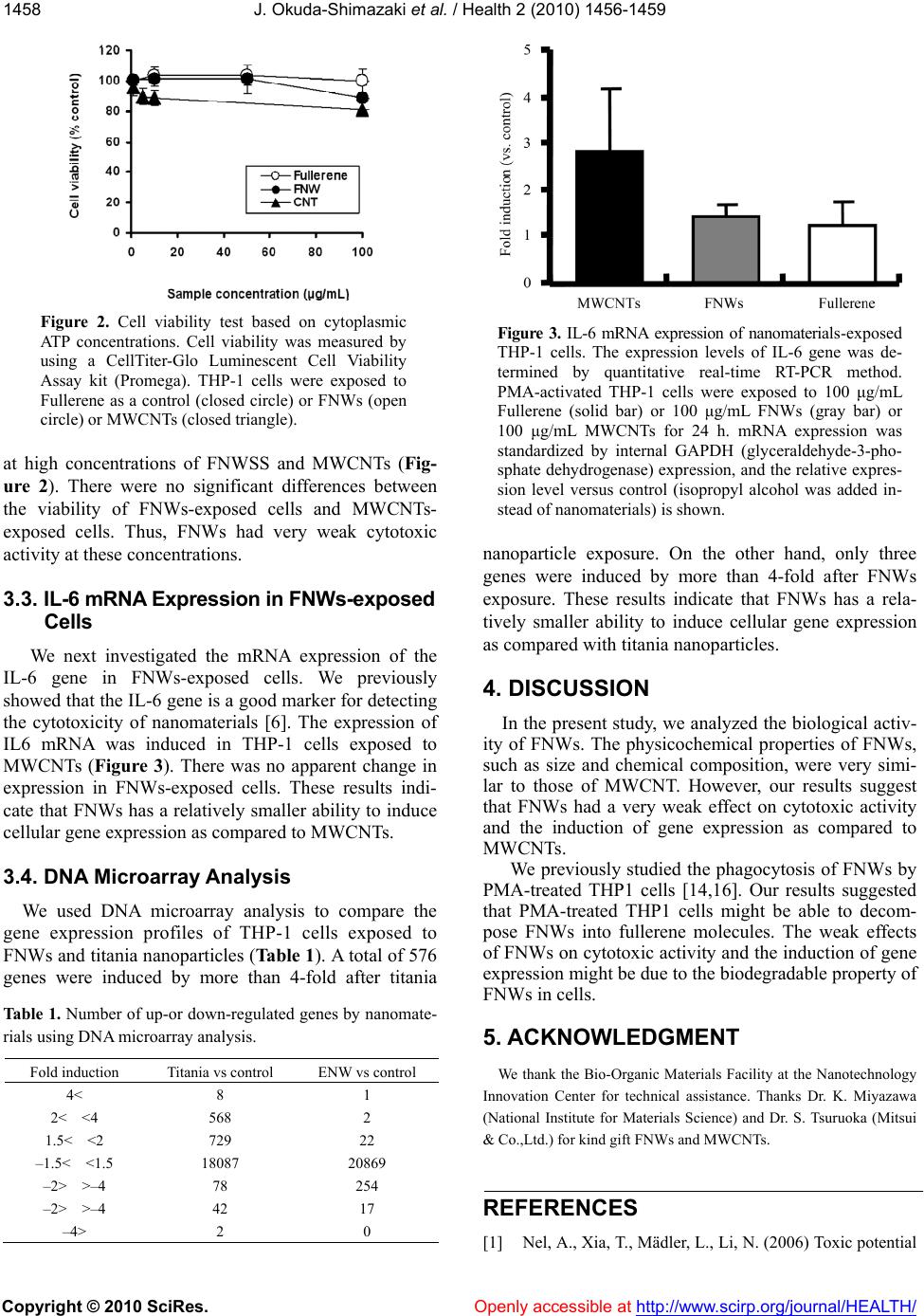 J. Okuda-Shimazaki et al. / Health 2 (2010) 1456-1459 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1458 Figure 2. Cell viability test based on cytoplasmic ATP concentrations. Cell viability was measured by using a CellTiter-Glo Luminescent Cell Viability Assay kit (Promega). THP-1 cells were exposed to Fullerene as a control (closed circle) or FNWs (open circle) or MWCNTs (closed triangle). at high concentrations of FNWSS and MWCNTs (Fig- ure 2). There were no significant differences between the viability of FNWs-exposed cells and MWCNTs- exposed cells. Thus, FNWs had very weak cytotoxic activity at these concentrations. 3.3. IL-6 mRNA Expression in FNWs-exposed Cells We next investigated the mRNA expression of the IL-6 gene in FNWs-exposed cells. We previously showed that the IL-6 gene is a good marker for detecting the cytotoxicity of nanomaterials [6]. The expression of IL6 mRNA was induced in THP-1 cells exposed to MWCNTs (Figure 3). There was no apparent change in expression in FNWs-exposed cells. These results indi- cate that FNWs has a relatively smaller ability to induce cellular gene expression as compared to MWCNTs. 3.4. DNA Microarray Analysis We used DNA microarray analysis to compare the gene expression profiles of THP-1 cells exposed to FNWs and titania nanoparticles (Table 1). A total of 576 genes were induced by more than 4-fold after titania Table 1. Number of up-or down-regulated genes by nanomate- rials using DNA microarray analysis. Fold induction Titania vs control ENW vs control 4< 8 1 2< <4 568 2 1.5< <2 729 22 –1.5< <1.5 18087 20869 –2> >–4 78 254 –2> >–4 42 17 –4> 2 0 Figure 3. IL-6 mRNA expression of nanomaterials-exposed THP-1 cells. The expression levels of IL-6 gene was de- termined by quantitative real-time RT-PCR method. PMA-activated THP-1 cells were exposed to 100 μg/mL Fullerene (solid bar) or 100 μg/mL FNWs (gray bar) or 100 μg/mL MWCNTs for 24 h. mRNA expression was standardized by internal GAPDH (glyceraldehyde-3-pho- sphate dehydrogenase) expression, and the relative expres- sion level versus control (isopropyl alcohol was added in- stead of nanomaterials) is shown. nanoparticle exposure. On the other hand, only three genes were induced by more than 4-fold after FNWs exposure. These results indicate that FNWs has a rela- tively smaller ability to induce cellular gene expression as compared with titania nanoparticles. 4. DISCUSSION In the present study, we analyzed the biological activ- ity of FNWs. The physicochemical properties of FNWs, such as size and chemical composition, were very simi- lar to those of MWCNT. However, our results suggest that FNWs had a very weak effect on cytotoxic activity and the induction of gene expression as compared to MWCNTs. We previously studied the phagocytosis of FNWs by PMA-treated THP1 cells [14,16]. Our results suggested that PMA-treated THP1 cells might be able to decom- pose FNWs into fullerene molecules. The weak effects of FNWs on cytotoxic activity and the induction of gene expression might be due to the biodegradable property of FNWs in cells. 5. ACKNOWLEDGMENT We thank the Bio-Organic Materials Facility at the Nanotechnology Innovation Center for technical assistance. Thanks Dr. K. Miyazawa (National Institute for Materials Science) and Dr. S. Tsuruoka (Mitsui & Co.,Ltd.) for kind gift FNWs and MWCNTs. REFERENCES [1] Nel, A., Xia, T., Mädler, L., Li, N. (2006) Toxic potential  J. Okuda-Shimazaki et al. / Health 2 (2010) 1456-1459 Copyright © 2010 SciRes. Openly accessible at http://www.scirp.org/journal/HEALTH/ 1459 of materials at the nanolevel. Science, 311, 622- 627. [2] Service, R.F. (2004) Nanotoxicology. Nanotechnology grows up. Science, 304, 1732-1734. [3] Oberdörster, G., Oberdörster, E., Oberdörster, J. (2005) Nanotoxicology: An emerging discipline evolving from studies of ultrafine particles. Environ. Health Perspect. 113, 823-839. [4] Poland, C.A., Duffin, R., Kinloch, I., et al. (2008) Car- bon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nat. Nanotechnol., 3, 423-428. [5] Takagi, A., Hirose, A., Nishimura, T., et al. (2008) Induc- tion of mesothelioma in p53+/- mouse by intraperitoneal application of multi-wall carbon nanotube. Journal of Toxicological Sciences, 33, 105-116. [6] Okuda-Shimazaki, J., Takaku, S., Kanehira, K., et al. (2010) Aggregate particle size effects of titanium dioxide nanoparticles on the gene expression. International Journal of Molecular Sciences, 11, 2383-2392. [7] Patlolla, A., Patlolla, B., Tchounwou, P. (2010) Evaluation of cell viability, DNA damage, and cell death in normal human dermal fibroblast cells induced by functionalized multiwalled carbon nanotube. Molecular and Cellular Biochemistry, 338, 225-232. [8] Jos, A., Pichardo, S., Puerto, M., et al. (2009) Cytotoxicity of carboxylic acid functionalized single wall carbon na- notubes on the human intestinal cell line Caco-2. Toxicol In Vitr o. 23, 1491-1496. [9] Lin, C., Fugetsu, B., Su, Y., Watari, F. (2009) Studies on toxicity of multi-walled carbon nanotubes on Arabidopsis T87 suspension cells. Journal of Hazard Mater. 170, 578-583. [10] Tong, H., McGee, J.K., Saxena, R.K., et al. (2009) In- fluence of acid functionalization on the cardiopulmonary toxicity of carbon nanotubes and carbon black particles in mice. Toxicology and Applied Pharmacology, 239, 224-232. [11] Belyanskaya, L., Weigel, S., Hirsch, C., et al. (2009) Effects of carbon nanotubes on primary neurons and glial cells. Neurotoxicology. 30, 702-711. [12] Miyazawa, K., Kuwasaki, Y., Obayashi, A., Kuwabara, M. (2002) C60 nanowhiskers formed by the liquid-liquid interfacial precipitation method. Journal of Materials Research, 17, 83-88. [13] Miyazawa, K., Cha, S., Ringor, C., et al. (2008) Synthe- sis of fullerene nanotubes and microtubes for materials storage, delivery and recovery. Nano, 3, 335-339. [14] Nudejima S., Miyazawa K., Okuda J., et al. (2009) Ob- servation of phagocytosis of fullerene nanowhiskers by PMA-treated THP-1 cells. Journal of Physics, 159, 1-6. [15] Okuda-Shimazaki, J., Yamamoto, A., Kuroda, D., et al. (2007) The effect of metal materials on heat shock pro- tein 70B’ gene expression. Open Biotechnology Journal, 1, 14-17. [16] Nudejima S, Miyazawa K, Okuda-Shimazaki J et al. (2010) Biodegradation of C60 fullerene nanowhiskers by macrophage-like cells. Advances In Biomedical Research, pp.89-94 |

