Current Distortion Evaluation in Traction 4Q Constant Switching Frequency Converters 71
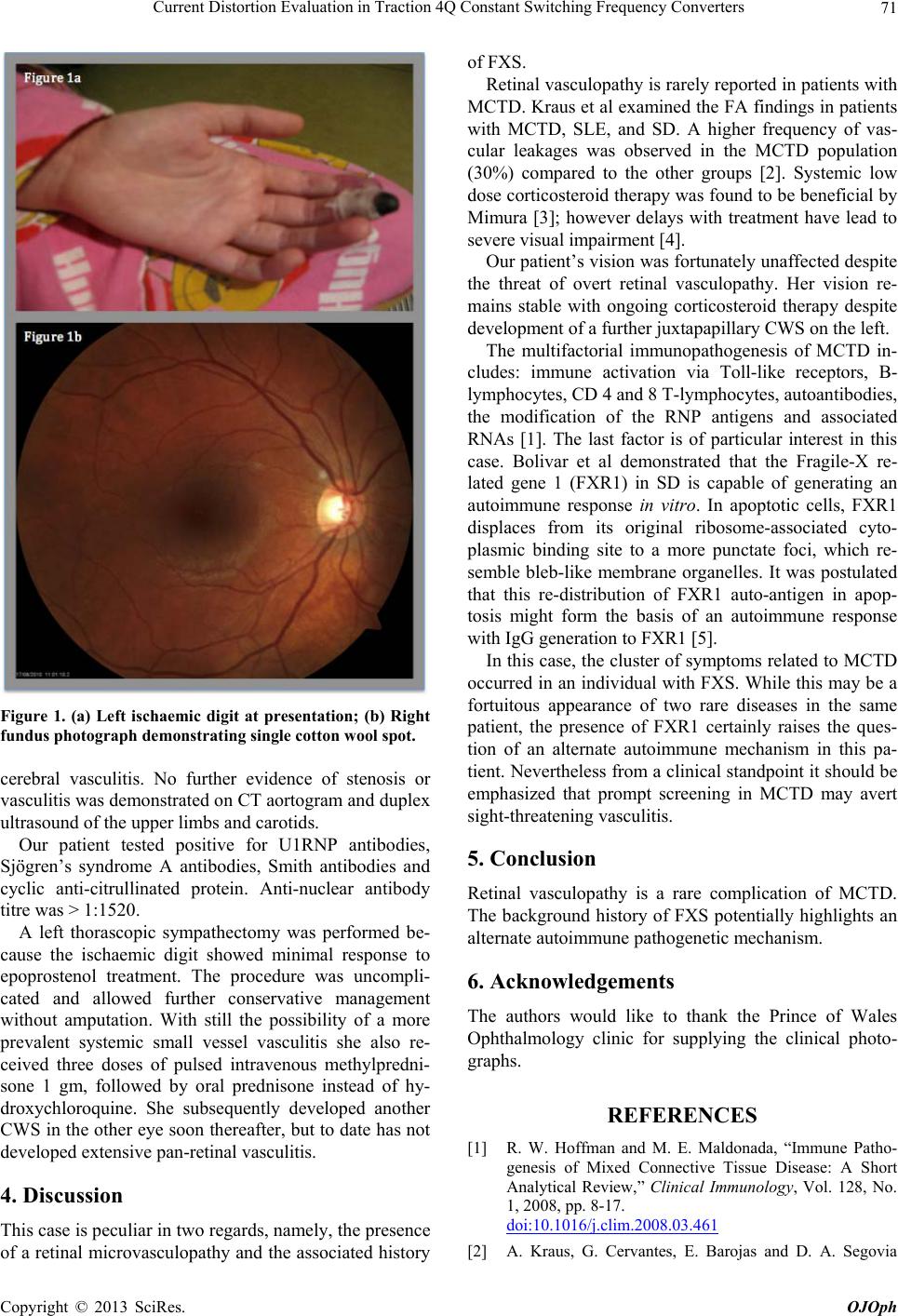
Figure 1. (a) Left ischaemic digit at presentation; (b) Right
fundus photograph demonstrating single cotton wool spot.
cerebral vasculitis. No further evidence of stenosis or
vasculitis was demonstrated on CT aortogram and duplex
ultrasound of the upper limbs and carotids.
Our patient tested positive for U1RNP antibodies,
Sjögren’s syndrome A antibodies, Smith antibodies and
cyclic anti-citrullinated protein. Anti-nuclear antibody
titre was > 1:1520.
A left thorascopic sympathectomy was performed be-
cause the ischaemic digit showed minimal response to
epoprostenol treatment. The procedure was uncompli-
cated and allowed further conservative management
without amputation. With still the possibility of a more
prevalent systemic small vessel vasculitis she also re-
ceived three doses of pulsed intravenous methylpredni-
sone 1 gm, followed by oral prednisone instead of hy-
droxychloroquine. She subsequently developed another
CWS in the other eye soon thereafter, but to date has not
developed extensive pan-retinal vasculitis.
4. Discussion
This case is peculiar in two regards, namely, the presen ce
of a retinal microvasculopathy and the associated history
of FXS.
Retinal vasculopathy is rarely reported in patients with
MCTD. Kraus et al examined the FA findings in patients
with MCTD, SLE, and SD. A higher frequency of vas-
cular leakages was observed in the MCTD population
(30%) compared to the other groups [2]. Systemic low
dose corticosteroid therapy was found to be beneficial by
Mimura [3]; however delays with treatment have lead to
severe visual impairment [4].
Our patient’s vision was fortunately unaffected despite
the threat of overt retinal vasculopathy. Her vision re-
mains stable with ongoing corticosteroid therapy despite
development of a further juxtapapillary CWS on the left.
The multifactorial immunopathogenesis of MCTD in-
cludes: immune activation via Toll-like receptors, B-
lymphocytes, CD 4 and 8 T-lymphocytes, autoantibodies,
the modification of the RNP antigens and associated
RNAs [1]. The last factor is of particular interest in this
case. Bolivar et al demonstrated that the Fragile-X re-
lated gene 1 (FXR1) in SD is capable of generating an
autoimmune response in vitro. In apoptotic cells, FXR1
displaces from its original ribosome-associated cyto-
plasmic binding site to a more punctate foci, which re-
semble bleb-like membrane organelles. It was postulated
that this re-distribution of FXR1 auto-antigen in apop-
tosis might form the basis of an autoimmune response
with IgG gener a t i on to F XR 1 [5] .
In this case, the cluster of symptoms related to MCTD
occurred in an individual with FXS. While this may be a
fortuitous appearance of two rare diseases in the same
patient, the presence of FXR1 certainly raises the ques-
tion of an alternate autoimmune mechanism in this pa-
tient. Nevertheless from a clinical standpoint it should be
emphasized that prompt screening in MCTD may avert
sight-threatening vasculitis.
5. Conclusion
Retinal vasculopathy is a rare complication of MCTD.
The background history of FXS potentially highlights an
alternate autoimmune pathogenetic mechanism.
6. Acknowledgements
The authors would like to thank the Prince of Wales
Ophthalmology clinic for supplying the clinical photo-
graphs.
REFERENCES
[1] R. W. Hoffman and M. E. Maldonada, “Immune Patho-
genesis of Mixed Connective Tissue Disease: A Short
Analytical Review,” Clinical Immunology, Vol. 128, No.
1, 2008, pp. 8-17.
doi:10.1016/j.clim.2008.03.461
[2] A. Kraus, G. Cervantes, E. Barojas and D. A. Segovia
Copyright © 2013 SciRes. OJOph