 Open Journal of Metal, 2013, 3, 1-7 http://dx.doi.org/10.4236/ojmetal.2013.32A1001 Published Online July 2013 (http://www.scirp.org/journal/ojmetal) Behavior of Heavy Metals during the Agro-Industrial Wastes Gasification Marcelo Echegaray, Marianela Costante, Alejandra Saffe, Carlos Palacios, Rosa Rodriguez Instituto de Ingeniería Química, Facultad de Ingeniería, Universidad Nacional de San Juan, San Juan, Argentina Email: rrodri@unsj.edu.ar Received May 24, 2013; revised June 27, 2013; accepted July 7, 2013 Copyright © 2013 Marcelo Echegaray et al. This is an open access article distributed under the Creative Commons Attribution Li- cense, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT The characterization analysis of three agro-industrial wastes was performed in order to study its thermal gasification. Some analyses such as determination of Ca, K and Mg concentration and determination of three representative toxic metals concentration Cd, Cr and Pb in all its oxidation states and the fundamental state, were carried out. The heavy metals concentration was also determined in the ashes obtained during the gasification process. The mobility of these elements was studied through three leaching tests. The behavior of heavy metals, sulfur and chlorine compounds, was predicted considering the presence of water vapor, syngas, Ca, Mg, K, Si, Al and other ash components. The heavy metals are not more concentrated in the gasification ash; these pollutants are released during this process. Ca, Mg and K presence in these residues would promote the pollutants retention. The ash of the studied waste can be disposed in controlled landfills or used in road construction, according to the obtained results during the leaching test DIN-DEV S4. The obtained results in the leaching test EPA 1311 TLCP classify these gasification ashes as no toxic waste. Keywords: Gasification; Agro-Ind ustrial Wastes; Ash 1. Introduction The agro-industrial sector produces a significant envi- ronmental impact in specific geographical areas, due to generated waste, such as the Cuyo Region, Argentina. A strategy in this sense is to propose an appropriate agro- industrial waste management in order to minimize the emitted pollutants, transforming them into high value- added products or renewable energy source, tending to “Zero-waste”. During the 2011 harvest, 690,000 tons were used to produce wine, generating nearly 200,000 kg of stalks without considering other solid wastes, such as marcs and wine dregs. The latter are generally used for the by- products re covery. However, reuse and/or disposal of exhausted marcs and wine dregs are a current problem in the region, be- cause their disposal in landfills is not environmentally convenient due to they are not fully reused and large volumes are generated, requiring significant areas of land for their disposal. The waste from the fruits and vegeta- bles canning industry have a high water content and, in many cases, significant amounts of lignocellulose mate- rials. The final disposal in landfills is also performed in this region. Moreover, there is a growing global interest in the technologies development for the exploitation of renew- able energy sources because of environmental and eco- nomic reasons. In particular, due to the continuous in- crease in the cost of fossil energy resources, biomass is considered as one of the most promising and viable al- ternatives. Energy from waste is an important component of integrated waste management. One of the major limi- tations in the use of biomass wastes for energy produc- tion is its availability and moderate calorific value re- sulting in a low production and high costs compared to fossil fuels. The reduction of gases emissions, such as SOx and greenhouse gases; however, is agreed with the policies of current pollution control [1]. The energy conversion technologies and the biomass- based systems are the only electricity renewable source excluding hydro power, a crucial fact for future electric- ity production. A technology with a great future is the gasification. After more than 30 years of research, there is now worldwide interest in the use of H2 as an alterna- tive transportation fuel [2 ]. The steam gasification of waste is an attractive process for producing H2-rich gas [3-5]. This process has been developed to reduce the amount of undesired products C opyright © 2013 SciRes. OJMetal  M. ECHEGARAY ET AL. 2 and the coke formation rate [6]. Furthermore, a vapor excess can easily be separated by condensation. Regard- ing existing gasification technologies, the fluidized bed is attractive because it provides a good contact between gas and solid, uniform temperatures and high reaction rates, compared to the fixed bed gasification [7]. Moreover, fluidized beds hav e a high flexibility in th e feed in terms of shape, size and composition, as well as a wide range of operational and safety capabilities [8]. Considering the heavy metals, they are enriched in the solid waste of gasification (fly ash and bottom), and they are also released in the gas stream or tar. Their vaporiza- tion depends on the initial chemical speciation, gasifica- tion atmosphere, the fluid dynamics, the kinetics of heavy metals diffusion in the solid particles and reaction kinetics between the heavy metals and major components of ash [9,10]. The ash disposal conditions as well as their reuse are established by the trace elements concentration and their mobility [8]. In view of these aspects, the heavy metals behavior during agro-industrial waste gasification, their mobility out the ash matrix and the toxicity determination of gen- erated solid waste during this process were studied. 2. Experimental Agro-industrial residues from canning and wine sector were used: peach pits, stalks and marc, respectively. These industries are located in the province of San Juan, Argentina. In order to obtain the ash, a differential reactor was used. It is constructed of AISI 316 stainless steel. It is constituted by a cylinder with 50 mm of diameter and 30 mm of length, heated by an electric resistance with elec- tronic temperature control. Figure 1 shows a used reactor scheme. According to Kurkela et al. [11] (2006), for feeds with high alkali content, low gasification temperatures (T = 750˚C - 850˚C) and the steam addition are recommended to prevent the agglomeration in the fluidized reactor. Skoulou et al. (2008) [12] studied the effect of tem- perature (T = 750˚C - 850˚C) and air equivalent ratio (ER = 0.2 - 0.4) in biomass gasification into a fluidized bed (ER is the ratio between the air sub-stoichiometric and the air required for complete combustion amounts). Ex- perimental results showed that working at 750˚C and ER equal to 0.2, the H2 optimal content in the syngas is ob- tained. Taking into account these experimental results ob- tained by other researchers, the used gasifying agent was the steam and air mixture. The ER was equal to 0.2 and the temperature equal to 750˚C. The gasifying agent (air- steam mixture) entered from the reactor bottom. The syngas exited at the top. Figure 1. Used reactor scheme. 1: water tank; 2: pump; 3: evaporator; 4: mixer; 5: power supply; 6: tempe rature con- troller; 7: electric resistanc e; 8: reactor ; 9: support steel; 10: porous metal mesh; 11: agro-industrial wastes; 12: gas out- put; 13: thermocouple. For each test, the reactor is loaded with 50 to 60 g of agro-industrial wastes. The obtained ashes in this reactor are considered with similar characteristics as the ash bot- tom obtained in a fluidized bed reactor [13]. 2.1. Agro-Industrial Wastes Characterization The weight loss at 105˚C (ASTM D3173-87, Standard Test Method for Moisture in the Analysis Sa mple of Coal and Coke, 1996), the ash and the org anic matter contents (ASTM D3172-89(2), Standard Practice for Proximate Analysis of Coal and Coke, 2002), the concentrations of Cd, Cr and Pb were determined for the studied agro-in- dustrial wastes. In or de r to d ete r mine the h ea vy me ta l co n- centrations, first, the samples were digested according to EPA digestion (US Environmental Protection Agency, 1982). Then, the heavy metals concentrations were de- termined using a v isible light spectrophotometer (HACH DR/2010 Spect ro p hotomet er Datal og gin g p ort able). Cd, Pb and Cr were considered due to their behaviors during heat treatments which are different. Cd vaporizes and it is not remained in the ash, Pb shows an intermedi- ate behavior, and Cr is remained in solid residue of gasi- fication [1 4]. In order to determine the influence of the presence of Ca, Mg and K in this biomass gasification, their concen- tration was determined in the studied agro-industrial wastes, using the atomic absorption method. 2.2. Heavy Metals Mobility of the Ash Mineral Matrix With the purpose of study the heavy metals mobility of Copyright © 2013 SciRes. OJMetal  M. ECHEGARAY ET AL. 3 the ash mineral matrix, the Cd, Pb and Cr concentration s were determined in the solid wastes of gasification, using the analytical techniques described above. This study is very important because it determines the final disposal and/or further use. Particularly, the use of the gasification ash in different applications contributes to the sustainability of biomass use in power generation. Several options are discussed: use as fertilizer, as a building material or as fuels [15]. The heavy metals mobility was studied by three different tests: The German test, DIN 38414 part 4 (DEV S4, Ger- man Standard Procedure for Water, Wastewater and Sediment Testing, 1984): It is used to classify the waste. The limits of heavy metals concentrations in the leaching solution are expressed in mg/l for dis- posal in landfill (Class 1) and for the ash use in road constructi o n [ 16]. The US EPA TCLP 1311 test (Toxicity Characteris- tic Leaching Procedure. Methods for Evaluating Solid Waste, 1992): It determines the potential leaching of organic and inorganic material in liquid, solid and multi-phase, of the residues in contact with ground- water. This test simulates landfill leaching conditions and the ash can be classified as toxic or not [17]. The Dutch NEN 7341 test (Determination of the Leaching Behavior of Granular Materials: Availabil- ity Test, 1993): This test determines the maximum proportion of heavy metals leached from different wastes such as ash. This is achieved by leaching of finely milled solid (maximizing the contact surface) and using a high liquid/so lid ratio. The metal fixation in the solid matrix is predicted with this test [18,19]. The DIN test uses the weaker leaching agent, distilled water, and the test NEN the stronger, nitric acid. 3. Results and Discussion The results of the agro-industrial wastes characterization are shown in Table 1. The highest ash and water content were found in the stalk. A high water content increases the energy requirements to carry out the gasification, decreasing the efficiency of the plant, but on the other hand, it improves the synthesis gas quality by increasing the content of CO2, CH4 and H2 [20] and decreasing hy- drocarbons and tars levels. In order to optimize the gasi- fier operation, Pfeifer et al. [21] determined the optimum content equal 20% to 40% by weight at low temperatures heating. Regarding the ash content, a low percentage of it will minimize the production of fly and the bottom ash. In general, these solids contain significant amounts of un- reacted carbon and sulfur [22]. Cd, Cr and Pb are present in the composition of stud- ied agro-industrial wastes. The stalks and the peach pits Table 1. Results of proximate analysis. Determination of heavy metals and Ca, K, and Mg concentrations in agro- industrial wastes. Stalk Marc Peach pits Weight loss at 105˚C (dry basis %) 73.23 55.06 35.57 Ash (dry basis %) 6.30 5.08 0.73 Organic matter (dry basis %) 93.7 94.92 99.27 Cd (mg/kg dry basis) 1.25 0.02 1.25 Cr (mg/kg dry basis) 25.00 37.50 3.125 Pb (mg/kg dry basis) 75.00 82.92 0.94 Ca (g/kg dry basis) 2.25 2.96 0.02 K (g/kg dr y ba s i s) 19.23 7.38 7.15 Mg (g/kg dry basis) 0.58 0.46 0.44 presented the highest Cd concentrations. With respect to Cr and Pb, the highest concentrations were found in winema ki ng waste. Considering the obtained results b y analyzing the ga si- fication ash (Table 2), the highest Cd and Pb concentra- tions were found in the marcs ash. For Cr, the highest concentrations were found in the stalks ash, in this case the metal concentrations is more variable (between 3.12 and 15.86 mg Cr/kg of dry weight waste). Comparing the found heavy metals concentrations in the stalks and their ash, the Cd concentration variation is very small and the Cr and Pb concentrations of stalks are higher than these concentrations in their ash. In the case of the marcs and their ash, the Cd is more concentrated in the gasification solid waste, but the Cr and Pb concentra- tions are higher in the marcs. Comparing the concentra- tions of three heavy metals found in peach pits and their ash, a significant variation is not observed. On this point, it is important to explain the heavy met- als behavior during the biomass thermal treatment. When organic matter is consumed during any heat treatment, heavy metals are exposed to a hot and oxygen-depleted atmosphere, adjacent to the particle, presenting one of the following behaviors [9]: 1) They vaporize directly in the initial chemical spe- cies; 2) They react with a compound present in the atmos- phere and then, they vaporize; 3) They remain unreacted in the mineral matrix. The vaporized species enter in the gas flow where they react or condense. The condensed species form new par- ticles (homogeneous nucleatio n) or they are deposited on the present particles surfaces (heterogeneous deposition ). Homogeneous nucleation gas explains the substantial amount of very fine metal particles (diameter between 0.02 to 1 microns) found in the effluent gases. The het- erogeneous deposition occurs in larger particles and they Copyright © 2013 SciRes. OJMetal  M. ECHEGARAY ET AL. 4 Table 2. Heavy metals concentrations in the ash. Ash Cd (mg/kg) Cr (mg/kg) Pb (mg/kg) Stalk 1.22 15.86 14.07 Marc 1.43 11.56 30.27 Peach pits 1.09 3.12 0.63 can be captured by the pollution control systems. To promote the heterogeneous deposition, it is necessary to limit the formation of fine metal particles. The species formation with lower oxidation states than the initial states are favored by the reducing conditions. Furthermore, these metals may react with other released elements, as chlorine or sulfur. These new species are generally more volatile than the metal species present in the agro-indus trial waste. The heavy metals vo latilization during the gasification depends of their speciation and the gasification atmosphere. Taking into account the heavy metals partition during gasification in fluidized bed reactor, the turbulence con- ditions during its operation cause a significant production of fly ash with high concentrations of these elements. The heavy metals partition during heat treatments in flu- idized bed is governed by the fluid dynamics, the kinetics of heavy metals diffusion in the ash particles and reaction kinetics between the heavy metals and the ash compo- nents [10]. The chemical composition of the mineral matrix has a great influence on the kinetics of heavy metals vaporiza- tion; it determines the bonding strength between the mineral matrix and these elements, as well as the time required for diffusion out of the particle. Thus, basic sp e- cies in the matrix (SiO2, Al2O3, CaO) can react with these metals encapsulating them in the particle center [8]. The CdO (s), Cr2O3 (s) and PbO (s) may react with HCl, ac- cording to the following reaction: 22 MeOs2HClgMeClgH Og (1) The used steam during the gasification phenomenon affects the reaction equilibrium and the heavy metals retention as oxides in produced solid waste [23]. A high water content of feed waste promotes this retention. Then, during the waste gasification using steam, the reaction is displaced to the left causing the formation of metal ox- ides. Notably, Moj tahedi and Salo [24] observ ed the pre- sence of heavy metal chlorides in volatile p hase when th e gasification was carried out at high temperature. According to Park et al. [25], MeO can react with the syngas according to the Reactions (2) and (3): 22 MeOs HgMes HOg (2) 2 MeOs COgMeCs Og (3) when these reactions occur, the heavy metal gradually These reactions can be inhibited by the addition of natu- ral zeolite. Vervaeke diffuses to the particle surface subsequently vaporized. et al. [26] observed the augmentation of Cd an heavy metals which remains in the ash- de the bed m er hand, some metals such as Ca inhibit the be hat most of the heavy metals ar d bi a fluidized bed re ert and no in shows the principal conditions of lixiviation te ese results, the three metals w lts, all metals had the highest mobility in the case of the peach pits ash. d Pb concentrations in the fly ash comparing with bot- tom ash, during wood gasification in fixed bed; however, Cr remained in the bottom ash. Pinto et al. [22] detected higher Pb concentrations in the ash captured by cyclones comparing with bottom ash, confirming the above men- tioned studies. The amount of creases when the working temperature is high, close to 900˚C, and the synthesis gas quality increases. According to Wei et al. [27], the sand, used as aterial, adsorbs heavy metals, decreasing their concen- trations in exit flow gas. The heavy metals release in- creases when the adsorption efficiency of this material decreases. On the oth d material agglomeration, maintaining the fluidization quality and sand mixed with the biomass to be gasified. Then, Ca improves the fluidization delaying the heavy metals release [28,29]. Cui et al. [30] observed t e enriched in the exit gas flow. The experimental re- sults are consistent with these observations. Approxi- mately 70% of trace elements found in the synthesis gas, including three studied heavy metals, come from the gas- ified biomass and about 25% from gasification system. Considering the alkali elements contents in the studie omass, the Ca concentrations vary in a small range, except for the peach pits. For K, the found concentration in the stalk is very high compared to these concentrations in the marc and peach pits. The Mg concentrations in all analyzed residues vary in a small range. If the gasification is carried out into actor, it is important to consider the biomass tendency to separate from the bed due to its low density, as well as the elutriation tendency of C small particle. On the other hand, the gasification ash are in volved in chemical equilibrium of the gasification reac- tions but, it may have a catalytic effect, accelerating the char gasification reaction with steam, especially when the ash contains metal oxides as K2O, CaO, MgO, P2O5, etc. [31]. Table 3 sts and relative ratio of the studied heavy metals found in the leaching solution. Taking into account th ere detected in leaching tests DIN-DEVS4, except to peach pits ash. Cd had higher mobility in leaching test DIN-DEVS4 for the marc ash. The Pb and Cr showed the highest mobility for the peach pits ash. Considering the EPA-TLCP test resu Copyright © 2013 SciRes. OJMetal 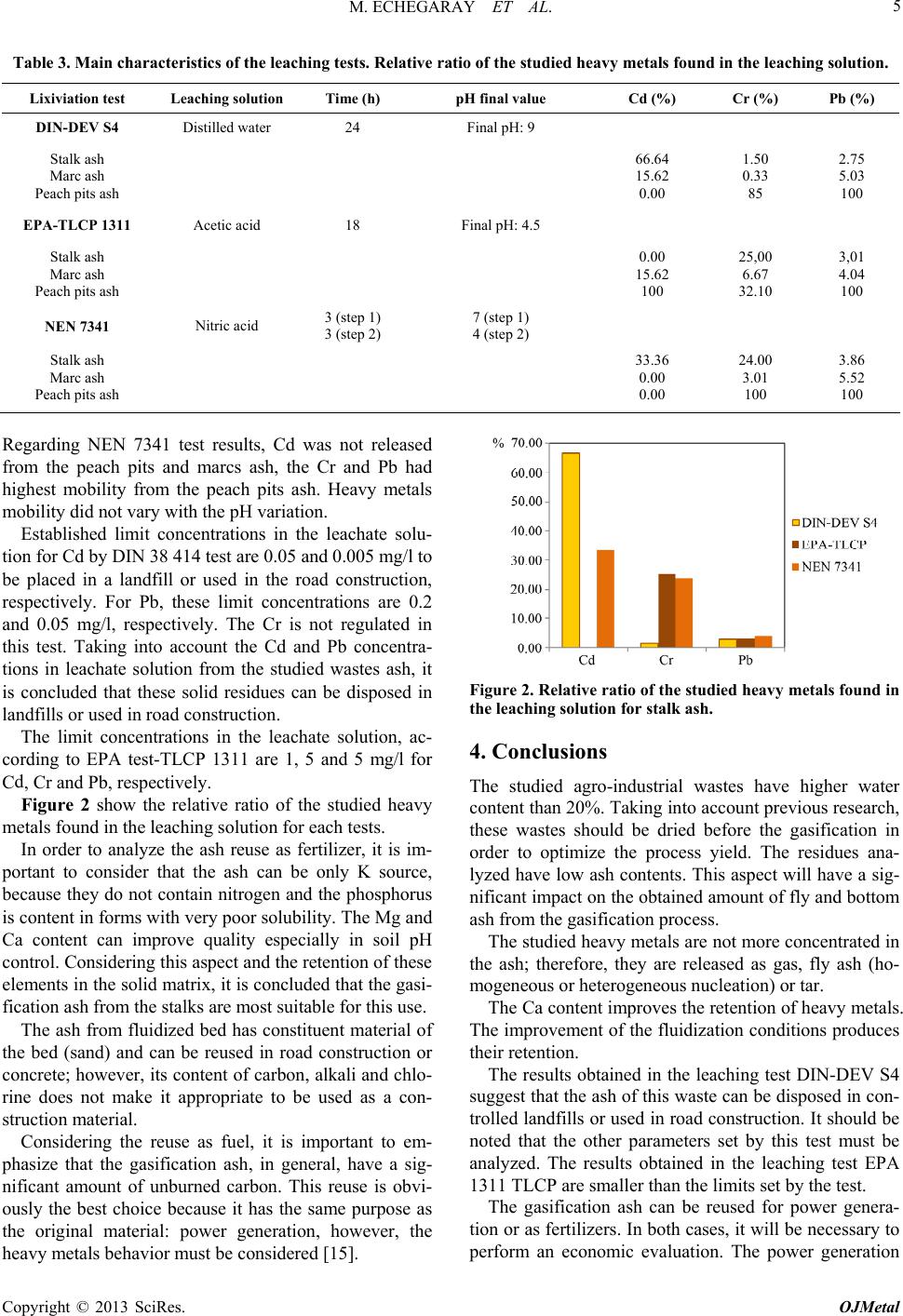 M. ECHEGARAY ET AL. Copyright © 2013 SciRes. OJMetal 5 Tatio of the studied heavy metals found in the leaching solution. able 3. Main characteristics of the leac hing tests. Relative r Lixiviation test Leaching solution Time (h) pH final value Cd (%) Cr (%) Pb (%) DIN-DEV S4 Distilled water 24 Final pH: 9 Stalk ash Marc ash 66.64 1.50 2.75 Peach pits ash 15.62 0.00 0. 33 85 5. 03 100 Acetic acid 18 Final pH: 4.5 E PA-TLCP 1311 Stalk ash Marc ash 0. 0025,00 3, 01 Peach pits ash 15.62 100 6. 67 32.10 4. 04 100 NEN 7341 Nitric acid 3 (s tep 1)7 (s tep 1) 3 (step 2) 4 (step 2) Stalk ash Marc ash 33.36 24.00 3. 86 Peach pits ash 0. 00 0.00 3. 01 100 5. 52 100 Reg 7341 test results, Cd was not released rom the peach pits and marcs ash, the Cr and Pb had 005 mg/l to be , 5 and 5 mg/l for C lution for each tests. urce, be co sification ash, in general, have a sig- ni arding NEN f highest mobility from the peach pits ash. Heavy metals mobility did not vary with the pH variation. Established limit concentrations in the leachate solu- tion for Cd by DIN 38 414 test are 0.05 and 0. placed in a landfill or used in the road construction, respectively. For Pb, these limit concentrations are 0.2 and 0.05 mg/l, respectively. The Cr is not regulated in this test. Taking into account the Cd and Pb concentra- tions in leachate solution from the studied wastes ash, it is concluded that these solid residues can be disposed in landfills or used in road construction. The limit concentrations in the leachate solution, ac- cording to EPA test-TLCP 1311 are 1 Figure 2. Relative ratio of the studied heavy metals found in the leaching solution for stalk ash. 4. Conclusions ount previous research, be dried before the gasification in he process yield. The residues ana- ention of heavy metals. Th DEV S4 su r parameters set by this test must be an d, Cr and Pb, respectively. Figure 2 show the relative ratio of the studied heavy metals found in the leaching so The studied agro-industrial wastes have higher water content than 20%. Taking into acc these wastes should order to optimize t In order to analyze the ash reuse as fertilizer, it is im- portant to consider that the ash can be only K solyzed have low ash contents. This aspect will have a sig- nificant impact on the obtained amount of fly and bottom ash from the gasification process. The studied heavy metals are not more concentrated in the ash; therefore, they are released as gas, fly ash (ho- mogeneous or heter ogeneous nucleation) or ta r. The Ca content improves the ret cause they do not contain nitrogen and the phosphorus is content in forms with very poor solubility. The Mg and Ca content can improve quality especially in soil pH control. Considering this aspect and the retention of these elements in the solid matrix, it is concluded that the gasi- fication ash from the stalks are most suitable for this use. The ash from fluidized bed has constituent material of the bed (sand) and can be reused in road construction or e improvement of the fluidization conditions produces their retention. The results obtained in the leaching test DIN- ncrete; however, its con tent of carbon, alkali and chlo - rine does not make it appropriate to be used as a con- struction material. Considering the reuse as fuel, it is important to em- phasize that the ga ggest that the ash of this waste can be disposed in con- trolled landfills or used in road construction. It should be noted that the othe alyzed. The results obtained in the leaching test EPA 1311 TLCP are smaller than the limits set by the test. The gasification ash can be reused for power genera- tion or as fertilizers. In both cases, it will be necessary to perform an economic evaluation. The power generation ficant amount of unburned carbon. This reuse is obvi- ously the best choice because it has the same purpose as the original material: power generation, however, the heavy metals behavior must be considered [15].  M. ECHEGARAY ET AL. 6 reuse is most appropriate because it increases the gasifi- ca eida, A. Bauen, F. Costa, J. Erics- son and J. Giegrich, “Total Costs and Benefits of Biomass in Selected Reg,” Energy 25, No. 11, 20 tion plant efficiency, but the heavy metals content in the ashes must be considered in order to minimize the environmental impact. REFERENCES [1] H. Groscurth, A. Alm ions of the European Union 00, pp. 1081-1095. , Vol. doi:10.1016/S0360-5442(00)00016-5 [2] A. Bridgwater, “The Technical and Economic-Feasibility of Biomass Gasificationfor Power-Generation,” Fuel, Vol. 74, No. 5, 1995, pp. 631-653. doi:10.1016/0016-2361(95)00001-L [3] M. Baratieri, P. Baggio, L. Fiori and M. Grigiante, “Bio- mass as an Energy Source: Thermodynamic Constraints on the Performance of the Conversion Process,” Biore- source Technology, Vol. 99, No. 15, 2008, pp. 7063-7073. doi:10.1016/j.biortech.2008.01.006 [4] C. Franco, F. Pinto, I. Guly urtlu and I. Cabrita, “The St udy of Reactions Influencing the Biomass Steam Gasification Process,” Fuel, Vol. 82, No. 7, 2003, pp. 835-842. doi:10.1016/S0016-2361(02)00313-7 [5] D. Ross, R. Noda, M. Horio, A. Kosminski, P. Ashman and P. Mullinger, “Axial Gas Profiles in a Bubbling Flu- idized Bed Biomass Gasifier,” Fuel, Vol. 86, No. 10-11, 2007, pp. 1417-1429. doi:10.1016/j.fuel.2006.11.028 [6] G. Taralas and M. Kontominas, “Pyrolysis of Solid Resi- dues Commencing from the Olive Oil Food Industry for Potential Hydrogen Production,” Journal of Analytical and Applied Pyrolysis, Vol. 76, No. 1-2, 2006, pp. 109- 116. doi:10.1016/j.jaap.2005.08.004 [7] P. Foscolo, A. Germanà, N. Jand and S. Rapagnà, “De- sign and Cold Model Testing of a Biomass Gasifier Con- sisting of Two Interconnected Fluidized Beds,” Powder Technology, Vol. 173, No. 3, 2007, pp. 179-188. doi:10.1016/j.powtec.2007.01.008 [8] R. Rodriguez and S. Udaquiola, “Gasificación Térmica de Residuos de la Agroindustria, San Juan,” Proceedings of 2th Congreso Iberoamericano, Hidrógeno y Fuentes Sus- tentables de Energía, Hyfusen, 2009, pp. 110-115. [9] S. Abadanes, “Comportement des Metauxlourdsdans les Procédéséchetsménagers,” Thèse du Doctorat, Université de Perpignan, 2001. [10] J. Toledo, J. Corella and L. Corella, “The Partitioning of Heavy Metals in Incineration of Sludges and Waste in a Bubbling Fluidized Bed. 2. Interpretation of Results with a Conceptual Model,” Journal of Hazardous Materials, Vol. 126, No. 1-3, 2005, pp. 158-168. doi:10.1016/j.jhazmat.2005.06.021 [11] E. Kurkela, M. Kurkela and A. Moilanen, “Fluidised-Bed Gasification of High-Alkali Biomass Fuels,” Proceedings of Science in Thermal and Chemical Biomass Conversion, 2006, pp. 662-676. [12] V. Skoulou, G. Koufodimos, Z. Samaras and A. Zaba- niotou, “Low Temperature Gasification of Olive Kernels in a 5-kW Fluidized Bed Reactor for H2-Rich Producer Gas,” International Journal of Hydrogen Energy, Vol. 33, No. 22, 2008, pp. 6515-6524. doi:10.1016/j.ijhydene.2008.07.074 [13] R. Rodriguez, M. Echegaray, R. Y. Castro and S. Uda- quiola, “Distribución Química de Plomo, Cromo y Cad- mio en lodos Cloacales y sus Cenizas,” Revista Acadé- mica de la Facultad de Ingen Autónoma de Yucatán, Vol. 11, No. 2 iería de la Universidad , 2007, pp. 31-38. rocedure for Water, Procedure. on of the Leaching Behavior of lmar, “Approach towards International Standariza- struction Ma- [14] R. Rodriguez, C. Palacios, S. Udaquiola, G. Flamant, O. Martínez and G. Mazza, “Estudio de la Vaporización de Elementos Traza Durante la Combustión de Barros Cloa- cales,” Rev. Facultad De Ingeniería -Universidad De Antioquia, Vol. 55, 2010, pp. 64-73. [15] J. Pels, D. De Nie and J. Kiel, “Utilization of Ashes from Biomass Combustion and Gasification,” Proceedings of the 14th European Biomass Conference & Exhibition, Bioenergy NoE Partner Publications, 2009. [16] DIN 38414-S4, “German Standard P Wastewater and Sediment Testing (Group S),” Determi- nation of Leachability by Water, Institutfür Normung, Berlín, Alemania, 1984. [17] EPA TCLP, “Toxicity Characteristic Leaching Method 1311. Test Methods for Evaluating Solid Waste,” US Environmental Protection Agency, Washington DC, 1992. [18] NEN 7341, “Determinati Granular Materials: Availability Test,” Netherlands Nor- malization Institute, Delft, Holanda, 1993. [19] H. Van Der Sloot, D. Kosson, T. Eighmy, R. Comans and O. Hje tion: A Concise Scheme for Testing of Granular Waste Leachability,” Proceeding of the International Confer- ence on Environmental Implications of Con terials and Technology Developments, Environmental As- pects of Construction with Waste Materials, Elsevier Sci- ence, 1994, pp. 453-466. doi:10.1016/S0166-1116(08)71478-X [20] L. Xie, T. Li, J. Gao, X. Fei, X. Wu and Y. Jiang, “Effe ct of Moisture Content in Sewage Sludge on Air Gasifica- tion,” Journal of Fuel Chemistry and Technology, Vol. 38, No. 5, 2010, pp. 615-620. doi:10.1016/S1872-5813(10)60048-5 [21] C. Pfeifer, S. Koppatz and H. Hofbauer, “Steam Gasifica- tion of Various Feedstocks at a Dual Fluidised Bed Gasi- fier: Impacts of Operation Conditions and Bed Materials,” Biomass Conversion and Bio pp. 39-53. refinery, Vol. 1, No. 1, 2011, 07-1doi:10.1007/s13399-011-00 0-1062. [22] F. Pinto, H. Lopes, R. Andre, I. Gulyurtlu and I. Cabrita, “Effect of Catalysts in the Quality of Syngas and By- Products Obtained by Co-Gasification of Coal and Wastes. 2: Heavy Metals, Sulphur and Halogen Compounds Abatement,” Fuel, Vol. 87, No. 7, 2008, pp. 105 doi:10.1016/j.fuel.2007.06.014 [23] R. Rodriguez, S. Acosta, A. Saffe and S. Udaquiola, “Predicción de la Partición de Cd, Cr y Pb Durante la Gasificación de Residuos Agroindustriales,” Proceedings of the Congreso Internacional de Ciencia y Tecnología Ambiental, Asociación Argentina para el Progreso de las Copyright © 2013 SciRes. OJMetal  M. ECHEGARAY ET AL. Copyright © 2013 SciRes. OJMetal 7 Ciencias, Mar del Plata, Buenos Aires, 2012, pp. 422- 427. [24] W. Mojtahedi and K. Salo, “Fate of Alkali and Trace Metals in Biomass Gasification,” Biomass and Bionergy, Vol. 15, No. 3, 1998, pp. 263-267. doi:10.1016/S0961-9534(98)00019-1 [25] K. Park, J. Hyun, S. Maken, S. Jang and J. Park, “Vitrifi- cation of Municipal Solid Waste Incinerator Fly Ash Us- ing Brown’s Gas,” Energy Fuels, Vol. 19, No. 1, 2005, pp. 258-262. doi:10.1021/ef049953z [26] P. Vervaeke, F. Tack, F. Navez, J. Martin, M. Verboo and N. Lust, “Fate of Heavy Metals during Fixed Bed Down- draft Gasification of Willow Wood Harvested from Con- taminated Sites,” Biomass Bioenergy, Vol. 30, No. 1, 2006, pp. 58-65. doi:10.1016/j.biombioe.2005.07.001 ls [27] X. Wei, U. Schnell and K. Hein, “Behaviour of Gaseous Chlorine and Alkali Metals during Biomass Thermal Utilization,” Fuel, Vol. 84, No. 7-8, 2005, pp. 841-848. [28] C. Lin, H. Kuo, M. Wey, S. Chang and K. Wang, “Inhibi- tion and Promotion: The Effect of Earth Alkali Meta and Operation Temperature on Particle Agglomeration/ Defluidization during Incineration in Fluidized Bed,” Powder Technology, Vol. 189, No. 1, 2009, pp. 57-63. doi:10.1016/j.powtec.2008.06.003 [29] C. Lin, M. Tsai and C. Chang, “The Effects of Agglom eration/Defluidization on Emission - of Heavy Metals for Various Fluidized Parameters in Fluidized-Bed Incinera- tion,” Fuel Processing Technology, Vol. 91, No. 1, 2010, pp. 52-61. doi:10.1016/j.fuproc.2009.08.012 [30] H. Cui, S. Turn, V. Keffer, D. Evans, T. Tran and M. Foley, “Study on the Fate of Metal Elements from Bio- mass in a Bench-Scale Fluidized Bed Gasifier,” Fuel, Vol. 108, 2013, pp. 1-12. doi:10.1016/j.fuel.2011.07.029 [31] J. Song, Y. Sung, T. Yu, Y. Choi and U. Lee, “Optimiza- tion of Biomass Gasification for F-T Bio-Diesel Synthe- sis,” Proceedings of the 20th International Conference on Fluidized Bed Combustion, Part 5, 2010, pp. 633-635.
|