Paper Menu >>
Journal Menu >>
 Materials Sciences and Applicatio ns, 2010, 1, 329-335 doi:10.4236/msa.2010.16048 Published Online December 2010 (http://www.scirp.org/journal/msa) Copyright © 2010 SciRes. MSA Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size High Purity Gold and Commercial Grade Zinc Grains* P. Sita Rama Rao, P. B. V. Prasad, P. B. Shashikanth S. R. Research Laboratory for Studies in Crystallization Phenomena, Khammam, India. E-mail: parchabs@gmail.com Received July 13th, 2010; revised October 28th, 2010; accepted November 26th, 2010. ABSTRACT The VTLLS microscopy studies were made on high purity gold and commercial grade zinc grains in a temperature range of 30-230˚C. Differential area ω and surface activity Sa were estimated from photomicrographs. The ω vs dT/dt (rate of heating) curve was seen to differ from those of silver and titanium. The nature of curve between normalized ω and dT/dt was seen to be non-exponential. The characteristic relation between sectorized differential area ωsec and mean temperature was examined. The present study further establishes the simplicity and versatility of the VTLLS tech- nique, in studying the defect-sub-structure of metal particles such as Au and Zn in presence of an imposed temperature gradient in a reasonable way. As such an attempt was made to connect the ω and defect-sub-structure related parame- ters. Keywords: Variable Temperature, Laser Light Scattering Microscopy, Au and Zn Grains, Defects 1. Introduction Studies on defects in materials and metals have been a fascinating subject and many techniques were developed to support such investigations. Triftschauser and Kogel [1] developed a positron annihilation technique to study defect structure in metals in the vicinity of surfaces. Recknagel and Wichert [2] reported a perturbed angular correlation study on point defects in metals. NMR stud- ies on point defects in Al and Cu were reported by Minier and Minier [3]. Mossbauer studies of defectimpu- rity interactions in metals were carried out by Vogl et al [4]. Peisl [5] made X-ray scattering studies on defects in metals. Earlier, the present authors [6] made variable temperature laser light scattering microscopy (VTLLSM) studies on relatively larger grains of aluminum, copper and silver in a small temperature interval and established the effectiveness of the technique. The technique was similar to the variable oblique incidence reflection mi- croscopy (OIRM) reported by the present authors [7]. Later on, the approaches were modified and the VTLLSM was used to investigate structural variations of high purity silver [8], titanium [8], gold [9] and (com- mercial grade) Pb-Sn alloy [9] grains having sub-milli- meter dimensions (within the range of 10-100 µm) as a function of the temperature (in a wider temperature ran- ge). The new technique consists of the Fourier space interference images (resulted from light scattered by par- ticles), which is the intermediate step in the formation of a holographic image. Such an image is composed of black and white contrast regions, which were attributed to surface micro-facets of the particles. The present au- thors used these contrasts and their variations with tem- perature as the signature of particle’s structural changes in terms of internal defects such as voids and disloca- tions. A large working distance (LWDO) optical microscope with water cooled heat shield [10,11], a zero thermal expansion (in upward direction) table (V+ZET) to func- tion as sample holder [12] and a beam path cooler (BPC) to stabilize image formed by scattered laser light at high temperature [13] were developed for the VTLLSM stud- *The study is part of a project funded by Defense Research and Devel- opment Organization, Ministry of Defense, Government of India.  Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size 330 High Purity Gold and Commercial Grade Zinc Grains ies. Gold and zinc are too well known; a complete intro- duction on them is therefore not attempted. Briefly, Doremus [14] studied optical adsorption by gold parti- cles. Granqvist and Hunderi [15] studied optical proper- ties of ultra fine gold particles. Hassam et al. reported [16,17] equilibrium phase diagram of some mixed sys- tems, that contained gold as a component. Investigations were made on 10-100 μm size gold and zinc grains, by employing variable temperature laser light scattering microscopy (VTLLSM). Results are pre- sented in this report. 2. Experimental As received Sigma-Aldrich (USA) made agglomerated (+ 99.999% pure) 10-100 μm size gold grains and com- mercial grade purity zinc grains (prepared one hour be- fore the commencement of each experiment) were em- ployed in the present study. The experimental procedure and data analysis were almost identical to those reported earlier [8,9,18]. A 10 mW, 670 nm laser, a zero thermal expansion hot stage (V+ZET) coupled with a beam path cooler (BPC), were employed [12,13]. Thermocouple based thermometry was used to measure the tempera- tures. Fused silica optical flat was used as sample holder; linear thermal expansion coefficients [19,20] of fused silica, gold and zinc are 0.4 × 10-6 k-1, 14 × 10-6 k-1 and 11.6 × 10-6 k -1 (┴C axis) and 64.20 × 10-6 k -1 (║C axis) respectively. In order to suppress the back ground scat- tering, amorphous MnO2 was employed in the form of thin layer. Reflection coefficients [21] of carbon black, gold and zinc are 0.003, 0.75 and 0.45 respectively in the visible spectrum. The coefficient of reflection of MnO2 may be equal or very close to that of carbon black. The metal grains were heated on V+ZET and a series of pho- tomicrographs were recorded at different temperatures. Measurements were made from such photographs. The same fused silica optical flat has been used in the studies on Ag, Ti, Pb-Sn, Al, Zn and Au [8,9,18], in order to eliminate the influence of substrate, if any, while com- paring the results on grains of different metals. 3. Results The VTLLS microscopy images of Au grains at three different temperatures are shown in Figure 1. The im- ages of zinc grains are similar to that of gold grains. The areas of bright patches (BPs [13]) were measured from a given photomicrograph and summed up, giving total area (ATOT). Differential area ω, defined as 2(Amax – Amin)/ (Amax + Amin), where Amax and Amin were maximum ATOT and minimum ATOT respectively in a heating-run, were estimated. The ω values, obtained in five heating-runs vs rate of heating (dT/dt) are shown in Figure 2. The curve ω vs dT/dt of Au and Zn (Figures 2(a,b)) shows an ini- tial increase and then a constant decrease in the average value of ω of two grains, as the rate of heating was pro- gressively increased. The relation between normalized differential area ω/aI vs dT/dt of Au is shown in Figure 3(a). The curve joining most of the points (dashed line; Figure 3(a)) has negative slope (similar to the curve in Figure 2(a), between dT/dt = 0.26 and 0.66). () 1 log tandT dt ωϕ − = (1) may describe the straight line (Figure 3(a)). It is a devia- tion from earlier observations, that exponential curves (connecting ω and dT/dt) with positive and negative slopes were noticed in case of grains of silver [8] and tita- nium [8]. In order to handle the experimental data, it may therefore be useful to use a general equation, such as, ( ) 1 1x YKeKmx φ ± =−+ ± (2) where Y ω = , x dT dt = and φ is a constant. K is a parameter that probably defines the state of a grain and can assume a value 0 or 1. It is assumed that the value of (a) (b) (c) Figure 1. VTLLS microscopy images of Au grains. (a) 29˚C, (b) 130˚C, (c) 230˚C; dT/dt = 0.46˚C/min. 500X. Copyright © 2010 SciRes. MSA  Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size 331 High Purity Gold and Commercial Grade Zinc Grains Figure 2. Differential area ω vs. dT/dt; (a) Case of Au and (b) Case of Zn. Figure 3(a). Normalized ω (curve a) and normalized Sa (curve b) of Au grains. In the graph: p × logX = 10-3 × [log(Sa/aI)] and q × logY = 10-5 × [log(ω/aI)]. Figure 3(b). Normalized Sa (curve a) and normalized ω (curve b) of Zn grains. In the graph: p × logX = 10-4 × [log(ω/aI)] and q × logY = 10-3 [log(Sa/aI)]. The profile of distribution of bright patches (BPs) was noticed to change with temperature [12]. A term surface activity Sa was defined, while taking the decompositions and recombinations of BPs into consideration. The sur- face activity Sa was proposed to be given by an empirical relation ()()( 21 11 aHBPs LBPsoTBPs SN NNN −− ⎡⎤ =− −− ⎣⎦ ) 1 () 1 11 mh TT − ⎡⎤ −+ − ⎣⎦ (3) where NHBPs and NLBPs are highest and lowest number of BPs respectively, found in photomicrographs of a heat- ing-run; No and NTBPs are total number of (photomicro- graph) frames and total number of bright patches respec- tively. Tm and Th are melting temperature (of the metal) and highest temperature reached (in a given heating-run) respectively and expressed in ˚C. It may be stated that Sa indicates the level of internal and external activity promoted by thermal pressure. It is known that atomic migration takes place at a much faster Copyright © 2010 SciRes. MSA 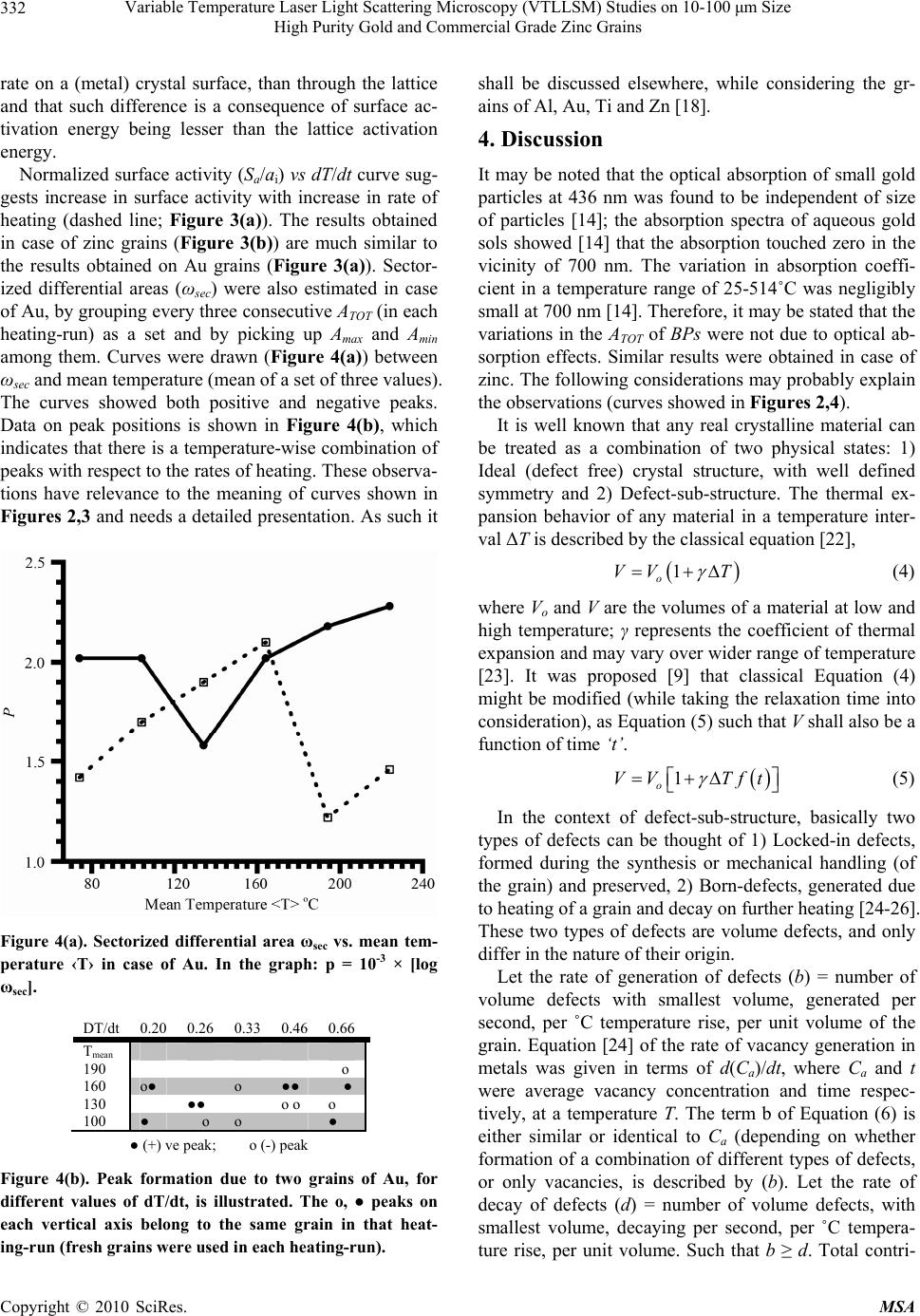 Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size 332 High Purity Gold and Commercial Grade Zinc Grains rate on a (metal) crystal surface, than through the lattice and that such difference is a consequence of surface ac- tivation energy being lesser than the lattice activation energy. Normalized surface activity (Sa/ai) vs dT/dt curve sug- gests increase in surface activity with increase in rate of heating (dashed line; Figure 3(a)). The results obtained in case of zinc grains (Figure 3(b)) are much similar to the results obtained on Au grains (Figure 3(a)). Sector- ized differential areas (ωsec) were also estimated in case of Au, by grouping every three consecutive ATOT (in each heating-run) as a set and by picking up Amax and A min among them. Curves were drawn (Figure 4(a)) between ωsec and mean temperature (mean of a set of three values). The curves showed both positive and negative peaks. Data on peak positions is shown in Figure 4(b), which indicates that there is a temperature-wise combination of peaks with respect to the rates of heating. These observa- tions have relevance to the meaning of curves shown in Figures 2,3 and needs a detailed presentation. As such it Figure 4(a). Sectorized differential area ωsec vs. mean tem- perature ‹T› in case of Au. In the graph: p = 10-3 × [log ωsec]. DT/dt 0.20 0.26 0.33 0.46 0.66 Tmean 190 o 160 o● o ●● ● 130 ●● o o o 100 ● o o ● ● (+) ve peak; o (-) peak Figure 4(b). Peak formation due to two grains of Au, for different values of dT/dt, is illustrated. The o, ● peaks on each vertical axis belong to the same grain in that heat- ing-run (fresh grains were used in each heating-run). shall be discussed elsewhere, while considering the gr- ains of Al, Au, Ti and Zn [18]. 4. Discussion It may be noted that the optical absorption of small gold particles at 436 nm was found to be independent of size of particles [14]; the absorption spectra of aqueous gold sols showed [14] that the absorption touched zero in the vicinity of 700 nm. The variation in absorption coeffi- cient in a temperature range of 25-514˚C was negligibly small at 700 nm [14]. Therefore, it may be stated that the variations in the ATOT of BPs were not due to optical ab- sorption effects. Similar results were obtained in case of zinc. The following considerations may probably explain the observations (curves showed in Figures 2,4). It is well known that any real crystalline material can be treated as a combination of two physical states: 1) Ideal (defect free) crystal structure, with well defined symmetry and 2) Defect-sub-structure. The thermal ex- pansion behavior of any material in a temperature inter- val ΔT is described by the classical equation [22], ( ) 1 o VV T γ = +Δ (4) where Vo and V are the volumes of a material at low and high temperature; γ represents the coefficient of thermal expansion and may vary over wider range of temperature [23]. It was proposed [9] that classical Equation (4) might be modified (while taking the relaxation time into consideration), as Equation (5) such that V shall also be a function of time ‘t’. () 1 o VV Tft γ =+Δ ⎡ ⎤ ⎣ ⎦ (5) In the context of defect-sub-structure, basically two types of defects can be thought of 1) Locked-in defects, formed during the synthesis or mechanical handling (of the grain) and preserved, 2) Born-defects, generated due to heating of a grain and decay on further heating [24-26]. These two types of defects are volume defects, and only differ in the nature of their origin. Let the rate of generation of defects (b) = number of volume defects with smallest volume, generated per second, per ˚C temperature rise, per unit volume of the grain. Equation [24] of the rate of vacancy generation in metals was given in terms of d(Ca)/dt, where Ca and t were average vacancy concentration and time respec- tively, at a temperature T. The term b of Equation (6) is either similar or identical to Ca (depending on whether formation of a combination of different types of defects, or only vacancies, is described by (b). Let the rate of decay of defects (d) = number of volume defects, with smallest volume, decaying per second, per ˚C tempera- ture rise, per unit volume. Such that b ≥ d. Total contri- Copyright © 2010 SciRes. MSA  Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size 333 High Purity Gold and Commercial Grade Zinc Grains T ) bution to the volume (of defect free metal grain) due to birth and decay of defects in a temperature interval (t – to = ΔT) is, () () 1 o VVbd=−Δ (6) It is assumed logically that , where rm is rate of migration of defects to surface, r ( ,, mr dfrrT= r ) } ⎤ ⎦ is the rate of recombination, and T is temperature. Let the concentra- tion of locked-in defects (number of defects per unit volume) = Г. Based on the fact that the physical proper- ties of metals vary, depending on the nature of prepara- tion [27], it may be stated that the value of Г depends on the process of synthesis of grains. Decay of locked-in defects due to rise of temperature may be represented by a decay factor D (= number of defects decaying per unit volume per ˚C rise of temperature). Therefore, total change in excess initial volume due to decay of locked-in defects is given by, () ( 2 o VV DT=Γ−Δ (7) The real volume of grain is given by the sum of Equtions (5-7) and may be written as: ()()( ) { 1 real o VV TftbdD γ =+Γ+Δ+−−⎡ ⎣ (8) On differentiating Equation (8) with respect to time and equating d(Vreal)/dt to zero (since expansion reaches maximum after a certain length of time) it may be writ- ten after simplification, () [] { 12 1 real oder dVVdT dtTF − =+Γ+ Δ } (9) where derivative factor ( ) ( ) der F dft bdDd t γ =+−− ⎡ ⎣⎤ ⎦ ) . It was shown [6] that the differential area ω, defined as ()( max minmax min 2AA AA−+, could give thermal volume-strain variation ( )( ) max minmaxmin 2VV VV ω =− +, where Vmax and Vmin were maximum and minimum total volumes respectively, exhibited by a grain, during a heat-run. It may therefore be written that maxreal dV V = min and V− () maxmin 2 o VV V≈+ , then Equation (9) may be written as () () [] { 12 25 der dT dtTF ω − =Γ+Δ } (10) By using Equation (10), values of Fder were calculated for a few assumed values of Г in case of the grains of zinc; the results are shown in Figure 5. Expectedly, Fder exhibits large variations, for relatively larger concentra- tion of locked-in defects. It may therefore be expected that, in case of small metal grains, variations in the volume with systematic changes in temperature, can be strongly influenced [28,29] by the crystal-defects related parameters. It may be Figure 5. Variation of Fder with dT/dt, for different values of Г; case of Zn. pointed out that, 1) Since no two metal grains (formed under identical conditions) can have identical de- fect-sub-structure, they exhibit non-identical response to the injected heat energy. (It may be recalled that non-uniform behavior is a frequent occurrence in crystal- lization [30-32], when individual cases are considered); and, 2) Yet, the defect-sub-structure parameters may vary with in an upper and lower limits, permitted by the parameters, controlling the nucleation and growth of grains. These two factors coupled with the macroscopic physical properties of a given material, lead to a sort of uniformity in diversity-like condition. Individual metal grains, studied by VTLLS microscopy, appear to have anarchic behavior. But, when the behavior of a good number of grains is considered, a systemization surpris- ingly surfaces. Such diversity in the behavior should be- come more dominant, with further reduction in the size of metal grains. It can be pointed out that the observed behavior of the grains is their intrinsic character and not due to any secondary influences. Because, unlike the conventional techniques, such as electron microscopy and X-ray techniques (where the probing beam can in- teract with the material and modify its state [33]), VTLLS microscopy is powerful, yet passive. In the sense that it does not modify the state of metal (or non-metal) grain, if the beam energy is not too high (as in the pre- Copyright © 2010 SciRes. MSA  Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size 334 High Purity Gold and Commercial Grade Zinc Grains sent case). The VTLLSM technique is most suited for studies on surfaces and small grains. 5. Conclusions The VTLLS microscopy technique can be effectively employed to study the response of small metal grains (in 10-100 μm size range) to an imposed temperature gradi- ent and the results can be interpreted in a reasonable way, by taking the defect-sub-structure related parameters into consideration. 6. Acknowledgements PBSK and PSR are grateful to Prof. Prasad (deceased) who lead the project. REFERENCES [1] W. Triftshauser and G. Kogel, In: J. I. Takamura, M. Doyama and M. Kiritani, Eds., Point Defects and Defect Interactions in Metals, University of Tokyo Press, North- Holland Publishing Co., Amsterdam, 1982, p. 15. [2] E. Recknagel and Th. Wichert, “Defects in Metals Studied by Implanted Radioactve Atoms,” Nuclear Instruments and Methods, Vol. 182, No. 1, 1981, pp. 439- 455. [3] M. Minier and C. Minier, “Screeing Charge Density around Several ΔZ = –1 Impurities in Copper: Nickel, Palladium, Platinum, and Vacancy,” Physical Review B, Vol. 22, No. 1, 1980, pp. 21-27. [4] G. Vogl, W. Petry, K. Sassa and S. Mantl, In: J. I. Takamura, M. Doyama and M. Kiritani, Eds., Point Defects and Defect Interactions in Metals, University of Tokyo Press, North-Holland Publishing Co., Amsterdam, 1982, p. 33. [5] H. Peisl, “Diffuse X-Ray Scattering from the Displa- cement Field of Point Defects and Defect Clusters,” Journal of Applied Crystallography, Vol. 8, No. 2, 1975, pp. 143-149. [6] P. B. V. Prasad, K. R. Gopal and Ch. V. Gopal, “Laser Light Scattering Studies on Small Metal Grains: Case of Al, Cu and Ag,” Proceedings of Solid State Physics (In- dia), Vol. 43, 2000, pp. 142-143. [7] P. B. Shashikanth, P. B. V. Prasad and G. S. Rao, “Oblique Incidence Reflection Microscopy (OIRM) on Hydrocarbon Films,” Crystal Research Technology, Vol. 34, No. 10, 1999, pp. 1287-1292. [8] P. B. V. Prasad and P. B. Shashikanth, “Variable Temperature Laser Light Scattering Microscopy Studies on High Purity 10-100 μm Ag and Ti Metal Particles,” Materials Science and Technology, DOI: 10.1179/ 026708310X12688283410361. [9] P. Sita Rama Rao, P. B. V. Prasad and P. B. Shashikanth, “On the Behaviour of Au and Pb-Sn Metal Grains Subjected to Heat: A Variable Temperature Laser Light Scattering Microscopy Study,” Materials Science: An Indian Journal, Vol. 5, No. 4, 2009, pp. 428-432. [10] P. B. V. Prasad and P. B. Shashikanth, “Design and Fab- rication of a 20 cm2 Table Having Zero Thermal Expan- sion in Positive Vertical Direction (V+ZET),” Proceed- ings of National Conference on Current Trends in Con- densed Matter Research, Warangal, 2004, p. 23. [11] P. B. V. Prasad and P. B. Shashikanth, “A Large Working Distance Microscope for High Temperature Studies,” Proceedings of National Conference on Perspectives in Engineering Optics & Spectroscopy, Meerut, 2004, p. 40. [12] P. B. V. Prasad and P. B. Shashikanth, “Laser Light Scattering Microscopy: Interpretation of Images,” Indian Journal of Engineering & Materials Science, Vol. 12, No. 6, 2005, pp. 591-594. [13] P. B. V. Prasad and P. B. Shashikanth, “On Imaging Metal Grains at High Temperature: Laser Light Scat- tering Microscopy,” Indian Journal of Engineering & Materials Science, Vol. 13, No. 2, 2006, pp. 162-166. [14] R. H. Doremus, “Optical Properties of Small Gold Parti- cles,” Journal of Chemical Physics, Vol. 40, No. 8, 1964, pp. 2389-2396. [15] C. G. Granqvist and O. Hunderi, “Optical Properties of Ultrafine Gold Particles,” Physical Review-B, Vol. 16, No. 8, 1977, pp. 3513-3534. [16] S. Hassam, M. Gambino and J. P. Bros, “The Ag+Au+Pb System: Determination of Liquidus Interface,” Thermo- chimica Acta, Vol. 257, 1995, pp. 83-92. [17] S. Hassam and Z. Bahari, “Equilibrium Phase Diagram of the Ag-Au-Pb Ternary System,” Journal of Alloys and Compounds, Vol. 392, No. 1-2, 2005, pp. 120-126. [18] P. B. Shashikanth and P. Sita Rama Rao, “Variable Temperature Laser Light Scattering Microscopy Studies on 10-100 μm Size Grains of Gold, Aluminum, Zinc and Titanium: Role of Relaxation Time in Thermally Triggered Volume Changes,” Materials Science an Indian Journal, Vol. 6, No. 1, 2010, pp. 68-70. [19] K. Koopman, In: K. Koopman, Ed., Informatie Boek Vwo-Havo Voor Het on Derwiji in De Natuurweten- schappen, Wolters-Noordhoff, Groningen, 1986, p. 89. [20] R. A. Walsh, “Machining and Metal Working Hand- book,” McGraw-Hill, Inc., New York, 1994. [21] R. C. Weast, “CRC Handbook of Chemistry and Phys- ics,” CRC Press, Florida, 1988. [22] D. Mckie and C. Mckie, “Crystalline Solids,” John Wiley & Sons Inc., New York, 1974. [23] V. E. Zinovev, “Handbook of Thermophysical Properties of Metals at High Temperatures,” Nova Science Pub- lishers, Inc., New York, 1996. [24] T. Kino and K. Ono, In: J. I. Takamura, M. Doyama and M. Kiritani, Eds., Point Defects and Defect Interactions in Metals, University of Tokyo Press, North-Holland Publishing Co., Amsterdam, 1982, p. 247. [25] M. Kiritani and H. Takaka, “Dynamic Studies of Defect Mobility Using High Voltage Electron Microscopy,” Jour- Copyright © 2010 SciRes. MSA  Variable Temperature Laser Light Scattering Microscopy (VTLLSM) Studies on 10-100 μm Size High Purity Gold and Commercial Grade Zinc Grains Copyright © 2010 SciRes. MSA 335 nal of Nuclear Materials, Vol. 69-70, 1978, pp. 277-309. [26] J. Takamura, In: J. I. Takamura, M. Doyama and M. Kiri- tani, Eds., Point Defects and Defect Interactions in Met- als, University of Tokyo Press, North-Holland Publishing Co., Amsterdam, 1982, p. 431. [27] E. Rose, “The Condensed Chemical Dictionary,” 7th Edition. Reinhold Publishing Co., New York, 1961. [28] J. P. Ganne and J. von Stebut, “Measurement of the intrinsic Thermal Expansion of Irradiation Defects in Aluminum at Low Temperatre,” Physical Review Letters, Vol. 43, No. 9, 1979, pp. 634-636. [29] R. W. Baluffi, “Vacancy Defect Mobilities and Binding Energies Obtained from Annealing Studies,” Journal of Nuclear Materials, Vol. 69-70, 1978, pp. 240-263. [30] A. R. Konak, “Single Versus Suspension Growth,” Jour- nal of Crystal Growth, Vol. 22, No. 1, 1974, pp. 67-68. [31] P. Bennema, “The Rate of Growth of Crystals from Slightly Supersaturated Solutions,” Ph.D. Thesis, Tech- nical University of Delft, Delft, 1965. [32] P. B. V. Prasad, “Dispersion in Crystal Growth Rates: Palmiticacid-Xylene System,” Journal of Crystal Growth, Vol. 102, No. 3, 1990, pp. 569-573. [33] M. Chen, E. Ma and K. Hemker, “Mechanical Behavior of Nanocrystalline Metals,” In: Y. Gogotsi, Ed., Nano- materials Handbook, Taylor & Francis, Boca Raton, 2006, pp. 407-529. |

