Paper Menu >>
Journal Menu >>
 Optics and Photonics Journal, 2013, 3, 229-232 http://dx.doi.org/10.4236/opj.2013.33037 Published Online July 2013 (http://www.scirp.org/journal/opj) Multispectral Imaging for Authenticity Identification and Quality Evaluation of Flos carthami* Cuiying Hu1, Qingxia Meng1#, Ji Ma2, Qichang Pang3, Jing Zhao4 1Department of Physics, Jinan University, Guangzhou, China 2College of Traditional Chinese Medicine, Southern Medical University, Guangzhou, China 3Department of Optoelec tronic Engineering, Jinan University, Guangzhou, China 4Department of Applied Physics, South China Agricultural University, Guangzhou, China Email: #hcyhome@163.com Received May 4, 2013; revised June 11, 2013; revised June 19, 2013 Copyright © 2013 Cuiying Hu et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT The identification and quality evaluation of Flos carthami were studied using tunable liquid spectral imaging instrument, to discuss the application range and advantages of spectral imaging technology in Chinese medicine identification and quality control field. The Flos carthami was indentified by extracting the normalized characteristic spectral curves of Flos carthami, Crocus sativus and Dendranthema morifo lium, which were standard samp les supplied by National Insti- tute for Drug Control. The qualities o f Flos carthamies collecting from different pharmacies were evaluated by extract- ing their normalized characteristic spectral curves. The imaging spectrum testing system was designed independently. The spectral resolution was 5 nm, and the spectral range was from 400 nm to 680 nm. The results showed that the nor- malized characteristic spectral curve of Flos carthami was significantly different from those of Crocus sativus’ and Dendranthema morifolium’s, and the fluorescence intensity of Flos carthami from different commercial sources were different. Spectral imaging technology could be used to identify and evaluate Flos carthami, and operation method was rapid, convenient and non-destructive. Keywords: Flos carthami; Spectral Imaging; Rapid Identification; Quality Evaluation 1. Introduction Flos carthami is the dried flower of Cartham u s tinctoriu s L. It is commonly used in traditional Chinese medicine. It contains a variety of ingredients such as flavonoids com- pounds, phenolic acids, fatty acids, volatile oils, polyace- tylene, adenosine, and so on. The main effects of Flos carthami are promoting blood circulation, dilation of blood vessels, improving microcirculation, eliminating free radicals, anti-inflammatory and other functions. The quality of Flos carthami is closely related with its effi- cacy. High-performance liquid chromatography (HPLC) can identify Flos carthami and evaluate its quality by detecting the content of the main component. However, HPLC methods can not be real-time detection, and it is time-consuming. Multispectral imaging is an emerging technology that integrates conventional imaging and spectroscopy to attain both spatial and spectral information from a sample. Multispectral imaging was originally developed for re- mote sensing applications [1] but has since found appli- cation in diverse fields such as environment, telemetry, agriculture and other fields [2-6]. A series of exploratory studies have been conducted about different kinds of Chinese he rb al medi cines by our rese arch group [7-11] . In this paper, The Flos carthami was indentified, and the qualities of Flos carthamies collecting from different pharmacies were evaluated using the multispectral imag- ing technolog y. The result shows th at multis pectral imag- ing technology provides an objective, time-saving, real- time detection, non-destructive and simple method for the identification and quality evaluation of Flos carthami . *This work was supported by the National Natural Science Foundation of China under Contract 60908038, Science and Technology Planning Project of Guangdong Province, China under Contract 2012B0403 02002 and Agricultural Science and Technology Project of Guangzhou China under Contract GZCQC1002FG080 1 5. #Corresponding author. 2. Materials and Methods 2.1. Samples Preparation The standard sample(SS)s of Flos carthami (FC), Crocus C opyright © 2013 SciRes. OPJ  C. Y. HU ET AL. 230 sativus (CS) and Dendranthema morifolium (DM) were supplied by Guangzhou Institute for Drug Control (GZIDC) on December 8, 2010 and April 27, 2011, re- spectively. The other Flos carthami samples collected from Guangzhou Tong Ren Tang pharmacy(GZTRTP), Guangzhou Bao Jian Tang pharmacy(GZBJTP), Guang- zhou Er Tian Tang pharmacy(GZETTP), Guangzhou Bao Zhi Lin pharmacy(GZBZLP), Guangzhou Oriental phar- macy(GZOP), respectively. The sample details are listed in Table 1. 2.2. Liquid Crystal Multispectral Imaging System The testing instrument was self-designed multispectral spectrum measurement system [12]. It is composed of the light source, a light source filter, a liquid crystal tunable filter (LCTF), the controller of LCTF, lens, CMOS sen- sor, data acquisition card and data processing software. The ray path of the testing system is shown in Figure 1. Table 1. Information of samples. Sample name Source Collecting time SS of FC (batch number: GDIDC 120907-200609) 2010.12.08 SS of CS (batch number: GZIDC 1009-200101) 2010.12.08 SS of DM (batch number: GZIDC 120995-200704) 2011.04.27 FC sample 1 GZTRTP 2010.12.12 FC sample 2 GZBJTP 2010.12.12 FC sample 3 GZETTP 2010.12.12 FC sample 4 GZBZLP 2010.12.12 FC sample 5 GZOP 2010.12.12 Figure 1. The ray path of the system. The centre wavelength of the light source is 254 nm. The LCTF is an important component of the system. It is a splitter component based on electrically controlled birefringence of the liquid crystal. It is used to divide the light coming from the samples in two dimensions. The working wavelength of LCTF is from 400 nm to 1100 nm, which is controlled by the controller of LCTF. The wavebands from 400 nm to 680 nm are chosen in our experiment. The samples partially reflect the light coming fro m the light source after the interaction of light and the sample. The light passes through LCTF carrying the information of the samples. The LCTF divides the light and focus it on the image sensor (CMOS). The images are captured by the image acquisition card and saved on the host computer, as JPG format. The two-dimensional spectrum data can be processed and the results are displayed on the monitor of the computer. 2.3. Methods The excitation light sources are two mercury lamps with the center wavelength of 254 nm in our research. The bandwidth of each lamp is 30 nm, and its optical power is 6 w. Single channel, continuous spectrum scan was used in the detection process. The spectral scan range was from 400 nm to 680 nm, controlled by the controller of LCTF. The two adjacent frames interval is 5 nm. The spectral resolution is up to 0.5 nm. The CMOS imaging camera was adjusted so that the focal plane coincided with the surface of the test samples at 55 0 nm waveband. The CMOS camera was set to continuous mode with the exposure time of 1000 ms, which was synchronized with spectral scanning time, and then the fluorescence images of the test sample were acquired at the whole wavebands. The captured images were stored in the computer with jpg format. A spectral cube of the test sample, formed by 57 frame spectral images, can be obtained in one test. The detected sample was placed on the substrate with- out any pre-treatment, and the two-dimension images of it at a number of narrow wavebands can be obtained. Removed noise in the images with a bandpass filter. Se- lected the same area in every image to calculate the ave- rage light intensity of the corresponding pixel and to normalize them according to Equation (1), then the cha- racteristic spectral curve of the sample was obtained. Figure 2 shows the normalized spectral curve of Flos carthami 1 max , N in iii n i iiNormalizedi II II NI (1) where, N is the number of the pixel, ii I is average light intensity of the ith image, , 1, 2,, 57imaxi I is the biggest light intensity among the 57 frame images. Copyright © 2013 SciRes. OPJ 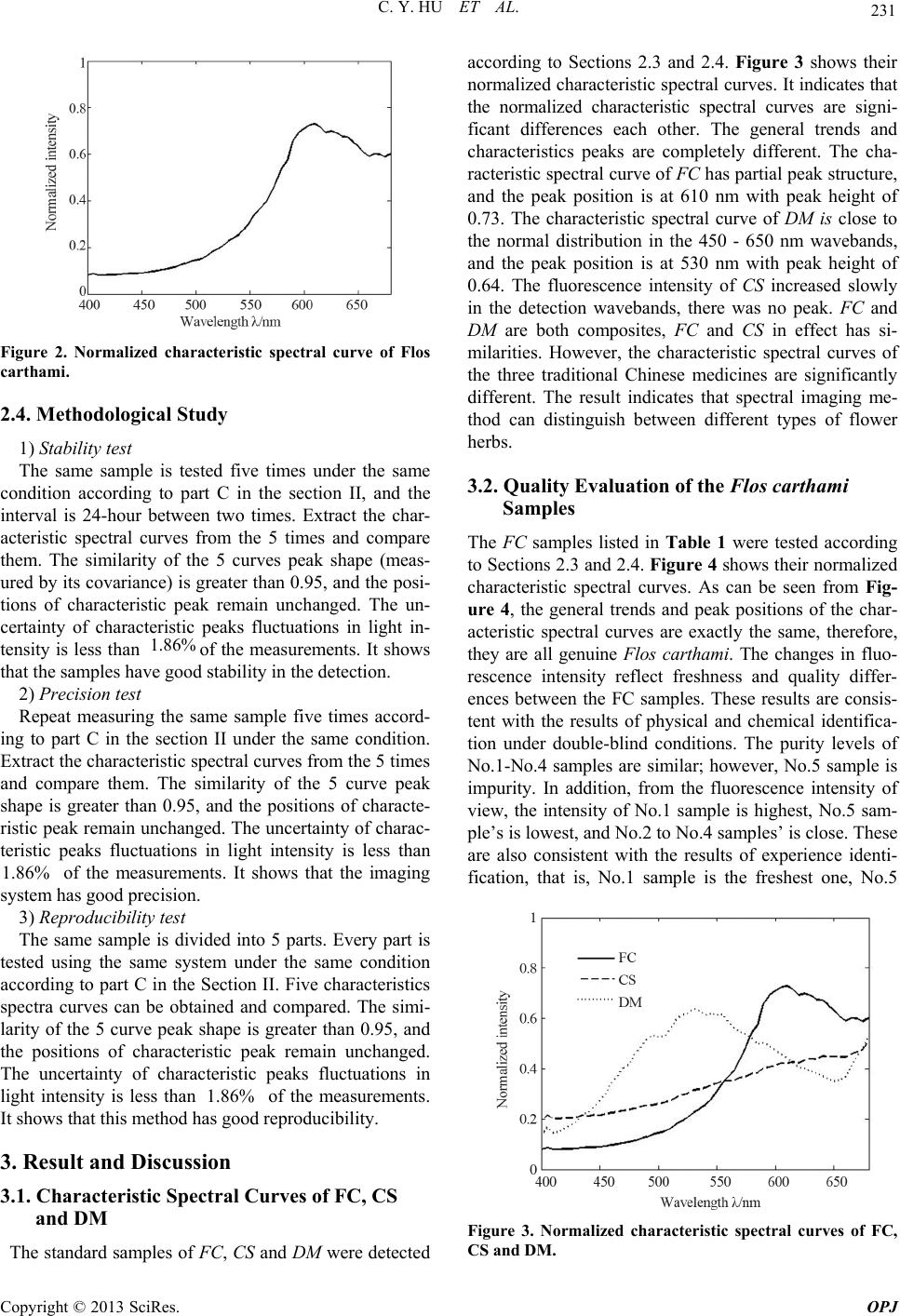 C. Y. HU ET AL. 231 Figure 2. Normalized characteristic spectral curve of Flos carthami. 2.4. Methodological Study 1) Stability test The same sample is tested five times under the same condition according to part C in the section II, and the interval is 24-hour between two times. Extract the char- acteristic spectral curves from the 5 times and compare them. The similarity of the 5 curves peak shape (meas- ured by its covariance) is greater than 0.95, and the posi- tions of characteristic peak remain unchanged. The un- certainty of characteristic peaks fluctuations in light in- tensity is less than of the measurements. It shows that the samples have good stability in the detection. %86.1 2) Precision test Repeat measuring the same sample five times accord- ing to part C in the section II under the same condition. Extract the characteristic spectral curves from the 5 times and compare them. The similarity of the 5 curve peak shape is greater than 0.95, and the positions of characte- ristic peak remain unchanged. The uncertainty of charac- teristic peaks fluctuations in light intensity is less than of the measurements. It shows that the imaging system has good precision. 1.86% 3) Reproducibility test The same sample is divided into 5 parts. Every part is tested using the same system under the same condition according to part C in the Section II. Five characteristics spectra curves can be obtained and compared. The simi- larity of the 5 curve peak shape is greater than 0.95, and the positions of characteristic peak remain unchanged. The uncertainty of characteristic peaks fluctuations in light intensity is less than of the measurements. It shows that this method has good reproducibility. 1.86% 3. Result and Discussion 3.1. Characteristic Spectral Curves of FC, CS and DM The standard samples of FC, CS and DM were detected according to Sections 2.3 and 2.4. Figure 3 shows their normalized characteristic spectral curves. It indicates that the normalized characteristic spectral curves are signi- ficant differences each other. The general trends and characteristics peaks are completely different. The cha- racteristic spectral curve of FC has partial peak structure, and the peak position is at 610 nm with peak height of 0.73. The characteristic spectral curve of DM is close to the normal distribution in the 450 - 650 nm wavebands, and the peak position is at 530 nm with peak height of 0.64. The fluorescence intensity of CS increased slowly in the detection wavebands, there was no peak. FC and DM are both composites, FC and CS in effect has si- milarities. However, the characteristic spectral curves of the three traditional Chinese medicines are significantly different. The result indicates that spectral imaging me- thod can distinguish between different types of flower herbs. 3.2. Quality Evalu at io n of the Flos carthami Samples The FC samples listed in Table 1 were tested according to Sections 2.3 and 2.4. Figure 4 shows their normalized characteristic spectral curves. As can be seen from Fig- ure 4, the general trends and peak positions of the char- acteristic spectral curves are exactly the same, therefore, they are all genuine Flos carthami. The changes in fluo- rescence intensity reflect freshness and quality differ- ences between the FC samples. These results are consis- tent with the results of physical and chemical identifica- tion under double-blind conditions. The purity levels of No.1-No.4 samples are similar; however, No.5 sample is impurity. In addition, from the fluorescence intensity of view, the intensity of No.1 sample is highest, No.5 sam- ple’s is lowest, and No.2 to No.4 samples’ is close. These are also consistent with the results of experience identi- fication, that is, No.1 sample is the freshest one, No.5 Figure 3. Normalized characteristic spectral curves of FC, CS and DM. Copyright © 2013 SciRes. OPJ 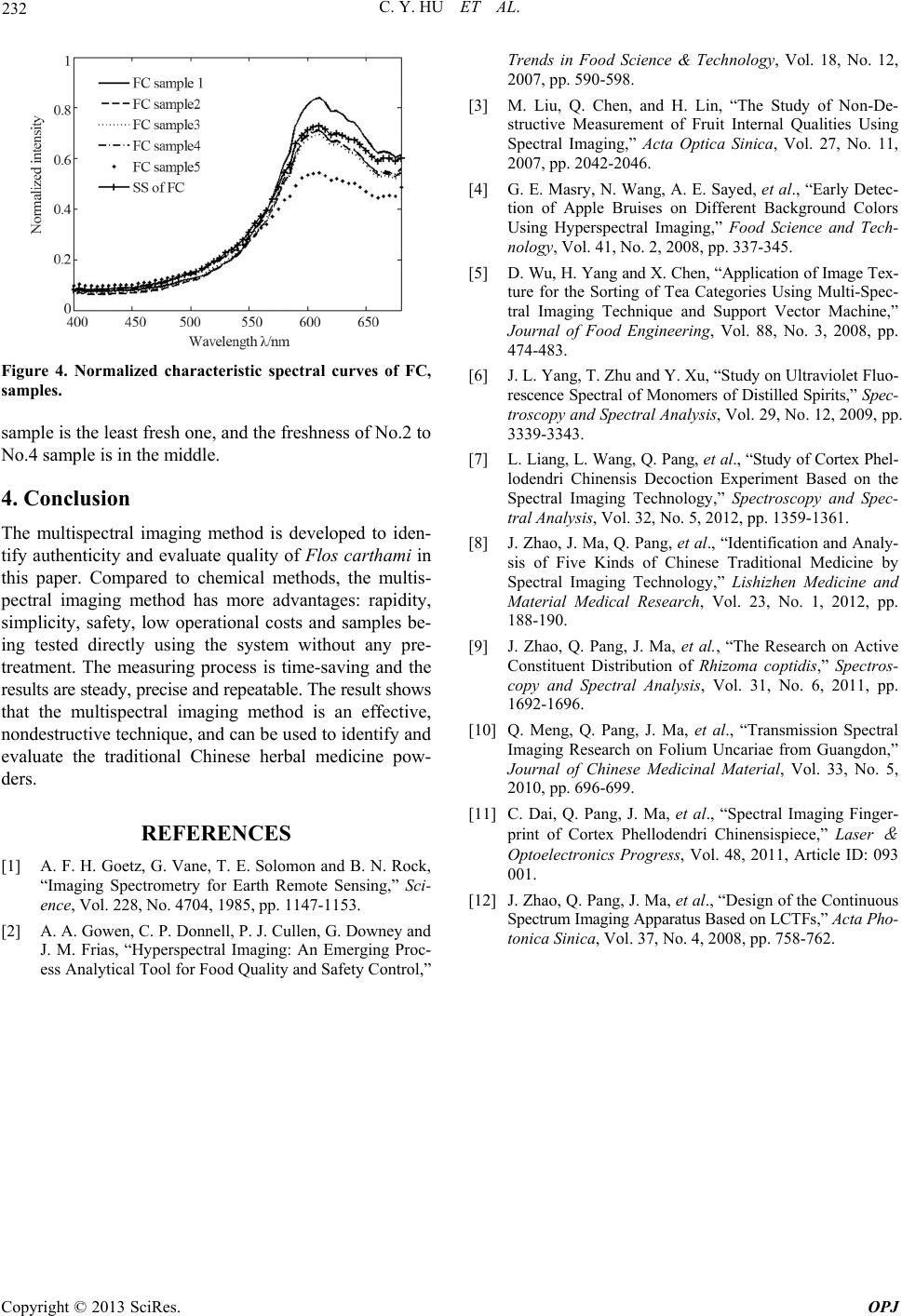 C. Y. HU ET AL. Copyright © 2013 SciRes. OPJ 232 Figure 4. Normalized characteristic spectral curves of FC, samples. sample is the least fresh one, and the freshness of No.2 to No.4 sample is in the middle. 4. Conclusion The multispectral imaging method is developed to iden- tify authenticity and evaluate quality of Flos carthami in this paper. Compared to chemical methods, the multis- pectral imaging method has more advantages: rapidity, simplicity, safety, low operational costs and samples be- ing tested directly using the system without any pre- treatment. The measuring process is time-saving and the results are steady, precis e and repeatab le. Th e result shows that the multispectral imaging method is an effective, nondestructiv e technique, and can be us ed to iden tify and evaluate the traditional Chinese herbal medicine pow- ders. REFERENCES [1] A. F. H. Goetz, G. Vane, T. E. Solomon and B. N. Rock, “Imaging Spectrometry for Earth Remote Sensing,” Sci- ence, Vol. 228, No. 4704, 1985, pp. 1147-1153. [2] A. A. Gowen, C. P. Donnell, P. J. Cullen, G. Downey and J. M. Frias, “Hyperspectral Imaging: An Emerging Proc- ess Analytical Tool for Food Quality and Safety Control,” Trends in Food Science & Technology, Vol. 18, No. 12, 2007, pp. 590-598. [3] M. Liu, Q. Chen, and H. Lin, “The Study of Non-De- structive Measurement of Fruit Internal Qualities Using Spectral Imaging,” Acta Optica Sinica, Vol. 27, No. 11, 2007, pp. 2042-2046. [4] G. E. Masry, N. Wang, A. E. Sayed, et al., “Early Detec- tion of Apple Bruises on Different Background Colors Using Hyperspectral Imaging,” Food Science and Tech- nology, Vol. 41, No. 2, 2008, pp. 337-345. [5] D. Wu, H. Yang and X. Chen, “Application of Image Tex- ture for the Sorting of Tea Categories Using Multi-Spec- tral Imaging Technique and Support Vector Machine,” Journal of Food Engineering, Vol. 88, No. 3, 2008, pp. 474-483. [6] J. L. Yang, T. Zhu and Y. Xu, “Study on Ultraviolet Fluo- rescence Spectral of Monomers of Distilled Spirits,” Spec- troscopy and Spectral Analysis, Vol. 29, No. 12, 2009, pp. 3339-3343. [7] L. Liang, L. Wang, Q. Pang, et al., “Study of Cortex Phel- lodendri Chinensis Decoction Experiment Based on the Spectral Imaging Technology,” Spectroscopy and Spec- tral Analysis, Vol. 32, No. 5, 2012, pp. 1359-1361. [8] J. Zhao, J. Ma, Q. Pang, et al., “Identification and Analy- sis of Five Kinds of Chinese Traditional Medicine by Spectral Imaging Technology,” Lishizhen Medicine and Material Medical Research, Vol. 23, No. 1, 2012, pp. 188-190. [9] J. Zhao, Q. Pang, J. Ma, et al., “The Research on Active Constituent Distribution of Rhizoma coptidis,” Spectros- copy and Spectral Analysis, Vol. 31, No. 6, 2011, pp. 1692-1696. [10] Q. Meng, Q. Pang, J. Ma, et al., “Transmission Spectral Imaging Research on Folium Uncariae from Guangdon,” Journal of Chinese Medicinal Material, Vol. 33, No. 5, 2010, pp. 696-699. [11] C. Dai, Q. Pang, J. Ma, et al., “Spectral Imaging Finger- print of Cortex Phellodendri Chinensispiece,” Laser & Optoelectronics Progress, Vol. 48, 2011, Article ID: 093 001. [12] J. Zhao, Q. Pang, J. Ma, et al., “Design of the Continuous Spectrum Imaging Apparatu s Ba sed on LCT Fs,” A cta Pho- tonica Sinica, Vol. 37, No. 4, 2008, pp. 758-762. |

