Paper Menu >>
Journal Menu >>
 Journal of Water Resource and Protection, 2013, 5, 653-668 http://dx.doi.org/10.4236/jwarp.2013.57066 Published Online July 2013 (http://www.scirp.org/journal/jwarp) Contamination Levels of Some Selected Wells in Ogbomoso South Local Government Area, Nigeria and the Implications on Human Health Samson O. Ojoawo*, T. Lateef Kolade Department of Civil Engineering, Ladoke Akintola University of Technology, Ogbomoso, Nigeria Email: *soojoawo@lautech.edu.ng Received April 15, 2013; revised May 22, 2013; accepted June 18, 2013 Copyright © 2013 Samson O. Ojoawo, T. Lateef Kolade. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Incident of water-borne diseases is becoming more rampant in developing countries in the 21st century. This paper ex- amines the contamination levels of some selected wells in Ogbomoso South Lo cal Government Ar ea (LGA), Oyo State, Nigeria. Methodology includes administering of structured questionnaire, field survey/sampling and laboratory analysis. The first set of questionnaires sought information on water sources, quality, supply and treatment. These were adminis- tered in 10 selected major locations which included: Ajegunle, Ar ink inkin, Aro wo mole, Caretake r, Esanu -Aje, I ta-O lola , Malete, Oke-Alapata, Ora-Gada and Sunsun. The other set which was administered in 6 randomly selected Health In- stitutions viz: Oyo State General Hospital, Alaafia Tayo, Oore-Ofe, Bethel, Favour, and Grace Hospitals had questions on health-related issues. Ten (10) wells were randomly selected for sampling, one in each location. Results were com- pared with WH O’s Drink ing Water Stand ards. Qu estionnaires r evealed th at residents depended mostly on w ells for w a- ter supply; the water quality was low; supply was irregular; quantity was inadequate and boiling was the commonest treatment method. Health Officials established that some residents consumed improperly treated well water and there were some incidences of water-related diseases. Laboratory analyses showed that samples from Ajegumle and Esanu- Aje had colour values above the standard. Also, the pH was observed to vary from slightly acidic to slightly alkaline with values ranging between 6.0 and 8.1. Only Arinkinkin and Ita-Olola areas had permissible values of total alkalinity and Oke-Alapata had the highest chloride value of 400 mg/l while the highest nitrate concentration was at Esanu-Aje (127.58 mg/l). Study concludes th at water supply in the area is no t fully supportiv e to health. To abate the prob lem, it is recommended that aside boiling, each household should endeavour to coagulate, filter, soften and disinfect well water before consumption. Also, personal hygiene within each household must always be emphasized and improved upon. Keywords: Contamination; Wells; Water Quality; Human Health 1. Introduction The importance of water to man cannot be over-empha- sized. It forms about 70 per cent of our body fluids and serves many other purposes among which are: drinking and culinary uses, washing, bathing and laundering, wa- tering lawns and gardens, protecting life and property against fire, etc. People in the rural areas in absence of potable water are exposed to the risk of water borne dis- eases like cholera, typhoid and other environmental im- plications [1]. There is evidence of widespread contami- nation of water resources in many areas of Nigeria. The Environmental Protection Agency’s National Water Qua- lity Inventory of 1994 has identified agriculture, urban runoff/storm water, and municipal point sources of the largest pollutant sources for rivers, lakes, and estuaries. Contaminants from these sources include pesticides, metal, nitrates, solvents, and other wastes. It should how- ever be noted that not all contamination events pose a threat to our health [2]. Water resources normally refer to the whole of the waters of a territory which can be used for suppo rting the diverse needs of individuals and communities [3]. Re- sources are all the attributes of our natural environment that can be utilized to promote human welfare at any place at a point in time. Resources vary in quantity, qual- ity, mutability, renewability, reusability and availability. *Corresponding a uthor. C opyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 654 The continued availability of adequate fresh water re- sources is a pre-requisite for sustaining life and devel- opment. It is imperative to plan and monitor the nation’s water resources to achieve sustainable development of the vital resources especially in the face of changing cli- mate. The development plan of water supply systems is important and must account the available water as a re- source which should be well developed planned and ex- ploited in order to obtain maximum returns at minimum cost. The major sources of water pollution are sewage or untreated faecal matter, domestic and industrial solid waste, sullage, and industrial waste water and sediment from soil erosion [4]. Sewage effluent and faeces when properly treated poses minimal pollution or health prob- lems though they require heavy financial investment but the advantage is greater than the cost. Domestic and in- dustrial solid wastes are found floating on rivers this pollutes the water and constricts the water flow. This eventually results in flooding which destroys lives and property. Sullage and industrial wastewater often contain significant amount of phosphate and oils and they con- stitute significant sources of water pollution. It is unfor- tunate that industrialization does not only increase human comfort but also spell doom on man. In an earlier work in South-Southern Nigeria, [5] reported that chemical characteristics including Nitrate oil and grease and met- als like Fe, Pb and Ni exceeded the acceptable limits in some or most of the wells sampled and this could portend serious health problems. This observation was more ob- vious around the refinery and water-side areas of Warri. Sediment of soil erosion is another source of pollution especially in the place where agriculture is being prac- ticed. In sloppy areas it is easy by gravitational force for liquid to spread to a wide expanse area of land. When a contaminant first enters the soil it will travel down vertically gravity until contact with groundwater. At this point it will begin to floor primarily in a horizon- tal direction. The contaminants will then spread out three-dimensionally like smoke from a chimney and is called a plume. Groundwater does not exhibit turbulent flows as found in surface water. The flow is defined by gravity, pressure and friction. It is much more constant than surface water. An aquifer can flow at a fraction of an much per day up to few of reducing contaminants in drinking water by controlling it at the source. Preven- tative measures such as modification of tilling methods and run-off, control in agriculture, the use of lined catch- ment ponds for treatment of wastes, and double lined underground storage tanks can greatly reduce contamina- tion of drinking water sources [2]. Wells are one of the oldest sources of water. In many rural areas, wells are the only reliable water sources while in most cases; well water is a safe and dependable option, in rare instances where contamination has oc- curred. A well can be contaminated by disease-producing pathogens and leachates from disposal of hazardous household wastes [6]. The level of contaminant in drink- ing water is sometimes high enough to cause acute (im- mediate) health effects such as nausea, lung irritation, skin rash, vomiting, dizziness, and even death. And more so, contaminants are more likely to cause chronic health effects occurring long after repeated exposure to small amount of a chemical. Examples of chronic health effects include cancer, liver and kidney damage, disorder and damage of the nervous system, damage to the immune system and birth defects [7]. Evidence relating chronic health effects to specific drinking water contaminants is limited. In the absence of exact scientific information, scientists predict the likely adverse effects of chemicals in drinking water using human data from clinical reports and epidemiological studies, and laboratory animal stud- ies. Radon is a radioactive contaminant that results from the decay of uranium in soils and rocks. It is usually more of a health concern when it enters a home as a gas than when it occurs in water supplies. Radon in air is associated with the possibility of contaminant in drinking water. Water supplies once considered to be pure may have various contaminants, often from natural sources these are usually at levels below those considered to be harmful. Water systems can be sensitive barometers of the health of the planet. Decreased fresh water supply has encumbered world food harvest, destroyed precious aquatic habitats and threatened biodiversity. WHO in 1985 estimated that 80% of all sickness and diseases in the world are attributable to unhygienic water. Water borne diseases are among the leading causes of death in many developing countries 10% of their productive time because of diseases related to poor and contaminated water programmes. Water from any sources must suit the purpose for which it is intended. Good quality water is water that is safe to use. Water intended for drinking must be free from pathogenic organisms. The chemical and physical qualities must also be suitable. Water must not contain any dissolved or suspended materials that are injurious to health of that would give an unpleasant taste. Physically safe water must be at suitable temperature and not have objectionable colour or cloudiness [8]. The quality of water according to [10] comprises considera- tion of many different factors. Information on and control of water quality is of great importance for a wide range of purpose including water supply, public health, agri- cultural and indu s trial u ses. As a result of human activities especially in urban ar- eas toxic materials discharged into the environment have affected the quality of water and rendered it unsafe. Good management of waste from households industries and Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 655 automobiles will safe-guard water contamination by im- proving its quality. Dissolved gases in water such as car- bon-dioxide (CO2) methane (CH4) and Hydrogen Sul- phide, for example at concentration greater than 1 mg/l are unfit for human consumption [9]. The World Health Or- ganization has set standards fo r water quality. This serves as a basis for approval of results of chemical analysis of in terms of suitability of water for various intended uses. A recent one is those established for drinking water in Geneva 2011. To date there is considerable controversy with regard to the specific organic constituents that should be included in drinking water standards and con- centration limits that should be established. For example total dissolved solids, Sulphate and Chloride consump- tion by humans thought water having concentration above WHO 2011 limits is not generally harmful [10]. Most often water when it is related to health laid more emphasis on the water chemistry this may be due to the fact that the world around us is made up of chemicals. In the bid to improve water quality disinfect water or our immediate environment and improvement in quality of living has resulted to introduction of toxic chemicals directly and indirectly to water available for our uses. Lead, Arsenic and Mercury are known to be harmful to humans even in small account. Also excessive Nitrates (N-NO3) cause metheno-globinemia in infants at a range more than 15 - 25 mg/l, it can affect livestock as well and can result in certain form of cancer [11]. Uses of chemi- cal to disinfect water require skill and experience other- wise its shortage of required quantity leave the water partially treated with the disease causing organism in the water increasing their resistant to such treatment. Over dosage will also have side effects, for instance excess io- dine in water causes goitre, while potassium permanga- nate adds colour to water and stains materials. Beside chemical elements in water, microbiological organisms as well as physical characteristics and radio- logical materials have their effects on health. The micro- biological quality of water determines and helps in diag- nosing various forms of diseases that can be linked to water either for its transmission sustenance or eradication. Drinking water should not contain pathogenic micro- organism, and should be free of bacteria that indicate excremental pollution. Water to be used for washing, ba- thing and cooking as well must be free of pathogens car- rying contagious diseases. It is therefore important that drinking water and house-hold use water samples from source to storage vessels be tested regularly for possible indicators of dangerous microbiological organisms. Such indicators include faecal streptococci, clostridium welchi and the coliform aerogens group; all these indicators are excreted in large numbers from warm blooded animals and their detection in water-denoted faecal pollution. Some macroscopic biological organism through their life cycle or activities especially aquatic lives contributes to health impacts of water on human being. Larvae crusta- cea and large numbers algae or filamentous growth (i.e. spirogyra) should not be allowed in house-hold use water. They affect taste, odour and appearance of water. Good water for domestic use must be free of disease-carrying organisms like bacteria-cholera and typhoid, protozoa- amoebic dysentery, viral infections hepatitis, worms-gui- nea worn, angus-ring worm of the foot, etc. Moreover, radiological materials are harmful; therefore their intake should be kept to a minimum. Radium and Strontium are particularly harmful when taken internally. Emission of radioactive materials gets into water unconsciously wher- ever it is used. In hospitals where it is used for X-Ray and curing of other ailments the storage vessel even wa- ter pipes made of metal not to mention plastics pipes can be penetrated by radioactive materials. Anchored on various records of water-related health crises being noted in the Ogbomoso South Local Gov- ernment (OSLG) Area, this research work focuses on the investigation of the contamination levels of some se- lected wells in the area and studying their associated health implications on human beings. With the present dearth of information on such occurrences in the study area, a study of this nature is expected to provide techni- cal explanations on the causes and implications of these incidents of water-borne diseases on human health. The OSLG has an area of 68 km² and with a population of 100,815 people [12]. With increase in agricultural activi- ties and domestic chores in the area, there is greater need for the adequate supply of good quality water for the growing population. As the well water is naturally pro- vided to every being, and then the environmental and health implications that were attached to this natural re- source cannot be over emphasized. This research there- fore ultimately targets minimizing the deleterious effects associated with the consumption of contaminated well water. 2. Methodology 2.1. Structured Questionnaire Two different sets of questionnaires were prepared ac- cording to the National Environmental Standard Regula- tion and Enforcement Agency (NESREA) guidelines format. The first set sought information which bothered on water sources, its quality, supply and treatment were administered in 10 selected major locations in the LGA which include: Ajegunle, Arinkinkin, Arowomole, Care- taker, Esanu-Aje, Ita-Olola, Malete, Oke-Alapata, Ora- Gada and Sunsun. The other set which was administered in the six (6) randomly select ed Health Institutions in the LGA viz: Oyo State General Hospital, Alaafia Tayo, Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 656 Oo re-Ofe, Bethel, Favour, and Grace Hospitals have ques- tions on water borne-diseases and health-related issues. 2.2. The Sampling Ten (10) wells were randomly selected, one in each ma- jor locations in the LGA viz: Ajegunle, Arinkinkin, Arowomole, Caretaker, Esanu-Aje, Ita-Olola, Malete, Oke-Alapata, Ora-Gada and Sunsun. One (1)-litre plastic containers were used to collect the water samples from the well visited. The containers were thoroughly washed in the laboratory with distilled water and labeled accord- ingly. At the sampling point, the containers were again rinsed with the samples to be collected . 2.3. Laboratory Analysis of Samples The well water samples collected were taken directly to the laboratory without any preservation for the physical and chemical analysis. The parameters of interest and their tests are as follows: 1) Temperature: This was carried out with the aid of a thermometer in situ while taking the samples. 2) pH: Some quantities of the water sample were poured into the beaker and the electrode of the pH meter dipped into the sample. The meter was switched on and allowed to stand for 30 minutes while the pH value was recorded. 3) Chloride: The apparatus were the weighing balance, syringes, plastic vessel, and small plastic containers. The chloride level in mg/l (ppm) was determined by mercuric nitrate titration. Two drops of diphenylcarbazone indica- tor were added and mixed carefully by swirling the ves- sel in circles. Nitric acid was added drop wise until the solution turns yellow. The titration syringe’s tip was in- serted into mercuric (nitrite solution) solution and the plunger pulled out until the lower edge of the plunger seal is on the 0 ml mark of the syringe. The titration solu- tion was slowly added drop wise and swirled until the colour changed from yellow to violet. The millilitres of titration solution from the syringe scale was read off and multiplied by 1000 to obtain mg/l (ppm) chloride. 4) Hardness: The apparatus used were titer syringe plastic vessel, small transparent plastic container and buffer solution. The cap from the small plastic syringe was removed and rinsed with the sample water. The sample was filled to 0.5 ml mark and the cap replaced. Five drops of hardness buffer solution were added and mixed carefully. One drop of calmagite indicator was then added. Take the titration syringe and push the plunger completely into the syringe. Tip of the syringe was inserted into ETDA solution and pulled out until its lower edge is on the 10 ml mark of the syringe. The EDTA titration solution is slowly added drop wise and swung to mix after each drop until the colour changes. The milliliters of titration solution were read off from the syringe scale and multiplied by 300 . 5) Alkalinity: Apparatus included transparent plastic vessel, calibrated syringe, bromophenol blue indicator, and titrant solution. The cap is removed from the plastic vessel rinsed with the sample and filled with the sample to 0.5 ml mark. The cap is replaced. Drops of Bromophe- nol blue indicator were added and the titration syringe filled to 10 ml with H13811.0 solution. Titration solution is added drop wise, mixed after each drop until a colour change is noted. The milliliters of titration solution used were read off from the syringe scale and multiplied by 300 to obtain mg/l (ppm ). 6) Conductivity: Digital conductivity meter was used. The conductivity was determined by dipping the elec- trode into the water sample directly in a beaker and al- lowed to stand for 30 minutes before taking the reading. 7) Colour: 10 ml of each sample was measured in the glass bottle given. Bottle was placed in the colour test machine. Read direct was pressed on the machine. Col- our reading was shown on the LCD screen of the ma- chine. 8) Nitrate: A 15 ml of water was prepared into two glasses each containing 5 ml of water sample. One of the glasses was placed into left hand opening of the checker disk. This was noted as the blank. A pocket of reagent was added to the other sample and properly mixed. The solution was marked as reacted sample and was placed at the right hand opening of the checker disk at a distance of about 30 to 40 cm away, and across a light source to illuminate the sample to be matched. The disk was ro- tated while looking at the sample through the colour test window until the colour is matched. Th e value in the test window is read as mg/l of nitrate or nitrogen (N-NO3). 9) Turbidity: 10 NTU (Nephelometric Turbidity Units) with 1 - 10 NTU graduated standard was used. A portion of water sample was poured into a clear and clean test tube was shaken thoroughly and later inserted into the sample chamber of standardized turbid meter (1 - 10 NTU graduated). The value was read off directly from the scale. This procedure was also repeated for other samples. 10) Iron: Apparatus used were colour comparator cube and plastic vessels. The cap was removed from the plastic vessel and rinsed with water sample. It was then filled to the 10 ml mark. 1 packet of the iron reagent HI 3834-0 was added. The cap was replaced and the solution mixed until solids dissolved. The cap was removed and the solution transferred into the colour comparator cube. The colour that matches the solution in the cube was re- corded as mg/l (ppm) iron. 11) Magnesium: Demineralizer bottle, plastic vessel, filter paper disc, funnel, pipette were the apparatus used. The cap was removed and the Demineraliser bottle filled Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 657 with tap water. The cap was replaced and the bottle shaken for at least 2 minutes to prepare the demineralised water. One of the large (50 mg/l) plastic vessels was filled with 25 ml of water sample, up to the mark. Four (4) drops of Buffer Reagent were added and swirled to mix. One (1) packet of HI 38079A-0 Oxalate reagent was added and stirred for 30 seconds by means of the plastic spoon. This was left for about 5 minutes for the reaction to complete. A folded filter paper into cone was placed in the funnel and the funnel placed over the other larger plastic vessel. The reacted water sample was poured through the filter paper. Using the 3 ml plastic pipette, 3 ml of the filtered clear water sample was carefully trans- ferred into the small (20 ml) plastic vessel. The top of the Demineraliser bottle cap was flipped open. By gently squeezing the bottle, the demineralised water was added to the vessel up to the 10 ml mark. Using the 1 mL plas- tic pipette, 1 mL of Buffer Solution was added and swirled to mix. One (1) drop of Calmagite indicator was added and swirled to mix. The syringe’s plunger was pushed completely into the HI 380798-0 EDTA solution bottle and pulled out un til th e lower edg e of the seal is o n the 0.0 ml mark of the syringe. Titration solution was slowly added drop by drop, swirling after each drop. As the colour changes from pink to purp le, it was swirled for 15 seconds after each additional drop, until the solution turns pure blue. The millilitre of titration solution was read off from the syringe. Calculation of the mg/l (ppm) of Magnesium in the sample was as follows: ppm of mg = ml of titrant × 243. 12) Sulfate: Apparatus: plastic vessel, spoon, plastic pipette, test tube. The plastic v essel was filled with 50 ml of the sample up to the mark. One (1) packet of HI 38000A-0 Sulfate Reagent was added and swirled gently to dissolve. Two (2) drops of Complexing Agent was added and swirled to mix. Two (2) spoons of HI 38000B- 0 reagent were added and swirled gently to mix. This was left for 5 minutes to allow reaction to occur. The test tube was kept on a white surface and viewed from the top at the black spot on the bottom. The plastic pipette was used to fill the tube with the reacted sample until the black spot has completely disappeared. The concentra- tion in mg/l (ppm) of sulfate was read in correspondence of the level of the liquid in the test tube. 13) Biological Oxygen Demand (BOD): The appara- tus used include: incubator bottles o f 125, 250 or 300 ml capacity with ground glass stopped; air-cooled insulator (this is thermostatically controlled at 20˚C); high quality distilled or deionized water; phosphate buffer solution; magnesium sulphate solution; calcium chloride solution; and iron (III) chloride solution. Preparation of dilution water was by storing the distilled water in a large incu- bator bottle whose mouth is plugged with clean cotton wool as to permit it to be saturated with sufficient Dis- solved Oxygen. To the distilled water was added 1 ml of each of the following reagents: phosphate buffer magne- sium sulphate, calcium chloride and iron (III) chloride solution. The water sample was pre-heated with 0.5 m acid or 1 M alkali if necessary to about pH 7 if it contains chloride compound s, to chlorin ate with ad equate qu antity of 0.125 M Na2SO3. Several solutions of the prepared sample were made so as to obtain adequate drop in oxy- gen content. The following dilutions were found ade- quate: 1% - 2% for raw wastewater; 2% - 3% for settled wastewater; 5% - 30% for biological treated wastewater; 3% - 7% for chemically treated wastewater; 35% - 100% for river water. Suitable amounts of sample were meas- ured into BOD bo ttles in dup licates by the aid of large tip volumetric pipette. Bottles were filled to the brim with dilution water in accordance with the percentage of dilu- tion required. The stoppers were inserted without leaving air bubbles. Dilution greater than 1% was made by dilut- ing the water in a volumetric flask before transferring it, carefully into BOD for final dilution. Since the sets of dilutions are made in duplicate, the initial DO was de- termined on the one set for 5 days in the dark at 20˚C in a cooled incubator. Thereafter, the DO in the incubated samples and the blank was determined. The BOD was then calculated. 14) Coliform Bacteria Determination: Apparatus used was Petri Dish Incubator. A 50 mg of MacConkey agar was weighed and 1 litre of deionized water added. This was allowed to so ak for 10 mins, swirled to mix and sterilized by autoclaving at 121˚C for 15 mins. The mix- ture was cooled to 47˚C and poured into petri dish. A dry surface was ensured before inoculation by partial expo- sure at 37˚C. A 0.5 ml portion of the sample was taken and poured into the petri dish, the prepared MacConkey agar solution was added and the dish covered. This was placed in the incubator for 24 hrs at 30˚C. This was later removed and placed on the colony counter for determina- tion of the number of coliform bacteria formed. 3. Results and Discussion 3.1. Questionnaires 1) Water supply: The questionnaires revealed that the major source of water supply in the area is from well which is most available throughout. Figure 1 illustrates the water supply pattern in the study area. It was also found out that the mode of collection of water from the source is majorly by head porterage with few uses of ve- hicle due to the distance of water source. Furthermore the respondent also testifies that water is always stored in- side the tank for later use and that they do not pay for the water since the wells are many and most of them are pri- vately owned. It is clearly shown from Figure 1 that even though th e Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE Copyright © 2013 SciRes. JWARP 658 area; effects of water-borne diseases and the availability of stagnant water in the surrounding. The numbers 1 to 10 in horizontal axes of the graphs in Figures 2 to 9 represents the 10 selected locations in this order; Aje- gunle, Arinkinkin, Arowomole, Caretaker, Esanu-Aje, Ita-Olola, Malete, Oke-Alapata, Ora-Gada and Sunsun. It could be observed from Figure 2 that more residents use their water directly without subjecting it to treat- ments as common in most rural areas of developing countries. Forty (40) is the maximum percentage of those treating their water before usage. This may be responsi- ble for the high incidents of water-related diseases noted in the area. Figure 1. Water supply patter n in the study area. Figure 3 shows that most residents believed have hu- man or animal wastes dominating the contamination of their water sources and this was closely followed by mu- nicipal wastes which perhaps generate leachate contami- nants. Since the level of industrial activities in the area is very low, it could account for the view that there is low industrial contamination . majority of the respondents depend on both well and tap water, well water remains the source of water with high- est sole patronage. 2) Water quality: The survey revealed that most resi- dents used water directly from the source without much treatment except a few who boil theirs. It was also found that refuse dumps are located on some uncompleted buildings within the range of 100 m or more to a few of the water sources. The respondents confirmed that there is likelihood of the contamination of their water by leachates emanating from these waste dumps. Again, according to the respondents there is no certification of good water quality by any authority. The respondents allude the ailments and water borne diseases which had affected the area in the past to this uncertified water quality. The results of responses on method of water treatment revealed that simple boilin g was adopted by about 60% - 70% of those treating their water before consumption in most locations. Residents of Esanu-Aje and Caretaker however adopt chemical treatment method of adding aluminium sulphate (alum) more than others using same treatment method. Treatments by UV light, Ozonation, Poly-electrolytes are not common in the area. As revealed in Figure 5, majority do not subject their water to any quality control or certification befo re its use. Physical screening is being used in only 5 locations whe- reas nearly 50% of the respondents assumed that their pipe-borne/tap water is already treated by the govern- ment. It is only in Ajegunle area that few residents claim to subject their water to lab oratory analysis, probably due Figures 2 to 9 illustrate patterns of other components of the questionnaire like the water use prior to or after treatments; types of contaminants; method of water treatment; water quality control; occurrence of wa- ter-borne diseases; proximity of refuse dump to the water source; incidences of water-borne disease outbreak in the Figure 2. Water use prior to or after treatments.  S. O. OJOAWO, T. L. KOLADE 659 Figure 3. Types of Contaminants. Figure 4. Method of water treatment. Figure 5. Water quality control. to their proximity to th e waterworks. All locations except one had the refuse dumps farther than 100 m from the water source. Equal numbers of residents have their dumps both far away from and near the water source in Malete area. From the result of the questionnaire survey presented Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 660 in Figure 7 indicates that residents in all the areas had at one time or the other experienced water-borne diseases. Those yet to experience it are however on the higher fig- ures, especially at Sunsun and Ajegunle areas. Even though this had not been a regular occurrence according to the respondents, yet at different times residents have fallen victims of the health challenges caused by con- taminated water. This is in agreement with an earlier re- search finding that contaminated water accounts for most of the diseases in the tropics [13]. As a rider to these in- cidences, Figure 8 displays the respondents’ awareness levels of the effects. Except at Ajegunle and Arinkinkin where equal numbers of residents were both affected and otherwise, in the rest locations more respondents had been affected by the water-related diseases in the previ- ous times. Most respondents at Arinkinkin, Malete and Ajegunle areas claimed to notice stagnant waters in their sur Figure 6. Proximity of refuse dump to the water source. Figure 7. Incidences of water-borne disease outbreak in the area. Figure 8. Effects of water-borne diseases. Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 661 roundings (Figure 9). This could be spring boards for mosquitoes and other vectors capable of affecting human health negatively. In six (6) other locations surveyed, equal numbers of respondents have and do not have stagnant waters within their neighbourhoo ds. 3.2. Results of the Laboratory Analysis on Water Samples from Selected Wells The results of the Laboratory analysis conducted on se- lected wells in Ogbomoso South Local Government were presented in Tables 1 and 2. The graphical representa- tions are in Figures 10 to 12. These were compared with the guidelines on drinking water by National Primary Drinking Water Regulations (NPDWRs, US), World Health Organization (WHO) and National Agency for Food and Drug Administration and Control (NAFDAC) on Ta b le 3 . The basis of comparison is to determine its potability of the water for intended use. 1) Appearance—All the ten samples gotten from the study area were observed to be clear. 2) Colour—Sample taken from Ajegunle and Esanu- Aje area were observed to have the colour value of 40 TCU and 70 TCU respectively, where other area has no observable colour. With reference to Figure 10, the standard was represented by a straight line graphs i.e. 15 TCU according to the specification given by National Primary Drinking Water Regulations, U.S (NPDW Rs). It is clearly shown that the sample taken from Ajegun le an d Figure 9. Availability of stagnant water in the surrounding. Figure 10. Physical parameters and locations. Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 662 Figure 11. The distribution of chemical parameters in the studied wells. Figure 12. Graph of the Coliform levels in the study area. Esanu-Aje areas have colour values above the standard as it is shown in Figure 10. Meanwhile other locations (i.e. Arinkinkin, Arowomole, Caretaker, Ita-olola, Male- te, Oke Alapata, Oora-Gada and Sunsun) have zero col- our value as clearly shown on the graph in the same Fig- ure. 3) Odour—No odour was noticed from the entire sample before and after the collection. Thus they have unobjectionable odour which meet the standard of both WHO and NAFDAC as case may be. 4) Turbidity—There was no wide variation in the measured value of the turbidity obtained, as the value ranges from 0.00 NTU - 13.25 NTU. It is clearly shown from the analysis that sample taken from Esanu-Aje area of Ogbomoso south Local Government is highly turbid. This value is above the recommended limit by WHO, NAFDAC and NPDWRs which is 5 NTU, while the sample from Sunsun is the least turbid. Ajegunle and Esanu-Aje areas have turbidity value above the standard, as indicated in Table 4. Sample taken from Aje g unl e area has 7.50 NTU while Sample from Esanu-Aje has 13.25 NTU, while other locations fell below the WHO, NAF- DAC and NPDWRs standard. By way of comparing tur- bidity with the colour it is observed that Location with high turbidity value has the highest colour value and vice-versa. 5) Temperature—Temperature is usually attrib uted to weather condition, season, and time of days. The samples Copyright © 2013 SciRes. JWARP 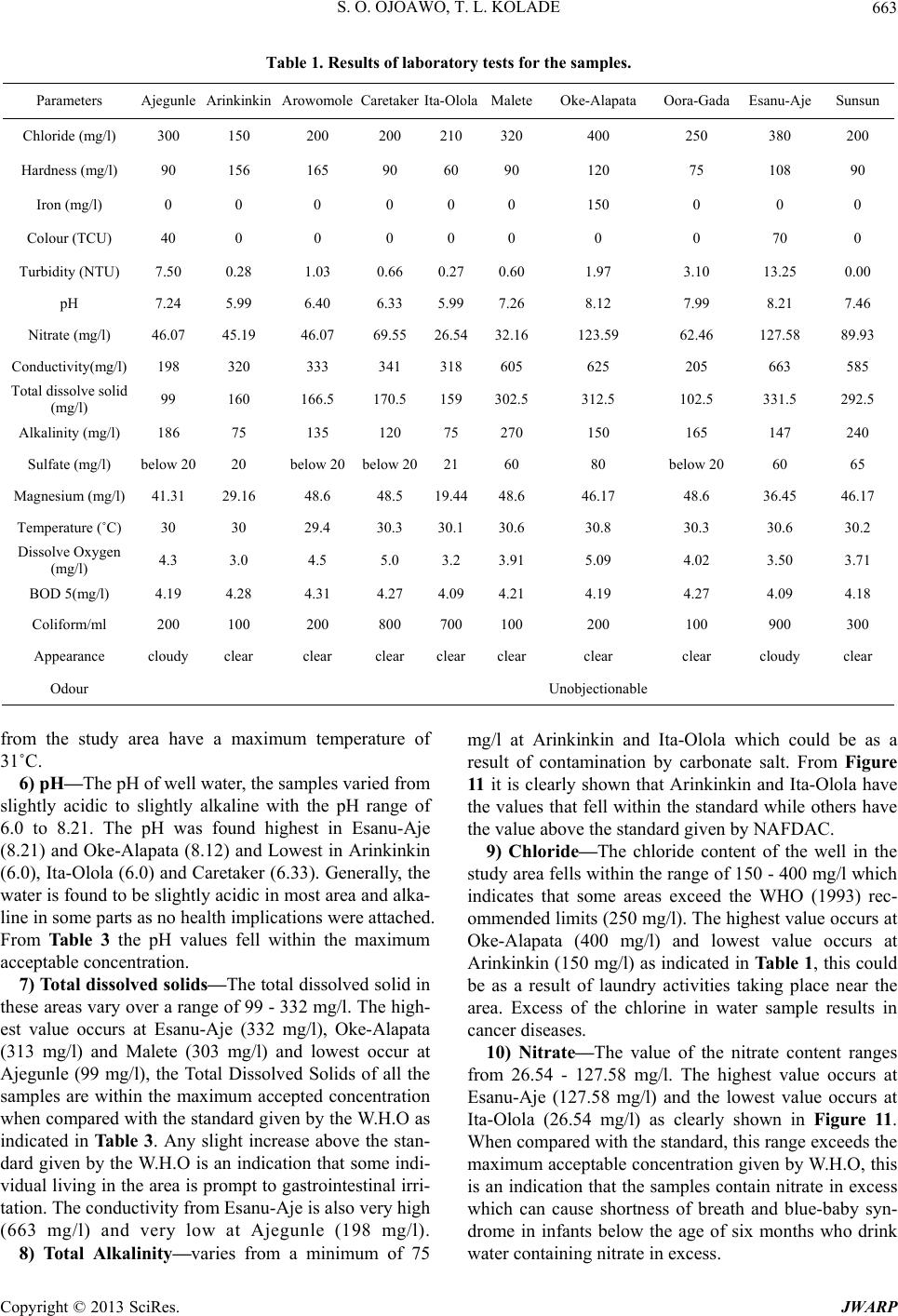 S. O. OJOAWO, T. L. KOLADE 663 Table 1. Results of laboratory tests for the sample s. Parameters Ajegunle Arinkinkin ArowomoleCaretakerIta-OlolaMaleteOke-Alapata Oora-Gada Esanu-Aje Sunsun Chloride (mg/l) 300 150 200 200 210 320 400 250 380 200 Hardness (mg/l) 90 156 165 90 60 90 120 75 108 90 Iron (mg/l) 0 0 0 0 0 0 150 0 0 0 Colour (TCU) 40 0 0 0 0 0 0 0 70 0 Turbidity (NTU) 7.50 0.28 1.03 0.66 0. 27 0.60 1.97 3.10 13.25 0. 00 pH 7.24 5.99 6.40 6.33 5.99 7.26 8.12 7.99 8.21 7.46 Nitrate (mg/l) 46.07 45.19 46.07 69.5526.5432.16123.59 62.46 127.58 89.93 Conductivity(mg/l) 198 320 333 341 318 605 625 205 663 585 Total dissolve solid (mg/l) 99 160 166.5 170.5159 302.5312.5 102.5 331.5 292.5 Alkalinity (mg/l) 186 75 135 120 75 270 150 165 147 240 Sulfate (mg/l) below 20 20 below 20 below 2021 60 80 below 20 60 65 Magnesium (mg/l) 41.31 29.16 48.6 48.5 19.4448.6 46.17 48.6 36.45 46.17 Temperature (˚C) 30 30 29.4 30.3 30.1 30.6 30.8 30.3 30.6 30.2 Dissolve Oxygen (mg/l) 4.3 3.0 4.5 5.0 3.2 3.91 5.09 4.02 3.50 3.71 BOD 5(mg/l) 4.19 4.28 4.31 4.27 4. 09 4.21 4.19 4.27 4.09 4.18 Coliform/ml 200 100 200 800 700 100 200 100 900 300 Appearance cloudy clear clear clear clear clear clear clear cloudy clear Odour Unobjectionable from the study area have a maximum temperature of 31˚C. 6) pH—The pH of well water, the samples varied from slightly acidic to slightly alkaline with the pH range of 6.0 to 8.21. The pH was found highest in Esanu-Aje (8.21) and Oke-Alapata (8.12) and Lowest in Arinkinkin (6.0), Ita-Olola (6.0) and Caretaker (6.33). Generally, the water is found to be slightly acidic in most area and alka- line in some parts as no health implications were attached . From Table 3 the pH values fell within the maximum acceptable concentration. 7) Tota l dissolved solids—The total d issolved solid in these areas vary over a range of 99 - 332 mg/l. The high- est value occurs at Esanu-Aje (332 mg/l), Oke-Alapata (313 mg/l) and Malete (303 mg/l) and lowest occur at Ajegunle (99 mg/l), the Total Dissolved Solids of all the samples are within the maximum accepted concentration when compared with the standard given by the W.H.O as indicated in Tabl e 3. Any slight increase above the stan- dard given by the W.H.O is an indication that some indi- vidual living in the area is prompt to gastro intestinal irri- tation. The conductivity from Esanu-Aje is also very high (663 mg/l) and very low at Ajegunle (198 mg/l). 8) Total Alkalinity—varies from a minimum of 75 mg/l at Arinkinkin and Ita-Olola which could be as a result of contamination by carbonate salt. From Figure 11 it is clearly shown that Arinkinkin and Ita-Olola have the values that fell within the standard while others have the value above the standard given by NAFDAC. 9) Chloride—The chloride content of the well in the study area fells within the range of 150 - 400 mg/l which indicates that some areas exceed the WHO (1993) rec- ommended limits (250 mg/l). The highest value o ccurs at Oke-Alapata (400 mg/l) and lowest value occurs at Arinkinkin (150 mg/l) as indicated in Table 1, this could be as a result of laundry activities taking place near the area. Excess of the chlorine in water sample results in cancer diseases. 10) Nitrate—The value of the nitrate content ranges from 26.54 - 127.58 mg/l. The highest value occurs at Esanu-Aje (127.58 mg/l) and the lowest value occurs at Ita-Olola (26.54 mg/l) as clearly shown in Figure 11. When compared with the standard, this range exceeds the maximum acceptable concentration given by W.H.O, this is an indication that the samples contain nitrate in excess which can cause shortness of breath and blue-baby syn- drome in infants below the age of six months who drink water containing nitrate in excess. Copyright © 2013 SciRes. JWARP 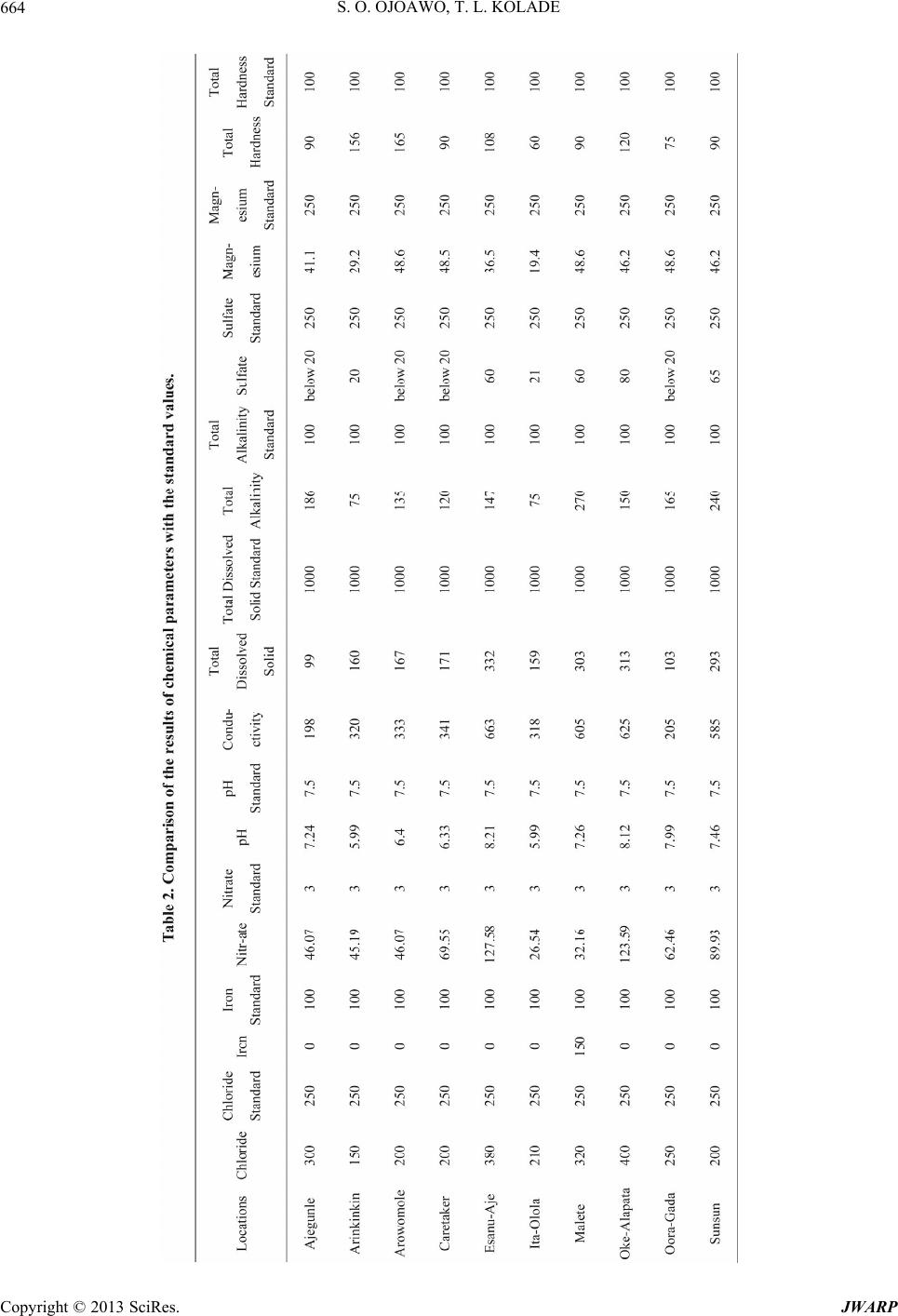 S. O. OJOAWO, T. L. KOLADE 664 Copyright © 2013 SciRes. JWARP 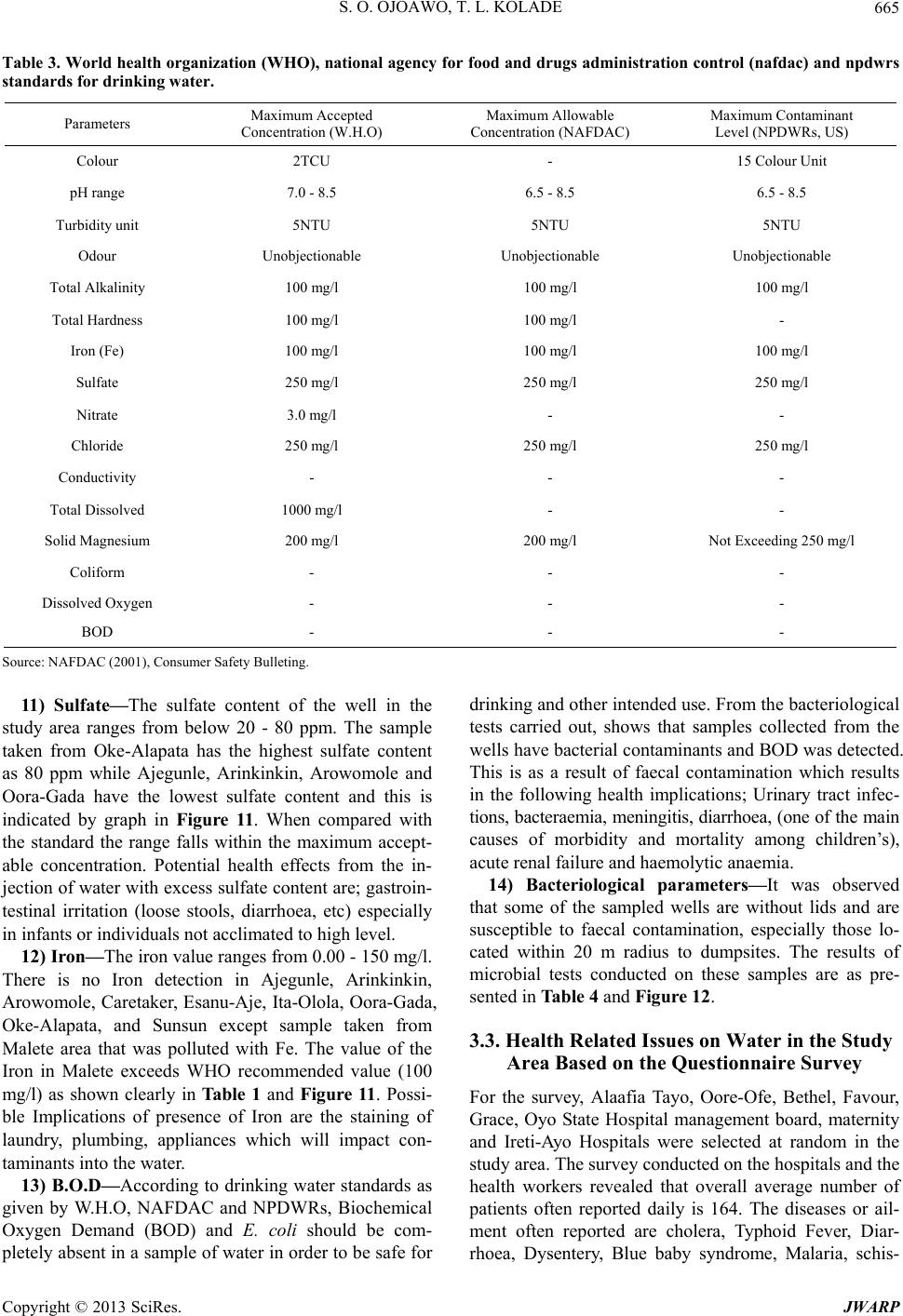 S. O. OJOAWO, T. L. KOLADE 665 Table 3. World health organization (WHO), national agency for food and drugs administration control (nafdac) and npdwrs standards for drinking water. Parameters Maximum Accepted Concentration (W.H.O) Maximum Allowable Concentration (NAFDAC) Maximum Contaminant Level (NPDWRs, US) Colour 2TCU - 15 Colour Unit pH range 7.0 - 8.5 6.5 - 8.5 6.5 - 8.5 Turbidity unit 5NTU 5NTU 5NTU Odour Unobjectionable Unobjectionable Unobjectionable Total Alkalinity 100 mg/l 100 mg/l 100 mg/l Total Hardness 100 mg/l 100 mg/l - Iron (Fe) 100 mg/l 100 mg/l 100 mg/l Sulfate 250 mg/l 250 mg/l 250 mg/l Nitrate 3.0 mg/l - - Chloride 250 mg/l 250 mg/l 250 mg/l Conductivity - - - Total Dissolved 1000 mg/l - - Solid Magnesium 200 mg/l 200 mg/l Not Exceeding 250 mg/l Coliform - - - Dissolved Oxygen - - - BOD - - - Source: NAFDAC (2001), Consumer Safety B ulleting. 11) Sulfate—The sulfate content of the well in the study area ranges from below 20 - 80 ppm. The sample taken from Oke-Alapata has the highest sulfate content as 80 ppm while Ajegunle, Arinkinkin, Arowomole and Oora-Gada have the lowest sulfate content and this is indicated by graph in Figure 11. When compared with the standard the range falls within the maximum accept- able concentration. Potential health effects from the in- jection of water with excess su lfate content are; gastroin- testinal irritation (loose stools, diarrhoea, etc) especially in infants or individuals not acclimated to high level. 12) Iron—The iron value ranges fro m 0.00 - 150 mg/l. There is no Iron detection in Ajegunle, Arinkinkin, Arowomole, Caretaker, Esanu-Aje, Ita-Olola, Oora-Gada, Oke-Alapata, and Sunsun except sample taken from Malete area that was polluted with Fe. The value of the Iron in Malete exceeds WHO recommended value (100 mg/l) as shown clearly in Ta ble 1 and Figure 11. Possi- ble Implications of presence of Iron are the staining of laundry, plumbing, appliances which will impact con- taminants into the water. 13) B.O.D—According to drinking water standards as given by W.H.O, NAFDAC and NPDWRs, Biochemical Oxygen Demand (BOD) and E. coli should be com- pletely absent in a sample of water in order to be safe for drinking and other intended use. From the bacteriological tests carried out, shows that samples collected from the wells have bacterial contaminants and BOD was detected. This is as a result of faecal contamination which results in the following health implications; Urinary tract infec- tions, bacteraemia, meningitis, diarrhoea, (one of the main causes of morbidity and mortality among children’s), acute renal failure and haemolytic anaemia. 14) Bacteriological parameters—It was observed that some of the sampled wells are without lids and are susceptible to faecal contamination, especially those lo- cated within 20 m radius to dumpsites. The results of microbial tests conducted on these samples are as pre- sented in Table 4 and Figure 12. 3.3. Health Related Issues on Water in the Study Area Based on the Questionnaire Survey For the survey, Alaafia Tayo, Oore-Ofe, Bethel, Favour, Grace, Oyo State Hospital management board, maternity and Ireti-Ayo Hospitals were selected at random in the study area. The survey conducted on the hospitals and the health workers revealed that overall average number of patients often reported daily is 164. The diseases or ail- ment often reported are cholera, Typhoid Fever, Diar- rhoea, Dysentery, Blue baby syndrome, Malaria, schis- Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 666 Table 4. Results of the DO, BOD and the coliforms. Locations Dissolved Oxygen mg/l BOD mg/l Coliform/ litre Ajegunle 4.3 4.19 200 Arinkinkin 3 4.28 100 Arowomole 4.5 4.31 200 Caretaker 5 4.27 800 Esanu-Aje 3.5 4.09 900 Ita-Olola 3.2 4.09 700 Malete 3.91 4.21 100 Oke-Alapata 5.09 4.19 200 Oora-Gada 4.02 4.27 100 Sunsun 3.71 4.18 300 tosomia, cancer, carcinogenic diseases. The respondent revealed that the major causes of the aforementioned diseases are; contaminated water, mosquito bites, tsetse flies and poor envir onmenta l managem e n t . As shown in Table 5, the respondents revealed that water can cause diseases and that water associated dis- eases are common in the study area. Figures 13 to 15 indicate the results of the responses of the Health Offi- cers on the water-related sicknesses . 1) Turbidity: is a measure of the degree to which the water loses its transparency due to the presence of sus- pended particulates. It is clearly shown from the results of analysis that sample taken from Esanu-Aje area is highly turbid and the water in the area is said to lose its transparency. 2) Chlorine: This causes environmental harm at low levels. Chlorine is especially harmful to organisms living in water and in soil. Results obtain from the analysis re- vealed that excessive chloride contents was noticed in Ajegunle, Arowomole, Caretaker, Esanu-Aje, Ita-Olola, Figure 13. Average number of patient reported by the hospitals in the study area. Figure 14. Possibility of water causing diseases. Copyright © 2013 SciRes. JWARP 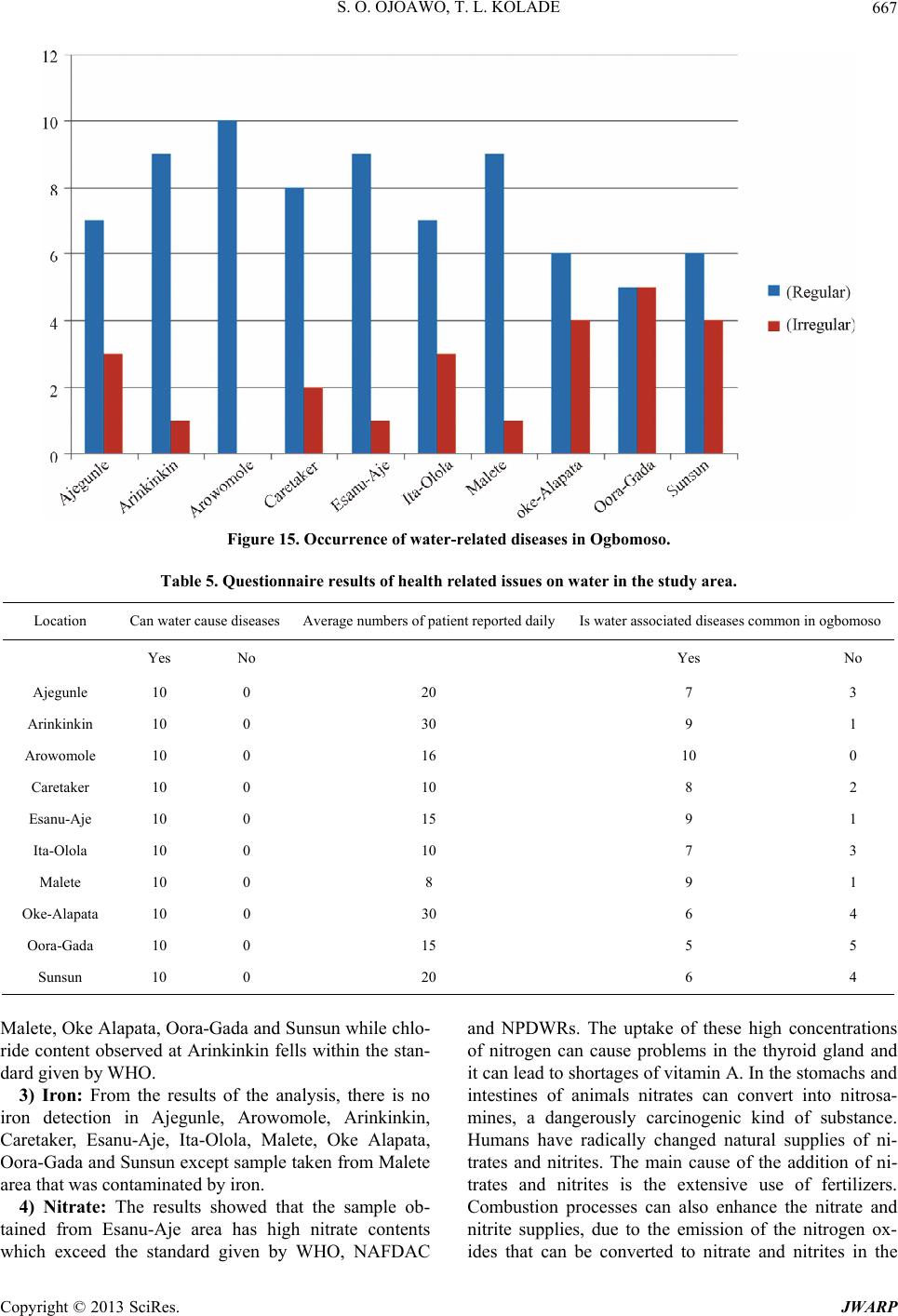 S. O. OJOAWO, T. L. KOLADE 667 Figure 15. Occurrence of water-related diseases in Ogbomoso. Table 5. Questionnaire results of health related issues on water in the study area. Location Can water cause diseases Average numbers of patient reported dailyIs water associated diseases common in ogbomoso Yes No Yes No Ajegunle 10 0 20 7 3 Arinkinkin 10 0 30 9 1 Arowomole 10 0 16 10 0 Caretaker 10 0 10 8 2 Esanu-Aje 10 0 15 9 1 Ita-Olola 10 0 10 7 3 Malete 10 0 8 9 1 Oke-Alapata 10 0 30 6 4 Oora-Gada 10 0 15 5 5 Sunsun 10 0 20 6 4 Malete, Oke Alapata, Oora-Gada and Sunsun while chlo- ride content observed at Arinkinkin fells within the stan- dard given by WH O. 3) Iron: From the results of the analysis, there is no iron detection in Ajegunle, Arowomole, Arinkinkin, Caretaker, Esanu-Aje, Ita-Olola, Malete, Oke Alapata, Oora-Gada and Sunsun except sample taken from Malete area that was contaminated by iron. 4) Nitrate: The results showed that the sample ob- tained from Esanu-Aje area has high nitrate contents which exceed the standard given by WHO, NAFDAC and NPDWRs. The uptake of these high concentrations of nitrogen can cause problems in the thyroid gland and it can lead to shortages of vitamin A. In the stomachs and intestines of animals nitrates can convert into nitrosa- mines, a dangerously carcinogenic kind of substance. Humans have radically changed natural supplies of ni- trates and nitrites. The main cause of the addition of ni- trates and nitrites is the extensive use of fertilizers. Combustion processes can also enhance the nitrate and nitrite supplies, due to the emission of the nitrogen ox- ides that can be converted to nitrate and nitrites in the Copyright © 2013 SciRes. JWARP  S. O. OJOAWO, T. L. KOLADE 668 environment. 5) Microbiological parameters: The pattern of the coliform levels in the samples is as presented in Figure 12. The measurement was exaggerated as coliform count per litre. The presence of coliform bacteria could be ac- counted for as a result of faecal contamination of the wells which are not covered. Again a few of the wells were carelessly located downstreams of the septic tanks in residential building s within a radius of 5 to 10 m. This gives rise to situations whereby traces of coliform were found in the samples thereby posing great health risks to the residents. 4. Conclusions The study reveals that one of the main sources of water supply to the residents of the study area is wells which fall into two categories namely shallow and deep well. The water supply in the study area is not fully sup portive to health as some measured parameters have values ex- ceeding those of the standards. The reason for this could be the fetching method through the use of ropes and buckets that exposes the wells to invisible contaminants and pollutants. Also the irregularity in the treatments of the wells is a major threat to the water quality. Water- related diseases like amoebic dysentery, typhoid fever, cholera etc. are therefore often reported at the surveyed hospitals. To abate the problems it is recommended that water for drinking should be subjected to treatment processes like coagulation and filtration to control excess turbidity and total solids, asid e boiling. Also, lime softening is also recommended for locations where samples indicate ex- cessive hardness in comparison with the standards. For areas with microbial contaminants, each household should endeavour to disinfect their wells least once in a month. Also, personal hygiene within each household must be improved and always emphasized. There is need for con- stant surveillance by public health officials to enforce water quality control measures and ensure that water delivered to consumers is at all times safe and potable. The personnel at the waterworks also need to optimize the available resources in terms of funds and equipment. Each component of water supply system from source through treatment process storage and distribution must be ascertained functioning well time to time without any risks or failure. REFERENCES [1] A. O. Olaniran and P. A. Oluwande, “Environmental and Health,” Nigerian Conservation Foundation, Vol. 3. No. 4, 1995, pp. 18-25. [2] P. A. Oluwande, “A Guide to Tropical Environment Health and Engineering,” National Institute of Science Education and Research, Ibadan, 1983, pp. 5-84. [3] O. Oyebande and A. Akinwale, “Nigeria’s Ground Water Resources-Extent, Value and Use,” Ultimate Water Tech- nology, Vol. 1, No. 1, 1996, pp. 41-45. [4] P. Morgan, “Drinking Water, Quality for Rural Areas,” Macmillan Publishers, Ibadan, 1990, pp. 248-252. [5] D. A. Aremu, “Impact of Petroleum Operation on Ground Water Quality in Warri,” M.S. Thesis, University of Iba- dan, Ibadan, 1988, pp. 45-56. [6] S. O. Ojoawo, O. A. Agbede and A. Y. San godoyin, “Co n- tamination Effects of Dumpsite Wastes on Surface and Groundwater Resource of Ogbomoso Local Government Areas,” LAUTECH Journal of Engineering and Technol- ogy, Vol. 6, No. 1, 2010, pp. 92-99. [7] J. O. Obemaeta, “Water Preservation and Treatment in: Water and Building Sanitation,” Claverium Press, Ibadan, 1991, pp. 20-41. [8] D. Chapman and V. Hall, “The Selection of Water Qual- ity Variables,” In: A Guide to the Use of Biota, Sediments and Water in Environmental Monitoring, World Health Organization, Geneva, 1987, p. 40. [9] E. A. Albert, “Chemical Composition of Bottled Mineral Water,” Archive of Environmental Health, Vol. 44, No. 2, 1987, p. 6. [10] USEPA, “Health Advisory Summaries on Water,” Office of Water, United States Environmental Protection Agency, Ohio, 2012, p. 23. [11] WHO, “Guidelines for Drinking Water,” World Health Organization, Geneva, 2011, p. 45. [12] NPC, “Population Figures,” National Population Com- mission,Abuja, 2006, p. 5. [13] S. Cairncross and R. G. Feachem, “Environmental Health Engineering in the tropics: An Introductory Text,” 2nd Edition, John Wiley, Chichester, 1997, p. 35. [14] NAFDAC, “Consumer Safety Bulletin,” National Agency for Food and Drug Administration and Control, Abuja, 2001, p. 7. Copyright © 2013 SciRes. JWARP |

