 Open Journal of Respiratory Diseases, 2013, 3, 44-51 http://dx.doi.org/10.4236/ojrd.2013.32008 Published Online May 2013 (http://www.scirp.org/journal/ojrd) Vitamin A, Vitamin E, Lutein and β-Carotene in Lung Tissues from Subjects with Chronic Obstructive Pulmonary Disease and Emphysema Michael W. Schäffer, Somdutta Sinha Roy, Shyamali Mukherjee, Salil K. Das* Department of Biochemistry and Cancer Biology, Meharry Medical College, Nashville, USA Email: *sdas@mmc.edu Received March 2, 2013; revised April 3, 2013; accepted April 10, 2013 Copyright © 2013 Michael W. Schäffer et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Vitamin A (VA) and its active metabolites play an essential role in lung airway function. Patients with moderate to se- vere chronic obstructive pulmonary disease (COPD) have a lower serum retinol concentration, and improvement of their 1-second Forced Expiratory Volume (FEV1) is achieved with VA supplementation. In order to test our hypothesis that the VA signaling pathway is compromised in COPD, we obtained 20 lung samples from COPD patients differing in the degree of emphysema as judged by their FEV% values. All were smokers or were exposed to secondhand smoke. Levels of VA (retinol/retinyl ester), tocopherols and carotenoids (lutein, beta-carotene) in these samples were deter- mined using HPLC. Additional analytes beside VA were included for their known roles as antioxidants and modulators of VA-action. VA levels (retinol/retinyl ester) decreased significantly with the increase in severity of emphysema. Among other analytes, α-tocopherol levels fell by 25.8% in the severe emphysema group in comparison to the mild emphysema group, and lutein levels similarly decreased in severe compared to moderate emphysema groups. However, beta-carotene levels remained unchanged. Thus there is a significant linear correlation between lung VA-levels and the severity of emphysema. There was also a significant reduction in the levels of α-, δ-tocopherol and lutein in the severe emphysema group of COPD patients who either smoked or were exposed to smoke. Keywords: COPD; β-Carotene; Emphysema; Lutein; Vitamin A; Vitamin E; HPLC 1. Introduction Chronic bronchitis and emphysema present two types of COPD. Presently, COPD is the fourth leading killer of adults in the US and likely to be the third leading cause of death by 2020 [1]. Approximately 12 million adults are diagnosed with COPD, 120,000 die from it each year, and an additional 12 million adults may have undiagnosed COPD [2]. As an added problem, smokers with COPD have a higher risk of lung cancer [3]. Previous studies suggested a link between a local res- piratory vitamin A deficiency and the severity of COPD [4]. The Second National Health and Nutrition Examina- tion Survey [5] documented that a smoker’s risk of de- veloping COPD is dose-dependently decreased by the intake of vitamin A. Vitamin A and its active metabolite all-trans retinoic acid (Tretinoin, ATRA) are important for growth and differentiation of many tissues/organs, including lung [6,7]. Further, retinoid signaling elements, such as receptors and binding proteins have been de- scribed in the adult lung [7]. Numerous findings suggest that ATRA may protect against emphysema [8,9]. Re- cently, several studies have shown that ATRA is effec- tive in promoting alveolization in papain-induced em- physema in dogs [10] and elastase-induced emphysema in adult rats [11]. We previously reported that exposure of guinea pigs to cigarette smoke caused an accumulation of lung retinol and a decrease in ATRA [12], suggesting an abnormality in retinoid metabolism and signaling. These findings raised the question as to whether smoke-associated COPD similarly result in abnormalities in retinoid me- tabolism and signaling by altering intracellular levels of vitamin A, and as a consequence ATRA, its active me- tabolite. Tocopherols (vitamin E) are a group of essential micronutrients that act mainly as lipophilic antioxidants which protect polyunsaturated fatty acids from oxidative damage, thus maintaining fluidity and integrity of cellu- *Corresponding author. C opyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. 45 lar membranes [13]. Since smokers subject their lungs to an increased burden of oxidative damage, a reduction in lung tocopherol-levels could be a consequence that con- tributes to the degree of severity of COPD. Although a systemic vitamin E deficiency is normally unseen in the Western world, circulating levels of vitamin E can be reduced in premature infants with bronchopulmonary dysplasia who are also selenium deficient [14]. Lutein, a dietary carotenoid that serves as a powerful antioxidant, plays an important role in ocular health and in the prevention of cardiovascular disease, stroke and lung cancer [15]. In previous studies we showed that lu- tein accumulates in guinea pig [16] and human lungs [17]. Although not much is known about the specific role of lutein in lung, a report [18] identified β-cryptoxanthin and lutein as natural ligands for the nuclear retinoic acid receptors (RAR)—especially RARα and RARγ. The ca- rotenoids β-carotene and lycopene failed to show a simi- lar activity. Although the role of dietary antioxidant β-carotene remains controversial [19]; an epidemiological study re- ported an inverse correlation between serum β-carotene and the incidence of lung cancer [20]. In contrast, two intervention trials (ATCB, CARET) showed that high doses of β-carotene given to smokers actually raised rather than lower their chances of developing lung cancer [21,22]. β-Carotene like lutein is deposited in human lung tissue [17]. We hypothesized that there is a lung deficiency of vi- tamin A and vitamin E and some carotenoids in COPD patients with emphysema, and further that there is a rela- tionship between the severity of emphysema and the lev- els of these nutrients. Dietary carotenoids were included because of their roles as potential effectors on the vita- min A-signaling pathway and contributors to the anti- oxidant capacity of the cells. We therefore analyzed lung tissues from 20 COPD pa- tients classified as having mild, moderate or severe em- physema. 2. Materials and Methods Human lung samples, preparation/extraction. Frozen human lung samples were obtained from the Lung Tissue Research Consortium (LTRC) of the National Heart, Lung, and Blood Institute, Bethesda, MD. Until extrac- tion for analysis, samples were stored at −80˚C. The LTRC provided us with de-identified information re- garding lung tissue donors including age, gender, smok- ing habits, FEV1% values, and the location from which the samples were taken. The characteristics of the human subjects whose biopsies were used for HPLC-analyses are described in Table 1. Nine subjects were categorized as having “severe emphysema” with FEV1% value <50 (average age was 52.6 ± 6.7 years), 8 subjects were cate- Table 1. Characteristics of subjects. Sample Tissue ID Gender* Age FEV1% Smoking Origin* 192361 F 50 12 yes RM 242420 M 63 14 yes LL 294945 F 52 15 yes RM 122541 F 55 19 yes RM 157421 M 46 20 yes RM 12861 M 51 22 yes LL 132625 F 63 24 yes RM 295167 M 49 27 yes LL 126571 M 44 40 yes RM 200930 F 78 52 yes RL 26458 M 73 54 yes RL 183561 M 83 57 yes RU 271851 M 77 68 yes LU 144462 F 73 69 yes RM 158231 F 61 72 yes LU 178967 F 72 72 yes RL 30753 M 75 77 no† LL 260111 F 61 84 yes RM 261361 F 63 89 yes RL 176562 M 73 105 yes LLO *Abbreviations: M = male; F = female; LLO = left lower; LL = left ling; LU = left upper; RL = right lower; RM = right middle; RU = right upper. †Secondhand smoke; FEV1%-range: <50 severe, 50 - 80 moderate, >80 mild. gorized as having “moderate emphysema” with FEV1% values ranging between 50 - 80 (average age was 72.3 ± 7.8 years), and 3 subjects were categorized as having “mild emphysema” with FEV1% values > 80% (average age was 65.7 ± 4.4 years). The average age of all 20 sub- jects was 62.9 ± 11.7 years; 52.4% were male and 47.6% were female. Amongst these subjects, only one was a non- smoker but was subjected to cigarette smoke at home. HPLC analysis, chemicals and reagents. Chemical and reagent resources, and High Pressure Liquid Chromatog- raphy (HPLC)-analyses were carried out as described earlier [17]. On average 100 mg of each lung was ho- mogenized, extracted and analyzed by HPLC for the analytes of 1) retinol and their found storage forms reti- nyl palmitate (RP) and retinyl stearate (RS), 2) toco- pherols (α, β/γ and δ) and 3) carotenoids (lutein and β-carotene). Values are expressed as mean ± standard error of the mean (SEM). Statistical analyses were per- formed using GraphPad Prism Version 5.04 (GraphPad software, San Diego, CA). Statistical significances (P- values) were calculated using an unpaired t-test (www. graphpad.com) to each of 8 analytes concentrations in Copyright © 2013 SciRes. OJRD 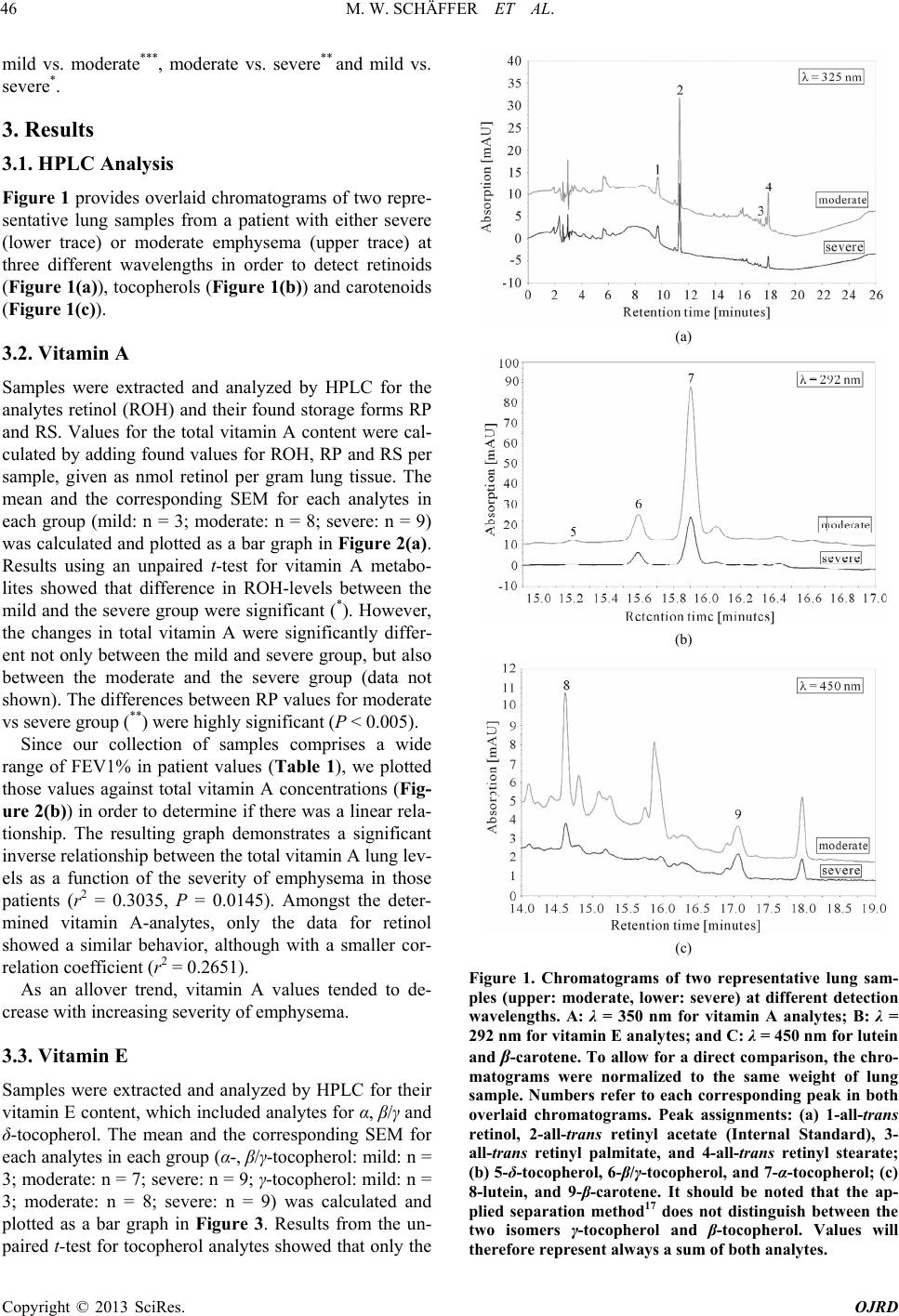 M. W. SCHÄFFER ET AL. 46 mild vs. moderate***, moderate vs. severe** and mild vs. severe*. 3. Results 3.1. HPLC Analysis Figure 1 provides overlaid chromatograms of two repre- sentative lung samples from a patient with either severe (lower trace) or moderate emphysema (upper trace) at three different wavelengths in order to detect retinoids (Figure 1(a)), tocopherols (Figure 1(b)) and carotenoids (Figure 1(c)). 3.2. Vitamin A Samples were extracted and analyzed by HPLC for the analytes retinol (ROH) and their found storage forms RP and RS. Values for the total vitamin A content were cal- culated by adding found values for ROH, RP and RS per sample, given as nmol retinol per gram lung tissue. The mean and the corresponding SEM for each analytes in each group (mild: n = 3; moderate: n = 8; severe: n = 9) was calculated and plotted as a bar graph in Figure 2(a). Results using an unpaired t-test for vitamin A metabo- lites showed that difference in ROH-levels between the mild and the severe group were significant (*). However, the changes in total vitamin A were significantly differ- ent not only between the mild and severe group, but also between the moderate and the severe group (data not shown). The differences between RP values for moderate vs severe group (**) were highly significant (P < 0.005). Since our collection of samples comprises a wide range of FEV1% in patient values (Table 1), we plotted those values against total vitamin A concentrations (Fig- ure 2(b)) in order to determine if there was a linear rela- tionship. The resulting graph demonstrates a significant inverse relationship between the total vitamin A lung lev- els as a function of the severity of emphysema in those patients (r2 = 0.3035, P = 0.0145). Amongst the deter- mined vitamin A-analytes, only the data for retinol showed a similar behavior, although with a smaller cor- relation coefficient (r2 = 0.2651). As an allover trend, vitamin A values tended to de- crease with increasing severity of emphysema. 3.3. Vitamin E Samples were extracted and analyzed by HPLC for their vitamin E content, which included analytes for α, β/γ and δ-tocopherol. The mean and the corresponding SEM for each analytes in each group (α-, β/γ-tocopherol: mild: n = 3; moderate: n = 7; severe: n = 9; γ-tocopherol: mild: n = 3; moderate: n = 8; severe: n = 9) was calculated and plotted as a bar graph in Figure 3. Results from the un- paired t-test for tocopherol analytes showed that only the (a) (b) (c) Figure 1. Chromatograms of two representative lung sam- ples (upper: moderate, lower: severe) at different detection wavelengths. A: λ = 350 nm for vitamin A analytes; B: λ = 292 nm for vitamin E analytes; and C: λ = 450 nm for lutein and β-carotene. To allow for a direct comparison, the chro- matograms were normalized to the same weight of lung sample. Numbers refer to each corresponding peak in both overlaid chromatograms. Peak assignments: (a) 1-all-trans retinol, 2-all-trans retinyl acetate (Internal Standard), 3- all-trans retinyl palmitate, and 4-all-trans retinyl stearate; (b) 5-δ-tocopherol, 6-β/γ-tocopherol, and 7-α-tocopherol; (c) 8-lutein, and 9-β-carotene. It should be noted that the ap- plied separation method17 does not distinguish between the two isomers γ-tocopherol and β-tocopherol. Values will therefore represent always a sum of both analytes. Copyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. 47 (a) (b) Figure 2. Vitamin A values (a) Bar graph of the mean and the corresponding SEM for the found values of retinol, ret- inyl palmitate and retinyl stearate, expressed in nmol reti- nol per gram wet weight lung tissue in each group (mild: n = 3; moderate: n = 8; severe: n = 9). Significances (P < 0.05) from unpaired t-test are indicated by *) for mild vs. severe and **) for moderate vs. severe values; (b) Demonstration of an inverse linear correlation between the degree of em- physema, expressed in FEV1%-values and the calculated amount of total vitamin A (= [ROH] + [RP] + [RS]) given in nmol retinol per gram wet weight lung tissue. changes in δ-tocopherol levels between the moderate and severe group (**, Figure 3(c)) were statistically highly significant (P < 0.005). Although not statistically sig- nificant, there was a decrease of 25.8% in α-tocopherol levels in the severe group compared to samples in the mild group (Figure 3(a)). Changes of β/γ-tocopherol- levels between the groups were not significantly different from each other, however amongst the tested vitamin E analytes; it is the only one that showed increased levels with increasing severity, for e.g. the β/γ-tocopherol-lev- els in the severe group were 21.8% higher on average than in the mild group (Figure 3(b)). 3.4. Carotenoids We also assayed for the carotenoids lutein and beta- carotene. The mean and the corresponding SEM for the analytes lutein (mild: n = 3; moderate: n = 7; severe: n = 9) and beta-carotene (mild: n = 3, moderate: n = 8, severe: n = 9) in each group was calculated and plotted as a bar graph (Figure 4). The levels of beta-carotene between (a) (b) (c) Figure 3. Vitamin E values. Bar graph of the mean and the corresponding SEM for the found values of (a) α-tocopherol; (b) β/γ-tocopherol and (c) δ-tocopherol, expressed in g per gram wet weight lung tissue in each group (α-, β/γ-toco- pherol: mild: n = 3; moderate: n = 7; severe: n = 9; δ-toco- pherol: mild: n = 2; moderate: n = 8; severe: n = 9). Signifi- cances (P < 0.05) from unpaired t-test are indicated by **) for moderate vs. severe. It should be noted that α-toco- pherol-levels are found to be an order of magnitude higher than those for γ/β-tocopherol, which are again an order of magnitude higher than in the detected δ-tocopherol-levels. the groups did not change. In contrast, changes in lutein levels between the moderate and severe group were sta- tistically significant (**, Figure 4). Interestingly, lung tissue harvested from subjects in the moderate group accumulated almost twice the lutein than samples from the other two groups. 4. Discussion There have been a number of human studies that have Copyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. 48 Figure 4. Carotenoid values. Bar graph of the mean and the corresponding SEM for the found values of lutein and β- carotene, expressed in μg per gram wet weight lung tissue in each (lutein: mild: n = 3, moderate: n = 7, severe: n = 9; β-carotene: mild: n = 3, moderate: n = 8, severe: n = 9), Significances (P < 0.05) from unpaired t-test are indicated by **) for moderate vs. severe lutein values. attempted to correlate intake of various micronutrients to the outcomes of COPD, asthma, bronchopulmonary dys- plasia and lung cancer using either by nutritional surveys or blood chemistries with a variety of outcomes and con- clusions [4,5]. They lacked, however, information about the concentration and composition of the analytes in the lung tissue, per se. To our knowledge, we report here for the first time the HPLC-analysis of micronutrients vita- mins A and E and the carotenoids lutein and β-carotene in lung samples from human COPD subjects with vary- ing degrees of emphysema. In the case of vitamin A and its metabolites, earlier studies showed that the risk for COPD increased with decreasing serum vitamin A [23,24]; and that daily oral doses of vitamin A (25,000 IE) for 30 days attenuated the symptoms [25]. Although the overall homeostatic regulation of plasma retinol values was unaffected by the pathologic condi- tions of COPD, Paiva et al. [4] found a positive correla- tion between serum retinyl esters and FEV1/FVC-values in healthy smokers and in patients with moderate to se- vere COPD; again there was an improvement in pulmo- nary functions after vitamin A supplementation. Al- though smokers with COPD usually do not have a sys- temic vitamin A deficiency [4], a local deficiency may occur as a result of chronic exposure to cigarette smoke. Our results show that lung vitamin A levels fell with the progression of disease, as judged by FEV1% values. This local vitamin A deficiency may be caused by an increased demand for its active form ATRA, followed by a depletion of intra-cellular retinyl ester stores. We have found a highly statistically significant decrease in RP levels in the severe vs. moderate groups. This could be explained by an increased preference of retinyl ester hy- drolase towards the palmitate-ester over the stearate-ester. Indeed, a report [26] showed that palmitate is the pre- ferred fatty acyl moiety over stearate. In addition, exposure to the environmental toxicant benzo [a] pyrene decreased the uptake of vitamin A into lung cells [27] and led to a local vitamin A deficiency in lung tissues [28]. The cellular uptake of retinol into tar- get tissue is mainly mediated by the membrane-bound STRA6-receptor [29]. Since vitamin A is depleted in lungs of COPD-patients, ATRA-levels could also be sig- nificantly reduced. This is important because ATRA is required for the activation of STRA6-gene expression [29]. Thus, it seems likely that reduced STRA6-levels would aggravate a local vitamin A-deficiency. We are currently investigating this question in our laboratory. Since a systemic supplementation may have severe toxic side effects, others have proposed the uptake of either vitamin A or ATRA in the lung by inhalation. Bi- esalski et al. [30] demonstrated that inhalation of an RP- aerosol can successfully improve the vitamin A status of preschool children. Furthermore, Kohlhäufl et al. [31] reported a complete reversal of metaplasia/dysplasia in 44% (and partial remission in 12%) of biopsies from smokers with premalignant lesions of the bronchial epi- thelium who were administered aerosolized vitamin A. Interestingly, a recent case study [32] on a 45-year-old patient with severe emphysema, a never smoker individ- ual on a 6-month controlled ATRA-inhalation therapy, showed signs of improvement in daily life in terms of the hours spent without oxygen supplementation and a re- duction in inflammatory markers such as serum IL-13. Although our data do not reveal if the local vitamin A-deficiency is a cause or a consequence of COPD, a therapy with inhalable formulations of vitamin A may be beneficial for the alleviation of symptoms while avoiding potential side effects by administration of this vitamin. However a recent study demonstrated the increased sus- ceptibility to the development of cigarette smoke-in- duced lung emphysema in mice with low but not defi- cient vitamin A status [33]. The most abundant tocopherol in the human lung, α- tocopherol, fell 25.8% from values in the mild to severe groups. The reduced availability of this antioxidant may be scientifically important, since these patients face an increased oxidative stress burden due to cigarette smoke. In a recent case-control study with 1088 lung cancer pa- tients and 1414 healthy matched controls, investigators [34] reported that an increased intake of dietary α-toco- pherol accounted for a 34% - 53% reduction in lung cancer risk. Thus the found 25.8%-reduction in α-toco- pherol levels in lungs in our study may be a critical risk factor for the development of lung cancer in COPD pa- tients. Since α-tocopherol inhibits retinyl ester hydrolase (REH) activity [35], the drop in α-tocopherol-levels may also reduce its ability to inhibit REH, resulting in a re- duction in RP. There is emerging evidence that tocopherols in addi- Copyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. 49 tion to their antioxidant properties have other distinct functions, for example they can have direct and indirect effects on several enzymes involved in signal transduc- tion [36] and anti-inflammatory effects [37]. Recent evi- dence suggested that δ- and α-tocopherol and their me- tabolites are more potent inhibitors of cyclooxygenase-2 (COX-2) than α-tocopherol [37,38]. Interestingly, an- other report [39] showed that the expression of COX-2 in lungs of COPD patients with lung cancer was signifi- cantly increased compared to control subjects. This study together with our observation of finding decreased α- and δ-tocopherol levels in these patients, suggest the in- volvement of low tocopherols and high COX-2 levels in COPD-related lung cancer. Our findings that lutein levels significantly decrease whereas beta-carotene levels do not change cannot be simply explained by differences in dietary consumption. There is increasing evidence that the uptake, distribution and their function in the body are tightly regulated by different enzymes. Beta-carotene, for e.g. is transported primarily in very low-density lipoproteins (VLDL) and low-density lipoproteins (LDL), whereas lutein has a preference for high-density lipoproteins (HDL) [40]. In combination with specific cell surface receptors, the apolipoprotein components of the lipoproteins assist the carotenoids to their specific target tissue. Cellular LDL- receptors mediate beta-carotene uptake [40], whereas lutein uses class B scavenger receptors [41]. Interestingly, the steroidogenic acute regulatory domain 3 (StARD3) protein, also known as MLN64, has recently been identi- fied as a lutein-binding protein in the human retinal mac- ula [40]. Furthermore, StARD3 is involved in intra-cel- lular cholesterol transport [41]. Since StARD3 is ex- pressed in many tissues including lung [42], we hypothe- size that this protein may mediate lutein uptake in lung and when its function is compromised under significant oxidative stress, this leads to a reduction of lung lutein levels. Our overall results suggest that with increasing sever- ity of emphysema in COPD patients, the levels of retinol and total vitamin A (retinol + ester) are reduced in lung. Together with changes seen in lutein and α- and δ-toco- pherol, critical components of the cellular antioxidative defense have been reduced. Among the COPD subjects included in this study, most were firsthand smokers, only one was secondhand smoker and none represented nonsmoker. Also there was no non-diseased control group. The limitation of the cur- rent study needs to be controlled by inclusion of these additional groups. It will be interesting to investigate whether blood levels of the nutrients of the COPD sub- jects vary with the degree of emphysema. Furthermore, we are currently investigating whether expression of cer- tain mediators of retinoid action (trafficking, metabolism and signaling) in lung of COPD patients vary with the degree of emphysema. 5. Acknowledgements This work was supported by NIH grant 1R03 HL095419- 01A1. REFERENCES [1] G. Veigi, A. Scognamiglio, S. Baldacci, F. Pistelli and L. Carrozzi, “Epidemiology of Chronic Obstructive Pulmo- nary Disease (COPD),” Respiration, Vol. 68, No. 1, 2001, pp. 4-19. doi:10.1159/000050456 [2] C. Roth, “Factsheet Chronic Obstructive Pulmonary Dis- ease (COPD),” National Institutes of Health, Vol. 10, 2010. http://report.nih.gov/NIHfactsheets/Pdfs/ChronicObstruct ivePulmonaryDisease(NHLBI).pdf [3] S. K. Das, “Harmful Effects of Cigarette Smoking,” Mo- lecular and Cellular Biochemistry, Vol. 253, No. 1, 2003, pp. 159-165. doi:10.1023/A:1026024829294 [4] S. A. Paiva, I. Godoy, H. Vannucchi, R. M. Favaro, R. R. Geraldo and A. O. Campana, “Assessment of Vitamin A status in Chronic Obstructive Pulmonary Disease Patients and Healthy Smokers,” American Journal of Clinical Nu- trition, Vol. 64, No. 6, 1996, pp. 928-934. [5] A. McDowell, A. Engle, J. T. Massey and K. R. Maurer, “Plan and Operation of the Second Health and Nutrition Examination Survey, 1976-80,” Vital and Health Statis- tics, Vol. 1, No. 15, 1981, pp. 81-1317. [6] D. E. Ong and F. Chytil, “Changes in Levels of Cellular Retinol- and Retinoic-Acid-Binding Proteins of Liver and Lung during Perinatal Development of Rat,” Proceedings of National Academy of Sciences USA, Vol. 73, No. 11, 1976, pp. 3976-3978. doi:10.1073/pnas.73.11.3976 [7] F. Chytil, “The Lungs and Vitamin A,” American Journal of Physiology, Vol. 261, No. 5, 1992, pp. L517-527. [8] N. R. MacIntyre, “Chronic Obstructive Pulmonary Dis- ease: Emerging Medical Therapies,” Respiratory Care, Vol. 49, No. 1, 2004, pp. 64-69. [9] W. C. Bailey and D. P. Tashkin, “Pharmacologic Therapy: Novel Approaches for Chronic Obstructive Pulmonary Disease,” Annals of the American Thoracic Society, Vol. 4, No. 7, 2007, pp. 543-548. doi:10.1513/pats.200701-017FM [10] F. S. Rosenthal, “Aerosol Probes of Emphysema Progres- sion in Dogs Treated with All-Trans Retinoic Acid—An Exploratory,” Journal of Aerosol Medicine, Vol. 20, No. 2, 2007, pp. 83-96. doi:10.1089/jam.2007.0581 [11] G. D. Massaro and D. Massaro, “Retinoic Acid Treatment Abrogates Elastase-Induced Pulmonary Emphysema in Rats,” Nature Medicine, Vol. 3, No. 6, 1997, pp. 675-677. doi:10.1038/nm0697-675 [12] S. Mukherjee, T. Nayyar, F. Chytil and S. K. Das, “Main- stream and Sidestream Cigarette Smoke Exposure Increases Retinol in Guinea Pig Lungs,” Free Radical Biology and Medicine, Vol. 18, No. 3, 1995, pp. 507-514. doi:10.1016/0891-5849(94)00161-C Copyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. 50 [13] H. J. Nelis, E. D’Haese and K. Vermis, “Vitamin E,” In: A. P. De Lenheer, W. E. Lambert and J. F. Van Bocxlaer, Eds., Modern Chromatographic Analysis of Vitamins, 3rd Edition, Marcel Dekker, New York, 2000, pp. 143-228. [14] H. S. Falciglia, J. R. Johnson, J. Sullivan, et al., “Role of Antioxidant Nutrients and Lipid Peroxidation in Prema- ture Infants with Respiratory Distress Syndrome and Bron- chopulmonary Dysplasia,” American Journal of Perina- tology, Vol. 20, No. 2, 2003, pp. 97-107. doi:10.1055/s-2003-38315 [15] F. M. Painter, “Lutein and Zeaxanthin,” Alternative Medi- cine Review, Vol. 10, No. 2, 2005, pp. 128-135. [16] M. W. Schäffer, S. Sinha Roy, M. Mukherjee and S. K. Das, “Identification of Lutein, a Dietary Antioxidant Ca- rotenoid in Guinea Pig Tissues,” Biochemical and Bio- physical Research Communications, Vol. 374, No. 2, 2008, pp. 378-381. doi:10.1016/j.bbrc.2008.07.030 [17] M. W. Schäffer, S. S. Roy, S. Mukherjee, et al., “Qualita- tive and Quantitative Analysis of Retinol, Retinyl Esters, Tocopherols and Selected Carotenoids Out of Various In- ternal Organs from Different Species by HPLC,” Ana- lytical Methods, Vol. 2, No. 9, 2010, pp. 1320-1332. doi:10.1039/c0ay00288g [18] A. Matsumoto, H. Mizukami, S. Mizuno, et al., “Beta- Cryptoxanthin, a Novel Natural RAR Ligand, Induces ATP-Binding Cassette Transporters in Macrophages,” Bio- chemical Pharmacology, Vol. 74, No. 2, 2007, pp. 256-264. doi:10.1016/j.bcp.2007.04.014 [19] J. E Dowling and G. Wald, “The Biological Function of Vitamin A Acid,” Proceedings of the National Academy of Sciences USA, Vol. 46, No. 5, 1960, pp. 587-608. doi:10.1073/pnas.46.5.587 [20] R. Ziegler, S. Mayne and C. Swanson, “Nutrition and Lung Cancer,” Cancer Causes Control, Vol. 7, No. 1, 1996, pp. 157-177. doi:10.1007/BF00115646 [21] The Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study Group, “The Effect of Vitamin E and Beta Caro- tene on the Incidence of Lung Cancer and Other Cancers in Male Smokers,” New England Journal of Medicine, Vol. 330, No. 15, 1994, pp. 1029-1035. doi:10.1056/NEJM199404143301501 [22] G. S. Omenn, G. E. Goodman, M. D. Thornquist, et al., “Effects of a Combination of β-Carotene and Vitamin A on Lung Cancer and Cardiovascular Disease,” New Eng- land Journal of Medicine, Vol. 334, 1996, pp. 1150-1155. doi:10.1056/NEJM199605023341802 [23] A. Morabia, A. Sorenson, S. K. Kumanyika and H. Abbey, “Vitamin A, Cigarette Smoking and Airway Obstruction,” American Review of Respiration Disease, Vol. 140, No. 5, 1989, pp. 1312-1316. doi:10.1164/ajrccm/140.5.1312 [24] A. Morabia, M. J. Menkes, G. W. Comstock and M. S. Tockman, “Serum Retinol and Airway Obstruction,” Ame- rican Journal of Epidemiology, Vol. 132, No. 1, 1990, pp. 77-82. [25] J. Lorenz and H. K Biesalski, “Vitamin A-Mangel und Bronchialkarzinom: Perspektiven der Chemoprevention bronchialer Tumoren,” Pneumologie, Vol. 47, No. 12, 1993, pp. 657-665. [26] J. R. Mata, N. L. Mata and A. T. C. Tsin, “Substrate Specificity of Retinyl Ester Hydrolase Activity in Retinal Pigment Epithelium,” Journal of Lipid Research, Vol. 39, 1998, pp. 604-612. [27] T. E. Edes, S. M. Kwan, C. S. Buckley and W. H. Thorn- ton, “Tissue Vitamin A Repletion Is Impaired by Expo- sure to Carcinogen,” International Journal of Cancer, Vol. 50, No. 1, 1992, pp. 99-102. doi:10.1002/ijc.2910500120 [28] T. E. Edes and D. S. Gysbers, “Carcinogen-Induced Tis- sue Vitamin A Depletion. Potential Protective Advan- tages of β-Carotene,” Annals of the New York Academy of Sciences, Vol. 686, 1993, pp. 203-212. doi:10.1111/j.1749-6632.1993.tb39176.x [29] R. Kawaguchi, J. Yu, J. Honda, et al., “A Membrane Receptor for Retinol Binding Protein Mediates Cellular Uptake of Vitamin A,” Science, Vol. 315, No. 5831, 2007, pp. 820-825. doi:10.1126/science.1136244 [30] H. K. Biesalski, R. Reifen, F. Pürst and M. Edris, “Reti- nyl Palmitate Supplementation by Inhalation of an Aero- sol Improves Vitamin A Status of Preschool Children in Gondar (Ethiopia),” British Journal of Nutrition, Vol. 82, No. 3, 1999, pp. 179-182. [31] M. Kohlhäufl, K. Häussinger, F. Stanzel, et al., “Inhalation of Aerosolized Vitamin A: Reversibility of Metaplasia and Dysplasia of Human Respiratory Epithelia a Prospec- tive Pilot Study,” European Journal of Medical Research, Vol. 7, No. 2, 2002, pp. 72-78. [32] M. Frankenberger, I. Heimbeck, W. Möller, et al., “Inhaled All-Trans Retinoic Acid in an Individual with Severe Em- physema,” European Respiratory Journal, Vol. 34, No. 6, 2009, pp. 1487-1489. doi:10.1183/09031936.00105309 [33] S. Van Eijl, E. Mortaz, C. Versluis, F. P. Nijkamp, G. Folkerts and N. Bloksma, “A Low Vitamin A Status In- creases the Susceptibility to Cigarette Smoke-Induced Lung Emphysema in C57BL/6J Mice,” Journal of Physi- ology and Pharmacology, Vol. 62, No. 2, 2011, pp. 175- 182. [34] S. Mahabir, K. Schendel, Y. Q. Dong, S. L. Barrera, M. R. Spitz and M. R. Forman, “Dietary α-, β-, γ- and δ-Toco- pherols in Lung Cancer Risk,” International Journal of Cancer, Vol. 123, No. 5, 2008, pp. 1173-1180. doi:10.1002/ijc.23649 [35] J. L. Napoli, A. M. McCormick, B. O’Meara and E. A. Dratz, “Vitamin A Metabolism: Alpha-Tocopherol Modu- lates Tissue Retinol Levels in Vivo, and Retinyl Palmitate Hydrolysis in Vitro,” Archives of Biochemistry and Bio- physics, Vol. 230, No. 1, 1984, pp. 194-202. doi:10.1016/0003-9861(84)90100-0 [36] J. M. Zingg, “Modulation of Signal Transduction by Vi- tamin E,” Molecular Aspects of Medicine, Vol. 28, No. 5-6, 2007, pp. 481-506. doi:10.1016/j.mam.2006.12.009 [37] E. Reiter, Q. Jiang and S. Christen, “Anti-Inflammatory Properties of α- and γ-Tocopherol,” Molecular Aspects of Medicine, Vol. 28, No. 5-6, 2007, pp. 668-691. doi:10.1016/j.mam.2007.01.003 [38] Q. Jiang, I. Elson-Schwab, C. Courtemanche and B. N. Ames, “Gamma-Tocopherol and Its Major Metabolite, in Contrast to Alpha-Tocopherol, Inhibit Cyclooxygenase Activity in Macrophages and Epithelial Cells,” Proceed- Copyright © 2013 SciRes. OJRD  M. W. SCHÄFFER ET AL. Copyright © 2013 SciRes. OJRD 51 ings of the National Academy of Sciences of the United States of America, Vol. 97, No. 21, 2000, pp. 11494-11499. doi:10.1073/pnas.200357097 [39] J. Roca-Ferrer, L. Pujols, C. Agusti, et al.., “Cyclooxi- genase-2 Levels Are Increased in the Lung Tissue and Bronchial Tumors of Patients with Chronic Obstructive Pulmonary Disease,” Archivos de Bronconeumología, Vol. 47, No. 12, 2011, pp. 584-589. doi:10.1016/j.arbres.2011.05.015 [40] C. M. Greene, D. Waters, R. M. Clark, J. H. Contois and M. L. Fernandez, “Plasma LDL and HDL Characteristics and Carotenoid Content Are Positively Influenced by Egg Consumption in an Elderly Population,” Nutrition & Me- tabolism, Vol. 3, No. 6, 2006, pp. 6-14. doi:10.1186/1743-7075-3-6 [41] B. Li, P. Vachali, J. M. Frederick and P. S. Bernstein, “Identification of StARD3 as a Lutein-Binding Protein in the Macula of the Primate Retina,” Biochemistry, Vol. 50, No. 13, 2011, pp. 2541-2549. doi:10.1021/bi101906y [42] H. Watari, F. Arakane, C. Moog-Lutz, et al.., “MLN64 Contains a Domain with Homology to the Steroidogenic Acute Regulatory Protein (StAR) that Stimulates Steroi- dogenesis,” Proceedings of the National Academy of Sci- ences of the United States of America, Vol. 94, No. 16, 1997, pp. 8462-8467.
|