Advances in Microbiology
Vol.2 No.4(2012), Article ID:26243,5 pages DOI:10.4236/aim.2012.24082
Occurrence of Methicillin-Resistant Staphylococcus aureus in Cheese Produced in German Farm-Dairies
1Faculty 2, Department of Microbiology,University of Applied Sciences and Arts, Hanover, Germany
2Animal Health and Animal Welfare, Faculty of Agricultural and Environmental Sciences, University of Rostock, Rostock, Germany
Email: *claudia.zinke@hs-hannover.de
Received September 18, 2012; revised October 23, 2012; accepted November 19, 2012
Keywords: Cheese Products; Staphylococcus aureus; MRSA; Small-Scale Processing Plant
ABSTRACT
The aim of this study was to determine if raw milk cheese or cheese to be made out of heated milk, manufactured in small German cheese dairies might be a reservoir of contaminations with methicillin-resistant Staphylococcus aureus (MRSA). Due to predominantly manual handling during cheese making, recontaminations with Staphylococcus (S.) aureus or MRSA cannot be excluded. Commercial cheese products (n = 72) available in the region of Hanover (Lower Saxony) and dairy products offered via internet were analyzed with regard to the occurrence of S. aureus and MRSA. Thereof, two cheese samples, manufactured from pasteurized milk and two samples, produced from raw milk, were S. aureus positive with CFU/g between 1.0 × 101 and 7.0 × 101. MRSA was not detected. All analysed cheese samples could be considered safe for consuming.
1. Introduction
Staphylococcus (S.) aureus is a Gram-positive, coagulase and katalase positive coccus producing several of virulence factors as well as toxins [1]. S. aureus is regularly detected in bulk milk samples as a major contagious pathogen causing intramammary infections in dairy cattle [2-4]. Pasteurization of milk leads to kill this micro-organism but due to the ability to produce heat-stable enterotoxins, S. aureus is a common cause of food poisoning [5]. Milk and milk products, such as cheese provide a good source of S. aureus contaminations. Therefore, European regulations require a concentration of S. aureus of <1.0 × 103 CFU/g in final cheese products manufactured from heated milk and for cheese products from raw milk, regulations require a limit value of <1.0 × 105 CFU/g [6]. Various studies indicated the presence of S. aureus in artisan cheese products manufactured in smallscale farm dairies [7,8]. Furthermore, André et al. [9] could isolate S. aureus from cheese manufacturers in a dairy processing plant. Approximately 75% (3/4) of the investigated persons were colonized with S. aureus. The strains were recovered from both hand and nose samples.
With the introduction of methicillin for antibiotic treatment of S. aureus infections in humans, 60 years ago, strains became resistant to this therapeutic agent. Today, methicillin-resistant Staphylococcus aureus (MRSA) is an important agent causing both hospital-acquired (haMRSA) and community-acquired (caMRSA) infections [10-12]. The methicillin resistance of S. aureus is caused by a specific penicillin-binding protein (PBP 2a) which is encoded by the mecA gene and has a low affinity to β- lactam antibiotics [13]. Due to limited options of treatment, MRSA infections turn out to be a high risk to human health because strains exhibit resistances to β-lactam antibiotics and other groups of antimicrobials.
First Voss et al. [14] reported on a new MRSA clone in humans called ST398 and associated with pig farming with a high potential of transmission to humans in contact with infected animals [15-17]. From that time on, the clone has also been detected in other livestock such as cattle and poultry [18-20]. Comprehensive data about the prevalence of ST398 are available in pigs. Various studies have been done on all production levels with detections of high incidence of this clone [21-24]. MRSA, predominantly ST398, could also be detected in raw meat products of different species, e.g. beef, pork, chicken and turkey [21].
MRSA ST398 has also been identified in dairy cow udders. Vicca et al. [20] reported of a prevalence of MRSA positive animals between 0 and approximately 14.0% of lactating dairy cows on herd-level. Overall, samples were taken from five dairies, which were proven positive for MRSA in previous studies. Isolates could be identified as ST398 spa t011, which also could be detected in pigs, poultry and in humans with close contact to infected animals [18,22-24]. MRSA ST398 spa t011 has also been isolated in three dairies in Southwest Germany. The prevalence on herd-level of this agent ranged between 5.1% and 16.7% [19]. In addition to meat products, MRSA contaminations were also recovered in dairy foodstuff e.g. bovine milk, mozzarella and pecorino cheeses [25].
Thus, it can be assumed that raw milk for cheese making could be a reservoir for MRSA contamination in final food products. In addition, MRSA-colonized staff could be considered as a source of contaminations in cheese. Particularly, due to predominantly manual operations during cheese making in farms with small-scale production, recontamination of final products by colonized staff could be a health risk for consumers. At present, reliable data remain unavailable.
The aim of the present study was therefore to determine if raw milk artisan cheese or cheese to be made out of pasteurized milk, manufactured in small German cheese dairies might be reservoir of contaminations with MRSA.
2. Materials
In a period of five month all available commercial cheese products were collected. Each product was included once in the study. Cause of low number of small-scale farm dairies in the region of Hanover, additional, all smallscale German processing plants which offered their cheese products by shipment via internet were examined. Numbers of cheese samples of same dairies varied between one and seven, depending on assortment of the retailers or manufacturers. A total of 72 cheese samples were obtained. 36 samples were purchased from dairy counters or refrigerated displays in grocery stores or farmer's markets. Another 36 samples were obtained directly from manufacturers via internet. Cheese samples originated from small-scale German processing plants in Lower Saxony, North Rhine-Westphalia, Baden-Württemberg, Hesse, Bavaria and Schleswig-Holstein. In total, eight cream cheeses, five soft cheeses, eight semi-hard cheeses, one hardand one processed cheeses, produced from pasteurized milk (Table 1) and nine soft cheeses, twentyeight semi-hardand twelve hard cheeses, made from raw milk, were evaluated (Table 2).
3. Methods
For quantitative analyses of S. aureus and MRSA, 25 g of each sample were homogenized in 225 mL bufferedpeptone water (Roth, Germany) in a stomacher (AES Chemunex) for 60 s. Decimal dilutions were plated onto Baird-Parker agar (Merck) with egg yolk tellurite emulsion (Oxoid, Germany) for detection of S. aureus (ISO 6888-1:1999). Inoculated agar plates were incubated at 37˚C for 48 h.
Quantitative detection of MRSA was carried out according to §64 LFGB L 00.100 (MPN technique). Threefold analysis of 1 mL of each decimal dilution was done by inoculating in 9 mL Mueller Hinton broth (Oxoid, Germany) with 6% NaCl, incubated at 37˚C for
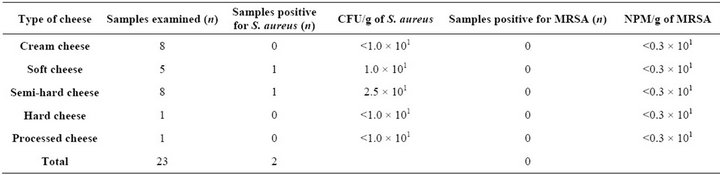
Table 1. Bacteria counts of S. aureus and MRSA recovered in cheese samples manufactured from pasteurized milk.

Table 2. Bacteria counts of S. aureus and MRSA recovered in cheese samples manufactured from raw milk.
48 h. Afterwards, samples were streaked in duplicate on Brilliance MRSA 2 agar (Oxoid, Germany) and incubated at 37˚C for 24 h. Blue colonies were considered as presumptive MRSA. For further analyses, colonies growing on Baird-Parker agar as well as the presumptive MRSA on Brilliance MRSA 2 agar were selected and subcultured on aesculin-blood agar (Oxoid, Germany) at 37˚C for 24 h. Gram-positive, catalase-positive and clumping factor A-positive (DiaMondiaL Staph Plus Kit, Sekisui Virotech, Germany) cocci were considered as S. aureus and MRSA, respectively. A mecA-positive S. aureus strain (ATCC 33591) was used as a positive control on Brilliance MRSA 2 agar. Regarding molecular biology, PCR analysis as recommended by Saiful et al. [26] was chosen to detect the mecA gene and thereby confirms the presence of MRSA. A sample was considered MRSApositive if at least one MRSA colony could be detected. MRSA counts were calculated in npm/g. Bacterial counts of S. aureus on Baird-Paker agar were calculated using the geometric means. For the analysis, agar plates with one up to 300 colonies were used.
4. Results
A total of 5.5% (4/72) cheese samples were positive for S. aureus. Thereof, two cheeses were manufactured from pasteurized milk and two samples were produced from raw milk. All samples originated from different smallscale dairies. One softand one semi-hard cheese sample produced from heated milk were contaminated with S. aureus at bacteria counts from 1.0 × 101 CFU/g and 2.5 × 101 CFU/g, respectively (Table 1). S. aureus could be detected in one semi-hard raw milk cheese at a level of 7.0 × 101 CFU/g, while one hard raw milk cheese contained 3.0 × 101 CFU/g (Table 2). Due to overgrowing of inoculated Baird-Parker agars with unwanted micro-organism (e.g. yeasts, streptococci, and Bacillus spp.), dilutions of 10–1 were used to calculate the bacterial counts of S. aureus.
Growth after incubation of samples in Mueller Hinton broth and streaking on Brilliance MRSA 2 agar occurred in 63 samples, which then showed blue colonies on the selective agar. After further testing, those colonies were identified as yeasts, streptococci, Bacillus spp. and Gramnegative rods. MRSA was not detectable in any of the 72 cheese samples.
Since no presumptive MRSA strains were encountered, molecular analyses were not conducted.
5. Discussion
All cheese samples examined in the current study were considered of no consequence for human health due to S. aureus. Detectable bacteria counts of S. aureus in cheese samples were below the standards established by the European Union for coagulase-positive staphylococci in cheese made from pasteurized milk or made from raw milk [6]. MRSA was also not detectable.
However, improper pasteurization or inadequate hygiene standards during manufacturing process are prone to cause contamination of cheese products with S. aureus. De Luca et al. [27] reported 16.3% (22/135) cheese samples (produced from pasteurized milk, purchased from grocery stores) positive for S. aureus, bacterial counts of S. aureus ranging from 1.0 × 101 to 3.15 × 105 CFU/g. Bulk milk appears to be a reservoir of S. aureus contamination in cheese products. Microbiological testing on a dairy processing plant manufacturing raw milk cheese indicated that S. aureus occurs both in bulk milk samples and cheese products after manufacture [7]. Based on molecular typing method, the authors inferred that bacteria were spread with milk to the equipment and the environment during milking and cheese production.
Bulk milk might be also considered a reservoir for MRSA contaminations in cheese or other dairy products since MRSA occur in dairy cattle [19,20]. Contamination of dairy products during manufacturing on small-scale farms is not entirely inconceivable, especially in raw milk cheeses. Deficient pasteurization or inadequate hygiene standards as well as colonized manufacturers could cause contamination with MRSA and/or non-MRSA S. aureus in final food products. Jones et al. [28] isolated caMRSA from pork barbecue as the origin of a foodborne illness. Those strains originated from a sales assistant in the supermarket, where the meat was purchased. Recontamination of food has been also causing an outbreak of MRSA in a hospital in the Netherlands [29]. Information assessment of our results cannot be accomplished because of lack of data about the appearance of MRSA in cheese products as well as information of required infection application rate of MRSA. Further investigations are needed.
REFERENCES
- E. Ortega, H. Abriouel, R. Lucas and A. Gálvez, “Multiple Roles of Staphylococcus aureus Enterotoxins: Pathogenicity, Superantigenic Activity, and Correlation to Antibiotic Resistance,” Toxins, Vol. 2, 2010, pp. 2117-2131. doi:10.3390/toxins2082117
- R. G. M. Olde Riekerink, W. Barkema, S. Veenstra, D. E. Poole, R. T. Dingwell and G. P. Keefe, “Prevalence of Contagious Mastitis Pathogens in Bulk Tank Milk in Prince Edward Island,” Canadian Veterinary Journal, Vol. 47, No. 6, 2006, pp. 567-572.
- F. Peles, M. Wagner, L. Varga, I. Hein, P. Rieck, K. Gutser, P. Keresztúri, G. Kardos, I. Turcsányi, B. Béri and A. Szabó, “Characterization of Staphylococcus aureus Strains Isolated from Bovine Milk in Hungary,” International Journal of Food Microbiology, Vol. 118, No. 2, 2007, pp. 186-193. doi:10.1016/j.ijfoodmicro.2007.07.010
- J. E. Virgin, T. M. van Slyke, J. E. Lombard and R. N. Zadoks, “Short Communication: Methicillin-Resistant Staphylococcus aureus Detection in US Bulk Tank Milk,” Journal of Dairy Science, Vol. 92, No. 10, 2009, pp. 4988-4991. doi:10.3168/jds.2009-2290
- Y. Le Loir, F. Baron and M. Gautier, “Staphylococcus aureus and Food Poisoning,” Genetics and Molecular Research, Vol. 2, 2003, pp. 63-76.
- Commission Regulation (EC) No 1441/2007 of 5 December 2007 Amending Regulation (EC) No 2073/2005 on Microbiological Criteria for Foodstuffs, Official Journal of European Union, L322/12, 2007.
- H. J. Jørgensen, T. Mørk and L. M. Rørvik, “The Occurrence of Staphylococcus aureus on a Farm with SmallScale Production of Raw Milk Cheese,” Journal of Dairy Science, Vol. 88, No. 11, 2005, pp. 3810-3817. doi:10.3168/jds.S0022-0302(05)73066-6
- Å. Rosengren, A. Fabricius, B. Guss, S. Sylvén and R. Lindqvist, “Occurrence of Foodborne Pathogens and Characterization of Staphylococcus aureus in Cheese Produced on Farm-Dairies,” International Journal of Food Microbiology, Vol. 144, 2010, pp. 263-269. doi:10.1016/j.ijfoodmicro.2010.10.004
- M. C. D. P. B. André, M. R. H. Campos, L. J. Borges, A. Kipnis, F. C. Pimenta and Á. B. Serafini, “Comparison of Staphylococcus aureus Isolates from Food Handlers, Raw Bovine Milk and Minas Frescal Cheese by Antibiogram and Pulsed-Field Gel Electrophoresis Following SmaI Digestion,” Food Control, Vol. 19, No. 2, 2008, pp. 200- 207. doi:10.1016/j.foodcont.2007.03.010
- M. C. Enright, D. A. Robinson, G. Randle, E. Feil, H. Grundmann and B. G. Spratt, “The Evolutionary History of Methicillin-Resistant Staphylococcus aureus (MRSA),” Proceedings of the National Academy of Sciences, Vol. 99, No. 11, 2002, pp. 7687-7692. doi:10.1073/pnas.122108599
- B. C. Herold, L. C. Immergluck, M. C. Maranan, D. S. Lauderdale, R. E. Gaskin, S. Boyle-Vavra, C. D. Leitch and R. S. Daum, “Community-Acquired Methicillin-Resistant Staphylococcus aureus in Children,” Japan Automobile Manufacturers Association, Vol. 279, No. 6, 1998, pp. 593-598. doi:10.1001/jama.279.8.593
- T. S. Naimi, K. H. LeDell, K. Como-Sabetti, S. M. Borchardt, D. J. Boxrud, J. Etienne, S. K. Johnson, F. Vandenesch, S. Fridkin, C. O’Boyle, R. N. Danila and R. Lynfield, “Comparison of Community and Health CareAssociated Methicillin-Resistant Staphylococcus aureus Infection,” Japan Automobile Manufacturers Association, Vol. 290, No. 22, 2003, pp. 2976-2984. doi:10.1001/jama.290.22.2976
- H. F. Chambers, “Methicillin Resistance in Staphylococci: Molecular and Biochemical Basis and Clinical Implications,” Clinical Microbiology Reviews, Vol. 10, No. 4, 1997, pp. 781-791.
- A. Voss, F. Loeffen, J. Bakker, C. Klaassen and M. Wulf, “Methicillinresistant Staphylococcus aureus in Pig Farming,” Emerging Infectious Diseases, Vol. 11, No. 12, 2005, pp. 1965-1966. doi:10.3201/eid1112.050428
- A. Moodley, E. C. Nightingale, M. Stegger, S. S. Nielsen, R. L. Skov and L. Guardabassi, “High Risk for Nasal Carriage of Methicillin-Resistant Staphylococcus aureus among Danish Veterinary Practitioners,” Scandinavian Journal of Work, Environment & Health, Vol. 34, No. 2, 2008, pp. 151-157. doi:10.5271/sjweh.1219
- I. van Loo, X. Huijsdens, E. Tiemersma, A. de Neeling, N. van de Sande-Bruinsma, D. Beaujean, A. Voss and J. Kluytmans, “Emergence of Methicillin-Resistant Staphylococcus aureus of Animal Origin in Humans,” Emerging Infectious Diseases, Vol. 13, No. 12, 2007, pp. 1834-1839. doi:10.3201/eid1312.070384
- M. Wulf, A. van Nes, A. Eikelenboom-Boskamp, C. Klaassen and A. Voss, “Difference in Carriage of MRSA in Organic and Regular Pig Farmers,” IPVS, Durban, 2008.
- M. Nemati, K. Hermans, U. Lipinska, O. Denis, A. Deplano, M. Struelens, L. A. Devriese, F. Pasmans and F. Haesebrouck, “Antimicrobial Resistance of Old and Recent Staphylococcus aureus Isolates from Poultry: First Detection of Livestock-Associated Methicillin-Resistant Strain ST398,” Antimicrobial Agents and Chemotherapy, Vol. 52, No. 11, 2008, pp. 3817-3819. doi:10.1128/AAC.00613-08
- M. Spohr, J. Rau, A. Friedrich, G. Klittich, A. Fetsch, B. Guerra, J. A. Hammerl and B.-A. Tenhagen, “MethicillinResistant Staphylococcus aureus (MRSA) in Three Dairy Herds in Southwest Germany,” Zoonoses Public Health, Vol. 58, No. 4, 2011, pp. 252-261. doi:10.1111/j.1863-2378.2010.01344.x
- J. Vicca, W. Vanderhaeghen, T. Cerpentier and P. Butaye, “Prevalence at Herd-Level of Methicillin Resistant Staphylococcus aureus in Milk Samples of Dairy Herds,” Mastitis Control-From Science to Practice, 2008, pp. 71- 75.
- E. de Boer, J. T. M. Zwartkruis-Nahuis, B. Wit, X. W. Huijsdens, A. J. de Neeling, T. Bosch, R. A. A. van Oosterom, A. Vila and A. E. Heuvelink, “Prevalence of Methicillin-Resistant Staphylococcus aureus in Meat,” International Journal of Food Microbiology, Vol. 134, No. 1-2, 2009, pp. 52-56. doi:10.1016/j.ijfoodmicro.2008.12.007
- A. J. de Neeling, M. J. M. van den Broek, E. C. Spalburg, M. G. van Santen-Verheuvel, W. D. C. Dam-Deisz, H. C. Boshuizen, A .W. van de Giessen, E. van Duijkeren and X. W. Huijsdens, “High Prevalence of Methicillin Resistant Staphylococcus aureus in Pigs,” Veterinary Microbiology, Vol. 122, No. 3-4, 2007, pp. 366-372. doi:10.1016/j.vetmic.2007.01.027
- B.-A. Tenhagen, A. Fetsch, B. Stührenberg, G. Schleuter, B. Guerra, J. A. Hammerl, S. Hertwig, J. Kowall, U. Kämpe, A. Schroeter, J. Bräunig, A. Käsbohrer and B. Appel, “Prevalence of MRSA Types in Slaughter Pigs in Different German Abattoirs,” Veterinary Record, Vol. 165, No. 20, 2009, pp. 589-593. doi:10.1136/vr.165.20.589
- E. van Duijkeren, R. Ikawaty, M. J. Broekhuizen-Stins, M. D. Jansen, E. C. Spalburg, A. J. de Neeling, J. G. Allaart, A. van Nes, J. A. Wagenaar and A. C. Fluit, “Transmission of Methicillin-Resistant Staphylococcus aureus Strains between Different Kinds of Pig Farms,” Veterinary Microbiology, Vol. 126, No. 4, 2008, pp. 383-389. doi:10.1016/j.vetmic.2007.07.021
- G. Normanno, M. Corrente., G. La Salandra, A. Dambrosio, N. C. Quaglia, A. Parisi, G. Greco, A. L. Bellacicco, S. Virgilio and G. V. Celano, “Methicillin-Resistant Staphylococcus aureus (MRSA) in Foods of Animal Origin Product in Italy,” International Journal of Food Microbiology, Vol. 117, No. 2, 2007, pp. 219-222. doi:10.1016/j.ijfoodmicro.2007.04.006
- A. J. Saiful, M. Mastura, S. Zarizal, M. I. Mazurah, M. Shuhaimi and A. M. Ali, “Detection of Methicillin-Resistant Staphylococcus aureus Using mecA/nuc Genes and Antibiotic Susceptibility Profile of Malaysian Clinical Isolates,” World Journal of Microbiology and Biotechnology, Vol. 22, No. 12, 2006, pp. 1289-1294. doi:10.1007/s11274-006-9174-9
- G. de Luca, F. Zanetti and S. Stampi, “Short Communication: Staphylococcus aureus in Dairy Products in the Bologna Area,” International Journal of Food Microbiology, Vol. 35, No. 3, 1997, pp. 267-270. doi:10.1016/S0168-1605(96)01229-9
- T. F. Jones, M. E. Kellum, S. S. Porter, M. Bell and W. Schaffner, “An Outbreak of Community Acquired Foodborne Illness Caused by Methicillin-Resistant Staphylococcus aureus,” Emerging Infectious Diseases, Vol. 8, No. 1, 2002, pp. 82-84. doi:10.3201/eid0801.010174
- J. Kluytmans, W. van Leeuwen, , W. Goessens, R. Hollis, S. Messer, L. Herwaldt, H. Bruining, M. Heck, J. Rost, N. van Leeuwen, A. van Belkum and H. Verbrugh, “FoodInitiated Outbreak of Methicillin-Resistant Staphylococcus aureus Analyzed by Phenoand Genotyping,” Journal of Clinical Microbiology, Vol. 33, No. 5, 1995, pp. 1121-1128.
NOTES
*Corresponding author.

