Paper Menu >>
Journal Menu >>
 Pharmacology & Pharmacy, 2010, 1, 69-74 doi:10.4236/pp.2010.12010 Published Online October 2010 (http://www.SciRP.org/journal/pp) Copyright © 2010 SciRes. PP 69 The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Olga G. Sitnikova1, Sergey B. Nazarov1, Irina V. Shikhanova2, Alexander V. Agafonov2, Jean A. Dyuzhev1, Irina G. Popova1 1Ivanovo State Research Institute of Maternity and Childhood (RIMC), Ivanovo, Russia; 2Institute of Solution Chemistry, Russian Academy of Sciences, Ivanovo, Russia. Email: ivgenlab@gmail.com Received July 20th, 2010; revised August 10th, 2010; accepted September 30th, 2010. ABSTRACT In this study investigation of influence of hybrid nanosilica-polyethylene glycols materials (molecular weight 1500, 6000 and 15000), prepared by sol-gel synthesis, on lipid peroxidation and antioxidant activity of human serum in vitro was performed. Methods included chemiluminescence analysis and quantitative malonic dialdehyde estimation. It was revealed that nanosilica-PEG materials with different molecular weight had certain biological activity. Powders of SiO2-PEG 1500 and SiO2-PEG 6000 manifest prooxidant effects, whereas mesoporous (calcine) powders produced antioxidant effects in blood serum in vitro. Keywords: Free-Radical Oxidation, Nanosilica, Chemiluminescence, Malonic Dialdehyde 1. Introduction The study of free-radical processes of lipid oxidation and antioxidant system is an important problem of modern biomedicine. Oxygen is a powerful oxidant and oxy- gen-mediated reactions are the main sources of energy for variety of biological species. Metabolic processes produce reactive oxygen species (ROSs), free radicals, peroxides, malonic dialdehyde, Schiff's bases, which damage membrane structures and lead to oxidative stress, being the causative factor for a lot of widely spread dis- eases, including reproductive disorders and prenatal inju- ries [1-3]. Concerning this the role of antioxidants is to neutralize toxic products of free-radical lipid peroxidation. In con- ditions of excessive peroxidation the capacity of antioxi- dant defense may decrease due to insufficient endogenic antioxidant production or unfavorable environment fac- tors [3]. Various substances are known to have antioxi- dant activity; their investigation is of certain importance for both biology and medicine [4]. Nowadays silica compounds attract considerable at- tention. Silica stimulates fibroblastic activity of mesen- chyma, promoting granulation and scarring. The lack of silica may lead to depression of leukocyte activity in in- flammation, poor wound scarring, anorexia, pruritus, tissue flexibility decrease, skin turgor decrease, vascular permeability increase and haemorrhagia as a result. Apart from this silica antioxidant activity stands [5]. Investigators of peroxidation showed that silica pow- ders stimulated active oxygen species and free radicals production in culture of epithelial cells, resulting in cas- pase activation and apoptosis [6]. Some authors experi- mentally found out that inhalation of crystalloid silica in animals lead to oxidative stress, inflammation and alveo- lar fibrosis [7,8]. As a catalysts and functional composite materials spe- cial attention is attracted by matrix hybrid silica derivates. In this study we presented physical-chemical properties of silica-based materials and their influence on peroxida- tion processes in human serum. Silica nanoparticles are biologically inert, have high adsorption rate, thermal and mechanic stability, and therefore are expected to mani- fest antioxidant activity. The aim of the work was to study the influence of or- ganic-innorganic silica and polyethylene glycols com- pounds on free-radical processes in vitro.  The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Copyright © 2010 SciRes. PP 70 2. Materials and Methods 2.1. Reagents for Silica Nanoparticles Synthesis Tetraethoxysilane (TEOS) (C2H5O)4Si (high purity grade, Ekos-1, Russia), diethylamine (С2Н5)2NH (moderate pu- rity grade, Aldrich), polyethylene glycols [-OCH2CH2-]n with molecular weight of 1500, 6000, 15000 (Aldrich) and rectified ethanol (96 wt%) were used without further purification. 2.2. Synthesis of Hybrid Organic-Inorganic SiO2-Polyethylene Glycol Materials The synthesis was carried out in ethanol media at tem- perature of 20℃. Polyethylene glycol was dissolved in required amount of water and was added to ethanol. Than a catalyst of tatraethoxysilane hydrolysis – diethyl amine – was added. The resulting mixture was homogenized for 15 min. After that small doses of tatraethoxysilane (0.5-1.0 ml) were injected into the system using syringe. Total synthesis time was 17 hrs. Particles were filtered from the original solution and dried out on open air at temperature of 70-100℃ until mass stability. We investigated two types of hybrid organic-inorganic SiO2-PEG materials: noncalcine and calcine in muffle furnace (800℃, 2 hrs). 2.3. IR Spectroscopy IR spectra of produced powders were recorded using Avatar 360 FT-IR ESP spectrometer (wave range: 400- 4000 cm-1). Preliminary samples of powders were grind- ed with KBr in agate mortar and pressed in discs. 2.4. Thermogravimetry Thermogravimetric measuring were performed on ther- moanalythical installation. The mass of a sample was 80-90 mg. The heating was carried out from room tem- perature up to 1000℃ with rate of 5℃ per min. 2.5. Brunauer-Emmett-Teller (BET) Surface Area Measurement First step of BET analysis was heating of samples to eliminate adsorbed impurities. Then argon at temperature of 77K was adsorbed on materials to form a monomo- lecular film on accessible area. The amount of adsorbed argon served to determine the BET surface area. In experiments we used native serum of 10 patients, administered in obstetrical or gynecological clinics of Ivanovo Research Institute of Maternity and Childhood. We decided not to use whole blood to prevent coagula- tion. Suspension of studied powders was added to 1 ml of native serum, which was then incubated at 4℃ for 1 hr. The ratio between powders and serum was 5.0 g/L for mesostructured (noncalcine) powder and 50.0 g/L for mesoporous (calcine) powder. At this stage of experi- ments we didn’t try to evaluate pharmacological doses of studied powders as the only aim of experiments was to determine any possible pro- or antioxidant properties of synthesized materials in vitro. The incubation tempera- ture was found to limit bacterial activity. Native serum without nanomaterials stood for control. After incubation samples were centrifuged at 3000 rpm for 10 min. Su- pernatants were transferred into clean dry tubes and un- derwent analysis. 2.6. Estimation of Peroxidation Intensity by Induced Chemiluminescence (CL) The method was based on catalytic reduction of perox- ides by bivalent ferric ions. Generated free radicals acted as oxidation initiators in biological substrate. Free radi- cals recombination followed by photon release, which could be registered in 40 s. This was a period of maximal intensity. We used hydrogen peroxide and ferric sulfate as biochemiluminescence inductors. 0.1 ml of serum was mixed with 0.4 ml phosphate buffer (pH 7.5), 0.4 ml fer- ric sulfate (0.01 mM), 0.2 ml 2% hydrogen peroxide and underwent CL. Intensity of luminescence was evaluated by BHL-06M biochemiluminometer (Russia). Registered parameters were as follows (Figure 1): peak intensity of luminescense (Imax) – the highest intensity of lumines- cense registered within first 40 seconds of Fenton’s reac- tion; light sum (S) – the area under the curve of lumines- cent signal; slope of the curve (tg α) – tangent of de- crease signal angle. Imax and S reflected potential for lipid peroxidation. Antioxidant activity was characterized by tg α and coeffi- cient K (Imax/S ratio). Figure 1. Typical kinetics of serum chemiluminescense. The registered parameters are as follows: peak intensity (Imax), light sum (S(t)), the slope of the curve (tg α).  The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Copyright © 2010 SciRes. PP 71 2.7. Malonic Dialdehyde Serum Concentration Measurement The method was based on reaction between malonic dialdehyde (MDA) and thiobarbituric acid [9]; the prod- uct of reaction had pink color and was identified by SF-46 spectrophotometer (Russia) on wave length 523 nm. 3. Results and Discussion Qualitative and quantitative structure of materials was investigated by IR spectroscopy and thermogravimetric analysis. Thermograms of hybrid organic-inorganic SiO2-PEG material (molecular weight 15000) are presented on Fig- ure 2. Burning of polymer component from matrix SiO2-PEG 15000 hybrid is described by the peak with maximum at 291℃ on differential thermogravimetric diagram. The amount of organic phase was 39 wt% in sample. Water content was 13.5 wt%. The curve of differential thermal analysis did not reveal any thermal effects. Figure 3 shows IR spectra of hybrid organic-inorganic (a) SiO2-PEG 15000 and (b) material calcined at 800℃. There were characteristic oscillations of Si-OH and Si-O-Si found in spectra. Also, in areas of wavenumbers 3600-3500, 1640 and 400-200 cm-1 oscillations of ad- sorbed H2O, isolated and linked OH groups were found. In spectrum of hybrid organic-inorganic SiO2-PEG 15000 oscillations of alkyl CH3, CH2 and CH groups [10-19] proved the presence of organic phase in compos- ite. Intensive flat halo on X-ray diagram of hybrid SiO2- PEG 15000 material reflected its amorphism (Figure 4). Results of sedimentation gave data on morphology of hybrid material powders. Sizes of particles of hybrid SiO2-PEG composite determined by sedimentation be- fore and after calcining were (Figure 5) as follows. Hy- brid material: 2-300 µm, with the prevalence of 24 and 150 µm fractions; tempered material: 2-110 µm, with modal size 28 µm. Specific areas of noncalcine and cal- cine hybrid organic-inorganic SiO2-PEG 15000 material powders were discovered by thermal desorption of argon (Table 1). Thus, there were synthesized two groups of composite materials with different structure: organic-inorganic compounds with amorphous structure and mesostructural materials with system of ordered mesochannels, filled by molecular organic templates. All materials were calcined at 800℃ for creating of high-porous silica specimens with the same structural features as their noncalcine an- cestors. Investigation of pro- and antioxidant properties Figure 2. Thermograms of hybrid organic-inorganic SiO2- PEG 15000 material. Table 1. Specific surface areas of hybrid SiO2-PEG 15000 and calcined at 800℃ materials. Material Specific surface area by BET, m2/g SiO2-PEG 15000 (noncalcine) 91 SiO2-PEG 15000 (calcine) > 800 of these powders was carried out in vitro. Biochemical study revealed different influence of in- vestigated powders on intensity of peroxidation in human serum (Tables 2 and 3). Parameters of chemilumines- cence revealed in native serum were estimated as 100%. Adding of SiO2-PEG powders with molecular weight 1500 and 6000 to experimental system lead to increased chemiluminescence, which proved high ROS and free radical (R-, OH-, RO-, RO2-, O2-) production in Fenton's reaction [20,21]. Recombination of radicals formed un- stable tetraoxide, which disintegrated with photon emis- sion and raised lightsum S (plus 23%, p = 0.0081 and 18%, p = 0.0131, comparing with controls, respectively for 1500 and 6000 powders), fast flash Imax (plus 26%, p = 0.0459 for 1500 powder). Investigation of SiO2-PEG with molecular weight 15000 didn’t reveal any signifi- cant CL changes. Study of mesoporous (calcine) SiO2- PEG powder revealed its antioxidant activity, which was developed by decreasing of peroxidation processes inten- sity in Samples 3 and 5 (Table 3). We found increasing of tg α (plus 15%, p = 0.0510) and decreasing of MDA. The conclusion was that nanosilica-PEG compounds with different molecular weight possessed specific oxidant and antioxidant activity. SiO2-PEG 1500 and SiO2-PEG 6000 powders lead to activation of free-radical oxidation, 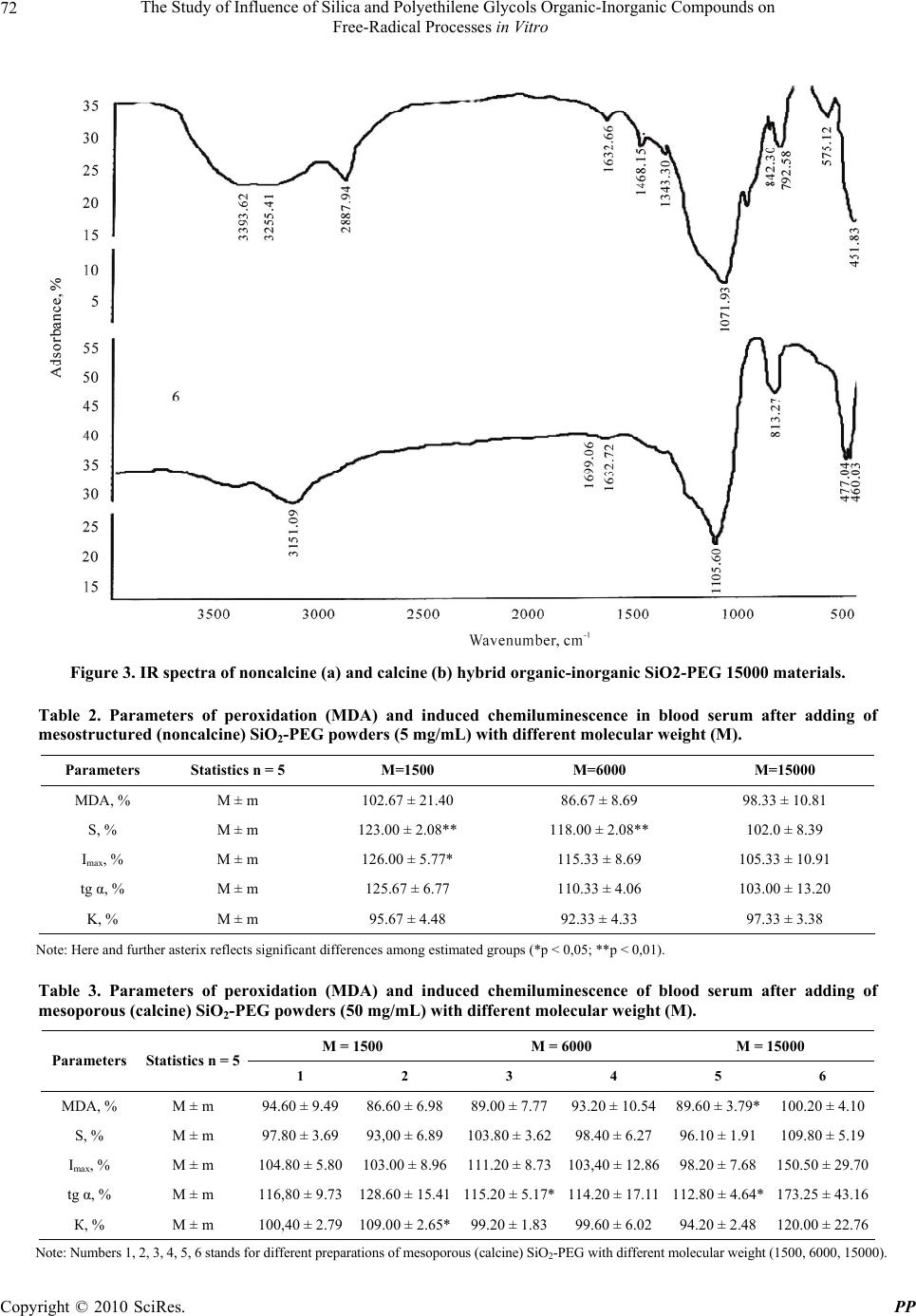 The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Copyright © 2010 SciRes. PP 72 Figure 3. IR spectra of noncalcine (a) and calcine (b) hybrid organic-inorganic SiO2-PEG 15000 materials. Table 2. Parameters of peroxidation (MDA) and induced chemiluminescence in blood serum after adding of mesostructured (noncalcine) SiO2-PEG powders (5 mg/mL) with different molecular weight (M). Parameters Statistics n = 5 M=1500 M=6000 M=15000 MDA, % M ± m 102.67 ± 21.40 86.67 ± 8.69 98.33 ± 10.81 S, % M ± m 123.00 ± 2.08** 118.00 ± 2.08** 102.0 ± 8.39 Imax, % M ± m 126.00 ± 5.77* 115.33 ± 8.69 105.33 ± 10.91 tg α, % M ± m 125.67 ± 6.77 110.33 ± 4.06 103.00 ± 13.20 K, % M ± m 95.67 ± 4.48 92.33 ± 4.33 97.33 ± 3.38 Note: Here and further asterix reflects significant differences among estimated groups (*р < 0,05; **р < 0,01). Table 3. Parameters of peroxidation (MDA) and induced chemiluminescence of blood serum after adding of mesoporous (calcine) SiO2-PEG powders (50 mg/mL) with different molecular weight (M). M = 1500 M = 6000 M = 15000 Parameters Statistics n = 5 1 2 3 4 5 6 MDA, % М ± m 94.60 ± 9.49 86.60 ± 6.9889.00 ± 7.7793.20 ± 10.5489.60 ± 3.79* 100.20 ± 4.10 S, % М ± m 97.80 ± 3.69 93,00 ± 6.89103.80 ± 3.6298.40 ± 6.2796.10 ± 1.91 109.80 ± 5.19 Imax, % М ± m 104.80 ± 5.80 103.00 ± 8.96111.20 ± 8.73103,40 ± 12.8698.20 ± 7.68 150.50 ± 29.70 tg α, % М ± m 116,80 ± 9.73 128.60 ± 15.41115.20 ± 5.17*114.20 ± 17.11112.80 ± 4.64* 173.25 ± 43.16 К, % М ± m 100,40 ± 2.79 109.00 ± 2.65*99.20 ± 1.8399.60 ± 6.0294.20 ± 2.48 120.00 ± 22.76 Note: Numbers 1, 2, 3, 4, 5, 6 stands for different preparations of mesoporous (calcine) SiO2-PEG with different molecular weight (1500, 6000, 15000). 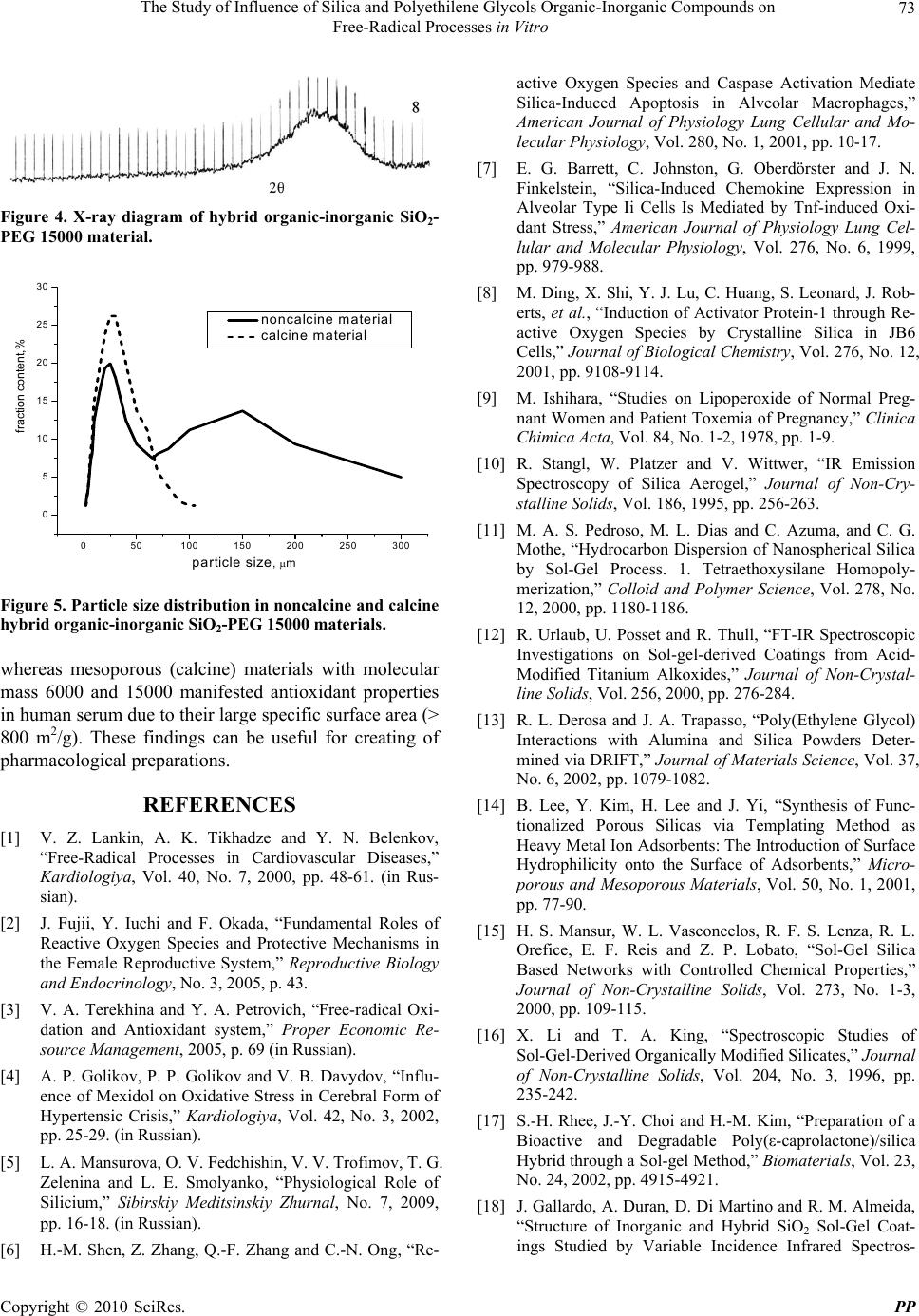 The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Copyright © 2010 SciRes. PP 73 Figure 4. X-ray diagram of hybrid organic-inorganic SiO2- PEG 15000 material. 050100 150 200 250 300 0 5 10 15 20 25 30 noncalcine material calcine material fraction content,% particle size, m Figure 5. Particle size distribution in noncalcine and calcine hybrid organic-inorganic SiO2-PEG 15000 materials. whereas mesoporous (calcine) materials with molecular mass 6000 and 15000 manifested antioxidant properties in human serum due to their large specific surface area (> 800 m2/g). These findings can be useful for creating of pharmacological preparations. REFERENCES [1] V. Z. Lankin, A. K. Tikhadze and Y. N. Belenkov, “Free-Radical Processes in Cardiovascular Diseases,” Kardiologiya, Vol. 40, No. 7, 2000, pp. 48-61. (in Rus- sian). [2] J. Fujii, Y. Iuchi and F. Okada, “Fundamental Roles of Reactive Oxygen Species and Protective Mechanisms in the Female Reproductive System,” Reproductive Biology and Endocrinology, No. 3, 2005, p. 43. [3] V. A. Terekhina and Y. A. Petrovich, “Free-radical Oxi- dation and Antioxidant system,” Proper Economic Re- source Management, 2005, p. 69 (in Russian). [4] A. P. Golikov, P. P. Golikov and V. B. Davydov, “Influ- ence of Mexidol on Oxidative Stress in Cerebral Form of Hypertensic Crisis,” Kardiologiya, Vol. 42, No. 3, 2002, pp. 25-29. (in Russian). [5] L. A. Mansurova, O. V. Fedchishin, V. V. Trofimov, T. G. Zelenina and L. E. Smolyanko, “Physiological Role of Silicium,” Sibirskiy Meditsinskiy Zhurnal, No. 7, 2009, pp. 16-18. (in Russian). [6] H.-M. Shen, Z. Zhang, Q.-F. Zhang and C.-N. Ong, “Re- active Oxygen Species and Caspase Activation Mediate Silica-Induced Apoptosis in Alveolar Macrophages,” American Journal of Physiology Lung Cellular and Mo- lecular Physiology, Vol. 280, No. 1, 2001, pp. 10-17. [7] E. G. Barrett, C. Johnston, G. Oberdörster and J. N. Finkelstein, “Silica-Induced Chemokine Expression in Alveolar Type Ii Cells Is Mediated by Tnf-induced Oxi- dant Stress,” American Journal of Physiology Lung Cel- lular and Molecular Physiology, Vol. 276, No. 6, 1999, pp. 979-988. [8] M. Ding, X. Shi, Y. J. Lu, C. Huang, S. Leonard, J. Rob- erts, et al., “Induction of Activator Protein-1 through Re- active Oxygen Species by Crystalline Silica in JB6 Cells,” Journal of Biological Chemistry, Vol. 276, No. 12, 2001, pp. 9108-9114. [9] M. Ishihara, “Studies on Lipoperoxide of Normal Preg- nant Women and Patient Toxemia of Pregnancy,” Clinica Chimica Acta, Vol. 84, No. 1-2, 1978, pp. 1-9. [10] R. Stangl, W. Platzer and V. Wittwer, “IR Emission Spectroscopy of Silica Aerogel,” Journal of Non-Cry- stalline Solids, Vol. 186, 1995, pp. 256-263. [11] M. A. S. Pedroso, M. L. Dias and C. Azuma, and C. G. Mothe, “Hydrocarbon Dispersion of Nanospherical Silica by Sol-Gel Process. 1. Tetraethoxysilane Homopoly- merization,” Colloid and Polymer Science, Vol. 278, No. 12, 2000, pp. 1180-1186. [12] R. Urlaub, U. Posset and R. Thull, “FT-IR Spectroscopic Investigations on Sol-gel-derived Coatings from Acid- Modified Titanium Alkoxides,” Journal of Non-Crystal- line Solids, Vol. 256, 2000, pp. 276-284. [13] R. L. Derosa and J. A. Trapasso, “Poly(Ethylene Glycol) Interactions with Alumina and Silica Powders Deter- mined via DRIFT,” Journal of Materials Science, Vol. 37, No. 6, 2002, pp. 1079-1082. [14] B. Lee, Y. Kim, H. Lee and J. Yi, “Synthesis of Func- tionalized Porous Silicas via Templating Method as Heavy Metal Ion Adsorbents: The Introduction of Surface Hydrophilicity onto the Surface of Adsorbents,” Micro- porous and Mesoporous Materials, Vol. 50, No. 1, 2001, pp. 77-90. [15] H. S. Mansur, W. L. Vasconcelos, R. F. S. Lenza, R. L. Orefice, E. F. Reis and Z. P. Lobato, “Sol-Gel Silica Based Networks with Controlled Chemical Properties,” Journal of Non-Crystalline Solids, Vol. 273, No. 1-3, 2000, pp. 109-115. [16] X. Li and T. A. King, “Spectroscopic Studies of Sol-Gel-Derived Organically Modified Silicates,” Journal of Non-Crystalline Solids, Vol. 204, No. 3, 1996, pp. 235-242. [17] S.-H. Rhee, J.-Y. Choi and H.-M. Kim, “Preparation of a Bioactive and Degradable Poly(ε-caprolactone)/silica Hybrid through a Sol-gel Method,” Biomaterials, Vol. 23, No. 24, 2002, pp. 4915-4921. [18] J. Gallardo, A. Duran, D. Di Martino and R. M. Almeida, “Structure of Inorganic and Hybrid SiO2 Sol-Gel Coat- ings Studied by Variable Incidence Infrared Spectros-  The Study of Influence of Silica and Polyethilene Glycols Organic-Inorganic Compounds on Free-Radical Processes in Vitro Copyright © 2010 SciRes. PP 74 copy,” Journal of Non-Crystalline Solids, Vol. 298, No. 2-3, 2002, pp. 219-225. [19] V. A. Maroni and S. J. Epperson, “An in Situ Spectro- scopic Investigation of the Pyrolysis of Ethylene Glycol Encapsulated in Silica Solidate,” Vibrational Spectros- copy, Vol. 27, No. 1, 2001. pp. 43-51. [20] A. I. Zhuravlev and A. I. Zhuravleva, “Hiperweak Lumi- nescence of Blood Serum and its Role in Complex Diag- nostics,” Meditsina Press, Moscow, 1976. (in Russian). [21] E. I. Kuzmina, A. S. Nelubin and M. K. Schennikova, “Application of Induced Chemiluminescence for Free- Radical Processes Estimation in Biological Substrates,” In: Biokhimiya i biofizika, Gorkiy, 1983, pp. 179-183. (in Russian). |

