Paper Menu >>
Journal Menu >>
 Journal of Biomaterials and Nanobiotechnology, 2010, 1, 1-6 doi:10.4236/jbnb.2010.11001 Published Online October 2010 (http://www.SciRP.org/journal/jbnb) Copyright © 2010 SciRes. JBNB 1 Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays Katia Campioni1, Cristina Morelli1, Antonio D’Agostino2, Lorenzo Trevisiol2, Pier Francesco Nocini2, Marco Manfrini1, Mauro Tognon1* 1Section of Cell Biology and Molecular Genetics, School of Medicine and Centre of Biotechnology, University of Ferrara, Ferrara, Italy; 2Department of Odontostomatology and Maxillo-Facial Surgery, School of Medicine and School of Dentistry, University of Verona, Verona, Italy. Email: *tgm@unife.it Received July 7th, 2010; revised July 28th, 2010; accepted July 29th 2010. ABSTRACT Many cellular models have been used to investigate bone sub stitutes including cora l-derived hydroxylapatite (HA). The aim of this study was to verify whether a new cellular model represented by Saos-eGFP cells can be used to test bioma- terials by in vitro assays. Saos-eGFP cells which express the enhanced Green Fluorescent Protein (eGFP), were de- rived from the osteoblast-like cell line Saos-2. To this purpose Saos-eGFP cells were employed to investigate the in-vitro bioactivity of a well characterized coral-derived HA biomaterial, in block and granule forms. This engineered cell line, by evaluatin g the emitted fluorescence, allo wed us to assay 1) cell adhesion, 2) cell proliferation and 3) colony capability. Electron microscopy analysis was employed to evaluate the 4) morphology of cells seeded on the biomate- rial surface. 5) Histological analysis of the bone grown after scaffold implantation was carried out in specimens from two clinical cases. Saos-eGFP cells indicate, as established before, that the coralline-HA biomaterial has a good in vitro cytocompatibility when tested with human osteoblast-like cells. Some differences in proliferation activity were detected for the two different forms assayed. Cytocompatibility data from in vitro analyses were confirmed by in vivo behaviour of biomaterials. Our tests suggest that the engineered cell line Saos-eGFP represents a suitable in vitro mode for studying the biocompatibility, the cell adhesion, spreading and proliferation on biomaterials developed for clinical applications. The main advantages of this cellular model are 1) less time consuming and 2) a reduced cost of the experiments. Keywords: Fluorescence, Osteoblast, Bi omaterial 1. Introduction Investigations into bone substitutes have been addressed to different biomaterials including coral-derived hy- droxylapatite (HA). One of the main goals of this bioma- terial, which is similar in its chemical composition to human bone, is to modulate cellular responses which control the interaction with the scaffold. Specifically, it may induce spontaneous three dimensional self-organi- sation in the new tissue, as it has been observed in the physiological environment [1,2]. Indeed, cell behaviour and phenotype are governed by responses to different types of signals that include mechanical forces, electrical stimuli, and various physical cues [3]. In addition, cells sense and respond to a variety of signals including solu- ble growth factors, differentiation factors, cytokines, and ion gradients [4-6]. Materials employed as scaffolds must possess specific features, such as cyto- and bio-compati- bility, osteo-inductivity and -conductivity, as well as the right mechanical strength to provide structural support during tissue growth and remodelling. Besides those of a bovine origin, natural HA biomaterials can also be de- rived from coral exoskeletons (genus Porites and Gonio- pora). Hydrothermal treatment (260°C.; 15,000 PSI) of the calcium carbonate exoskeletal microstructure of these corals results in conversion into hydroxylapatite [7]. Different hydrothermal coral treatments have resulted in only partial conversion of its calcium carbonate to HA [8]. As a result, its HA/CaCO3 composite is resorbed faster than pure HA. Like natural bone, this HA may contain minor elements such as Mg, Sr, F, and CO3 and has a completely interconnected porosity which is similar to trabecular bone. 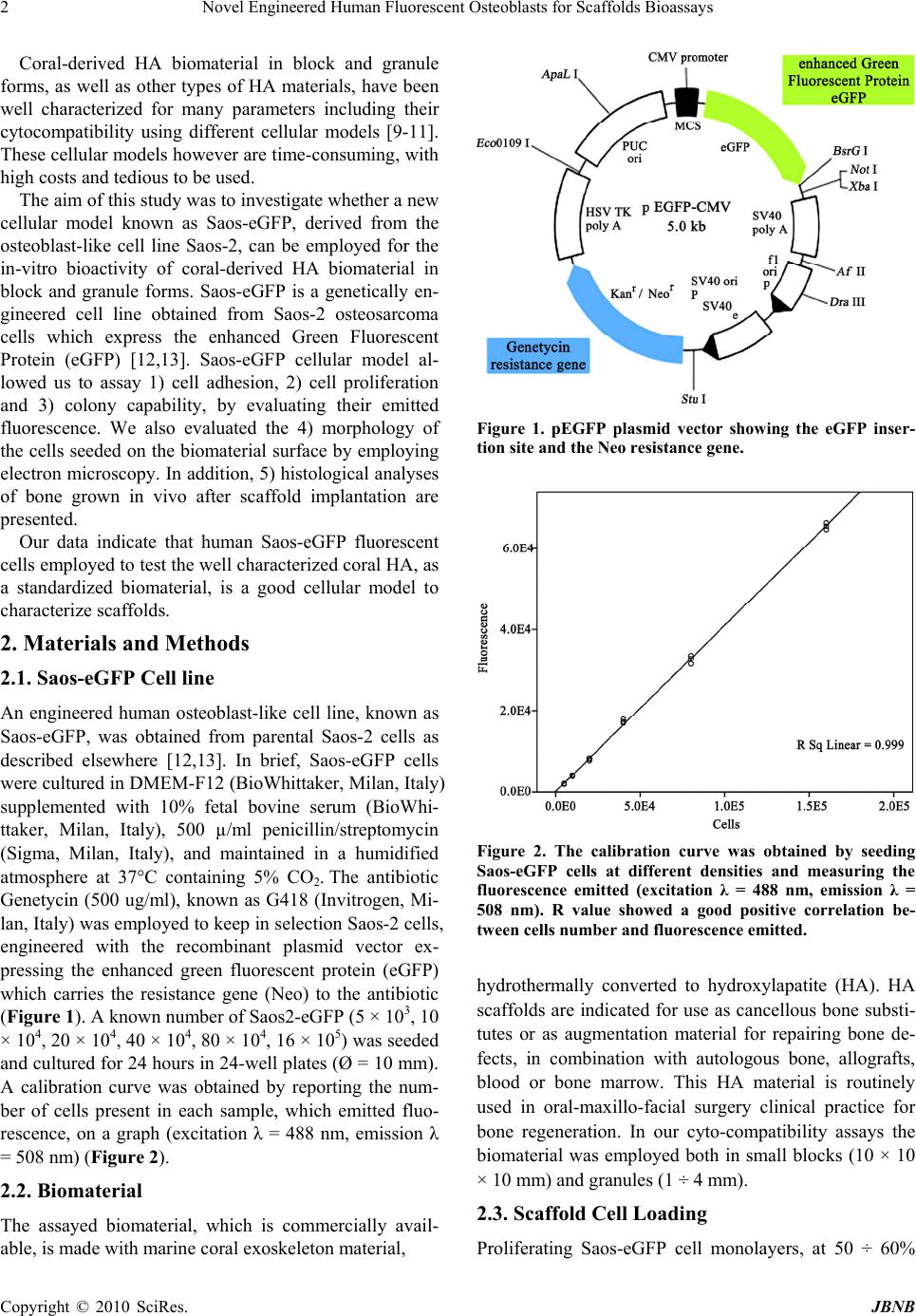 Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays 2 Coral-derived HA biomaterial in block and granule forms, as well as other types of HA materials, have been well characterized for many parameters including their cytocompatibility using different cellular models [9-11]. These cellular models however are time-consuming, with high costs and tedious to be used. The aim of this study was to investigate whether a new cellular model known as Saos-eGFP, derived from the osteoblast-like cell line Saos-2, can be employed for the in-vitro bioactivity of coral-derived HA biomaterial in block and granule forms. Saos-eGFP is a genetically en- gineered cell line obtained from Saos-2 osteosarcoma cells which express the enhanced Green Fluorescent Protein (eGFP) [12,13]. Saos-eGFP cellular model al- lowed us to assay 1) cell adhesion, 2) cell proliferation and 3) colony capability, by evaluating their emitted fluorescence. We also evaluated the 4) morphology of the cells seeded on the biomaterial surface by employing electron microscopy. In addition, 5) histological analyses of bone grown in vivo after scaffold implantation are presented. Our data indicate that human Saos-eGFP fluorescent cells employed to test the well characterized coral HA, as a standardized biomaterial, is a good cellular model to characterize scaffolds. 2. Materials and Methods 2.1. Saos-eGFP Cell line An engineered human osteoblast-like cell line, known as Saos-eGFP, was obtained from parental Saos-2 cells as described elsewhere [12,13]. In brief, Saos-eGFP cells were cultured in DMEM-F12 (BioWhittaker, Milan, Italy) supplemented with 10% fetal bovine serum (BioWhi- ttaker, Milan, Italy), 500 µ/ml penicillin/streptomycin (Sigma, Milan, Italy), and maintained in a humidified atmosphere at 37°C containing 5% CO2. The antibiotic Genetycin (500 ug/ml), known as G418 (Invitrogen, Mi- lan, Italy) was employed to keep in selection Saos-2 cells, engineered with the recombinant plasmid vector ex- pressing the enhanced green fluorescent protein (eGFP) which carries the resistance gene (Neo) to the antibiotic (Figure 1). A known number of Saos2-eGFP (5 × 103, 10 × 104, 20 × 104, 40 × 104, 80 × 104, 16 × 105) was seeded and cultured for 24 hours in 24-well plates (Ø = 10 mm). A calibration curve was obtained by reporting the num- ber of cells present in each sample, which emitted fluo- rescence, on a graph (excitation λ = 488 nm, emission λ = 508 nm) (Figure 2). 2.2. Biomaterial The assayed biomaterial, which is commercially avail- able, is made with marine coral exoskeleton material, Figure 1. pEGFP plasmid vector showing the eGFP inser- tion site and the Neo resistance gene. Figure 2. The calibration curve was obtained by seeding Saos-eGFP cells at different densities and measuring the fluorescence emitted (excitation λ = 488 nm, emission λ = 508 nm). R value showed a good positive correlation be- tween cells number and fluorescence emitted. hydrothermally converted to hydroxylapatite (HA). HA scaffolds are indicated for use as cancellous bone substi- tutes or as augmentation material for repairing bone de- fects, in combination with autologous bone, allografts, blood or bone marrow. This HA material is routinely used in oral-maxillo-facial surgery clinical practice for bone regeneration. In our cyto-compatibility assays the biomaterial was employed both in small blocks (10 × 10 × 10 mm) and granules (1 ÷ 4 mm). 2.3. Scaffold Cell Loading Proliferating Saos-eGFP cell monolayers, at 50 ÷ 60% Copyright © 2010 SciRes. JBNB  Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays3 confluence, were detached from the culture flasks and re-suspended in culture medium to obtain a cell suspen- sion with a density of 104 cells/cm2 surface area, in 1 ml, for each scaffold. A tissue culture polystyrene (TCPS) vessel was employed as control. The scaffold was then placed in wells (Ø = 10 mm) filled with the proper cell suspension and incubated for 2 h. To maximize cell- scaffold interaction, cell suspension was subjected to pipetting every 15 min. After the incubation period, scaffolds were placed in empty wells. Fresh culture me- dium, 1 ml, was added to each scaffold. 2.4. Cell Adhesion and Proliferation Saos-eGFP cells were loaded onto scaffolds and TCPS (control), in 24-well culture plates (Ø = 10 mm) and cul- tured as described above. In order to mimic the clinical application environment, the same volume of bone void filler was used for each type of scaffold. To analyze cell adhesion on each biomaterial and on the control, samples were incubated for 24 h at 37°C, 5% CO2. Attached cells per scaffolds were detected by measuring the fluores- cence (excitation λ = 488 nm, emission λ = 508 nm) emitted by the viable cells. Then, cells were re-fed with fresh culture medium and cultured at 37°C in a humidi- fied atmosphere with 5% CO2. Assays were carried out to evaluate cells attached on biomaterials and on the con- trol, at 24 h, 48 h, and 96 h. 2.5. Cell Spreading Saos-eGFP cells were loaded onto the scaffolds and TCPS, in 24-well culture plates (Ø = 10 mm) and cul- tured as described above. Samples were incubated for 24 h, 48 h ad 96 h at 37°C, 5% CO2. Direct observation of the living cells on the biomaterial was carried out by fluorescence microscopy in order to determine their dis- tribution, colonization and morphology on the biomate- rials. 2.6. Scanning Electron Microscopy (SEM) For the SEM analysis, Saos-eGFP (104 cells/well) were cultured on HA scaffolds for 48 h. Cells which were at- tached to the biomaterials were washed with PBS 1X solution and fixed for 1 h by 2.5% glutaraldehyde in a phosphate buffer and then for 4h with a 1% osmium so- lution in a phosphate buffer. Specimens were coated with colloidal gold and analyzed using scanning electron mi- croscopy (SEM, Cambridge UK, model Stereoscan S- 360). 2.7. Histology Granules were used for maxillary sinus augmentation in patients with bone resorption in the posterior segment of the upper jaw for implant-prosthetic rehabilitation. Blocks were used as interpositional materials in orthog- nathic surgery patients when the upper jaw was down grafted in order to increase the stability of the mobilized segment. Biopsies were taken in accordance with the local ethical committee and after patients’ written con- sent. In one clinical case (patient 1), biomaterial granules were used for bone regeneration in a patient who had undergone maxillary sinus augmentation for pre-prosthetic surgical rehabilitation. In this patient, a bone biopsy was taken four months after the first surgical procedure, during implant surgery. In another clinical case, (patient 2), to correct dentoskeletal deformities, blocks of biomaterial were employed as interpositional grafts in a patient treated with LeFort I osteotomy. Bone biopsies were taken from this patient, during plate re- moval, one year after surgery. Tissues were fixed in 4% formaldehyde and dehydrated in a graded series of alco- hols. Then, all specimens were embedded in methyl- methacrylate resin. Undecalcified sections were obtained using a water-cooled diamond saw. Slides were ground to a final thickness of about five μm, placed on glass slides, stained with hematoxilin- eosin and viewed and photographed in a Leitz Orthoplan photomicroscope. 2.8. Statistical Analysis All data were obtained from five independent experi- ments and expressed as a mean value ± SD. ANOVA testing followed by post-hoc Bonferroni testing was em- ployed to evaluate differences in the mean values among groups. Statistically significant findings were considered when p < 0.05. All data elaboration was computed by SPSS (SPSS Inc., Chicago, IL, U.S.A.). 3. Results 3.1. Cell Adhesion and Proliferation The results obtained showed a good adhesive capability of Saos-eGFP cells to the biomaterial. Indeed, Saos- eGFP cells attached both to block and granules of the biomaterial with a prevalence of 67% and 64%, respec- tively, compared to seeded cells. In the control experi- ment, 94% of the seeded cells were attached to the plastic well, showing a statistically significant difference (p < 0.01) as regards the two scaffolds (Figure 3). Cells pro- liferated in each sample during the assay as in the control (p < 0.01). At 48 h, the fluorescence detected for the granules was lower than the fluorescence detected for the control and block (p < 0.01). Cells grown on blocks be- haved differently, as shown by the data obtained at 96 incubation h. Indeed at this point, the fluorescence de- tected on these samples diminished with statistically sig- nificant differences as regards the control and granules (p < 0.05) (Figure 4). Copyright © 2010 SciRes. JBNB 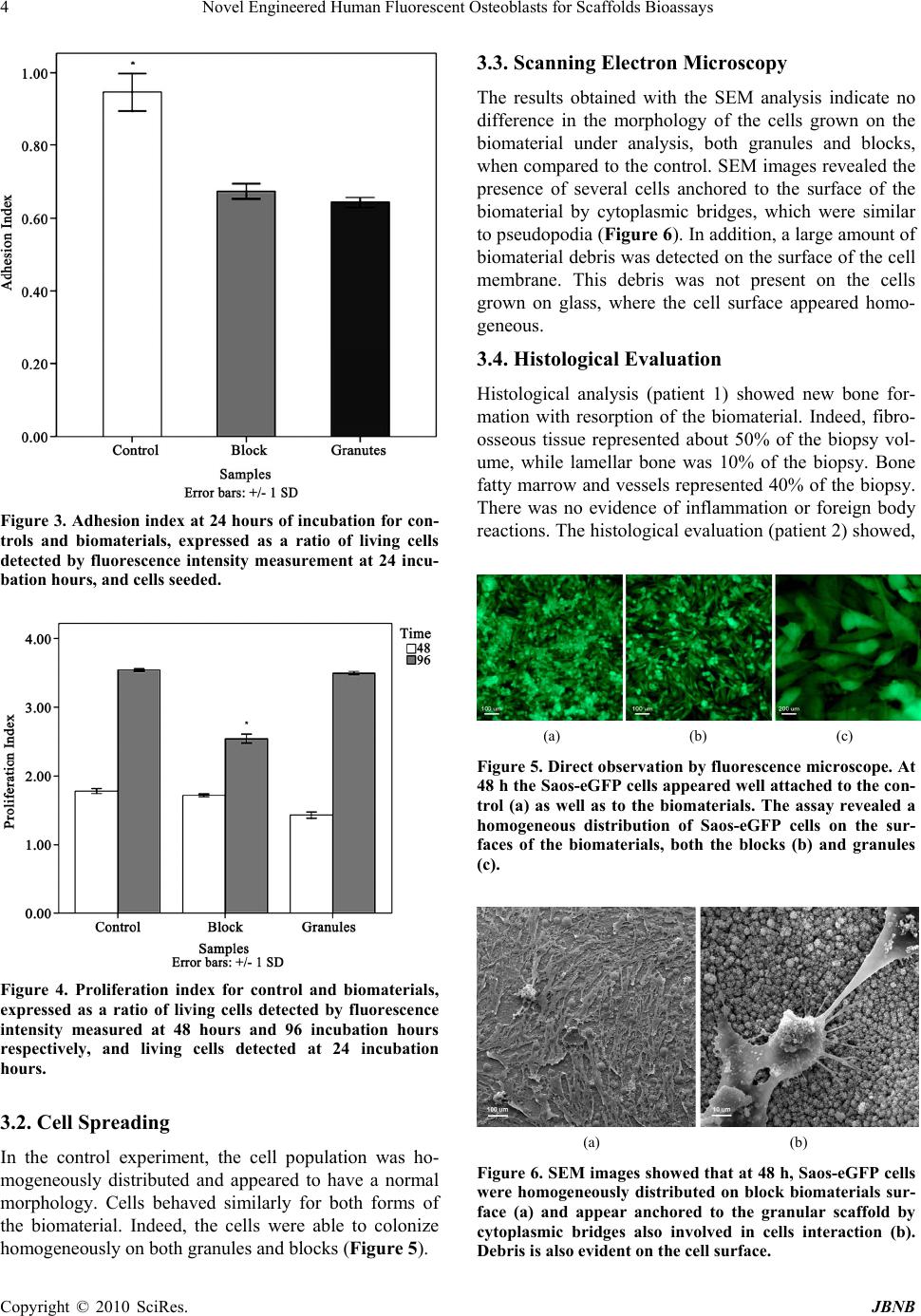 Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays 4 Figure 3. Adhesion index at 24 hours of incubation for con- trols and biomaterials, expressed as a ratio of living cells detected by fluorescence intensity measurement at 24 incu- bation hours, and cells seeded. Figure 4. Proliferation index for control and biomaterials, expressed as a ratio of living cells detected by fluorescence intensity measured at 48 hours and 96 incubation hours respectively, and living cells detected at 24 incubation hours. 3.2. Cell Spreading In the control experiment, the cell population was ho- mogeneously distributed and appeared to have a normal morphology. Cells behaved similarly for both forms of the biomaterial. Indeed, the cells were able to colonize homogeneously on both granules and blocks (Figure 5). 3.3. Scanning Electron Microscopy The results obtained with the SEM analysis indicate no difference in the morphology of the cells grown on the biomaterial under analysis, both granules and blocks, when compared to the control. SEM images revealed the presence of several cells anchored to the surface of the biomaterial by cytoplasmic bridges, which were similar to pseudopodia (Figure 6). In addition, a large amount of biomaterial debris was detected on the surface of the cell membrane. This debris was not present on the cells grown on glass, where the cell surface appeared homo- geneous. 3.4. Histological Evaluation Histological analysis (patient 1) showed new bone for- mation with resorption of the biomaterial. Indeed, fibro- osseous tissue represented about 50% of the biopsy vol- ume, while lamellar bone was 10% of the biopsy. Bone fatty marrow and vessels represented 40% of the biopsy. There was no evidence of inflammation or foreign body reactions. The histological evaluation (patient 2) showed, (a) (b) (c) Figure 5. Direct observation by fluorescence microscope. At 48 h the Saos-eGFP cells appeared well attached to the con- trol (a) as well as to the biomaterials. The assay revealed a homogeneous distribution of Saos-eGFP cells on the sur- faces of the biomaterials, both the blocks (b) and granules (c). (a) (b) Figure 6. SEM images showed that at 48 h, Saos-eGFP cells were homogeneously distributed on block biomaterials sur- face (a) and appear anchored to the granular scaffold by cytoplasmic bridges also involved in cells interaction (b). Debris is also evident on the cell surface. Copyright © 2010 SciRes. JBNB 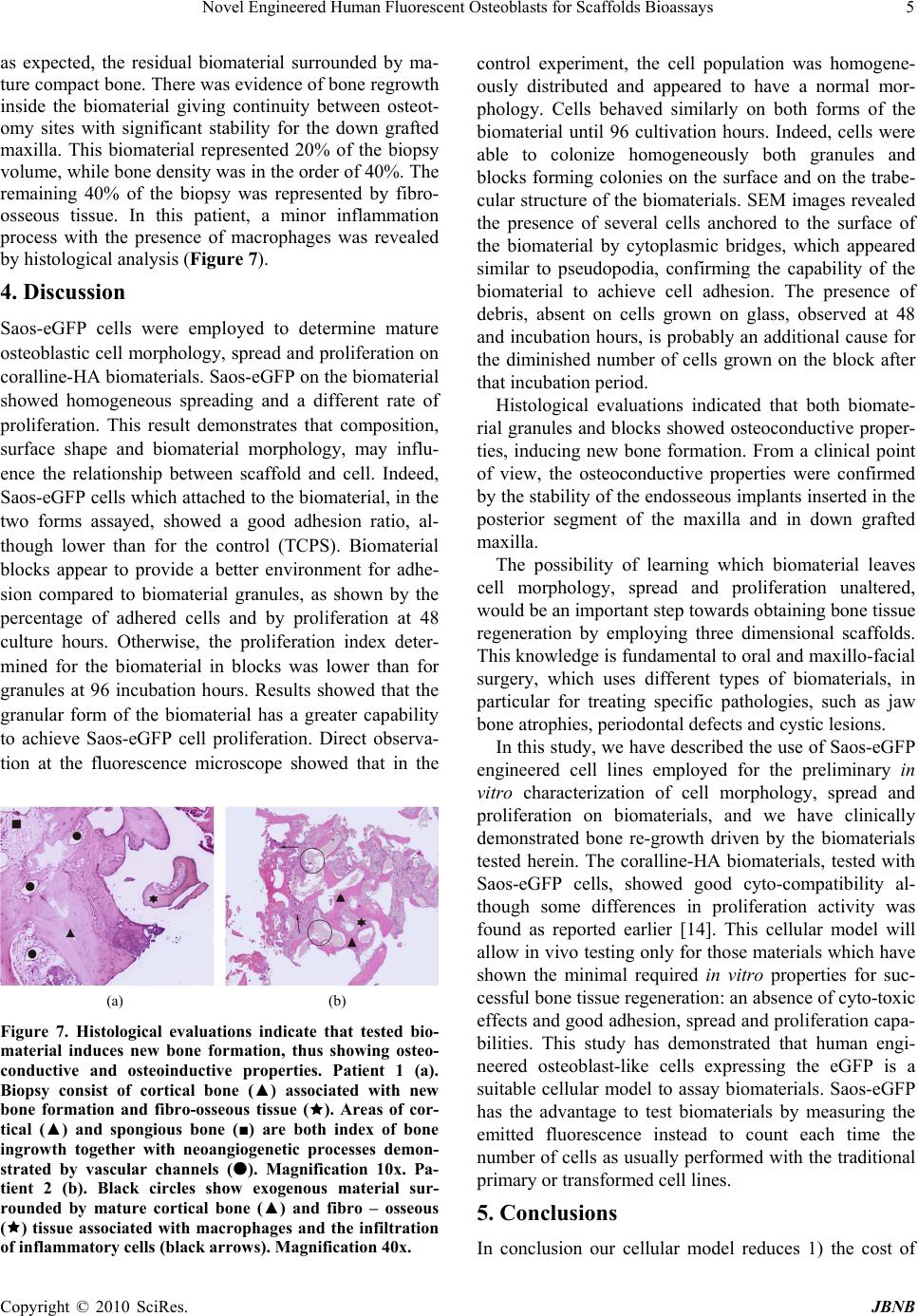 Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays5 as expected, the residual biomaterial surrounded by ma- ture compact bone. There was evidence of bone regrowth inside the biomaterial giving continuity between osteot- omy sites with significant stability for the down grafted maxilla. This biomaterial represented 20% of the biopsy volume, while bone density was in the order of 40%. The remaining 40% of the biopsy was represented by fibro- osseous tissue. In this patient, a minor inflammation process with the presence of macrophages was revealed by histological analysis (Figure 7). 4. Discussion Saos-eGFP cells were employed to determine mature osteoblastic cell morphology, spread and proliferation on coralline-HA biomaterials. Saos-eGFP on the biomaterial showed homogeneous spreading and a different rate of proliferation. This result demonstrates that composition, surface shape and biomaterial morphology, may influ- ence the relationship between scaffold and cell. Indeed, Saos-eGFP cells which attached to the biomaterial, in the two forms assayed, showed a good adhesion ratio, al- though lower than for the control (TCPS). Biomaterial blocks appear to provide a better environment for adhe- sion compared to biomaterial granules, as shown by the percentage of adhered cells and by proliferation at 48 culture hours. Otherwise, the proliferation index deter- mined for the biomaterial in blocks was lower than for granules at 96 incubation hours. Results showed that the granular form of the biomaterial has a greater capability to achieve Saos-eGFP cell proliferation. Direct observa- tion at the fluorescence microscope showed that in the (a) (b) Figure 7. Histological evaluations indicate that tested bio- material induces new bone formation, thus showing osteo- conductive and osteoinductive properties. Patient 1 (a). Biopsy consist of cortical bone (▲) associated with new bone formation and fibro-osseous tissue (). Areas of cor- tical (▲) and spongious bone (■) are both index of bone ingrowth together with neoangiogenetic processes demon- strated by vascular channels (). Magnification 10x. Pa- tient 2 (b). Black circles show exogenous material sur- rounded by mature cortical bone (▲) and fibro – osseous () tissue associated with macrophages and the infiltration of inflammatory cells (black arrows). Magnification 40x. control experiment, the cell population was homogene- ously distributed and appeared to have a normal mor- phology. Cells behaved similarly on both forms of the biomaterial until 96 cultivation hours. Indeed, cells were able to colonize homogeneously both granules and blocks forming colonies on the surface and on the trabe- cular structure of the biomaterials. SEM images revealed the presence of several cells anchored to the surface of the biomaterial by cytoplasmic bridges, which appeared similar to pseudopodia, confirming the capability of the biomaterial to achieve cell adhesion. The presence of debris, absent on cells grown on glass, observed at 48 and incubation hours, is probably an additional cause for the diminished number of cells grown on the block after that incubation period. Histological evaluations indicated that both biomate- rial granules and blocks showed osteoconductive proper- ties, inducing new bone formation. From a clinical point of view, the osteoconductive properties were confirmed by the stability of the endosseous implants inserted in the posterior segment of the maxilla and in down grafted maxilla. The possibility of learning which biomaterial leaves cell morphology, spread and proliferation unaltered, would be an important step towards obtaining bone tissue regeneration by employing three dimensional scaffolds. This knowledge is fundamental to oral and maxillo-facial surgery, which uses different types of biomaterials, in particular for treating specific pathologies, such as jaw bone atrophies, periodontal defects and cystic lesions. In this study, we have described the use of Saos-eGFP engineered cell lines employed for the preliminary in vitro characterization of cell morphology, spread and proliferation on biomaterials, and we have clinically demonstrated bone re-growth driven by the biomaterials tested herein. The coralline-HA biomaterials, tested with Saos-eGFP cells, showed good cyto-compatibility al- though some differences in proliferation activity was found as reported earlier [14]. This cellular model will allow in vivo testing only for those materials which have shown the minimal required in vitro properties for suc- cessful bone tissue regeneration: an absence of cyto-toxic effects and good adhesion, spread and proliferation capa- bilities. This study has demonstrated that human engi- neered osteoblast-like cells expressing the eGFP is a suitable cellular model to assay biomaterials. Saos-eGFP has the advantage to test biomaterials by measuring the emitted fluorescence instead to count each time the number of cells as usually performed with the traditional primary or transformed cell lines. 5. Conclusions In conclusion our cellular model reduces 1) the cost of Copyright © 2010 SciRes. JBNB  Novel Engineered Human Fluorescent Osteoblasts for Scaffolds Bioassays Copyright © 2010 SciRes. JBNB 6 the experiments; 2) the time of the execution; 3) while giving the same kind of results obtained with other cel- lular models, as demonstrate herein in assaying the well characterized HA biomaterials [9-11,14]. 6. Acknowledgements This study was supported in part by grants to M.T and P.F.N. from the Ministero della Università e Ricerca, University of Ferrara and University of Verona. Italy. Dr. Katia Campioni was supported by a post-doc fello- wship of the Fondazione Cassa di Risparmio di Cento, Italy. SEM analyses were carried out at the Centre of Elec- tron Microscopy, University of Ferrara, Ferrara, Italy. REFERENCES [1] J. A. Hubbell, “Bioactive Biomaterials,” Current Opinion in Biotechnology, Vol. 10, No. 2, 1999, pp. 123-129. [2] L. F. Reichardt, “Introduction: Extracellular Matrix Mole- cules. Guidebook to the Extracellular Matrix, Anchor, and Adhesion Proteins,” Oxford University Press, New York, 1999, p. 335. [3] E. H. Danen and A. Sonnenberg, “Integrins in Regulation of Tissue Development and Function,” The Journal of Pathology, Vol. 201, No. 4, 2003, pp. 632-641. [4] M. M. Sandberg and H. T. Aro, “Gene Expression during Bone Repair,” Clinical Orthopaedics and Related Re- search, Vol. 289, 1993, pp. 292-312. [5] T. T. Kon and J. Cho, “Expression of Osteoprotegerin, Receptor Activator of NF-kappaB Ligand (Osteopro- tegerin Ligand) and Related Proinflammatory Cytokines During Fracture Healing,” Journal of Bone and Mineral Research, Vol. 16, No. 6, 2001, pp. 1004-1014. [6] J. R. Lieberman and A. Daluiski, “The Role of Growth Factors in the Repair of Bone. Biology and Clinical Ap- plications.” Journal of Bone Joint and Surgery, Vol. 84, No. A-6, 2002, pp. 1032-1044. [7] D. M. Roy and S. K. Linnehan, “Hydroxyapatite Formed from Coral Skeletal Carbonate by Hydrothermal Ex- change,” Nature, Vol. 247, No. 438, 1974, pp. 220-222. [8] A. R. Vaccaro, K. Chiba, et al., “Bone Grafting Alternatives in Spinal Surgery,” The Spine Journal, Vol. 2, No. 3, 2002, pp. 206-215. [9] O. Shamsuria, A. S. Fadilah, A. B. Asiah, M. R. Rodiah, A. H. Suzina and A. R. Samsudin “In Vitro Cytotoxicity Evaluation of Biomaterials on Human Osteoblast Cells CRL-1543; Hydroxyapatite, Natural Coral and Polyhy- droxybutarate,” Medical Journal of Malaysia, Vol. 59, Suppl. B, 2004, pp. 174-175. [10] M. Barron, L. Franklin, J. Woodall Jr., S. Wingerter, H. Benghuzzi and M. Tucci, “Comparison of Osteoconduc- tive Materials on MG63 Osteoblast Cell Function,” Bio- medical Sciences Instrumentation, Vol. 43, 2007, pp. 248-253. [11] J. C. Fricain, J. Alouf, et al., “Cytocompatibility Study of Organic Matrix Extracted from Caribbean Coral Porites Astroides,” Biomaterials, Vol. 23, No. 3, 2002, pp. 673-679. [12] C. Morelli, G. Barbanti-Brodano, et al., “Cell Morphol- ogy, Markers, Spreading, and Proliferation on Orthopae- dic Biomaterials. An Innovative Cellular Model for the In Vitro Study,” The Journal of Biomedical Materials Re- search, Vol. 83, No. 1, 2007, pp. 178-183. [13] M. Tognon, C. Morelli, et al., “A Novel Genetically En- gineered Human Osteoblasts for the in Vitro Study of Biomaterials,” In: N. Ashammakhi, R. Reis and F. Chiel- lini, Eds., Topics in Tissue Engineering, 2008, pp. 1-13. [14] C.T. Begley, M. J. Doherty, D. P. Hankey and D. J. Wil- son, “The Culture of Human Osteoblasts upon Bone Graft Substitutes,” Bone, Vol. 14, No. 4, 1993, pp. 661-666. |

