Journal of Biomedical Science and Engineering
Vol.7 No.4(2014), Article ID:43593,8 pages DOI:10.4236/jbise.2014.74021
A Carbon Monoxide Sensing Film Based on Hemoglobin Allostery
YorikoTakada1,RisaOtsuka2, KosukeTsukada1,2
1Department of Applied Physics and Physico-Informatics, Faculty of Science and Technology, Keio University, Yokohama, Japan
2Graduate School of Fundamental Science and Technology, Keio University, Yokohama, Japan
Email: ktsukada@appi.keio.ac.jp
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 30 January 2014; revised 2 March 2014; accepted 9 March 2014
ABSTRACT
Carbon monoxide (CO) is a gaseous mediator,which is generated via anenzymatic reaction of hemeoxygenase, and it plays physiological roles to regulatecellular respiration and blood flow in the liver.The concentration and distribution of CO molecules in theliving body is unknown owing to a lack of a suitabletechnique for measuringthem in vivo. A needle-type CO sensor has been used for bioinstrumentation, but it is inappropriate for implantation in vivo and long-term monitoring. We developed a CO sensor sheet based on hemoglobin (Hb) allostery, asHbundergoes a conformational changeon CO binding.Hb was extracted from mice blood and mixed withagarose gel with a reducer to stabilize deoxy-Hb in the gel. CO-releasing molecules (CORM) were used to mimic CO-generating tissue, and the sensitivity of the Hbgelcould be regulated by Hb concentration. We defined the CO–Hb index, an absorbance ratio at 539 and 557 nm, to estimate the accumulation of captured CO in the gel.Itcorrelatively increased with CORM dose, indicating that gel-embedded Hb underwent a conformational change on CO binding, thereby acting as a CO sensor.We subsequently used the Hb-sensorsheet for two-dimensional imaging of CO distribution. CORM-containing gels with different sizes and doses were layered on this sheet. Size-and dose-dependentCO distribution was visualized by scanning the CO-Hb index in the sheet. Our Hb-based CO sensor sheet is composed ofbiocompatible materials and can be applied to detect low-level CO sources in the living body.
Keywords:Carbon Monoxide; Gas Sensor;Hemeoxygenase; Tumor Hypoxia

1. Introduction
Carbon monoxide (CO) is generated viaanenzyme reaction in which hemeoxygenase (HO) catalyzes the oxidative conversion of hemeto biliverdinIXα and ironin vivo[1] [2] . Because CO is a gaseous molecule, it freely diffuses in the living body and is used for the regulation of physiological functions. CO regulates vessel diameter and blood flow in the liver [3] and exerts protective effects on cells[4] . In particular,in tumor environments,heterogeneouslydistributed hypoxic areas are caused by an imbalance between oxygen supply and consumption in tumor tissues because of immature neovessels and blood flow disturbance [5] . Hypoxia inducible factor-1 (HIF-1) is a transcription factor that transactivates genes encoding proteins contributing to homeostatic responses to hypoxia [6] . HO-1 is located downstream to HIF-1;thus,CO gases can generate in hypoxic tumor tissues[7] .It is proposed that CO molecules generated in a tumor bind to hemoglobin (Hb) in erythrocytes and diffused in blood. However, the details of the pathological roles of CO in cancer remain unknown[8] .Extraneous CO gas, e.g.,which generated upon incomplete combustion,is taken up viapulmonary respiration and binds to Hb. Becausethe affinity of CO for Hb is 240 times stronger than that for oxygen [9] , excess binding of CO to Hbimpairs oxygen transport to tissues, leading to CO poisoning.
Commerciallydeveloped CO sensors are mainly of the needle type.When they are insertedin the living body, invasion is inevitable; further,this sensor type is unsuitable for long-term monitoring. Because the amount of CO produced in vivois infinitesimal, the sensor has higher sensitivity and biocompatibility. Weapplied the conformational change of Hb by CO bindingas a CO gas sensor. Itis well known that thedifference in Hbspectra between oxy-Hb and deoxy-Hb is used in pulse oximetry because the spectra differ substantially.The spectrum change depends on allosteric change of Hb conformation:relaxed (R)state in oxy-Hb and tense (T) state in deoxy-Hb. CO-Hb, similar to oxy-Hb,assumes the Rstate,and therefore,the absorption spectrum of CO-Hb is similar to that of oxy-Hb. Reports have described hypoxia sensing with Hbin vivo[10] [11] . However, Hb has not been used as a CO sensor because distinction between the spectra of CO-Hb and oxy-Hb is technically difficult.
In this study, we developed a sensor sheet made with biocompatible materialsfor detecting CO based on Hballostery. All Hb molecules in the sensor sheet are stabilized to the Tstate by inclusion of a reducing agent, and only when CO bindsto Hb, the conformation changes to the Rstate (Figure 1). CO-releasing molecules(CORM) were used as a CO source to mimic living tissues, such as tumor tissues. We determined the sensitivity of the sensor sheet, and CO distribution was two-dimensionally(2D) imaged by scanning the absorption spectra.
2. Materials and Methods
2.1. Hb Preparation and CORM-Containing Gel as a CO Source
All experimental protocols were approved by the Animal Care Committee of Keio University School of Medicine. Fresh heparinized venous blood was obtained from male BALB/c mice. Erythrocytes were separated by centrifugation at 2500 rpm for 5 min. Plasma and buffy coat were removed, and erythrocytes were washed thriceusing phosphate-buffered saline (PBS). The packed erythrocytes were diluted using 10 times theirvolume of distilled water in order to lyse them.After centrifugation at 10,000g for 30 min, the supernatant, without membrane component, was sampled.Agarose powder (500 mg) was suspended in 500 ml of PBS and dissolved by
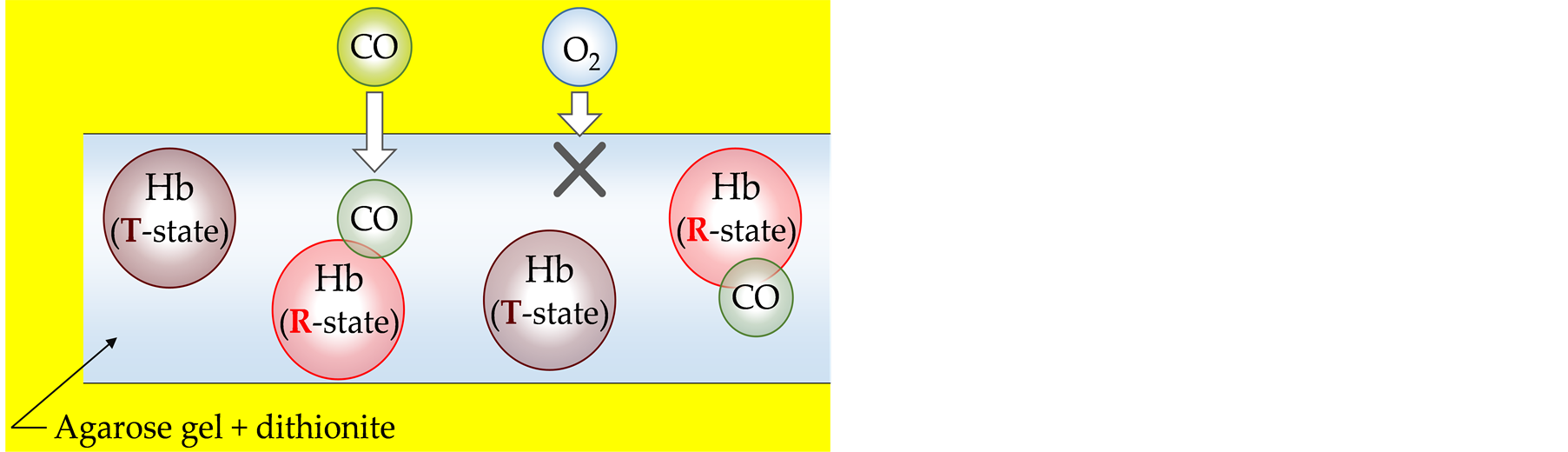
Figure 1.Schematic illustration of Hb-based CO sensor in gel. Conformation of Hb is stabilized by dithionite to the Tense (T) state and changed to the Relaxed (R) state only when CO binds to Hb.
heating in a microwave oven. When the temperature decreased to50˚C, Hb solution and dithionite [12] -[14] were added to the agarose solution, and the mixture was stored at 4˚C until use.By mixing dithionite in the agarose gel, Hb conformation was stabilized to the Tstate until CO gas bound to the heme.For 2D imaging of COHb,the Hbgel was made in the form of a sheet(thickness, 2.5 mm; diameter, 35 mm). For measurement of the absorption spectra, the Hbgelwas placedin an optical cell. CORMwas used as a low-levelCO generation source. CORM was dissolved in agarose at various concentrationsand placed on the Hbgel for spectrum analysis and CO imaging under a microscope.
2.2. Maintaining Hb Absorption Spectra and Defining CO-Hb Index
The absorption spectra of the Hb gel weremeasured using a spectrophotometer (Smart Spec Plus, Biorad).Figure 2 shows the spectra of air-saturated oxy-Hb,dithionite-treateddeoxy-Hb, and CORM-layered CO-Hb. As noted above, dithionite changed all oxy-Hb (Rstate) todeoxy-Hb (Tstate) so that only deoxy-Hb or CO-Hbcouldbe presentin the gel. Theproportionof CO-Hb present in the gel was quantified by theratio of absorbance at 539 nm, the absorbance peak of CO-Hb, to that at557 nm, the absorbance peak ofdeoxy-Hb, according to the following Formula (1).
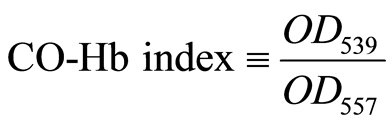 (1)
(1)
The Hb gels in optical cuvettes for spectralmeasurement areshown in Figure 3. Oxy-Hb and CO-Hbwere bright red, indicatingthe Rstate, whereas deoxy-Hbwas dark purple, indicating that Hb conformation
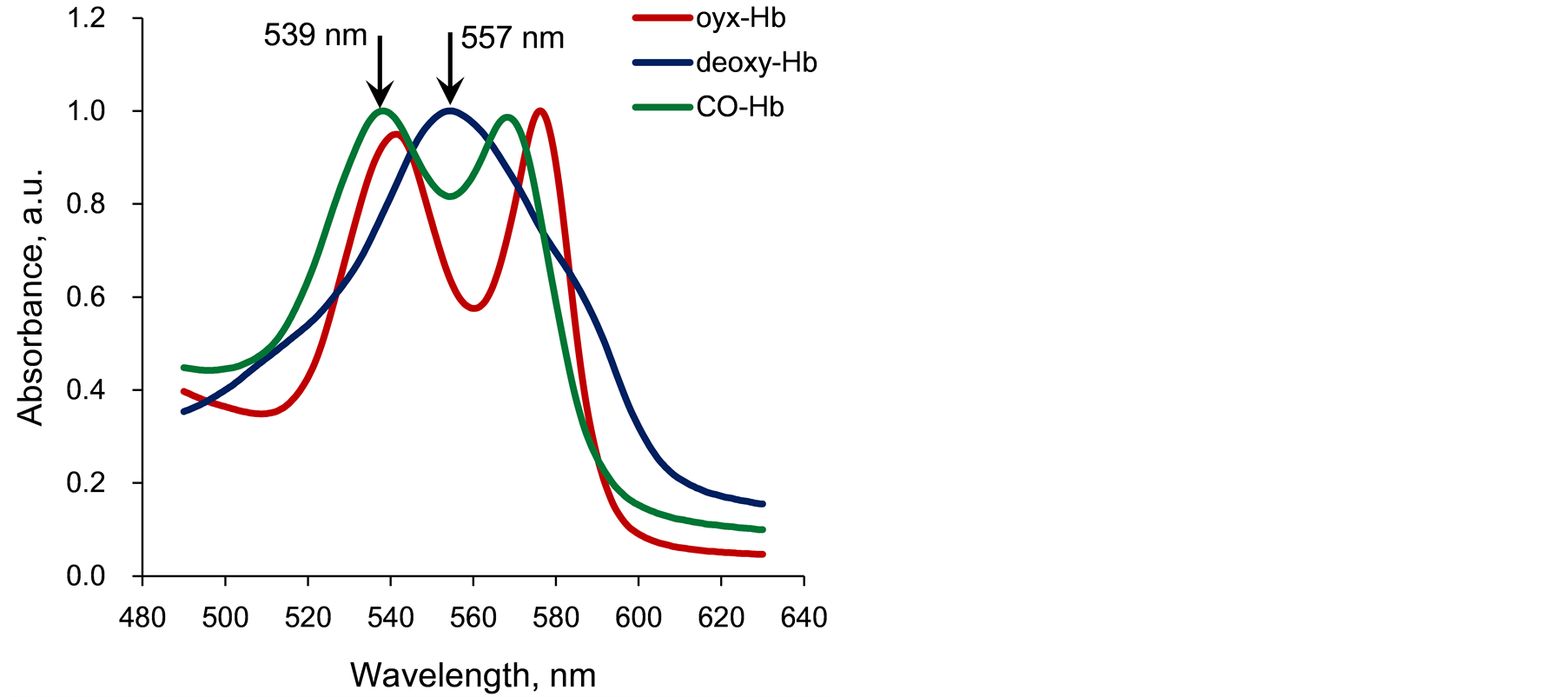
Figure 2.Absorption spectra of air-saturated oxy-Hb, deoxy-Hb, and CO-Hb extracted from mice erythrocytes. Absorption ratio at 539 and 557 nm was used for CO-Hb index.

Figure 3.Images of Hbgel in glass cells for spectrum analysis. From the left, air-saturated oxy-Hb, deoxy-Hbdeoxygenated with dithionite, and CO-Hb covered with a layer of CORM-containing gel.
changed due todeoxygenation and CO binding to Hb molecules.
2.3. Fabrication of the HbGel Sheet for CO-Hb Imaging
The Hbgel sheet was transformed intoa disk (thickness, 2.5 mm;diameter, 35 mm)in a cell culture dish. CORMgel was set into circular sheets with a diameter of 3.5, 5.2, and7.5 mm at a fixed CORM concentration of 10 mM.In a similar manner, gels were set usingCORM concentrations of 5, 10, and 20 mM at a fixed diameter of 9 mm. These CO-releasing gel sheets were layered on the Hbgel sheet for2D imaging of CO-Hb.
2.4. Optical Setup for CO Imaging on Sensor Film
The optical setup for imaging CO-Hb in the gel sheet isshown in Figure 4. The Hbgel was placedunder an objective lens (20×) and irradiated with white light transmitted from anLED under the Hbgel. The light transmitted through the Hbsheet passed through the objective,andabsorption spectra were obtained with a multi-channel CCDvia a polychrometer (C5094, Hamamatsu Photonics).The CO-Hb index was calculated from the absorption spectra. The Hbgel sheet was scanned in 20 mm squarewith a spatial resolution of 0.5 mmusinga X-Ystage installed in the microscope. The CORM-containing gel,as asource of CO gas,was placedon aHbgel sheet for 15 min until scanning.Two sets of CO-releasing gel sheets were prepared as follows:sampleswithvarying CORMconcentrations of5,10,and 20mMat a constantsize of 9 mm, and samples with varying gelsizes of 3.5, 5.2, and 7.5 mm at a constant CORM concentration of 10 mM.The scanned CO-Hb index was imaged in pseudocolor.
3. Results
3.1. Time Course Change in CO-Hb Index Depending on CORM Concentration
Figure 5 shows the timecourse change in CO-Hb index in anoptical cell with aHb concentration of 7.5%. CORM gels with varyingconcentrations were placedon the Hbgel in the optical cell at time 0. CO-Hb index increased in a short time with an increase in the concentration of CORM, but no change was observed in the absence of CORM.At a CORM concentrationof 1.0 mM, CO-Hb index continued to increase over 3 h and stabilized after 10 h. However, at a low concentration of 0.3mM, CO-Hb index didnot reach CO-Hb index 1.2, owing to the complete release of CO molecules from CORM after 3 h. This experiment showed that the Tstate molecules bound minute amounts of CO gas and changed to the Rstate.CO-Hb index represents the total amount of CO molecules gradually released from CORM molecules, indicating that this Hb-gel sensor can sense a CO source thatcontinuously and infinitesimallygenerates CO, such astumor tissues, in theliving body.
Figure 6 shows the timecourse change in CO-Hb index with Hb concentration in the gelsheet at a fixed CORM concentration of 0.5 mM.The lowerthe Hb concentration in the gel, the more rapidly CO-Hb index responded and became saturated. At 7.5% Hb, the index continued to increase after 3 h.The Hb concentration corresponds to the numberof acceptors for CO molecules;a higher concentration of Hb in the gel leads to
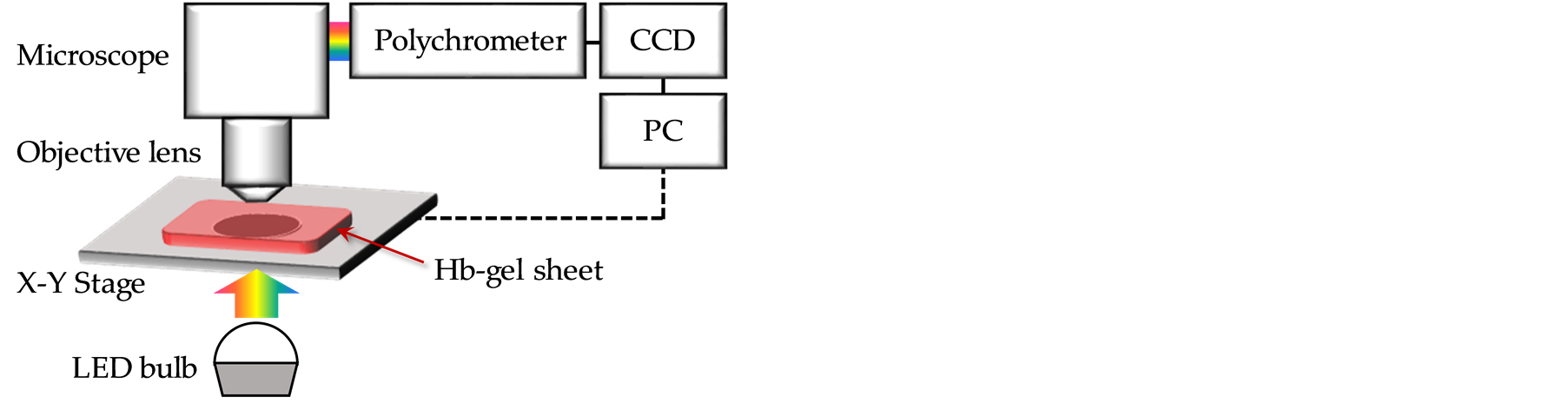
Figure 4.Microscope setup for 2D imaging of CO-Hb in the gel sheet.White light transmitted from an LED passed through the Hbgel sheet and was corrected with an objective lens.The spectrum was obtained using a polychrometer. X-Y stage was controlled to visualize 2D image of CO–Hb index.
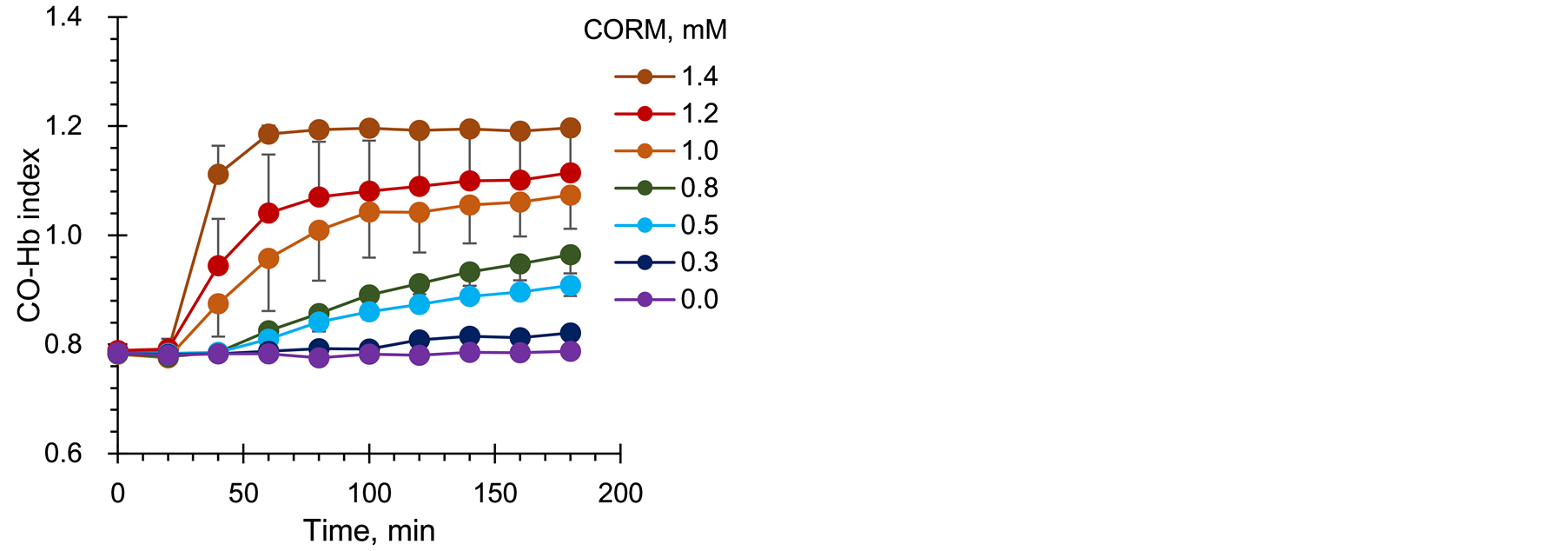
Figure 5.Time course change in CO-Hb index with CORM concentration in the layered gel on the Hb gel.
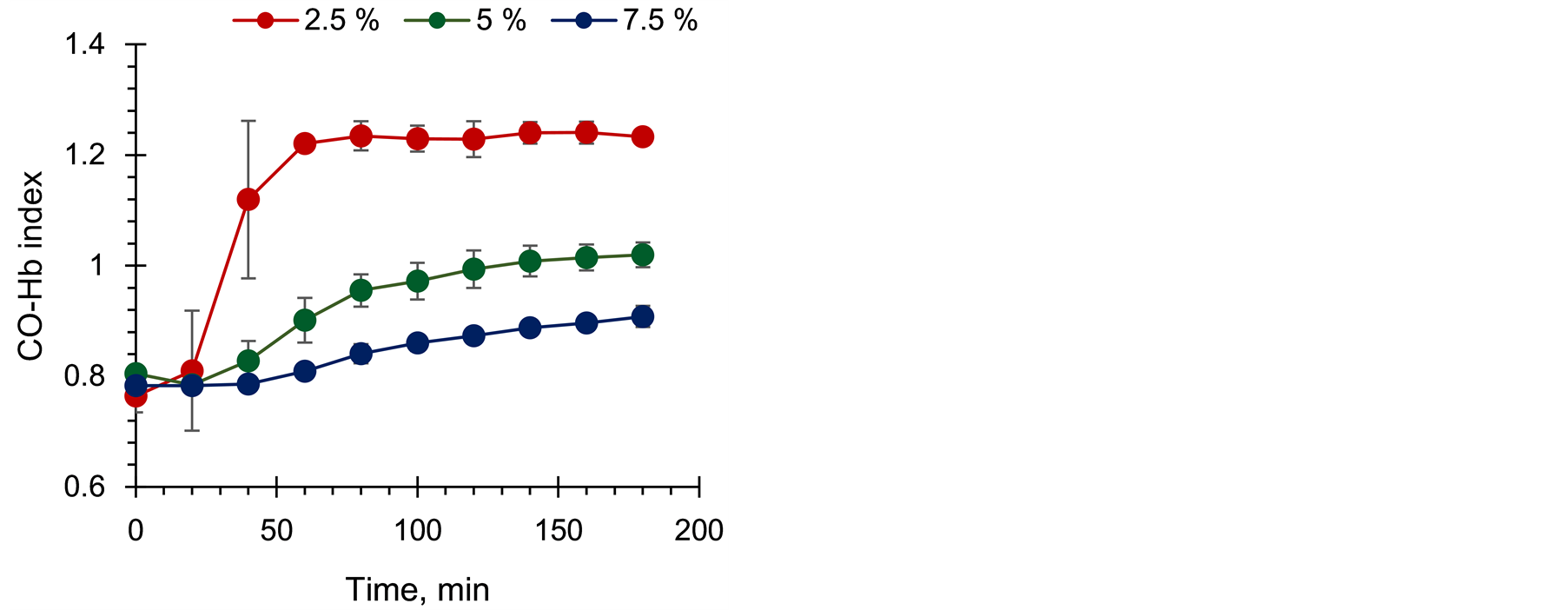
Figure 6.Time course change in CO-Hb index with Hb concentration in the Hbgel.
agradual increase in CO-Hb indexwith aconstant CO binding rate. Therefore, the Hb concentration in the gel is a determinant of sensitivity of the gel sheet.
3.2. CO-Dependent Change in CO-Hb Index atFixed Time Interval
Figure 7 shows the change in CO-Hb index with a CORM concentration at 5% on theHb gel. CO-Hb index didnot change in the absence ofCORM,as shown inFigure 5. At180 min, the index increaseddose-dependently at a low CORM concentration, indicatingthatHb in the gel sheet continuallybound to the released CO. These results indicate that the Hbgel sheet can be applied to detect low levels of CO on long-term accumulation. Because Hb concentration determines the sensor sensitivity as shown in Figure 6, it is important to establish theHb concentration required for measurementbyconsidering the amounts of CO released from the target. CO-Hb index should be below 1.2 at the end point of measurement for accurate measurement.
3.3. Spatial Imaging of CO Concentration on Sensor Film
The 2D change in CO-Hb indexwas imaged by scanning the Hbgel sheet. A double-layeredCORM-containing gel on the Hbgel was removed after 15 min and absorption spectra were obtained with the microscope systemillustrated in Figure 4. Figure 8(a) shows a pseudocolor image of CO-Hb index in theHbgel sheet. CORM concentrationsin the upper gels varied between5, 10, and 20 mM with the same diameter(9 mm). Circular changes inthe Hbindex depending on CORM concentration were observed. Higher CORMdosesresulted in an increase in the size,owing to the diffusion of CO gas in the Hbgel sheet. Figure 8(b) illustrates thesize-de-
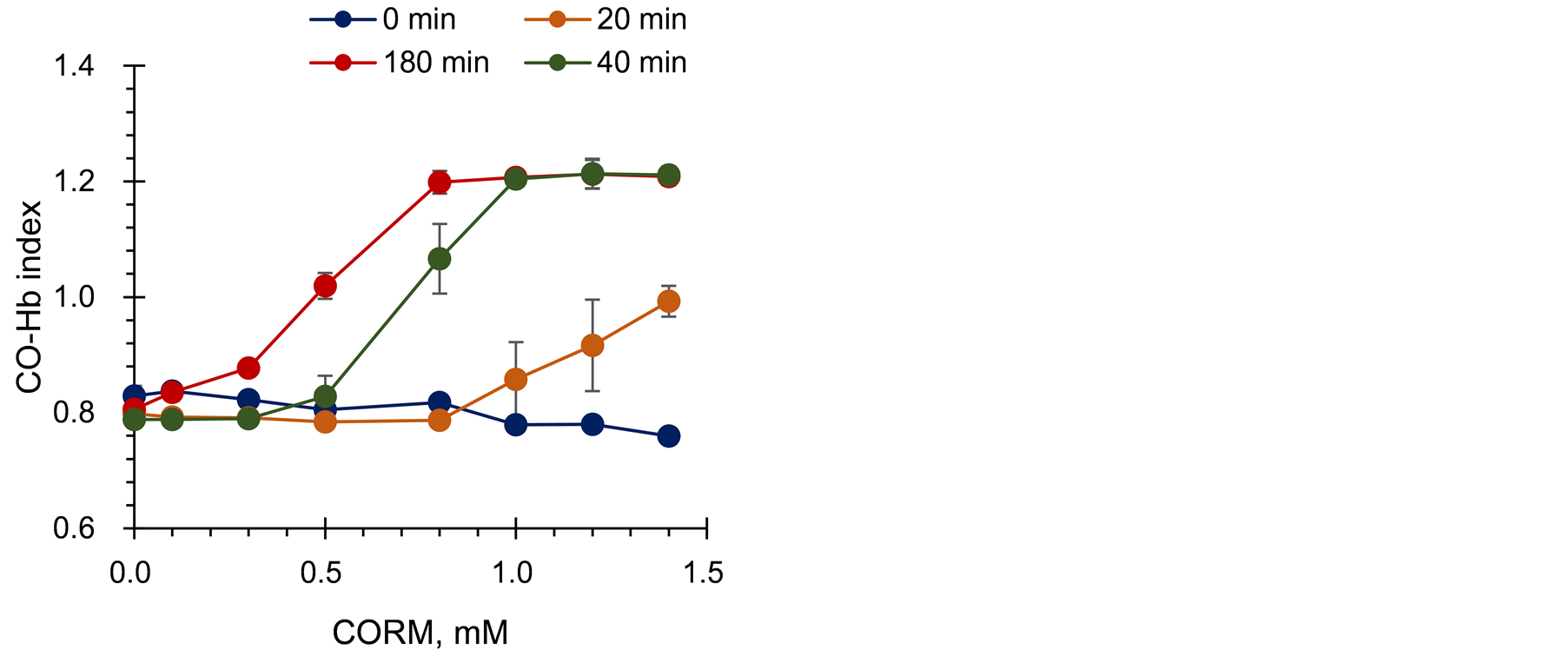
Figure 7.Relationship between CORM concentration and CORM index at the contact time of the Hb gel with CORM-containing gel.

Figure 8.Visualization of CO-Hb index in the Hbgel sheet. Circular gels with a diameter of 9mm at CORM concentration of 5, 10, and 20 mM were layered on the Hbgel sheet (a);Likewise, gels of diameter 3.5, 5.2and 7.5 mm at a fixed concentration of CORM at 10 mM were imaged (b).
pendentchange in CO-Hb index for3.5, 5.2, and 7.5-mm gels at a constant CORM concentration. These results indicate that theHbgel sheet can detect and image CO-releasing substrates with different sizes and concentrations.
4. Discussion
Here we developed a CO sensor based on Hballostery. CO-Hb index derived from theratio of Hb absorptions at 539 and 557 nm was effectivefor estimatingCO concentrations. Although severaltypes of gas sensors have been developed [15] -[17] , in general, CO gas sensors have beenlimited to the needle type for bioinstrumentation. Because our sensor was basically composed ofmaterials with a high biocompatibility, it can be implanted for long-term monitoring of CO generation in vivo. We used CORM as a small generator of CO for potential biometric applications. However, our Hbgel sheet can be applied for industrialor generaluse by increasing the Hb content in the gel.
The addition of a reducer tothe gel removed oxygen molecules and stabilized Hb in the Tstate. An increase inthe Rstate, equivalent to an increase in CO-Hb index, corresponds toaconformational change of Hb by binding CO, and not oxygen. Although the absorption spectra of oxy-Hb and CO-Hb are similar, the absorption peaks of CO-Hb shift toward shorter wavelengths compared with oxy-Hb[18] . The quantification of CO-Hbusing thisshift has been described[19] . However, acquiring Hb spectra is required forestimatingthe amount of the shift, consuming time for measurement, and data processing. In particular, when CO-Hb is scanned for 2D imaging, absorption data should be processed at each point (pixel) in the scan area. In our method, because oxy-Hbwasnot presentin the gel, only absorption at twowavelengths, those characterizing the R and Tstates, needto be measured. We used a polychrometer to obtain Hb spectra for CO imaging. However, CO-Hb index couldbe directly calculated using twophotodetectors with filters for 539 and 557 nm, allowing simultaneous measurement. This method should be appropriate for fast scanning of CO-Hb in the gelsheet.
To remove oxygen and to change oxy-Hb to deoxy-Hb in the gel, sodium dithionite was used as a reducing agent[12] -[14] , and all Hb was stabilized to the Tstate. However, dithionite does not durably remove oxygen and the effect fades with time. In addition, escape of dithionite fromthe gel could reducethe Tstate stabilization effect. We applied glucose oxidase and catalase system [20] to remove oxygen molecules, but glucose oxidaseis yellow, which affects absorption measurements (data not shown). Another method should be contrived to maintain the Tstate of Hb without absorption at 539 and 557 nm.
The sensitivity can be adjusted by changing Hb concentration in the gelsheet, as shown in Figure 6. If Hb concentration is lowered, CO-Hb index will increase at low CO levels, corresponding to enhancedsensitivity. However, in general, lower Hb concentrations lead to a reducedsignal-to-noise ratioowingto low absorbance. Some type of noise reduction is required, e.g., by averaging signals. Our sensor sheet detects cumulative amounts of CO-Hband not momentary absolute values. Itshould thus be suitable for CO detection in tissues that continuously generate minute amounts of COover long periods, such astumor tissues.
Here we used Hb extracted from mouseblood by centrifugation and membrane removal. However, the extract contained not onlyHb butalso several ions and proteins, such as glycolytic enzymes, in erythrocytes. Wesupposedthat molecules other than Hb were not in sufficient abundance to affect the absorption used for determining CO-Hb index. However, the dissociation curve of Hb shifted by binding 2,3-bisphosphoglycerate, an allosteric effector, so thatpurification could change the property of R–T transition by CO binding. In addition, the increase of content of met-Hbwithrepeated purification decreases sensitivity, indicating that the relationship between purification and sensitivity should be determined for future practical application. Finally, the durability of the sensor should be tested for long-term use because the Hb protein can denature or degrade with time.
5. Conclusion
We developed a CO sensor based on Hballostery and successfully imaged CO-generating gels, mimicking CO sourcessuch as tumor tissues in theliving body. Further studies of Hb purification, quantification of CO, and durability of the gel sheet are needed for practical application.Because thesensor has a high biocompatibility, it maybe expected to be useful forlong-term monitoring in vivo as well as for other general uses.
References
- Maines, M.D. (1997) TheHemeOxygenase System: A Regulator of Second Messenger Gases. Annual Review of Pharmacology and Toxicology, 37, 517-554.http://dx.doi.org/10.1146/anurev.pharmtox.37.1.517
- Kikuchi, G., Yoshida, T. and Noguchi, M. (2005) HemeOxygenaseand Heme Degradation. Biochemical and Biophysical Research Communications, 338, 558-567.http://dx.doi.org/10.1016/j.bbrc.2005.08.020
- Suematsu, M., Tsukada, K., Tajima, T., Yamamoto, T., Ochiai, D., Watanabe, H., Yoshimura, Y. andGoda, N. (2005) Carbon Monoxide as a Guardian against Hepatobiliary Dysfunction.Alcoholism: Clinical and Experimental Research, 29, 134S-139S.http://dx.doi.org/10.1097/01.alc.0000189273.49148.87
- Leffler, C.W., Parfenova, H. andJaggar, J.H. (2011) Carbon Monoxide as an Endogenous Vascular Modulator. The American Journal of Physiology—Heart and Circulatory Physiology, 301, H1-H11. http://dx.doi.org/10.1152/ajpheart.00230.2011
- Jain, R.K. (2008) Taming Vessels to Treat Cancer. Scientific American,298, 56-63. http://dx.doi.org/10.1038/scientificamerican0108-56
- Semenza, G.L. (2012) Molecular Mechanisms Mediating Metastasis of Hypoxic Breast Cancer Cells. Trends in Molecular Medicine, 18, 534-543. http://dx.doi.org/10.1016/j.molmed.2012.08.001
- Jozkowicz, A., Was, H. andDulak, J. (2007) HemeOxygenase-1 in Tumors: Is It a False Friend? Antioxid Redox Signal, 9, 2099-2117.http://dx.doi.org/10.1089/ars.2007.1659
- Coburn, R.F., Forster, R.E. and Kane, P.B. (1965) Considerations of the Physiological Variables That Determine the Blood Carboxy Hemoglobin Concentration in Man. Journal of Clinical Investigation, 44, 1899-1910. http://dx.doi.org/10.1172/JCI105296
- Leach, R.M., Rees, P.J. andWilmshurst, P. (1998) Hyperbaric Oxygen Therapy. BMJ, 317, 1140-1143. http://dx.doi.org/10.1136/bmj.317.7166.1140
- Liu, P., Zhu, Z., Zeng, C. andNie, G. (2012) Specific Absorption Spectra of Hemoglobin at Different PO2Levels: Potential Noninvasive Method to Detect PO2 in Tissues. Journal of Biomedical Optics, 17, 125002. http://dx.doi.org/10.1117/1.JBO.17.12.125002
- Sorg, B.S., Moeller, B.J., Donovan, O., Cao, Y. and Dewhirst, M.W. (2005) HyperspectralImaging of Hemoglobin Saturation in Tumor Microvasculature and Tumor Hypoxia Development. Journal of Biomedical Optics, 10, 44004. http://dx.doi.org/10.1117/1.2003369
- Hamada, K., Okazaki, T., Shukuya, R. andKaziro, K. (1962) Thedeoxygenation of Dilute Oxyhemoglobin by Sodium Dithionite. The Journal of Biological Chemistry, 52, 374-376.
- Dalziel, K. and O’Brien, J.R.P. (1961) TheKinetics of Deoxygenationof Human Haemoglobin. The Journal of Biological Chemistry, 78, 236-245.
- Dalziel, K. and O’Brien, J.R.P. (1957) Side Reactions in the Deoxygenationof Dilute Oxyhaemoglobin Solutions by Sodium Dithionite.The Journal of Biological Chemistry, 67, 119-124.
- Sharma, S. and Madou, M.(1967) A New Approach to Gas Sensing with Nanotechnology.Philosophical Transactions. Series A, Mathematical, Physical, and Engineering Sciences, 370, 2448-2473.
- Bârsan, N. and Weimar, U. (2003)Understanding the Fundamental Principles of Metal Oxide Based Gas Sensors; the Example of CO Sensing with SnO2Sensors in the Presence of Humidity. Journal of Physics: Condensed Matter,15, R813-R839.http://dx.doi.org/10.1088/0953-8984/15/20/201
- Arafat, M.M., Dinan, B., Akbar, S.A. andHaseeb, A.S.M.A. (2012) Gas Sensors Based on One Dimensional Nanostructured Metal-Oxides: A Review.Sensors, 12, 7207-7258.http://dx.doi.org/10.3390/s120607207
- Klendshoj, N.C., Feldstein, M. and Sprague, A.L. (1950) TheSpectrophotometric Determination of Carbon Monoxide. The Journal of Biological Chemistry, 183, 297-303.
- Hunter, F.T.(1951) TheQuantitation of Mixtures of Hemoglobin Derivatives by Photoelectric Spectrometry. Charles C. Thomas, Springfield.
- Wilson, R. and Turner, A.P.F. (1992) Glucose Oxidase: An Ideal Enzyme. Biosensors and Bioelectronics, 7, 165-185. http://dx.doi.org/10.1016/0956-5663(92)87013-F

