Natural Science
Vol.6 No.12(2014), Article ID:48818,18 pages DOI:10.4236/ns.2014.612090
Global Proteomics of the Extremophile Black Fungus Cryomyces antarcticus Using 2D-Electrophoresis
Kristina Zakharova1, Katja Sterflinger1, Ebrahim Razzazi-Fazeli2, Katharina Noebauer2, Gorji Marzban3
1Austrian Center of Biological Resources and Applied Mycology, Department of Biotechnology, University of Natural Resources and Life Sciences, Vienna, Austria
2Proteomics Unit, VetCore Facility for Research, University of Veterinary Medicine, Vienna, Austria
3Plant Biotechnology Unit, Department of Biotechnology, University of Natural Resources and Life Sciences, Vienna, Austria
Email: gorji.marzban@boku.ac.at
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 3 June 2014; revised 5 July 2014; accepted 15 July 2014
Abstract
The microcolonial black fungus Cryomyces antarcticus is an extremophile organism growing on and in rock in the Antarctic desert. Ecological plasticity and stress tolerance make it a perfect model organism for astrobiology. 2D-gel electrophoresis and MALDI-TOF/TOF mass spectrometry were performed to explore the protein repertoire, which allows the fungus to survive in the harsh environment. Only a limited number of proteins could be identified by using sequence homologies in public databases. Due to the rather low identification rate by sequence homology, this study reveals that a major part of the proteome of C. antarcticus varies significantly from other fungal species.
Keywords: Microcolonial Fungi, Extremophiles, Proteomics, Protein Characterization

1. Introduction
The discovery of extreme earth’s environments and the organisms that inhabit them has awakened curiosity of humans on the limits of life on and outside our planet and even through the universe. For a long time desert regions including the hot deserts of North America (Great Basin desert), Africa (Sahara desert, Kalahari desert), Asia (Gobi desert), the Middle East (Syrian desert), Australia (Great Victoria desert) as well as the permafrost environments up to the cold, dry valleys of Antarctica, Arctic and Patagonia were believed to be too hostile for any forms of life [1] . Today, we know that deserts are full of life forms with representatives of all domains and throughout all kingdoms of life [2] . Doubtlessly, deserts together with the deep biosphere provide the most extreme conditions for life on Earth and triggered manifold adaptation mechanisms in plants, animals and microorganisms [3] [4] .
Antarctic ice-free deserts are among the closest analogues of Mars due to very cold and very dry conditions, extremely poor nutrient availability, and large fluxes of UV-light [5] . Despite of the hostile conditions, life is present in cryptic niches: microorganisms are organized in cryptoendolithic communities mostly dominated by lichens and by non-lichenized fungi and cyanobacteria [6] . Some of the components of those communities are black meristematic fungi, also known as microcolonial fungi (MCF) [7] [8] .
The enormous stress resistance of MCF makes them important models for understanding stress resistance and adaptations in Eukaryont in general. Some special characteristics of the black fungi assumed to be involved in stress resistance are: 1) the cell composition of the fungi which consists mostly of very complex lipids (e.g. mono-, diand tri-acylglycerols, phosphotidilcholine, phosphatidylethanolamines, sterols, sterol ethers, phosphatidilrthanolamines and free fatty acids); 2) pigments with their various protective effects as e.g. melanins, carotenoids in colourless and brown-red formations; and 3) the dense cell wall built of chitin, melanin and polysaccharides [9] . All those characteristics make the fungi able to survive diverse stress factors [10] -[12] ; they are even known to be survivals of outer space and Mars like environments [13] [14] .
At the same time the described characteristics of these fungi make it difficult to perform analysis on the cellular level. For instance the cell wall is the main obstacle in protein extraction from fungal biomass. Moreover, due to rigid cell wall and melanin content, also DNA extraction and purification have not been convenient to obtain genomic data of MCFs [15] . Up to now whole genome sequences of only two strains of black microclonial fungi—Coniosporium apollinis and Cryomyces antarcticus—exist in the public databases, however without annotation. In addition, the studied MCF strains have an extremely slow growth e.g. they need at least 30 days to reach the minimal quantity of biomass level for any type of experiment.
Up till now there are few proteomic data obtained from MCF showing protein patterns and their changes under varying conditions [12] [14] [16] [17] . However, no identification of proteins was done in any of the black MCF. Thus it was the aim of this study to perform the first protein identifications of the very particular fungal species C. antarcticus cultivated under laboratory conditions as a basis for a deeper understanding of the systems biology of this fungus and related extremophile genera.
2. Materials and Methods
2.1. Model Organisms
C. antarcticus, MA 5682, is one of the most extremophilic eukaryote on Earth was used as a model organism in this and previous studies. The strain was kindly provided by the Italian National Museum of Antarctica and was preserved in the ACBR culture collection (Austrian Centre of Biological Resources and Applied Mycology, http://www.acbr-database.at/). Inoculi were prepared as cell suspensions and drop-inoculated onto sterilized cellophane membranes (Model 583 Gel Dryer Backing; Bio-Rad, USA) placed on the surface of 2% malt-extract agar (MEA, Applichem, Germany). The fungus was grown at its optimal growth temperature: 15˚C (288 K) for 30 days, and the biomass was collected by scratching the material from the plates using a sterile scalpel, transferred into a sterile tube (Eppendorf, Germany), immediately frozen in liquid nitrogen and stored at −80˚C (193 K) for further analysis. All the experiments were performed in triplicate.
2.2. Proteomics
2.2.1. Protein Extraction
Protein extraction from biomass and the procedure of 2D gel electrophoresis was optimized for black fungi. Prior to the extraction, the biomass was disrupted in liquid nitrogen with mortar and pestle. Approximately 400 mg of the biomass were transferred into ice-cold 2 ml O-ring screw-capped microfuge tube (ribolyzer-tubes, Biozym Scientific GmbH, Germany). Subsequently the samples were washed two times with 500 μl of sterile saline solution (NaCl 0.9%, sodium chloride, Applichem, Germany), vortexed shortly and centrifuged for one minute after the first and 2 minutes after the second washing at maximal speed (200 × g) at 4˚C and supernatant was discarded. During the whole procedure, samples were kept on ice. Then approximately 400 mg of acidwashed 0.5 mm diameter glass beads (0.25 - 0.5 cm, Roth, Germany) were added. Prior to extraction, glass beads were washed as follows: approximately 100 g of glass beads were put into 200 ml distilled H2O into a beaker and stirred using a glass rod. Then Triton X-100 (Merck, Germany) 100 ml was added under stirring until the detergent was completely dissolved. The Supernatant was carefully poured off and precipitate washed for 2 times 10 min each with distilled H2O, followed by washing two times with 95% ethanol (Merck, Germany) under stirring for 2 minutes. Then beads were rinsed 2 times with distilled H2O, then 2 times with 6 N HCl (hydrogen chloride, Merck, Germany), then rinsed 1 time with distilled H2O and washed 2 times with 6 N HNO3 (nitricacid, Merck, Germany). The cleaning of beads is completed after 3 times washing with distilled H2O until the pH reached the value of approximately 6. Then the supernatant was discarded and the beads in the beaker were dried at 80˚C for 36 hours. The beads were added to the sample together with 1200 μl of Lysis Buffer (tris 0.6 g (Invitrogen, USA), EDTA disodium salt 186 mg (Calbiochem, Germany), KCl 745.5 mg (potassium chloride, Merck, Germany), PVPP 1 g (polyvinylpolypyrrolidone, BioChemica, AppliChem, Germany), Sucrose 30 g (Merck, Germany)) in 100 ml MilliQ H2O (Millipore, MA, USA). Lysis buffer should be stored at +4˚C for next use. The biomass was disrupted using Bead Beater/Rybolyzer (MP Bio, USA) and the tubes kept on ice between all the steps.
After disruption the lysate was transferred into 15 ml centrifugation tubes (Corning, Mexico) and 3 ml of tris-buffered phenol pH 8.0 (Sigma, USA) was added (mix the buffer before pipetting). Two ml of the buffer were added directly into the tube and with the rest the homogenized material was washed off the Rybolyzer tube. The tube was vortexed for 15 min at room temperature, followed by centrifugation for 10 min at 200 × g at 4˚C. The phenol phase (upper phase) was transferred into a fresh pre-weighed tube (Corning, Mexico) and added 5 volumes of cold precipitation solution (ammonium acetate 7.7 g (AppliChem, Germany)) in 100% methanol (Merck, Germany). The precipitation was done overnight at −20˚C.
After the precipitation, samples were centrifuged for 30 min at 500 × g at 4˚C. Supernatant was discarded and the pellet washed once with 2 ml cold 100% methanol (Merck, Germany) and then with cold 80% acetone (Merck, Germany). The pellet was air-dried at −20˚C overnight. Dried pellet was dissolved in Modified Sample Buffer (MSB, thiourea 6.09 g (Fluka, Germany), urea 16.9 g (AppliChem, Germany), CHAPS 1.6 g (Sigma, USA), DTT 400 mg (D,L-Dithiothreitol, Sigma, USA), carrier ampholytes pH 2 - 11 800 μl (Servalyt® 2 - 11, Serva, Germany)), MilliQ water (Millipore, USA) was added to volume of 40 ml and stored at −20˚C in 1 ml aliquots. For each tube 400 μl of MSB was used.
2.2.2. Protein Determination
The Bradford [18] protein Assay (BioRad, USA) was performed to determine the concentration of proteins in fungal extracts according to manufacturer’s recommendation. Reactions were carried out in 96-welled microtiter plates (Nunclon Delta Surface, Denmark). A standard curve was established using serial dilutions from 0.8 µg∙ml−1 to 100 µg∙ml−1 of bovine serum albumin (BSA, Pierce, USA). The resulting optical density (OD) at 595 nm was measured with a plate reader (Tecan Austria, Austria). The protein concentration of three samples (experiment was performed in triplicate) was in a range 1010 - 1200 µg∙ml−1.
2.2.3. 2D-Electrophoresis
All 2DE experiments were carried out in triplicates. For the analysis 13 cm strips non-linear pH 3 - 10 were used (GE-Healthcare Immobiline DryStrip pH 3 - 10 NL, 13 cm, Sweden). Using protein concentration value we calculated the sample amount which was loaded for the strip aiming to get the desirable amount of proteins which is normally 20 µg [12] [17] . However our pre-experiments showed that application of 20 µg of total protein to each gel was too low for the protein identification of this fungus using mass spectrometry. Therefore 200 µg of total protein were used to generate preparative 2D gels. The calculated sample amount and Reswelling solution ((urea 6 g (AppliChem, Germany), CHAPS 0.5 g (Sigma, USA), DTT 37.5 mg (D,L-Dithiothreitol, Sigma, USA), bromphenol blue 1 ml (Serva, Germany), ampholytes 125 µl Servalyt® 2 - 11 (Serva, Germany) dissolved in 25 ml dH2O) has to be stored at −20˚C) were mixed and applied to the gel in final volume 255 µl. Strip rehydration took approximately 20 hours.
Iso-electric focusing was performed using Protean IEF Cell (BioRad, USA) according to the manufacturer’s instructions overnight. The re-equilibration was carried out prior to second dimension using an orbital shaker (Heidolph Unimax 1010, Germany) at room temperature as follows. Two ml of equilibration Solution A (tris 10 ml (UltraPure™, Invitrogen, USA), urea 36 g (AppliChem, Germany), glycerol 30 ml (AppliChem, Germany/Sigma, USA), SDS 2 g (sodium dodecyl sulphate, Sigma, USA), DTT 2 g (D,L-Dithiothreitol, Sigma, USA) in dH2O 100 ml) were added to each strip and incubated for 15 minutes. Then Solution A was replaced by Solution B (tris 10 ml (Sigma, USA), urea 36 g (AppliChem, Germany), glycerol 30 ml (AppliChem, Germany/ Sigma, USA), SDS 2 g (sodium dodecyl sulphate, Sigma, USA), IAA 2.5 g (iodoacetamide, Sigma, USA), bromphenol blue 100 µ (Serva, Germany) in 100 ml dH2O) and let incubate for 15 minutes. After the re-equilibration strips were applied to second dimension electrophoresis. Acrylamide solution (10%) (for 10 ml: dH2O 4.8 ml, acrylamide 2.5 ml (ready to use solution 40%, Merck, Germany), tris 2.5 ml (UltraPure™, Invitrogen, USA), SDS 0.1 ml (sodium dodecyl sulphate, Sigma, USA), TEMED 0.004 ml (Serva, Germany), APS 0.1 ml (ammonium persulfate, AppliChem, Germany), for 1 gel approximately 30 ml of acrylamide solution is needed) was prepared in a beaker.
The gel chamber was thermostatized using a Heto water cooling (Heto CBN 8 - 30, Denmark). The second dimension was performed under following conditions: 160 V, 35 mA, 5 W for around 4 hours. Visualization of protein spots was performed by high sensitive mass spectrometric compatible silver staining [19] . Gels were washed for 1 min in MilliQ water at room temperature followed by incubation for 20 min in fixing solution (methanol 500 ml (Merck, Germany), acetic acid 50 ml (AppliChem, Germany), in MilliQ water to the volume of 1 L, store at room temperature). Then gels were incubated at room temperature for 10 min in washing solution (methanol 500 ml (Merck, Germany) in MilliQ water to the volume of 1 L, store at room temperature). Then gels were washed overnight in MilliQ water at 4˚C with gentle agitation; incubated for 1 min in sensitizing solution (sodium thiosulfate 0.2 g (Merck, Germany) in MilliQ water to the volume of 1 L, has to be prepared fresh each time) followed by washing 2 times in MilliQ water 1 min each. Then gels were stained for 20 min at 4˚C in silver solution (silver nitrate 1 g (Merck, Germany) in MilliQ water to the volume of 1 L, has to be prepared fresh each time) followed by washing 2 times 1 min each in MilliQ water. Developing of the gels was performed in developing solution (sodium carbonate 20 g (Merck, Germany), formalin (35% formaldehyde, AppliChem, Germany) in MilliQ water to the volume of 1 L, has to be prepared fresh each time) and took 5 - 10 min. Then gels were washed 3 times 1 min each in stop solution (acetic acid 50 ml (AppliChem, Germany) in MilliQ water to the volume of 1 L, store at room temperature).
After staining, the gels were scanned as 16 bit TIFF images and stored at +4˚C in MilliQ water for further analysis. Gel images were matched by warping (Image Master 2D Platinum version 5.0, Amersham Biosciences, Swiss Institute of Bioinformatics, Geneva, Switzerland) and the sum of all spots which were present at least in two gels was taken into account for the protein pattern analyses as described in previous studies [12] [17] . The protein spots were excised from the gels by a manual spot-picker and transferred into a sterile tube (Eppendorf, Germany) containing 100 µl of MilliQ water (Millipore, MA, USA) and frozen at −80˚C for further MS analyses.
3. Protein Identification
3.1. Mass Spectrometry Using MALDI-TOF/TOF
Prominent 2-DE spots were excised by scalpel, washed, destained, reduced with DTT (D,L-Dithiothreitol, Sigma, USA) and alkylated with IAA (iodoacetamide, Sigma, USA). In-gel digestion was performed with trypsin (Trypsin Gold, Mass Spectrometry Grade, Promega) [19] . Dried peptides were concentrated and de-salted using Zip-Tips C18 (microbed) (Millipore) according to the manufacturer’s instructions.
3.2. Derivatisation for de Novo Sequencing
In order to enhance the quality of tandem mass spectrometry (MS/MS) spectra for de novo sequencing, N-terminal chemical modification using 4-sulfophenyl isothiocyanate (SPITC) was carried out with some modifications [20] [21] . The dried peptides were used for derivatisation with SPITC (4 µL of 10 mg/ml SPITC in 50 mM Tris-HCl buffer [pH 8.2]) for 30 min at 56˚C. Following acidification of the solution to stop the reaction with 6 µl 0.1% TFA, the peptides were de-salted using ZipTips C18 (microbed).
3.3. Spotting and Mass Spectrometry
Desalted peptides (0.5 µl) were spotted onto a disposable AnchorChip MALDI target plate pre-spotted with acyano-4-hydroxycinnamic acid (PAC target, Bruker Daltonics). Data were acquired on a Matrix Assisted Laser Desorption Ionisation Tandem Time-of-Flight (MALDI-TOF/TOF) mass spectrometer (Ultraflex II, Bruker Daltonics) in MS and MS/MS modes. Spectra processing and peak annotation were carried out using FlexAnalysis and Biotools (Bruker Daltonics).
3.4. Data Analysis and de Novo Peptide Sequencing
Standard databases were used to search for processed spectra. Each spectrum was compared via an in-house Mascot server (Matrix Science) to the Swiss-Prot database, NCBInr and to a generic database-based on genomic data of the organism-using the search parameters like taxonomy all entries; global modifications carbamidomethylation on cysteine; variable modifications oxidation on methionine; MS tolerance 100 - 150 ppm; MS/MS tolerance 1 Da; one missed cleavage allowed. Identifications were considered statistically significant where p < 0.05. Peptide sequencing was carried out manually using FlexAnalysis. These sequences were then used for a homology search using MS-homology search (http://prospector.ucsf.edu/prospector/cgi-bin/msform.cgi?form=mshomology).
4. Results
In order to visualize and investigate the proteome of C. antarcticus, 2-DE was performed using an optimized protein extraction protocol for MCFs. The main aim of the experiment was to separate the total protein repertoire of C. antarcticus, which was grown under laboratory conditions as a major basis to study the adaptations of the fungus to the extreme environment of Antarctica. Due to the numerous obstacles caused by the dense and melanised cell wall the protein extraction was, therefore, adapted and optimized to bypass the complex matrix of MCFs [15] . In order to increase the success with protein identification, 10 times higher amounts of protein (200 µg of total protein instead of 20 µg) were loaded to each gel. Taking into consideration all possibilities and limitations, our results allowed a first insight into the protein expression pattern in C. antarcticus. The 2-DE electrophoresis delivered 200 well defined protein spots by the orthogonal properties of isoelectric point and molecular weight. However, the results showed that some of the spots include more than one protein. All spots were visualized using a MS compatible silver staining and are shown in Figure 1.
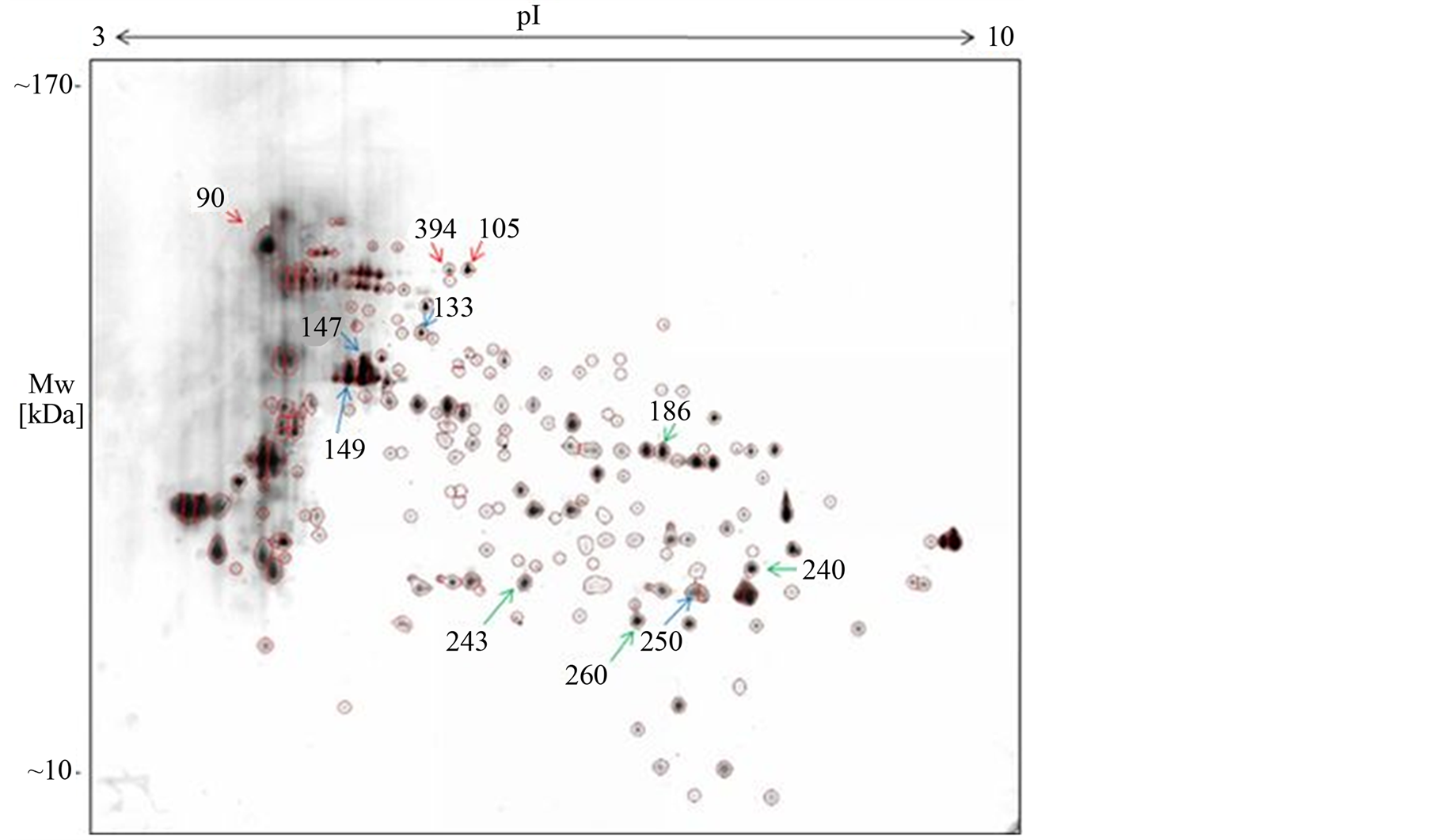
Figure 1. 2-DE pattern of Cryomyces antarcticus separated by two-dimensional electrophoresis. Red arrows: chaperons and heat shock proteins; blue arrows: enzymes and green arrows: other proteins.
The protein spots were distributed alongside the whole pI range of 3 - 10. A few high abundant and high molecular weight proteins were found in the acidic ranges between pI of 4 to 6.5. The resolution of the gel was very high and the spots could be excised without contamination. All protein spots were analyzed by MS-MALDITOF/TOF, however, only 11 proteins could be identified based on sequence homology with other species.
Although missing an annotated genome of C. antarcticus as a reference, some protein spots could be identified by de novo sequencing with high reliability (Mascot score 84% - 531%). The identified proteins are divided into three main categories: 1) heat shock proteins and 2) functional enzymes and 3) other proteins (Tables 1-3). Given the fact that C. antarcticus is an inhabitant of extreme environment it seems that its systems biology maintains the cell function by the expression of some prominent and conserved enzymes and commonly known heat shock genes even when growing under optimal temperature conditions.
Table 1. Chaperons and heat shock proteins identified in Cryomyces antarcticus. The scores of protein spots without MASCOT match are given only in homology score.
SC: sequence coverage; U: unique peptide; Red letters: exchanged amino acids in the homologue protein; Grey letters: homology searches in different species, or isoforms of the same protein, and peptides with an upper case u: are exceptional for C. antarcticus.
Table 2. Metabolic enzymes as identified by de novo sequencing and sequence homology to other species. The scores of protein spots without MASCOT match are given only in homology score.
SC: sequence coverage; U: unique peptide; Red letters: exchanged amino acids in the homologue protein; Grey letters: homology searches in different species, or isoforms of the same protein, and peptides with an upper case u: are exceptional for C. antarcticus.
4.1. Heat Shock Proteins
(HSP) are a group of proteins induced when cells are exposed to low or high temperature or other environmental stress. The most prominent members of this group are a class of functionally related proteins involved in the folding and unfolding of other proteins [22] . This increase in expression is regulated normally by gene transcription [23] . Expression of high levels of heat shock proteins are stimulated by different types of environmental conditions, like exposure of the cell to toxins (ethanol and trace metals) or to ultraviolet light, starvation, or water deprivation. As a result, the heat shock proteins are called stress proteins and their up-regulation sometimes described more generally as part of the stress response-network [24] .
Molecular chaperones are originally defined as proteins that mediate the correct assembly of other proteins, but are not themselves components of the final structure. They bind to and stabilize an otherwise unstable conformation, and through a controlled cycle of binding and release facilitate the correct fate in vivo [25] . One major function of chaperones is to prevent both newly synthesized polypeptide chains and assembled subunits from aggregating into nonfunctional structures. For this reason, many chaperones are also known as heat shock proteins due to the increased protein aggregation by stresses [26] .
Table 3. Other proteins, which are identified in C. antarcticus. The scores of protein spots without MASCOT match are given only in homology score.
SC: sequence coverage; U: unique peptide; Red letters: exchanged amino acids in the homologue protein; Grey letters: homology searches in different species, or isoforms of the same protein, and peptides with an upper case u: are exceptional for C. antarcticus.
4.2. Heat Shock Protein 70
The C. antarcticus proteome contains the heat shock 70 kDa protein (spot 90), identified by several peptides and a Mascot score of 176.0. The expression of the HSP having a molecular mass of about 70 kDa, was observed in hyphomycetous fungi as e.g. Fusarium oxysporum f. sp. niveum, Neurospora crassa and Saccharomyces cerevisiae [27] .
Proteins of HSPs70 family are highly conserved in eukaryotic organisms. Their expression of HSP70 as a response to several stresses is universal. The respond was studied solely at the cytological level and several important observations were made: induces by other stress treatments; produced within a few minutes; associated with newly synthesized RNA, found in many different tissues, accompanied by the disappearance of previously active protein synthesis are suppressed. HSP70 chaperones make up a set of prominent cellular machines that assist with a wide range of protein folding processes in almost all cellular compartments. They are also involved in protein import and translocation processes, and in facilitating the proteolytic degradation of unstable proteins by targeting the proteins to lysosomes or proteasomes. HSP70 is also involved in the modulation of signal transducers such as protein kinase A, protein kinase C and protein phosphatise. In this respect, HSP70 chaperones might play a broad role by participating in modulating the expression of many downstream genes in signal transduction pathways both during stress and under normal growth conditions [20] .
HSP70 shows essential functions in preventing aggregation and in assisting refolding of non-native proteins under both normal and stress conditions [20] . Under non-stressful conditions, HSP70 cognate functions in concert with a variety of co-chaperones facilitate folding of de novo synthesized proteins assisting the transport of precursor proteins into organelles and help to target damaged proteins for degradation. Stress-induced HSP70 functions to mitigate aggregation of stress-denatured proteins and to refold non-native proteins restoring their biological function through iterative cycles of adenine nucleotide hydrolysis-dependent peptide binding and release [28] . Molecular chaperones were defined as proteins that mediate the correct assembly of other proteins, but are not themselves components of the final structure. Those proteins bind and stabilize an unstable conformation, and through a controlled cycle of binding and release facilitate the correct fate in vivo [25] .
Chaperones do not convey information directing a protein to correct molecular pathway. Instead, they reduce non-productive interactions. Both folding and aggregation can be very rapid, and thus the rates of chaperone recognition and binding must be high to intercept the de processes. Chaperones block entry into side reactions by their property of recognizing non-native polypeptide structures. Recognition must lead to tight binding and shielding of hydrophobic surfaces, so that aggregation of folding are prevented. In addition, binding must be more rapid than competing reactions that are essentially irreversible [25] .
4.3. HSP70 kDa Chaperone Precursor
Both HSP70 and HSP70 chaperon precursor could be identified in C. antarcticus. The HSP70 chaperones have implications on protein folding, assembly/disassembly of oligomeric complexes, protein synthesis, degradation, and the translation of polypeptides across cellular membranes [28] . As it was shown by Glick [28] , ATP hydrolysis allows luminal HSP70 to bind strongly both to the precursor chain and to a membrane partner protein. HSP70 would then undergo a conformational change, thereby pulling a segment of the precursor chain through the translocation channel. Finally replacement of the bound ADP with ATP would release HSP70 from the precursor and from the membrane partner protein and the cycle could begin again. Translocation would be unidirectional because luminal HSP70 is anchored to the membrane in a defined orientation and because interactions of the precursor chain with the translocation channel would inhibit “backsliding” of the precursor between rounds of HSP70 binding. This mechanism for protein import is reminiscent of the ATP-driven translocation of actin filaments by myosin. Thus, even though the N-terminal domain of HSP70 is structurally related to actin, HSP70 proteins may be functionally analogous to myosin.
4.4. Chaperone Protein HSP31
Under stress many cellular proteins lose their native configuration and tend to aggregate. This could pose a serious threat to the cellular functionality. Chaperones provide the essential assistance by promoting refolding of stress-denaturated proteins and preventing their aggregation. The predominant HSPs are classified by their molecular masses as HSP100, HSP90, HSP70, HSP60, HSP40 and small HSP (sHSPs) [29] .
The functions of sHSPs, as ATP independent molecular chaperones, in protein folding and, as antistressors, in acquired thermotolerance have been well established [30] . Upon heat-shock or exposure to other stressors, sHSPs capture unfolded proteins and form high molecular weight multimeric complexes, thus prevent their irreversible aggregation by holding them in a soluble and folding competent state. Then, refolding of the captured proteins most likely occur with the assistance of the ATP dependent molecular chaperones, such as ClpB (or HSP104) and/or the DnaK/DnaJ/GrpE (or HSP70/HSP40) system [30] .
4.5. Metabolic Enzymes
Most of the identified proteins of the C. antarcticus proteome belong to the category 2 and are essential metabolic enzymes. These enzymes catalyze reactions, which allow organisms to grow and reproduce, maintain their structures, and respond to their environments. The protein spots identified by de novo sequencing comprise a series of key enzymes, which are described in the following (Table 2).
4.6. V-Type Proton ATPas Subunit B
Spot number 133 contain at least two proteins, a V-type ATPase and with a lower rate of homology Vacuolar ATP synthase subunit B. V-type ATPase is a multisubunit enzyme (approximately 500 kDA) composed of a water-soluble catalytic sector and an integral membrane proton channel complex [31] . V-ATPases have an important role in both endocytosis and intracellular transport [32] . The V-type H+ ATPase comprises an ATP driven enzyme that transforms the energy of ATP hydrolysis to electrochemical potential differences of protons across diverse biological membranes via the primary active transport of H+ [33] .
The vacuolar (H+) ATPase or V-ATPases are a family of ATP-driven proton pumps responsible for acidification of a variety of intracellular compartments in eukaryotic cells, including clathrin-coated vesicles, endosomes, lysosomes, Golgi, secretory vesicles. The V-ATPases function to acidify intracellular compartments in eukaryotic cells, playing an important role in such processes as receptor-mediated endocytosis, intracellular membrane traffic, protein degradation and coupled transport. V-ATPases in the plasma membrane of specialized cells also function in renal acidification, bone resorption and cytosolic pH maintenance. The V-ATPases are composed of two domains. The V1 domain is a 570 kDa peripheral complex composed of 8 subunits (A-H) of molecular weight 70 - 13 kDa which is responsible for ATP hydrolysis [34] [35] . The protein identified from the C. antarcticus belongs to subunit B.
V-ATPases play a role in energizing secondary transport and maintenance of solute homeostasis. Under stress conditions such as salinity, drought, cold, acid stress, anoxia, and others, survival of the cells depends strongly on maintaining or adjusting the activity of the V-ATPase. Regulation of gene expression and activity are involved in adapting the V-ATPase on longand short-term bases [36] .
4.7. ATP Synthase
Changes in metabolism under the influence of stress conditions include almost all the processes and pathways within the cells. The proper course of metabolic processes requires sufficient amount of biologically useful energy, usually in the form of high-energy phosphate bonds between residues in the ATP [37] .
Adenosine triphosphate (ATP) synthase contains a rotary motor involved in biological energy conversion. ATP is the universal biological energy currency. ATP synthase produces ATP from adenosine diphosphate (ADP) and inorganic phosphate with the use of energy from a transmembrane proton-motive force generated by respiration or photosynthesis [38] . Recently two models are proposed to explain the requirement of ATP during protein import to mitochondria involving the action of HSP70 as the center of the protein translocase machinery. Both models agree that HSP70 proteins or homologues of them should bind to the pre-sequence of the pre-protein being imported with the binding cycle regulated by ATP hydrolysis [39] . Majority of experimental data support the proposal that ATP hydrolysis is essential for chaperone activity [39] .
4.8. Сarboxy Peptidase Y
The principle of self-assembly in protein structure states that the native three-dimensional structure of a protein are determined solely by the primary amino acid sequence and that folding of the peptide chain. This phenomenon is a spontaneous process which does not require input of energy or accessory factors. Some proteins can apparently not be re-natured once they have been fully denatured. Such proteins are frequently synthesized as zymogens that are later matured to active enzymes by proteolysis. The function of the function of the propeptides is not only to render the proenzymes catalytically inert but also to act as so-called co-translational chaperones, since they mediate correct folding without being components of the final three-dimensional structure. The folding is in some cases not dependent on the covalent linkage of the pro-peptide to the segment that becomes later a mature protein. Similar as described above, carboxypeptidase Y (CPY) is synthesized as a zymogen. CPY is a vacuolar protein that was extensively studied as a model for eukaryotic intracellular protein transport and targeting. The gene for CPY encodes a preproenzyme containing a signal peptide of 20 amino acid residues that directs the precursor across the endoplasmic reticulum membrane. In the endoplasmic reticulum the signal peptide is cleaved off and folding takes place. A 4-amino acid segment of the CPY propeptide serves as signal for vacuolar targeting. Upon arrival of pro-CPY in the vacuole it is activated by degradation of the propeptidein a process involving the vacuolar endopeptidase proteinase A [40] [41] .
4.9. Tri-(Tetra-)Hydrohyl Naphthalene Reductase
It was reported that polyhydroxylnaphthalene reductase is described as an important element in melanin biosynthesis in pathogenic fungus Magnaporthe grisea [42] . Cell walls of C. antarcticus are incrusted with melanin which gives it black colour. During the biosynthesis of fungal melanin, tetrahydroxylnaphthalene reductase catalyzes the NADPH-dependent reduction of 1,3,6,8-tetrahydroxylnaphthalene (T4 HN) into (+)-scytalone and 1,3,8-trihydroxylnaphthalene into (−)-vermelone [43] . Melanin, a high-molecular mass black pigment, is synthesized in numerous pathogenic fungi, such as Verticillium dahlia, Cochliobolus miyabeanus and Magnaporthe grisea and others. Mutants of those pathogens lacking the capability to synthesize melanin lose their pathogenicity [43] .
4.10. NADPH-Ubiqinone Oxidoreductase 29.9 kDa Subunit
NADPH oxidases (Nox), dedicated enzymes for reactive oxygen species (ROS) production, are widely distributed in eukaryotic cells [44] . In eukaryotic cells many Nox genes control different processes, including cell proliferation, apoptosis and hormone response in animals [44] , and programmed cell death, hormone signalling and root hair tip growth in plants [45] . Nox can serve both defense and differentiation signalling roles [46] .
The enzymes of this group in filamentous fungi play a key role in multi-cellular development and defense [46] . The study of Kayano and colleagues [47] suggests that NoxA and associated regulators are essential for polarized hyphal growth under conditions of nutrient starvation NADPH oxidase gene (NoxA) found in Aspergillus nidulans blocks differentiation of sexual fruiting bodies (cleistothecia) but has no effect on hyphal growth or asexual development [48] . Fungal NADPH oxidase catalysed production of ROS is also an important signalling mechanism for plant pathogens. Performed functional analysis emphasize the importance of ROS signalling for many different physiological and developmental processes in fungi [46] .
4.11. Peptidase M18 Aminopeptidase I
Aminopeptidases catalyze the sequential removal of amino acids from unblocked N termini of peptides and proteins. These enzymes are widely distributed in eukaryotes and prokaryotes as either integral membrane or cytosolic proteins. Aminopeptidases are generally classified in terms of their substrate specificities-preference for a neutral, acidic, or basic amino acid in the P1 position. In addition to their role in general protein and peptide metabolism, aminopeptidases have more specific functions: activation and inactivation of biologically active peptides; removal of the N-terminal methionine of newly synthesized proteins; and possibly trimming of antigens for presentation by the major histocompatibility complex-1 system [49] .
4.12. Other Proteins
Four proteins belong to different functional groups were identified by de novo sequencing and sequence homologies (Table 3). These proteins are shared among a wide variety of species from microorganisms to humans and are likely to be essential for surviving of any given organism on the earth.
4.13. Actin
Actin plays a crucial role in elementary eukaryotic cellular processes such as motility, cell growth regulation, cell differentiation and provides structural stability. Apparently these functions have not allowed actin of different species to diverge significantly, since the actin protein is highly conserved [50] . Plamann and colleagues reported that cytoplasmic dynein and actin-related protein Arp1 are required for normal nuclear distribution in filamentous fungi [51] . There are some studies showing that actin is present in many filamentous fungi [52] [53] .
4.14. Mitochondrial Processed Peptide
Mitochondrial processing peptidase (MPP) consists of α and β subunits that catalyze the cleavage of N-terminal mitochondrial-targeting sequence (N-MTSs) and deliver proproteins to the mitochondria [54] . The majority of mitochondrial proteins are synthesized as precursors on cytosolic ribosomes and transported post-translationally into the mitochondria. This process is facilitated by specific matrix-targeting signal sequences which are normally part of the N-termini of these proteins prior to their transportation. These proteins are unfolded and imported into the mitochondrial matrix across a double membrane through protein translocation machinery comprising translocases of the outer and inner mitochondrial membrane. After precursor translocation, the targeting signal is no longer necessary and is proteolitically removed. Although several mitochondrial peptidases participate in pre-protein processing, the most important role seems to belong to the mitochondrial processing peptidase (MPP), since its deletion is lethal. Indeed, no inherited disorders have been linked with any mutants of MMP, indicating that its biological function is so vital that even relatively moderate disruption to its activity are also likely to produce non-viable organisms. This may be linked to its essential role in mitochondrial biogenesis [55] .
The ability of the pre-sequence to adopt context-dependent conformations during different steps of MPP’s action is a basic requirement for substrate recognition processing [55] . The mitochondrial precursors are translocated across the outer and the inner mitochondrial membrane via the translocase of the outer membrane (TOM) and the translocase of the inner membrane (TIM) machineries, respectively. Transport across the outer membrane does not require energy, whereas transport across the inner membrane is an energy-dependent process that requires a membrane potential across the inner membrane. Inside the mitochondrial matrix, the presequence of the imported precursor protein is cleaved off by the mitochondrial processing peptidase (MPP). In yeast and mammals, MPP is localized to the matrix, whereas in plants MPP is integrated into the cytochrome bc1 complexof the respiratory chain as core proteins of the complex facing the matrix. Cleavage by MPP may be followed by additional trimming of some precursors in the mitochondrial matrix by the mitochondrial intermediate peptidase (MIP) or the Intermediate cleaving peptidase of 55 kDa, Icp55 [56] .
4.15. 14-3-3 Protein
The cellular decision to live or die is finely controlled by tightly regulated signaling pathways. 14-3-3 proteins have a key role in this decision by controlling many of the signaling pathways that mediate this process. 14-3-3 proteins comprise a large family of acidic proteins that are expressed within all eukaryotic cells and function as homodimers and heterodimers. 14-3-3 dependent protein regulation occurs through phosphorylation-dependent binding which results in the release of survival signals in the cells [57] .
14-3-3 proteins represent a family of highly homologous proteins that were described in all eukaryotic organisms from fungi to humans. These small acidic proteins have been implicated in a wide variety of cellular processes including signal transduction, cell cycle regulation and transcriptional regulation [58] . 14-3-3 monomers form cup-shaped homoand hetero-dimers are able to bind protein ligands. Usually phosphorylated on serine or threonine residues or consensus binding motifs. 14-3-3 proteins are involved in altering the ability of the target protein to interact with other proteins; in modifying the subcellular portioning of a target, in enhancing/ inhabiting the catalytic activity of the target; in protecting a target from dephosphorylation/proteolysis; and in acting as a scaffold. However, 14-3-3 proteins do not appear to be involved in the same process in different fungi [59] .
The 14-3-3 protein family is shown to implicate in the dynamic nucleocytoplasmic transport of a number of proteins. They specifically recognize one or more short phosphoserine/theonine-containing sequence motifs on target proteins [60] .
The members of this family interact with hundreds of protein partners, participate in regulation of many cellular processes like apoptosis, cell division, transcription, trafficking [61] . Moreover 14-3-3 proteins play an important role in regulation of signal transduction, apoptosis, cell cycle control, and nutrient-sensing pathways.
It was found that 14-3-3 itself can undergo different posttranslational modifications (including phosphorylation) strictly regulating its cellular functions. Phosphorylation seems to be the most important way of 14-3-3 regulation and can affect either direct interaction of 13-4-4 with its targets or induce dissociation of 14-3-3 dimers. As a rule, phosphorylation negatively affects interaction of 14-3-3 with its target [62] .
The data of the study by Zakharova and colleagues showed that the basic level of sugars was high in fully hydrated C. antarcticus culture [12] . Trehalose is a naturally occurring non-reducing sugar found in a wide variety of organisms where it protects proteins and membranes from various stress conditions like dehydration, heat, cold, oxidation and desiccation and serves as a carbon and energy source. It was suggested that in yeast and plants it serves as a regulatory and signaling molecule to direct certain metabolic pathways or to affect growth. The neatral trehalase Nth1 of yeast is reported to be activated by 14-3-3 protein [63] .
Among the many specialized cellular signal transduction models, the family of 14-3-3 protein occupies a remarkably ubiquitous role as downstream effectors of phosphorylation events. Essentially soluble dimers of single phosphor-Ser/Thr binding domains are reported to bind to this protein. The 14-3-3 proteins are additionally described to bind hundreds of different cellular protein, although there are also examples of binding to nonphosphorylated target proteins. Upon binding to their phosphor-recognition site, they are reported to affect the target protein (TP) by modulation of its activity, interaction with other molecules, intracellular localization and stability [64] .
4.16. Pre-mRNA Splicing Factor
As it was shown by Keightley and colleagues the regulation of pre-mRNA splicing is critical for correct lineage specification in haematopoiesis [65] . Spliceosomes are composed of uridineuridine-rich small nuclear ribonuclearprotein particles (U-snRNPs) and multiple associated proteins. The vast majority of introns in pre-mRNA are excited by U2-dependent major spliceosomes, which contain U1, U2, U4/U6 and U5 snRNPs. Transcription and pre-mRNA splicing have emerged as highly coordinated processes. The close function coupling between the two processes is achieved through multiple mechanisms. A key role is played by the CTD domain of the largest subunit of RNA polymerase II, which both interacts with splicing factors and may function as a scaffold for cotranscriptional pre-mRNA splicing [66] . CPY is synthesized as a pre-enzyme containing an N-terminal 20- amino acid signal peptide, which directs pro-CPY to the lumen of the endoplasmic reticulum [66] .
5. Discussion
C. antarcticus is an Antarctic rock-inhabiting microorganism, which lives at the absolute edge of life under the most extreme conditions known on the earth [67] . These fungi live between the limit of adaptability and near-death, barely surviving and rarely regenerating. They are assumed to be the most stress resistant eukaryotic organisms known on the earth [68] . In this context it is interesting that only a number of 200 different protein spots were exhibited and visualized on the gel although a high amount of total protein was loaded on the gel. This finding is in accordance with previous studies that have shown that a low number of proteins are expressed in Antarctic black fungi as Cryomyces and the second endemic genus Friedmanniomyces—also under optimal and eutrophic growth conditions. This can be interpreted as a high adaptation and narrow ecological amplitude in comparison to mesophilic but stress-tolerant meristematic fungi as e.g. fungi Coniosporium sp. [12] [16] [17] which might correspond to the number of essential proteins necessary for surveillance of this very particular microorganism. As a response to the short growth periods of the Antarctic summer and the extremely low nutrient availability these fungi express only those proteins and cellular functions that are absolutely necessary to survive. Recent studies on the whole genome of the fungus have shown that the genome size is approximately 28 kb and thus in average range for fungi (Sterflinger, unpublished data). The results of this study suggest that only a limited part of the genome is transcribed and most of it might have undergone genome silencing due to the “energy saving mode”. Interestingly, HSPs and particular chaperons—which are expressed to compensate the risks of undesired protein denaturation and inactivations—were the most abundant proteins found although the fungus was grown under optimal temperature conditions (15˚C). This indicates that the fungus keeps up a basic level of these protective proteins also under non-stress conditions. On the one hand this could be explained by the fact that the fungus lost the ability to downscale these proteins due to a constant need in the natural environment. On the other hand a basic level of those proteins could enable the fungus to withstand rapid environmental changes without need for fast reactions on the cellular level. Previous studies showed that C. antarcticus reacts to rehydration after complete desiccation more slowly than other MCF [12] . This was explained by the fact that C. antarcticus has to respond to long-yearly cycles of anhydrobiosis and its activity is depending on the Antarctic seasons whereas MCFs from more moderate climatic regions have much shorter cycles depending on weather and rain events [11] .
After de novo sequencing and sequence homology searching in the public domain databases, only a minor portion of protein spots could be identified. The identified proteins belong mostly to very high conserved protein families, which are encoding key proteins guaranteeing the maintenance of the central metabolism for growth and regeneration after state of dormancy. However, the rest of the protein spots remained unidentified, since they show no similarities to the available data. This suggests that the proteome of C. antarcticus must comprise a high number of novel proteins with unknown functions. Also the phylogenetic position of the fungus confirms its special role within the fungal kingdom: together with Cryomyces minteri, C. antarcticus forms a distinct clade within the ascomycetes without any obvious direct ancestor [69] .
The better characterization of the hitherto unidentified proteins will be possible after genome annotation of the whole genome and transcriptomic studies which are on the way (Sterflinger, paper in preparation). Since black fungi have numerous outstanding abilities including the survival of radioactivity and the capacity to use radioactivity as an energy source for ATP generation—so-called “radiotropism” [70] , the whole protein pool might be a treasure box containing novel proteins with interesting and novel functions as well as cell protective agents. These proteins will be of high interest in view of pharmaceutical biotechnological applications and are in the focus of future studies.
Acknowledgements
The work was financed by the Austrian Science Fund FWF under project number P 24206-B16. The ION Proton facility of the VIBT Extremophile Center was financed by the VIBT-Equipment GmbH, the Vienna “Zentrum für Innovation und Technology” (ZID) and the Austrian Ministry of Science, Research and Economy. We further thank the Italian National Program for Antarctic Researches and the Italian National Museum of Antarctica for kindly providing the strain.
References
- Cary, S.C., McDonald, I.R., Barrett, J.E. and Cowan, D.A. (2010) On the Rocks: The Microbiology of Antarctic Dry Valley Soils. Nature Review Microbiology, 8, 129-138. http://dx.doi.org/10.1038/nrmicro2281
- Cockell, C.S., Lee, P., Schuerger, A.C., Hidalgo, L., Jones, J.A. and Stokes, M.D. (2001) Microbiology and Vegetation of Micro-Oases and Polsar Desert, Haughton Impact Crater, Devon Island, Nunavut, Canada. Antarctic and Alpine Research, 33, 306-318.
- Navarro-Gonzales, R., Rainey, F.A., Molina, P., Bagaley, D.R., Hollen, B.J., De la Rosa, J., Small, A.M., Quinn, R.C., Grunthaner, F.J., Cacaeres, L., Gomez-Silva, B. and McKay, C.P. (2003) Mars-Like Soils in the Atacama Desert, Chile, and the Dry Limit of Microbial Life. Science, 302, 1018-1021. http://dx.doi.org/10.1126/science.1089143
- Direito, S.O.L., Ehrenfreund, P., Marees, A., Staats, M., Foing, B. and Roling, W.F.M. (2011) A Wide Variety of Putative Extremophiles and Large Beta-Diversity at the Mars Desert Research Station (Utah). International Journal of Astrobiology, 10, 191-207. http://dx.doi.org/10.1017/S1473550411000012
- Onofri, S., Selbmann, L., De Hoog, G.S., Grube, M., Barreca, D., Ruisi, S. and Zucconi, L. (2007) Evolution and Adaptation of Fungi at Boundaries of Life. Advances in Space Research, 40, 1657-1664.http://dx.doi.org/10.1016/j.asr.2007.06.004
- Friedmann, I.E. (1982) Endolithic Microorganisms in the Antarctic Cold Desert. Nature, 215, 1045-1053.
- Selbmann, L., De Hoog, G.S., Mazzaglia, A., Friedmann, E.I. and Onofri, S. (2005) Fungi at the Edge of Life— Cryptoendolothic Fungi from the Antarctic Desert. Studies in Mycology, 51, 1-32.
- Selbmann, L., De Hoog, G.S., Gerrits van den Ende, A.H.G., Ruibal, C., De Leo, F., Zucconi, L., Isola, D., Ruisi, S. and Onofri, S. (2008) Drought Meets Acid: Three New Genera in a Dothidealean Clade of Extremotolerant Fungi. Studies in Mycology, 61, 1-20. http://dx.doi.org/10.3114/sim.2008.61.01
- Gorbushina, A.A., Kollova, E.R. and Sherstneva, O.A. (2008) Cellular Responses of Microcolonial Rock Fungi to Long Term Desiccation and Subsequent Rehydration. Studies in Mycology, 61, 91-97. http://dx.doi.org/10.3114/sim.2008.61.09
- Dadachova, E., Bryan, R.A., Huang X., Moadel, T., Schweitzer, A.D., Aisen, P., Nosanchuk, J.D. and Casadeval, L.A. (2007) Ionizing Radiation Changes the Electronic Properties of Melanin and Enhances the Growth of Melanized Fungi. Plos One, 5, Article ID: e457. http://dx.doi.org/10.1371/journal.pone.0000457
- Sterflinger, K., Tesei, D. and Zakharova, K. (2012) Fungi in Hot and Cold Deserts with Particular Reference to Microcolonial Fungi. Fungal Ecology, 5, 453-462. http://dx.doi.org/10.1016/j.funeco.2011.12.007
- Zakharova, K., Tesei, D., Marzban, G., Dijksterhuis, J. and Wyatt, T. (2013) Microcolonial Fungi on Rocks: A Life in Constant Drought? Mycopathologia, 175, 537-547. http://dx.doi.org/10.1007/s11046-012-9592-1
- Onofri, S., Barreca, D., Selbmann, L., Isola, D., Rabbow, E., Horneck, G., De Vera, J.P.P., Hatton, J. and Zucconi, L. (2008) Resistance of Antarctic Black Fungi and Cryptoendolithic Communities to Simulated Space and Martian Conditions. Studies in Mycology, 61, 99-109. http://dx.doi.org/10.3114/sim.2008.61.10
- Zakharova, K., Marzban, G., De Vera, J.-P., Lorek, A. and Sterflinger, K. (2014) Protein Patterns of Black Fungi under Simulated Mars-Like Conditions. Scientific Reports, 4, 5114. http://dx.doi.org/10.1038/srep05114
- Marzban, G., Tesei, D. and Sterflinger, K. (2013) A Review beyond the Borders: Proteomics of Microcolonial Black Fungi and Black Yeasts. Natural Sciences, 5, 640-645.
- Isola, D., Marzban, G., Selbmann, L., Onofori, S., Laimer, M. and Sterflinger, K. (2011) Sample Preparation and 2-DE Procedure for Protein Expression Profiling of Black Microclonial Fungi. Fungal Biology, 115, 971-977. http://dx.doi.org/10.1016/j.funbio.2011.03.001
- Tesei, D., Marzban, G., Zakharova, K., Isola, D., Selbman, L. and Sterflinger, K. (2012) Alteration of Protein Patterns in Black Rock Inhabiting Fungi as a Response to Different Temperatures. Fungal Biology, 116, 932-940. http://dx.doi.org/10.1016/j.funbio.2012.06.004
- Bradford, M.M. (1976) A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Analytical Biochemistry, 72, 248-254. http://dx.doi.org/10.1016/0003-2697(76)90527-3
- Shevchenko, A., Wilm, M., Vorm, O. and Mann, M. (1996) Mass Spectrometric Sequencing of Proteins from Silver Stained Polyacrylamide Gels. Analytical Chemistry, 68, 850-858. http://dx.doi.org/10.1021/ac950914h
- Wang, W., Vinocur, B., Shoseyov, O. and Altman, A. (2004) Role of Plant Heat-Shock Proteins and Molecular Chaperones in the Abiotic Stress Response. Trends in Plant Science, 9, 244-252. http://dx.doi.org/10.1016/j.tplants.2004.03.006
- Zhang, H.Y., He, H., Chen, L.B., Liang, M.Z., Wang, X.F., Liu, X.G., He, G.M., Chen, R.S., Ma, L.G. and Deng, X.W. (2008) A Genome-Wide Transcription Analysis Reveals a Close Correlation of Promoter INDEL Polymorphism and Heterotic Gene Expression in Rice Hybrids. Molecular Plant, 1, 720-731. http://dx.doi.org/10.1093/mp/ssn022
- De Maio, A. (1999) Heat Shock Proteins: Facts, Thoughts, and Dreams. Shock, 11, 1-12.
- Wu, C. (1995) Heat Shock Transcription Factors: Structure and Regulation. Annual Review of Cell and Developmental Biology, 11, 441-469. http://dx.doi.org/10.1146/annurev.cb.11.110195.002301
- Santoro, M.G. (2000) Heat Shock Factors and the Control of the Stress Response. Biochemical Pharmacology, 59, 55-63. http://dx.doi.org/10.1016/S0006-2952(99)00299-3
- Miernyk, J.A. (1997) The 70 kDa Stress-Related Proteins as Molecular Chaperones. Trends in Plant Science, 2, 180-187. http://dx.doi.org/10.1016/S1360-1385(97)85224-7
- Pauwels, K., Van Molle, I., Tommassen, J. and Van Gelder, P. (2007) Chaperoning Anfinsen: The Steric Foldases. Molecular Microbiology, 64, 917-922. http://dx.doi.org/10.1111/j.1365-2958.2007.05718.x
- Freeman, S., Ginzburg, C. and Katan, J. (1989) Heat Shock Protein Synthesis in Propagules of Fusarium oxysporum f. sp. niveum. Physiology and Biochemistry, 79, 1054-1058.
- Glick, B.S. (1995) Can HSP70 Proteins Act as Force-Generating Motors? Cell, 80, 11-14. http://dx.doi.org/10.1016/0092-8674(95)90444-1
- Feder, M.E. and Hofmann, G.E. (1999) Heat-Shock Proteins, Molecular Chaperones, and the Stress Response: Evolutionary and Ecological Physiology. Annual Review in Physiology, 61, 243-282. http://dx.doi.org/10.1146/annurev.physiol.61.1.243
- Kocabiyik, S. and Aygar, S. (2012) Improvement of Protein Stability and Enzyme Recovery under Stress Conditions by Using a Small HSP (tpv-HSP 14.3) from Thermoplasma volcanium. Process Biochemistry, 47, 1676-1683. http://dx.doi.org/10.1016/j.procbio.2011.11.014
- Gogarten, J.P., Kibak, H., Dittrich, P., Taiz, L., Bowman, E.J., Bowman, B.J., Manolson, M.F., Poole, R.J., Date, T., Oshima, T., Konishi, J., Denda, K. and Yoshida, M. (1989) Evolution of the Vacuolar H+-ATPase: Implications for the Origin of Eukaryotes. Proceedings of the National Academy of Sciences of the United States of America, 86, 6661-6665. http://dx.doi.org/10.1073/pnas.86.17.6661
- Nishi, T. and Forgas, M. (2002) The Vacuolar (H+)-ATPasas-Nature’s Most Versatile Proton Pumps. Nature, 3, 94-103.
- Beyenbach, K.W. and Wieczorek, H. (2006) The V-Type H+ ATPase: Molecular Structure and Function, Physiological Role and Regulation. The Journal of Experimental Biology, 209, 577-589. http://dx.doi.org/10.1242/jeb.02014
- Forgas, M. (1998) Structure, Function and Regulation of the Vacuolar (H+)-ATPases. FEBS Letters, 440, 258-263. http://dx.doi.org/10.1016/S0014-5793(98)01425-2
- Sagermann, M., Stevens, T.H. and Matthews, B.W. (2001) Crystal Structure of the Regulatory Subunit H of the V-Type ATPasa of Saccharomyces cerevisiae. Proceedings of the National Academy of Sciences of the United States of America, 98, 7134-7139. http://dx.doi.org/10.1073/pnas.131192798
- Dietz, K.J., Tavakoli, N., Kluge, C., Mimura, T., Sharma, S.S., Harris, G.C., Chardonnens, A.N. and Golldack, D. (2001) Significance of the V-Type ATPase for the Adaptation for the Stressful Growth Conditions and Its Regulation on the Molecular and Biochemical Level. Journal of Experimental Botany, 52, 1969-1980. http://dx.doi.org/10.1093/jexbot/52.363.1969
- Wojtyla, L., Kosmala, A. and Garnczarska, M. (2013) Lupine Embryo Axes under Salinity Stress. II. Mitochondrial Proteome Response. Acta Physiologiae Plantarum, 35, 2383-2392. http://dx.doi.org/10.1007/s11738-013-1273-2
- Stock, D., Leslie, A.G.W. and Walker, J.E. (1999) Molecular Architecture of the Rotary Motor in ATP Synthase. Science, 286, 1700-1705. http://dx.doi.org/10.1126/science.286.5445.1700
- Rial, D.V., Arakaki, A.K. and Ceccerelli, E.A. (2000) Interaction of the Targeting Sequence of Chloroplast Precursors with HSP70 Molecular Chaperones. European Journal of Biochemistry, 267, 6239-6248. http://dx.doi.org/10.1046/j.1432-1327.2000.01707.x
- Spormann, D.O., Heim, J. and Wolf, D.H. (1991) Carboxypeptidase yscS: Gene Structure and Function of the Vacuolar Enzyme. European Journal of Biochemistry, 197, 399-405. http://dx.doi.org/10.1111/j.1432-1033.1991.tb15924.x
- Winther, J. and Sørensen, P. (1991) Propeptide of Carboxypeptidase Y Provides a Chaperone-Like Function as Well as Inhibition of the Enzymatic Activity. Proceedings of the National Academy of Sciences of the United States of America, 88, 9330-9334. http://dx.doi.org/10.1073/pnas.88.20.9330
- Andersson, A., Jordan, D., Achneider, G. and Lindqvist, Y. (1996) Crystal Structure of the Ternary Complex of 1,3,8-Trihydroxynaphthalene Reductase from Magnaporthe grisea with NADPH and an Active-Site Inhibitor. Structure, 4, 1161-1170. http://dx.doi.org/10.1016/S0969-2126(96)00124-4
- Vidal-Cros, A. and Boccara, M. (1998) Identification of Four Chitin Synthase Genes in the Rice Blast Disease Agent Magnaporthe grisea. FEMS Microbiology Letters, 165, 103-109. http://dx.doi.org/10.1111/j.1574-6968.1998.tb13133.x
- Sumimoto, H. (2008) Structure, Regulation and Evolution of Nox-Family NADPH Oxidases That Produce Reactive Oxygen Species. FEBS Journal, 275, 3249-3277. http://dx.doi.org/10.1111/j.1742-4658.2008.06488.x
- Torres, M.A., Jones, J.D.G. and Dangl, J.L. (2005) Pathogen-Induced, NADPH Oxidase-Derived Reactive Oxygen Intermediates Suppress Spread of Cell Death in Arabidopsis thaliana. Nature Genetics, 37, 1130-1134. http://dx.doi.org/10.1038/ng1639
- Takemoto, D., Tanaka, A. and Scott, B. (2007) NADPH Oxidases in Fungi: Diverse Roles of Reactive Oxygen Species in Fungal Cellular Differentiation. Fungal Genetics and Biology, 44, 1065-1076. http://dx.doi.org/10.1016/j.fgb.2007.04.011
- Kayano, Y., Tanaka, A., Akano, F., Scott, B. and Takemoto, D. (2013) Differential Roles of NADPH Oxidases and Associated Regulators in Polarized Growth, Condidation and Hyphal Fusion in the Symbiotic Fungus Epichloë festucae. Fungal Genetics and Biology, 56, 87-97. http://dx.doi.org/10.1016/j.fgb.2013.05.001
- Lara-Ortiz, T., Riveros-Rosas, H. and Aguirre, J. (2003) Reactive Oxygen Species Generated by Microbal NADPH Oxidase NoxA Regulate Sexual Development in Aspergillus nidulans. Molecular Microbiology, 50, 1241-1255. http://dx.doi.org/10.1046/j.1365-2958.2003.03800.x
- Wilk, S., Wilk, E. and Magnusson, R.P. (1998) Purification, Characterization, and Cloning of a Cytosolic Aspartyl Aminopeptidase. The Journal of Biological Chemistry, 273, 15961-15970. http://dx.doi.org/10.1074/jbc.273.26.15961
- Wery, J., Dalderup, M.J.M., Linde, J.T., Boekhout, T. and Van Ooyen, A.J.J. (1998) Structural and Phylogenetic Analysis of the Actin Gene from the Yeast Phaffia rhodozyma. Yeast, 12, 641-651. http://dx.doi.org/10.1002/(SICI)1097-0061(19960615)12:7<641::AID-YEA952>3.0.CO;2-2
- Plamann, M., Minke, P.F., Tinsley, J.H. and Bruno, K.S. (1994) Cytoplasmic Dynein and Actin-Related Protein Arp1 Are Required for Normal Nuclear Distribution in Filamentous Fungi. The Journal of Cell Biology, 127, 139-149. http://dx.doi.org/10.1083/jcb.127.1.139
- Bourett, T.M. and Howard, R.J. (1991) Ultrastructural Immunolocalization of Actin in a Fungus. Protoplasma, 163, 199-202. http://dx.doi.org/10.1007/BF01323344
- Helgason, T., Watson, I.J. and Young, J.P.W. (2006) Phylogeny of the Glomerales and Diversisporales (Fungi: Glomeromycota) from Actin and Elongation Factor 1-Alpha Sequences. FEMS Microbiology Letters, 229, 127-132. http://dx.doi.org/10.1016/S0378-1097(03)00802-4
- Mach, J., Poliak, P., Matusková, A., Zársky, V., Janata, J., Lukes, J. and Tachezy, J. (2013) An Advanced System of the Mitochondrial Processing Peptidase and Core Protein Family in Trypanosoma brucei and Multiple Origins of the Core I Subunit in Eukaryotes. Genome Biology and Evolution, 5, 860-875. http://dx.doi.org/10.1093/gbe/evt056
- Kucera, T., Otyepka, M., Matusková, A., Samad, A., Kutejová, E. and Janata, J. (2013) A Computational Study of the Glycine-Rich Loop of Mitochondrial Processing Peptidase. PLoS One, 8, Article ID: e74518. http://dx.doi.org/10.1371/journal.pone.0074518
- Teixeira, P.F. and Glaser, E. (2013) Processing Peptidases in Mitochondria and Chloroplasts. Biochimica et Biophysica Acta, 1833, 360-370. http://dx.doi.org/10.1016/j.bbamcr.2012.03.012
- Pozuelo-Rubio, M. (2012) 14-3-3 Proteins Are Regulators of Autophagy. Cells, 1, 754-773.
- Dougherty, M.K. and Morrison, D.K. (2004) Unlocking the Code of 14-3-3. Journal of Cell Science, 117, 1875-1884. http://dx.doi.org/10.1242/jcs.01171
- Mielnichuk, N. and Pérez-Martín, J. (2008) 14-3-3 Regulates the G2/M Transition in the Basidiomycete Ustilago maydis. Fungal Genetics and Biology, 45, 1206-1215. http://dx.doi.org/10.1016/j.fgb.2008.05.010
- Conti, A., Sueur, C., Lupo, J., Brazzolotto, X., Burmeister, W.P., Manet, E., Gruffat, H., Morand, P. and Boyer, V. (2013) Interaction of Ubinuclein-1, a Nuclear and Adhesion Junction Protein, with the 14-3-3 Epsilon Protein in Epithelial Cells: Implication of the PKA Pathway. European Journal of Cell Biology, 92, 105-111. http://dx.doi.org/10.1016/j.ejcb.2012.12.001
- Mackintosh, C. (2004) Dynamic Interactions between 14-3-3 Proteins and Phosphoproteins Regulate Diverse Cellular Processes. Biochemical Journal, 381, 329-342. http://dx.doi.org/10.1042/BJ20031332
- Sluchanko, N.N. and Gusev, N.B. (2012) Oligomeric Structure of 14-3-3 Protein: What Do We Know about Monomers? FEBS Letters, 586, 4249-4256. http://dx.doi.org/10.1016/j.febslet.2012.10.048
- Macakova, E., Kopecka, M., Kukacka, Z., Veisova, D., Novak, P., Man, P., Obsil, T. and Obsilova, V. (2013) Structural Basis of the 14-3-3 Protein Dependent Activation of Yeast Neutral Trehalase Nth1. Biochimica et Biophysica Acta, 1830, 4491-4499. http://dx.doi.org/10.1016/j.bbagen.2013.05.025
- Kleppe, R., Ghorbani, S., Martinez, A. and Haavik, J. (2013) Modelling Cellular Signal Communication Mediated by Phosphorylation Dependent Interaction with 14-3-3 Proteins. FEBS Letters, 588, 92-98.
- Keightley, M.C., Crowhurst, M.O., Layton, J.E., Beilharz, T., Markmiller, S., Varma, S., Hogan, B.M., Jong-Curtain, T.A., Heath, J.K. and Lieschke, G.J. (2013) In Vivo Mutation of Pre-mRNA Processing Factor 8 (Prpf8) Affects Transcript Splicing, Cell Survival and Myeloid Differentiation. FEBS Letters, 587, 2150-2157. http://dx.doi.org/10.1016/j.febslet.2013.05.030
- Orvain, C., Matre, V. and Gabrielsen, O.S. (2008) The Transcription Factor c-Myb Affects Pre-mRNA Splicing. Biochemical and Biophysical Research Communications, 372, 309-313. http://dx.doi.org/10.1016/j.bbrc.2008.05.054
- Onofri, S., Selbmann, L., Zucconi, L. and Pagano, S. (2004) Antarctic Microfungi as Models for Exobiology. Planetary and Space Science, 52, 229-237. http://dx.doi.org/10.1016/j.pss.2003.08.019
- De Hoog, G.S. and Grube, M. (2008) Black Fungal Extremes. Studies in Mycology, 61, 198.
- Van Leeuwen, M.R., Wyatt, T.T., Golovina, E.A., Stam, H., Menke, H., Dekker, A., Stark, J., Wösten, H.A.B. and Dijksterhuis, J. (2013) Germination of Conidia of Aspergillus niger Is Accompanied by Major Changes in RNA Profiles. Studies in Mycology, 74, 59-70. http://dx.doi.org/10.3114/sim0009
- Dadachova, E. and Casadevall, A. (2008) Ionizing Radiation: How Fungi Cope, Adapt, and Exploit with the Help of Melanin. Current Opinion in Microbiology, 11, 525-531. http://dx.doi.org/10.1016/j.mib.2008.09.013


