Advances in Bioscience and Biotechnology
Vol.3 No.1(2012), Article ID:17185,6 pages DOI:10.4236/abb.2012.31003
Adsorption of cytochrome c by succinic anhydride-modified peanut shells
![]()
Key Laboratory of Analytical Chemistry, The State Ethnic Affairs Commission, College of Chemistry and Materials Science, South-Central University for Nationalities, Wuhan, China
Email: *libh@mail.scuec.edu.cn
Received 8 October 2011; revised 13 November 2011; accepted 16 December 2011
Keywords: Peanut Shells; Modification; Adsorption; Purification; Cytochrome c; Enzyme Activity
ABSTRACT
A novel adsorbent based on peanut shells modified with glutaraldehyde and succinic anhydride was prepared. Factors affecting the adsorption capacity, such as the pH, temperature, adsorption time, initial cytochrome c (cyt c) concentration and NaCl ionic strength, were extensively investigated. The results showed that the maximum adsorption capacity of the modified peanut shells (MPSs) was 432.6 mg/g when 10 mL of cyt c solution was adsorbed by 20 mg of MPSs at pH 5.0 for 3 h. In contrast, the adsorption capacities of the unmodified peanut shells (PSs), alkaline peanut shells (APSs) and crosslinked peanut shells (CPSs) were only 100.6, 180.3, and 173.0 mg/g, respectively, 4.3-, 2.4-, and 2.5-fold lower, respectively, than that of the modified shells. The desorption rate reached 89.9% with 1.5 mol/L NaCNS as an eluent, because the electrostatic attraction between the positive charges of the protein and the negative charges of the MPSs was reduced when the ionic strength was increasing. The MPSs were used to separate and purify cytochrome c from pig myocardium. A purification of 13.5-fold in a single step with a total enzyme activity recovery of 74.0% was achieved.
1. INTRODUCTION
Cyt c, a type of spherical electronic-transfer protein in the biological respiratory chain that has high biological stability, is not only an alkaline respiratory enzyme with an iron porphyrin group but is also an important cell respiration activator. It is readily soluble in water and acidic solutions and is abundant in yeast and animal myocardium. Typically, crude cyt c is obtained from animal myocardium by salting-out and then further purified by column chromatography. However, this process is time consuming and has a low throughput. In contrast, adsorption techniques using adsorbents with affinity ligands allow a high throughput and a short purification circle. Recently, experiments on the purification cyt c with different adsorbents have been reported. These adsorbents include polyhydroxylethyl methylacrylic acid bonded with dimethyleneimine [1], Fe3O4 containing nanomaterials modified with Cl-NH2 [2] and magnetic materials bonded with Cu2+ [3] None of these adsorbents is a biomaterial, and they all have a low adsorption capacity for cyt c. In contrast, we demonstrated that affinity ligandmodified peanut shells have much better adsorption properties for cyt c. PSs are used to treat wastewater in development research pertaining to agricultural waste [4]. The use of similar agricultural wastes in deodorisation [5] and in the adsorption of oil [6] and hyperoxide [7] have also been reported. However, the use of chemically modified peanut shells to adsorb bioactive substances has not been studied previously. The primary components of peanut shells are cellulose and hemicellulose. Therefore, there are a large number of hydroxyls located on the surface of peanut shells that can be effectively activated and easily modified with carboxyl, amidogen and affinity dye ligands. The affinities of peanut shells for cyt c differ based on the modified group. Based on this, we can achieve the selective adsorption of cyt c and lysozyme over haemocyanin and ovalbumin. In this study, we prepared a novel adsorbent from peanut shells modified with succinic anhydride and then compared its adsorption of cyt c with that of unmodified shells. In addition, using optimised parameters, cyt c was purified from a solution crude extract of pig myocardium using this novel adsorbent.
2. MATERIALS AND METHODS
2.1. Materials
Glutaraldehyde was purchased from Jingchun Chemical Co., and cytochrome c was obtained from Sigma (USA). All other chemicals were of analytical grade. Pig myocardium and peanut shells were purchased from a farmer’s market in Wuhan, China. The collected biomaterial was extensively washed with tap water to remove soil and dust, sprayed with distilled water and then dried in an oven at 50˚C to a constant weight. The dry biomass was crushed to form a powder and sieved with 100 - 140 mesh.
2.2. Preparation of the Adsorbent
PSs (7.5 g) were mixed with 200 mL of NaOH solution (20 wt%) with stirring at 25˚C for 16 h. The mixture was then filtered, rinsed with water until the pH was 7, ovendried and stored in a desiccator. This protocol yielded the APSs.
To obtain CPSs, 1.5 g APSs, 2 mL 50% glutaraldehyde and 48 mL distilled water were added to a flask and shaken for 12 h at 30˚C. Then, the mixture was rinsed with distilled water. Afterwards, the filter residue was dried under vacuum at 60˚C to a constant weight.
Then, 1.0 g CPSs was added to 50 mL pyridine in which 3.0 g succinic anhydride had been dissolved. The mixture was reacted with agitation at 75˚C for 24 h. Then, the products were rinsed, leached and oven-dried to obtain MPSs. The MPSs were treated with saturated NaHCO3 for one hour and then oven-dried, weighed (1.45 g) and stored in a desiccator. The ligand-binding rate (45%) was calculated by weighing the peanut shells before and after modification.
2.3. Equipment and Characterisation Methods
XPS (VGMultilab 2000 X-ray photoelectron spectrometer) was used to analyse the surfaces of the different types of biomass: PSs, APSs, CPSs and MPSs. The analysis was performed with an Mg X-ray source to determine the percentages of C, O and N atoms on the surface of the samples. During each measurement, the pressure in the analysis chamber was maintained at less than 10−8 Torr. All binding energies were referenced to the neutral C (1 s) peak at 284.6 eV to compensate for the surface charge effects.
Infrared spectra of PSs, CPSs and MPSs were obtained using an FT-IR spectrophotometer (Nicolet NEXUS 470, Nicolet Co., Ltd., USA) with KBr disks. A Systronics microprocessor pH meter (pHS-3C, Shanghai Leizi Instrument Factory, China) was used to take the pH measurements. A temperature-controlled water bath flask shaker (SHZ-03, Shanghai Kanxin Instrument Factory, China) was used to mix all solutions. The concentration of cyt c in each solution was determined using a UV-vis spectrophotometer (LAMBDA B10 35, PerkinElmer, USA).
2.4. Adsorption and Desorption Performance
The adsorption of cyt c in aqueous solution was performed by adding 20 mg PSs, APSs, CPSs or MPSs to each of a series of 50 mL conical flasks containing 10 mL of single cyt c solution (0.5 mg/mL). Then, the flasks were placed in a flask shaker at 120 rpm for 3 h before centrifugation. The cyt c concentration was determined with a UV-vis spectrophotometer at the wavelength of 408 nm. The amount of adsorbed cyt c was determined by calculating the concentration difference between the initial and residual solutions. The cyt c adsorption rate or adsorption capacity of the adsorbent was calculated as follows.
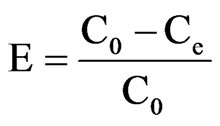 (1)
(1)
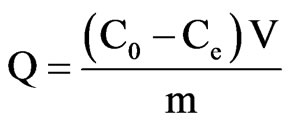 (2)
(2)
where E is the adsorption rate; Q is the adsorption capacity; C0 and Ce are the cyt c concentrations before and after adsorption, respectively; m is the mass of the biomass; and V is the volume of the cyt c solution.
After adsorption, the solution was centrifuged, and the supernatant was discarded. The desorption of cyt c was performed by putting 10 mL eluent into each flask, and the flasks were then shaken at 120 rpm for 3 h. The concentration and the desorption rate of cyt c were determined.
2.5. Cytochrome c Purification from Pig Myocardium
The method for the preparation of the crude pig myocardium extract for the purification of cyt c can be found in Zhang [8].
2.6. Cytochrome c Activity Measurement
The cyt c activity was determined based on its protoheme activity toward hydrogen peroxide according to the method of Li [9]. One cyt c unit was defined as the amount of enzyme that caused an increase or decrease of 0.001 per minute in the absorbance at 426 nm using 20 mmol/L o-diaminobenzene and 20% H2O2 as the substrate.
3. RESULTS AND DISCUSSION
3.1. Adsorbent Characterisation
3.1.1. FT-IR Spectra
Infrared spectroscopy provided information on the chemical structure of the adsorbent materials. As shown in Figure 1, the 3360/cm band was attributed to the stretching vibrations of the hydroxyl group (-OH); the 2930/cm
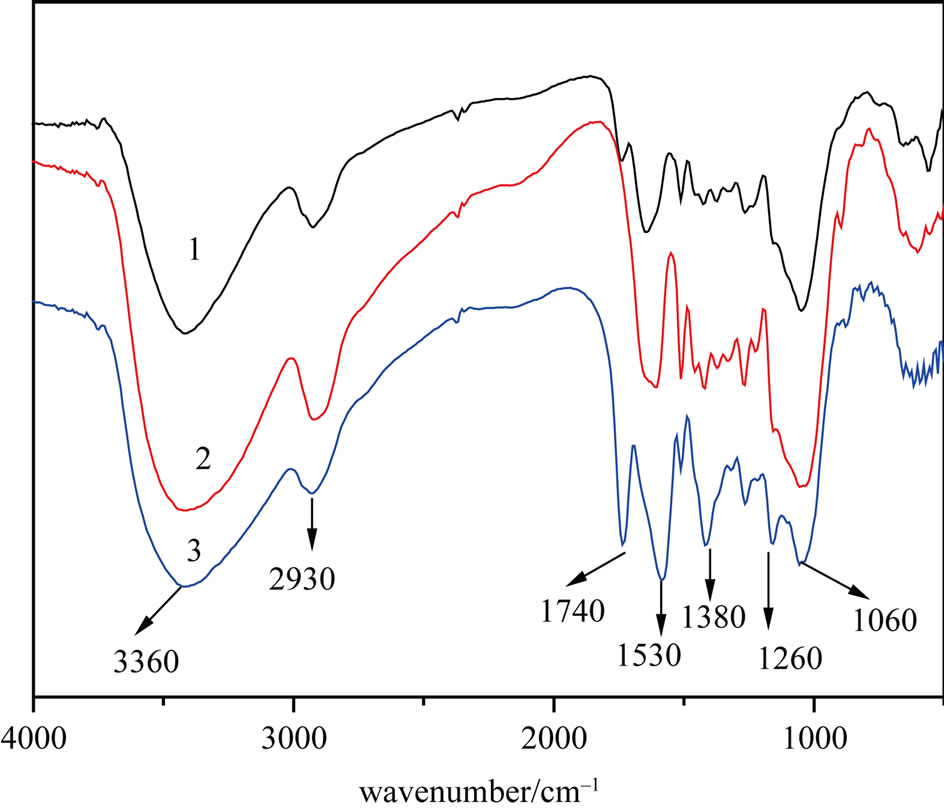
Figure 1. FT-IR spectra of the PSs (1), CPSs (2) and MPSs (3).
band was attributed to the stretching vibrations of -CH; the 1380/cm band was attributed to the deformation vibration of -CH in cellulose and hemicellulose; the 1260/cm band was attributed to the vibration of C-O in lignose; and the 1060/cm band was attributed to the stretching vibrations of C-O in cellulose and hemicellulose [10-12]. Compared with that of CPSs, the FT-IR spectrum of MPSs contained a new peak at 1740/cm that was due to the stretching vibrations of carboxyl groups (C=O) in carboxylates [13]. The results indicate that succinic anhydride was grafted on to the shells, leading to an increase in the weight of the biomass.
3.1.2. X-Ray Photoelectron (XP) Spectra
The C:O:N ratios on the surfaces of the PSs, APSs, CPSs and MPSs were 72.31:24.94:2.75, 73.07:24.32:2.62, 70.91: 27.09:2.0 and 65.42:33.33:1.26, respectively. There was a significant 6.24% increase in the percentage of oxygen atoms and a 5.49% decrease in the percentage of carbon atoms in the MPSs compared with the CPSs. Moreover, we found that the area ratio of the O(1 s) spectra for the MPSs was higher than that for the unmodified shells. These results confirmed that the biomass was indeed modified by succinic anhydride through the modification reaction.
3.2. Adsorption Experiments
3.2.1. Effect of pH on Adsorption
Figure 2 illustrates the effect of pH on cyt c adsorption. This adsorption primarily depends on the physicochemical properties of cyt c and its degree of ionisation under specific pH conditions. When the pH was between 4.0 and the pI of cyt c (10.7), cyt c was positively charged, whereas the carboxyl groups on the surface of the adsorbent dissociated to form -COO–, allowing cyt c to be
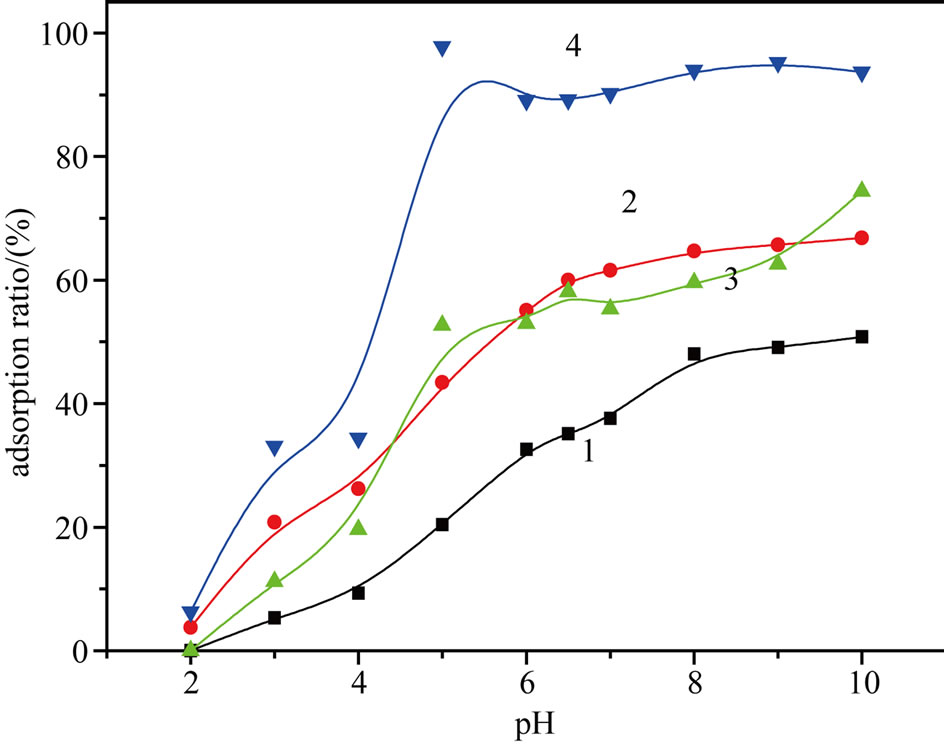
Figure 2. Effect of pH on the cyt c adsorption by PSs (1), APSs (2), CPSs (3) and MPSs (4).
adsorbed through an electrostatic interaction. From Figure 2, we can see that the adsorption ratio of cyt c increased along with the increase in pH in this range. When the pH was 5.0, adsorption ratio of cyt c reached a maximum. At pH equal to pI, the net charge of cyt c is zero. At this pH, cyt c could be only be adsorbed through hydrophobic interactions with its hydrophobic surface patch and exposed intramolecular hydrophobic residues; the resulting adsorption ratio of cyt c was also high when pH = 10.7.
3.2.2. Effects of Adsorption Time and Temperature
We investigated the effect of the adsorption time on the adsorption rate. The adsorption rate increased and reached equilibrium within 3 h. This result could be explained by the fact that at the beginning, more adsorption sites on the peanut shell surface were exposed and the concentration of cyt c in the liquid phase was higher. However, as more sites were gradually occupied by cyt c and the concentration of cyt c in the liquid phase decreased, the adsorption equilibrium was reached.
Our experiments also showed that the temperature had no influence on the adsorption ratio of cyt c in the range of 15˚C - 40˚C.
3.2.3. Effect of the Initial Concentration of Cytochrome c
As shown in Figure 3, an increase in the initial cyt c concentration resulted in an increase in the adsorption capacity of the four types of adsorbent. When the initial concentration was 1.5 mg/mL, the maximum adsorption capacity was 432.6 mg/g for the MPSs. Compared with the PSs, APSs and CPSs, the adsorption capacity of the MPSs was significantly greater. This greater capacity was clearly due to the specific interaction between cyt c and -COO– on the surface of the modified peanut shells, indicating that the non-specific interactions between cyt c and the unmodified peanut shells resulted in only a low adsorption capacity.
3.2.4. Effect of Ionic Strength
The results presented in Figure 4 show the effect of ionic strength, adjusted using NaCl, on cyt c adsorption; the adsorption capacity significantly decreased with increasing ionic strength of the binding buffer (acetic acid buffer). As the NaCl solution concentration changed from 0.05 M to 1.0 M, the adsorption rate of cyt c decreased by 93.2%, a change that may be attributed to the repulsive electrostatic interactions between the MPSs and the protein molecules. As the ionic strength increased, the width of the electrical double layer around the molecules increased, thereby decreasing the electrostatic forces between molecules.
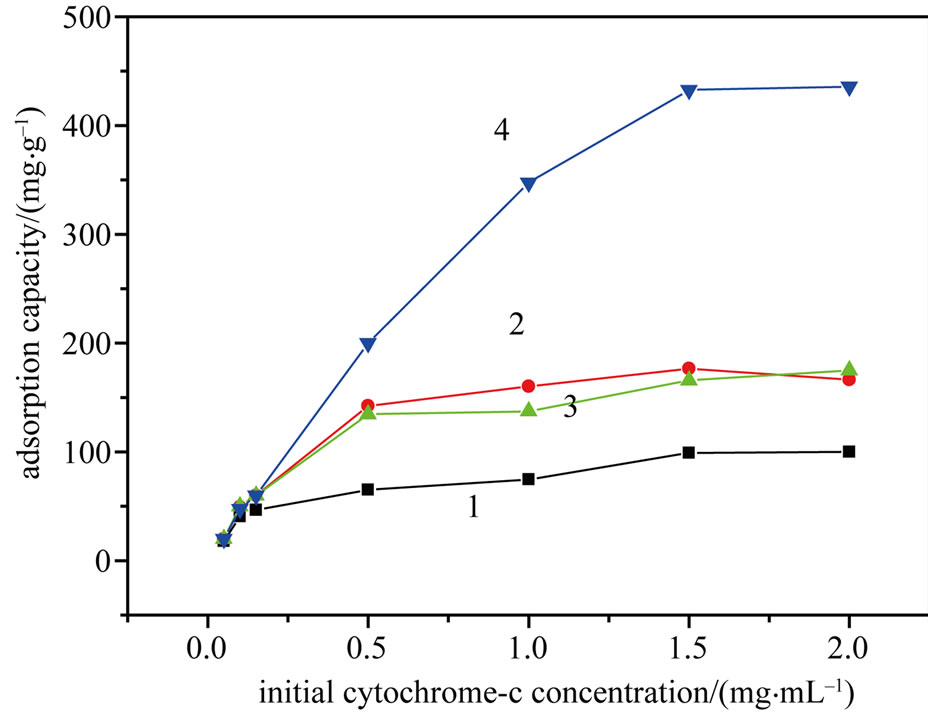
Figure 3. Effect of the initial cyt c concentration on the adsorption capacity of PSs (1), APSs (2), CPSs (3) and MPSs (4). (Adsorption time, 3 h; pH 5.0; temperature, 25˚C).

Figure 4. Effect of the NaCl concentration on the cyt c adsorption by PSs (1), APSs (2), CPSs (3) and MPSs (4). (Adsorption time, 3 h; initial cyt c concentration, 0.1 mg/mL; pH 5.0; temperature, 25˚C).
3.2.5. Adsorption Isotherm
The results in Figure 3 suggest that at low concentrations, the adsorption behavior occurred primarily on the adsorbent surface and chemisorption was dominant. The Langmuir [14] and Freundlich [15] adsorption isotherm equations were used to model the adsorption processes. The Langmuir and Freundlich adsorption constants were calculated from the isotherms along with the correlation coefficients (Table 1). With R2 > 0.99, the Langmuir isotherm gave a better fit than the Freundlich isotherm did. This result suggests that the cyt c adsorbed onto the surface of the modified biomass in a monolayer. Moreover, the ratio of the qm value of the MPSs to that of the PSs was 4.3, demonstrating the significantly improved adsorption capacity of the MPSs. Based on this model, the theoretical results were consistent with the experimental data.
3.3. Desorption Experiments and Reusability
To study the reusability of the MPSs, two eluents, aqueous solutions containing NaCl or NaSCN, were used in desorption experiments. As shown in Figure 5, 89.9% and 76.2% of the adsorbed cyt c was eluted with 1.5 M NaSCN and 1.5 M NaCl, respectively. When the ionic strength was changed by increasing the NaSCN concentration from 0.5 M to 1.5 M, the electrostatic attraction between the positive charges of the protein and the negative charges of the MPSs was reduced, leading to desorption. Moreover, increasing the ionic strength of a solution would increase the thickness of the electrical double layer, thereby shielding the original charges and decreasing the strength of the electrostatic forces.
When using 10 mL of pure cyt c solution (0.5 mg/mL) as the sample, we found that the cyt c adsorption capacity of the MPSs decreased by only 3% after three cycles

Figure 5. Effect of varying eluents on the cyt c elution rate (MPSs). (Elution time, 3 h; pH 7.0; temperature, 25˚C).
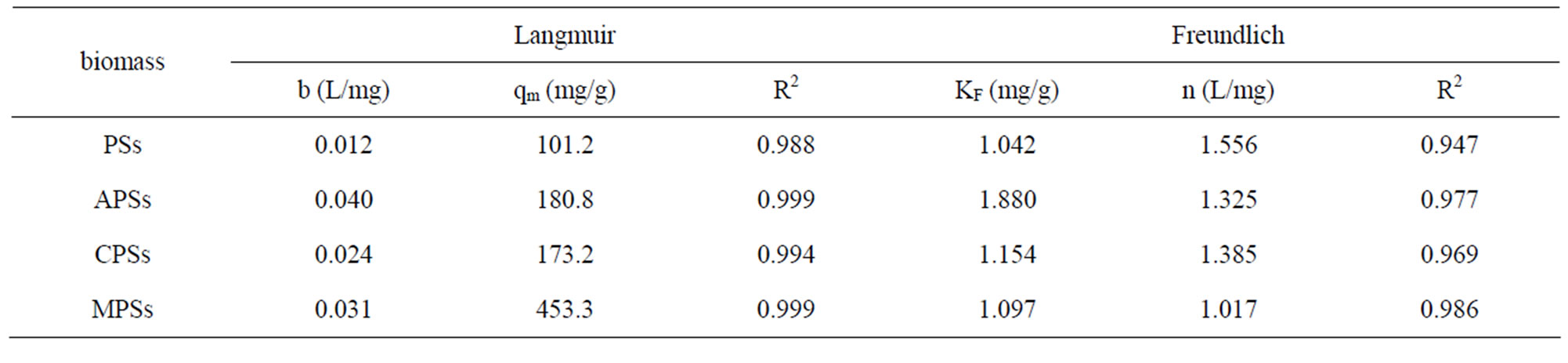
Table 1. Langmuir parameters for the adsorption of cytochrome c by the different types of biomass.
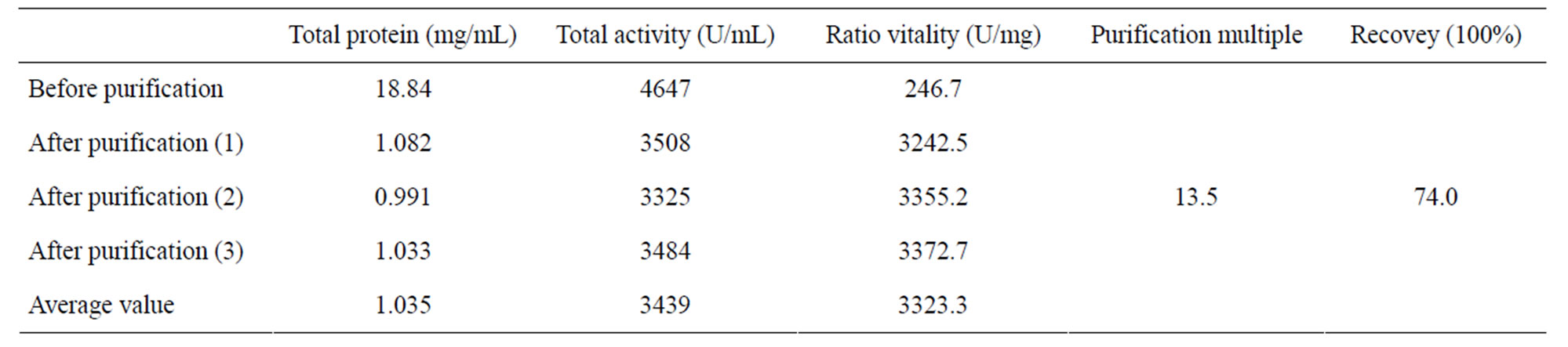
Table 2. Purification of cytochrome c from pig myocardium (MPSs).
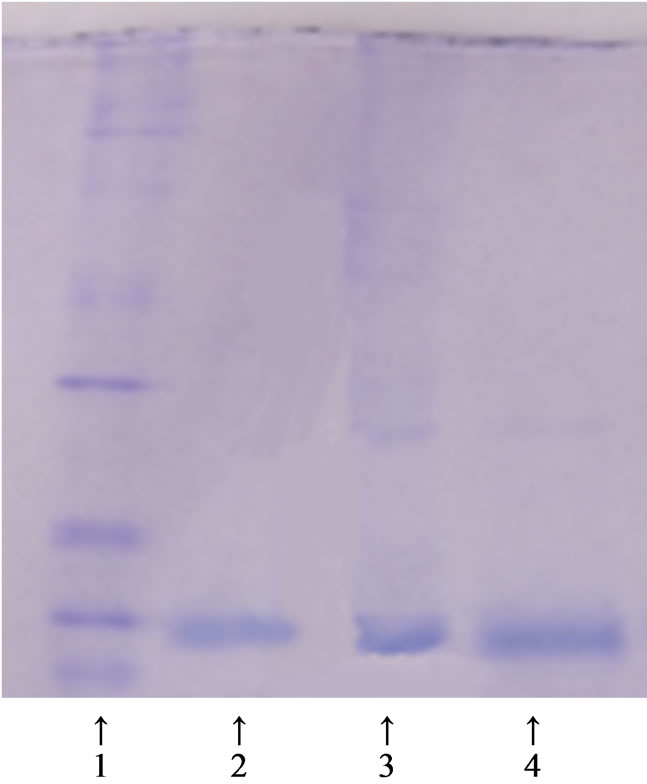
Figure 6. SDS-PAGE (MPSs). 1. Molecular weight markers; 2. Cyt c standard; 3. Crude pig myocardium extract; 4. Fraction eluted with 1.5 M NaCN.
of adsorption and desorption, indicating that the MPSs has a good chemical stability and that it can be re-used with little loss of adsorption capacity.
3.4. Purification of Cytochrome c from Pig Myocardium
Our results show that the activities of pure cyt c before and after the adsorption were 5118 U/mg and 4809 U/mg respectively, and the percentage of retained activity was 94.0%. Therefore, the use of MPSs is an efficient method to purify cyt c from pig myocardium. Regarding the separation and purification of cyt c from pig myocardium in a single step, Table 2 shows that the recovery of cyt c activity was 74.0% and the purification fold was 13.5. Figure 6 shows the electrophoretic patterns. The highly purified cyt c product is presented in the Figure 6(4).
4. CONCLUSIONS
In this study, we developed a novel adsorbent, succinic anhydride-modified peanut shells, that could selectively and efficiently separate cyt c from pig myocardium. The preparation process was simple, and the required materials are readily available, implying that this method has many advantages for protein purification and other applications. The cyt c adsorption capacity of the MPSs was 432.6 mg/g, much better than any adsorbent tested in previous studies. Moreover, the cyt c activity was well maintained during the adsorption process. With the MPSs, the total activity recovery of cyt c after extraction from pig myocardium was 74.0%, and the purification fold was 13.5, demonstrating the high purification efficiency of this method.
5. ACKNOWLEDGEMENTS
We are grateful to the College of Chemistry and Materials, SouthCentral University for Nationalities, for providing financial support for this research project.
REFERENCES
- Turkmen, D., Yavuz, H. and Denizli, A. (2006) Synthesis of tentacle type magnetic beads as immobilized metal chelate affinity support for cytochrome c adsorption. International Journal of Biological Macromolecules, 38, 126-133.
- Jang, J.H. and Lim, H.B., (2010) Characterization and analytical application of surface modified magnetic nanoparticles. Microchemical Journal, 94, 148-158. doi:10.1016/j.microc.2009.10.011
- Akkaya, B., Uzun, L., Candan, F. and Denizli, A. (2007) N-methacryloyl-(L)-histidine methyl ester carrying porous magnetic beads for metal chelate adsorption of cytochrome c. Materials Science and Engineering: C, 27, 180-187. doi:10.1016/j.msec.2006.04.009
- Zhang, Q.F., Zhu, Y.B. and Li, J.P. (2009) Compared experiment on heavy metal removal by modified peanut shell and corncob. Journal of Peanut Science, 38, 6-10.
- Sekine, H. (2000) Gas treatment medium using fiber assembled body and gas treatment apparatus using the medium, Jpn. Kokai Tokkyo Koho JP patent No. 2000- 237530.
- Hacher, T., Roller, P. and Schmauder, R. (2000) Oil spill adsorbent based on straw. Ger. Offen. DE 19903862.
- Gnanasambandam, R. and Proctor, A. (1997) Soy hull as adsorbent source in processing soy oil. Journal of the American Oil Chemists’ Society, 74, 685-692.
- Zhang, L.X., Zhang, T.F. and Li, L.Y. (1997) Experimental methods and techniques of biochemistry. 2nd Edition, Higher Education Press, Beijing, 124-125.
- Li, R.Q., Chen, Y., Jiang, F.Y., Fang, L. and Zhang, X.F. (2005) Measurement of the quality of purified cyt c products using kinetic spectrophotometry. Journal of Analytical Science, 21, 463-464.
- Pandey, K.K. and Pitman, A.J. (2003) FTIR studies of the changes in wood chemistry following decay by brown-rot and white-rot fungi. International Biodeterioration and Biodegradation, 52, 151-160. doi:10.1016/S0964-8305(03)00052-0
- Jeffrey, A.B. and Ronaid, J.S.(2002) Chemical composition and bioavailability of thermally altered pinus resinosa (red pine). Wood Organic Geochemistry, 33 1093- 1109. doi:10.1016/S0146-6380(02)00062-1
- Gong, R.M., Sun, Y.Z. and Chen, J. (2005) Effect of chemical modification on dye adsorption capacity of peanut hull. Dyes and Pigments, 67, 175-181. doi:10.1016/j.dyepig.2004.12.003
- Zheng, L., Dang, Z. and Yi, X. (2010) Equilibrium and kinetic studies of adsorption of Cd(II) from aqueous solution using modified corn stalk. Journal of Hazardous Materials, 176, 650-656. doi:10.1016/j.jhazmat.2009.11.081
- Richard, S., Blackburm, H.A. and Lorna, L.K. (2006) Sorption of poly (hexamethylenebiguanide) on cellulose: Mechanism of binding and molecular recognition. Langmuir, 22, 5636-5644. doi:10.1021/la053002b
- Tan, I.A.W., Hameed, B.H. and Ahmad, A.L. (2007) Equilibrium and kinetic studies on basic dye desorption by oil palm fiber activated carbon. Chemical Engineering Journal, 27, 111-119. doi:10.1016/j.cej.2006.09.010
NOTES
*Corresponding author.

