Advances in Bioscience and Biotechnology
Vol.2 No.3(2011), Article ID:5042,4 pages DOI:10.4236/abb.2011.23020
Changes in the organization of isozyme loci Me1 and Adh1induced with Triton X-100 in common wheat lines
![]()
1Institute of Plant Biology and Biotechnology, Almaty, Kazakhstan;
2Institute of Cytology and Genetics, Novosibirsk, Russia.
E-mail: carinamakh@mail.ru, levites@bionet.nsc.ru
Received 17 March 2011; revised 21 April 2011; accepted 25 April 2011.
Keywords: Common Wheat; Triton X-100; DNA; ISSR-Amplification; Isozymes
ABSTRACT
The study of PCR-profiles of isozyme loci Me1 and Adh1 in common wheat lines obtained by means of treatment of initial cultivar Alem with detergent Triton X-100 was carried out by the modified ISSRamplification method. It was demonstrated that exposure to Triton X-100 causes changes of PCR-profiles of enzyme loci. The obtained data are suggestive of the role of chromatin and nuclear membrane interaction in structural-functional genome organization.
1. INTRODUCTION
An important moment in the study of inherited variability is the induction of changes by means of different impacts [1-6]. It is known that inherited changes in plants may be induced, for example, by means of temperature or humidity changes and by changes of nutrition components ratio [1,7,8]. It enables us to assume the presence of a great number of mechanisms by means of which an organism responds to environmental stresses. It is known that induced morphological changes are connected with this or that transformation in plant genome, for example, with DNA methylation [9-11], or histone acetylation [11,12]. Very often these changes are connected with the appearance of iterations in different genome sites, their size and percentage in genome and also with the character of high and few copying sites distribution in genome limits [13,14]. However, a contribution to genome transformation may be brought even by means of changes in the structural organization of solitary loci. That is why, to gain a better understanding of common regularities underlying of plant organismal responses to environmental stresses, it is necessary to extend the search of variability inductors and to improve the methods of individual genes DNA analysis.
It is known that chromosomes are connected with the nuclear membrane [15] and this connection plays a big role in genes function [16]. We reported earlier that Triton X-100 affecting the condition of membranes and proteins connected with them caused inherited changes of morphological traits in common wheat and sugar beet plants [4-6]. In common wheat plants the induced changes of spike morphology are inherited within 5 generations [5]. This stability of changed traits arouses interest to the obtained common wheat lines from the practical and theoretical point of view and it makes the necessity for researches in molecular bases of such variability.
To study the DNA structure, a modified ISSR-amplification method is convenient where in a pair with a specific isozyme gene primer microsatellite one is used [17- 19]. By means of this method differences among plants carrying the same allele of isozyme locus were shown and also differences in the structure of marker isozyme locus evincing in different tissues of the same plant were revealed [19]. The differences of PCR-profiles of isozyme loci of common wheat genotrophs obtained by means of different epimutagens including nicotinic acid were shown [20]. In the aforementioned article, it was shown that natural epimutagens act more specifically causing the appearance of certain bands in PCR-profiles of investigated genes. At the same time, using artificial epimutagens, such a precise dependence is not found. This approach is also effective for studying the variability mechanism in common wheat under Triton X-100 treatment. Exactly this approach served as the base of the given article: comparison of expression of isozyme loci and features of DNA structures in the initial form and common wheat lines obtained from it under Triton X-100 treatment of the surface-active material.
2. MATERIALS AND METHODS
The objects of our investigation were spring common wheat cultivar Alem and lines 99-19, 99-52 and 99-54. These lines were obtained on the base of cv. Alem by its pre-sowing treatment with Triton X-100 [4]. To obtain these lines, in 2005 cv. Alem seeds were soaked in 0.1% Triton X-100 solution (sample 99-19) and in 0.01% Triton X-100 solution (samples 99-52 and 99-54). The seeds were exposed to the temperature of 25 - 30ºС for 24 hours. During tillering plants grown from the treated seeds were sprayed with 0.1% Triton X-100 solution (sample 99-19) and 0.01% Triton X-100 solution (samples 99-52 and 99-54) once for the vegetation period. The following generations were not treated any more. Dry seeds and water-soaked seeds served as control. Plantlets of seeds obtained from the self-pollination of isolated individual plant spikes of the 4th post-treatment generation were used in our research.
To reveal the polymorphism of common wheat plant PCR-profiles, the modified ISSR-amplification method was used. This method consists in the thing that, for PCR-amplification in a pair with a microsatellite primer, the primer specific to the investigated isozyme locus was used [17]. Assisted with the common wheat DNA-sequences available in the Internet, the primers for isozyme loci controlling alcohol dehydrogenase (ADH1) and malic-enzyme (ME1) were chosen. Mic 2 (5’-gacagacaga-cagac-a-3’) was used as a microsatellite primer.
DNA extraction, PCR-amplification. The total plant DNA was extracted from the plantlets by means of CTAB-method [21].
In the pair of microsatellite primer mic 2, the following specific primers were used: 1) adh1 (direct orientation), which is specific for locus Adh1; 2) malic1 (direct orientation), which is specific for locus Me1. The primers with direct orientation allowed us to amplify the structural part of the gene.
PCRs were carried out in 20 µl of reaction mixture containing 10 - 200 ng of total DNA, 65 mM tris-HCl (pH 8.0), 16 mM (NH4)2SO4, 0.05% twin-20, 1.5 mM MgCl2, 0.2 mM of each dNTP, 1 μM of each primer, 2.5 units of Taq-polymerase activity. The following temperature regime was used: preliminary denaturation – 94ºС (4 min), then 30 cycles – 94ºС (1 min), 52ºС (42 sec), 72ºС (4 min) and the last cycle – 72ºС (7 min).
The amplification products were separated in 5% polyacrylamide gel (0.5 X TBE buffer) and then they were dyed with ethidium bromide.
The electrophoregrams of the PCR-profiles were scanned with Biodoc2 device.
3. RESULTS AND DISCUSSION
The treatment of common wheat with detergent Triton X-100 results in different changes of PCR-profiles of marker isozymes which consist both in the change of separate spectrum bands frequency and in the appearance of additional bands.
So, while analyzing PCR-profiles obtained for Me1 locus with the help of primers malic1 and mic2, it was found out that, at 0.1% solution treatment, the number of bands increases in the middle part of PCR-profiles (Figure 1, 10-21). Under the treatment with Triton X-100 lesser concentration (0.01%), the occurrence frequency of phenotypes having two bands in the middle part of PCR-profile increases (Figure 1, 22-35). On the whole, we may establish the fact that Triton X-100 impact upon plants results in the increase of bands number in malic-enzyme PCR-profiles. The obtained data confirm the thing that a greater part of investigated plants demonstrates changes in enzyme locus Me1 DNA-organization.
Primer adh1 permitting to amplify the structural part of locus Adh1 was used as a specific to study locus Adh1 variability.
In the initial cultivar Alem, three types of PCR-profiles of locus Adh1 are revealed: the first type is path 1; the second type is path 2; the third type is path 6 (Figure 2: 1, 2, 6). The lines obtained on the base of this cultivar also have these profiles, though their frequencies vary. The following tendency is revealed, for example if, in
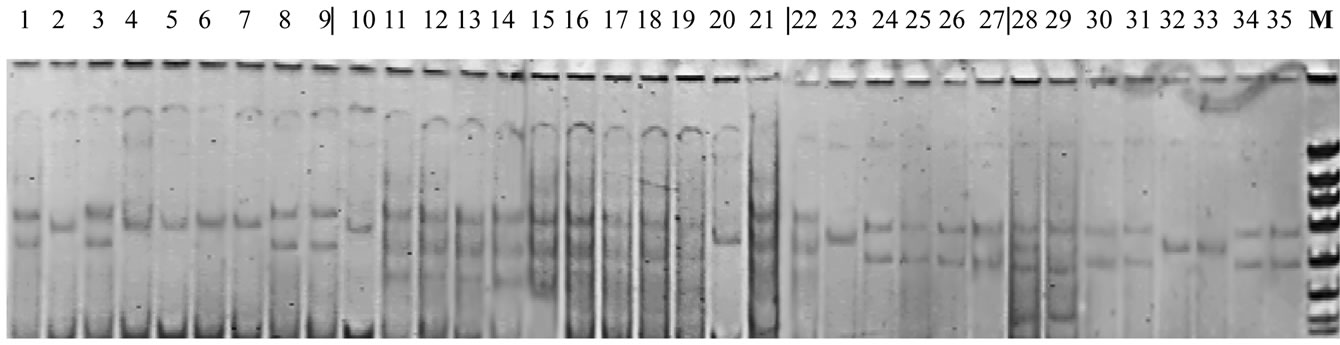
Figure 1. PCR-profiles of locus Me1 in control and treated with Triton X-100 solutions plants (Triticum aestivum L.). 1-9 – control, cultivar Alem; 10-21 – line 99-19 (treatment with 0.1% Triton X-100 solution); 22-27 – line 99-52 (treatment with 0.01% Triton X-100 solution); 28-35 – line 99-54 (treatment with 0.01% Triton X-100 solution); М – marker (pBluescript-Msp I).
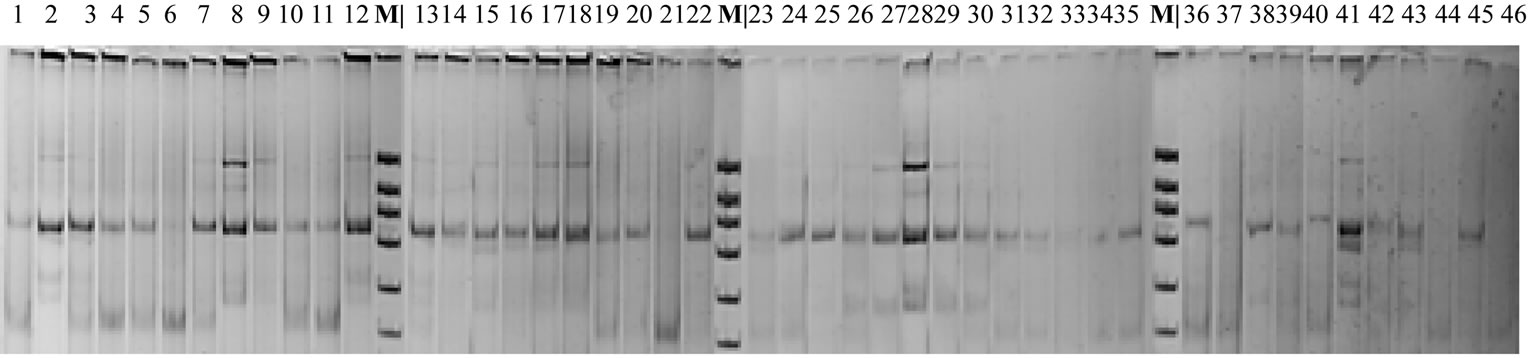
Figure 2. PCR-profiles of locus Adh1 in control and treated with Triton X-100 solutions plants (Triticum aestivum L.). 1-12 – control, cultivar Alem; 13-22 – line 99-19 (treatment with 0.1% Triton X-100 solution); 23-35 – line 99-52 (treatment with 0.01% Triton X-100 solution); 36-46 – line 99-54 (treatment with 0.01% Triton X-100 solution); М – marker (pBluescript-Msp I).
the control, the first type occurs more often then in line 99-19, and the profile that is absent in the control occurs more often in line 99-19 (Figure 2, 13-18). In line 99-52, the revealed profiles are an intermediate type between the first and the second types found in the control. PCRprofiles in line 99-54 are similar to those in line 99-52, but they have a noticeable variation of bands comparative intensity in the spectrum. The obtained data confirm the impact of Triton X-100 upon the structural part of locus Adh1. Some differences in PCR-profiles of lines 99-52 and 99-54 obtained by means of treatment with the same Triton X-100 solution (0.01%) concentration are suggestive of the role of genotypic medium in response of the structural organization of enzyme loci to environmental stresses. In some PCR-profiles of the treated plants, the occurrence of additional band ~ 330 n.p. is observed (Figure 2, 15-17, 31, 32, 41, 43).
The marker enzymes used in our contribution were different in their functions. So, alcohol dehydrogenase is the enzyme participating in anaerobic glycolysis and it is located in the soluble fraction of cytoplasm [22]. At the same time, malic enzyme is the one participating in interconversion of malic acid and pyruvic acid and it is connected with cell organelles [23]. The obtained data confirm the thing that the loci controlling enzymes, so different in their function, react to Triton X-100 impact similarly. In PCR-profiles of these loci, the separate bands appear or disappear.
The presented data demonstrate significant changes of PCR-profiles of enzyme loci in common wheat lines caused by detergent Triton X-100 treatment.
One can hypothesize that similar processes may also take place in the loci controlling morphological traits. This supposition is proved with the discovery of inherited changes of morphological traits [5] in line 99-19 obtained after the treatment of cultivar Alem with 0.1% Triton X-100 solution. The presence, in one and the same line, of both changes in morphological traits and PCR-profiles of enzyme loci enables us to hypothesize the thing that in the base of all these changes lie the similar processes where the significant role belongs to DNA interaction with the nuclear membrane.
4. CONCLUSIONS
The changes of PCR-profiles of enzyme loci under Triton X-100 treatment points out the changes in DNA organization. It can be suggestive of the significant role of the connection between chromatin and nuclear membrane in the structural and functional genome organization.
5. ACKNOWLEDGEMENTS
Investigations of Russian researchers were carried out within Integration Project #99 for 2009-2011, supported by Siberian Branch of the Russian Academy of Sciences.
REFERENCES
- Durrant, A. (1962) The environmental induction of heritable change in Linum. Heredity, 17, 27-61. doi:10.1038/hdy.1962.2
- Bogdanova, E.D. (1992) Wheat genetic variability induced by nicotinic acid and its derivatives. PH.D. Thesis, Institute of Cytology and Genetics SB RAS, Novosibirsk.
- Bogdanova, E.D. (2003) Epigenetic variation induced in Triticum aestivum L. by nicotinic acid. Russian Journal of Genetics, 39, 1221-1227. doi:10.1023/A:1025775101592
- Makhmudova, K.Kh. (2007) Inheritance of epigenetic alterations in common wheat (Triticum aestivum L.). Thesis of Candidate of Sciences, Kazakh National University, Almaty.
- Makhmudova, K.Kh., Bogdanova E.D. and Levites E.V. (2009) Triton X-100 induces heritable changes of morphological characters in Triticum aestivum L. Russian Journal of Genetics, 45, 495-498. doi:10.1134/S1022795409040152
- Kirikovich, S.S. and Levites, E.V. (2009) Effect of epimutagene Triton X-100 on morphological traits in sugar beet (Beta vulgaris L.). Sugar Tech, 11, 307-310.
- Durrant, A. and Timmis, J.N. (1973) Genetic control of environmentally induced changes in Linum. Heredity, 30, 369-379. doi:10.1038/hdy.1973.45
- Cullis, C.A. (1973) DNA differences between flax genotrophs. Nature, 243, 515-516. doi:10.1038/243515a0
- Richards, E. (1997) DNA methylation and plant development. Trends in Genetics, 13, 319-323. doi:10.1016/S0168-9525(97)01199-2
- Vanyushin, B.F. and Kirnos, M.D. (1988) DNA methylation in plants. Gene, 74, 117-121.
- Lusser, A. (2002) Acetylated, methylated, remodeled: chromatin states for gene regulation. Current Opinion in Plant Biology, 5, 437-443. doi:10.1016/S1369-5266(02)00287-X
- Pfluger, J. and Wagner, D. (2007) Histone modifications and dynamic regulation of genome accessibility in plants. Current Opinion in Plant Biology, 10, 645-652. doi:10.1016/j.pbi.2007.07.013
- Cullis, C.A. (1981) Environmental induction of heritable changes in flax: defined environments inducing changes in r-DNA and peroxidase isozyme band pattern. Heredity, 47, 87-94. doi:10.1038/hdy.1981.61
- Cullis, C.A. (2005) Mechanisms and control of rapid genomic changes in flax. Annuals of Botany, 95, 201- 206. doi:10.1093/aob/mci013
- Mosolov, A.N. (1972) New approach to decide a problem of spatial disposition of chromosomes in interphase nucleus (polar model of interphase nucleus). Cytology, 14, 542-552.
- Taddei, A., Hediger, F., Neumann, F.R. and Gasser, S.M. (2004). The function of nuclear architecture: A genetic approach. Annual Review of Genetics, 38, 305-345. doi:10.1146/annurev.genet.37.110801.142705
- Vinichenko, N.A., Kirikovich, S.S. and Levites E.V. (2006) The genetic instability of the Adh1 locus alleles in sugar beet agamospermous progeny. Sugar Tech, 8, 288-291. doi:10.1007/BF02943570
- Vinichenko, N.A., Kirikovich, S.S. and Levites, E.V (2008) Polymorphism of PCR profiles and expression of alleles at the locus Adh1 in agamospermous progeny of sugar beet Beta vulgaris L. Russian Journal of Genetics, 44, 1092-1095. doi:10.1134/S1022795408090123
- Vinichenko, N.A., Kirikovich, S.S. and Levites, E.V. (2007) Tissue distinctions in the organization of sugar beet locus Adh1. Achievements and Problems of Genetics, Breeding and Biotechnology, 2, 247-251.
- Vinichenko, N.A, Bogdanova, E.D, Makhmudova, K.Kh., Kirikovich, S.S. and Levites, E.V. (2010) Molecular differences in genotroph forms of common wheat (Triticum aestivum L.) and their initial cultivars. Advances in Bioscience and Biotechnology, 1, 162-166. doi:10.4236/abb.2010.13022
- Doyle, J.J. and Doyle, J.L. (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochemical Bulletin, 19, 11-15.
- Scandalios, J.G. (1967) Genetic control of alcohol dehydrogenase isozymes in maize. Biochemical Genetics, 1, 1-9. doi:10.1007/BF00487731
- Famiani, F., Walker, R.P., László, T., Chen, Z.H., Proietti, P. and Leegood, R.C. (2000) An immunohistochemical study of the compartmentation of metabolism during the development of grape (Vitis vinifera L.) berries. Journal of Experimental Botany, 51, 675-683. doi:10.1093/jexbot/51.345.675

