Journal of Environmental Protection
Vol. 3 No. 4 (2012) , Article ID: 18456 , 12 pages DOI:10.4236/jep.2012.34039
A Toxicological Assessment of Endocrine Disrupting Chemicals Found in the BMW (Border, Midland and Western) Region of Ireland
![]()
Endocrine Disruption Group, Department of Life and Physical Sciences, School of Science, Athlone Institute of Technology, Athlone, Ireland.
Email: *cbrougham@ait.ie
Received January 20th, 2012; revised February 14th, 2012; accepted March 19th, 2012
Keywords: Endocrine Disrupting Chemicals (EDCs); Proliferation; Transactivation; Mutagenicity; Genotoxicity; Sewage Treatment Plant (STP); Border, Midlands and Western (BMW) Region of Ireland
ABSTRACT
A battery of tests was established to determine the oestrogenic, mutagenic and genotoxic potential of two categories of endocrine disrupting chemicals (EDCs), phthalates and alkylphenols. Diisononylphthalate (DINP), diethylhexylphthalate (DEHP), dibutylphthalate (DBP), diisododecylphthalate (DIDP) and 4-nonylphenol (4-NP) were oestrogenic in the yeast estrogen screen (YES) assay and potently oestrogenic in the MVLN and E-SCREEN assays at environmentally relevant concentrations. DINP and 4-NP were mutagenic in the Ames assay and also induced significant levels of unscheduled DNA synthesis and DNA strand breakage. Significant induction in the percentage of cells containing micronuclei was observed after treatment with DINP, DEHP and 4-NP. In addition, sewage effluents from sewage treatment plants (STPs) in the Border, Midlands and Western (BMW) region of Ireland were significantly oestrogenic in the YES assay. Moreover, analysis of levels of phthalates and alkylphenol identified in Irish rivers receiving treated effluent showed potent oestrogenicity in the YES assay. The proliferative and genotoxic ability of the phthalates and alkylphenol, and the oestrogenicity of the treated effluents reported here, is significant as these EDCs and EDCs within the effluent may play a role in the etiology of human abnormalities.
1. Introduction
Endocrine disrupting chemicals (EDCs), defined as any substance man-made or natural that interferes with hormones in the body, are being released into the environment. Pollution of natural waters with waste effluents arising from diverse industries has become a significant problem in Ireland and globally, as industrial growth and development have been on a very large scale. Replacement of endogenous hormones with foreign mimetics can result in an agonistic or antagonistic profile. The consequences of developmental exposure may result in irreversible deleterious effects in a number of reproductive and non-reproductive processes. The chemical diversity of EDCs, with respect to their molecular structure, precludes the precise prediction of oestrogenic activity on this basis.
Sewage, a complex mixture of organic and inorganic chemicals, is considered to be a major source of environmental pollution. In the UK a random screen of 20 organic man-made chemicals present in liquid effluents revealed that half appeared to interact with the oestrogen receptor [1] and results obtained from fifteen sewage treatment plants (STPs) showed that exposure of male trout to effluent resulted in a very pronounced increase in plasma vitellogenin concentration indicating exposure to a substance or substances oestrogenic to fish [2]. More recently studies in the UK have reported a reduction in egg production in fish after exposure to wastewater treatment effluent [3] and that the life stage at which male fish are exposed can have dramatic consequences for sexual disruption [4]. The UK Environmental Protection Agency have concluded that the weight of evidence for endocrine disruption of fish, with particular attention to the phenomenon of feminisation of male fish, is sufficient to develop a risk management strategy for oestrogenically active effluents that discharge to the aquatic environment [5].
Markman et al., 2007 [6] have reported on the accumulation of EDCs in earthworms within the sewage treatment system and that animals can accumulate EDCs to levels significantly higher than those in wastewater.
A combination of selected test systems has been undertaken to aid in the screening and assessment of the oestrogenic and genotoxic activity of EDCs found in Irish waterways. Reporter bioassays that are based on the ability of a compound to stimulate ER-dependent transcriptional activity were exploited here. The yeast estrogen screen (YES) [7] and the MVLN assays [8] were used to assess ER-dependent transcriptional expression of the EDCs. The E-SCREEN assay [9] was used to assess proliferation of the EDCs. The proliferation observed in the E-SCREEN assay is thought to be a nonreceptor mediated response and the hallmark of oestrogen activity [10].
Testing of substances for genotoxicity is a reliable tactic for the evaluation of genetic hazard and indicating possible tumourigenic potential. The assessment of possible drug-related genotoxic potential requires the use of various short-term tests suitable for the detection of all types of genetic endpoints. The EDCs were investigated for genotoxicity, using the comet assay [11], the in vitro unscheduled DNA synthesis assay (UDS) [12] and the in vitro cytokinesis-block micronucleus technique (CBMN) [13]. The EDCs were also investigated for the potential to induce mutagenesis using the well established bacterial reverse-mutation test, the hallmark of mutagencity [14- 17].
The Yes assay was employed to assess the oestrogenic potency of environmental samples from the BMW region (Roscommon, Athlone, Tullamore and Longford) in Ireland. Yeast is more resistant to environmental contaminants such as heavy metals and is well suited for monitoring the oestrogenic potential of natural specimens such as effluent. Composite effluents from wastewater plants (tertiary treatment) were sampled throughout the year to assess seasonal variation. The receiving waters tested were the Hind River upstream and downstream of Roscommon STP and the Camlin River upstream and downstream of Longford STP. In addition levels of selected EDCs recovered from freshwater samples in the BMW of Ireland [18,19], were tested in the YES assay for oestrogenic potency.
2. Materials and Methods
2.1. Materials
Dimethyl sulphoxide, diisononylphthalate (≥99%), diethylhexylphthalate (≥99%), diisododecylphthalate (≥ 99%), dibutylphthalate (≥99%), L-glutamine, sodium bicarbonate, D-glucose, sulphorhodamine B, TCA, tris base, phenol red-free Dulbecco’s modified eagle medium (DMEM), potassium phosphate monobasic anhydrous, ammonium sulphate, potassium hydroxide pellets, magnesium sulphate anhydrous, iron (III) sulphate pentahydrate, L-leucine, L-histidine free base, adenine free base, L-arginine hydrochloride, L-methionine, L-tyrosine free base, L-isoleucine, L-lysine monohydrochloride, Lphenylalanine, L-glutamic acid free acid, L-valine, Lserine, thiamine hydrochloride, pyridoxine, D-pantothenic acid hemicalcium salt, inosital, d-biotin, D-(+)-glucose anhydrous, L-aspartic acid free acid, L-threonine, copper (II) sulphate anhydrous, sodium hydroxide pellets, sodium chloride, EDTA dihydrate, triton X-100, sigma 7-9R tris, ethidium Bromide, electrophoresis film, hydrogen peroxide, Hams F-12 nutrient mixture, cytochalasin B, acridine orange, formaldehyde, methanol, hydroxyurea crystalline, nitroquinoline–N-oxide, acetic acid, non-essential amino acids and glycerol purchased from Sigma Aldrich (Ireland). 4-Nonylphenol (≥98%), 2-aminoanthracene and sodium azide purchased from Lennox (Ireland). 2-Nitrofluorene purchased from Merck (Ireland). 17β-Oestradiol (≥97%) purchased from Merck (Germany). Hylcone foetal bovine serum, sodium pyruvate, L.M.P. agarose, scintillation cocktail—Ecoscint, Nunc 96 microwell plates and white solid 96 microwell plates purchased from Bio Sciences (Ireland). Linbro 24 well tissue culture plates and DMEM purchased from MP Biomedicals (UK). 3H Thymidine purchased from Amersham (UK). CPRG purchased from Fannin Healthcare (Ireland). Luciferase cell culture lysis reagent and Bright glo luciferase assay system purchased from Medical Supply (Ireland). Quadriperm plus dishes purchased from Sartorius (UK). Petri dishes, 6 well TC plate and cell culture discs purchased from Sarstedt (Ireland). Corning 12 well TC plates purchased from Fannin (Ireland). Nutrient agar and nutrient broth Oxoid No. 2 purchased from Fannin (Ireland). NADPH reagent “A”, NADPH reagent “B” and S9 fraction purchased from Mol. Tox. Inc. (USA).
2.2. Propagation of MCF-7 Bos and MVLN Cells
MCF-7 Bos cell and MVLN cells were cultivated in DMEM supplemented with sodium bicarbonate and 5% Hyclone foetal bovine serum. The cell lines were maintained in a cell incubator with humidified air and a CO2 concentration of 5%.
2.3. E-SCREEN Assay
Preparation and storage of media, charcoal-dextran stripped serum and assay procedure for the E-SCREEN assay was carried out according to Soto et al., 1995 [20] with the following deviation. The bioassay was terminated on day six by carrying out a sulphorhodamine B (SRB) Protein/biomass estimation assay.
2.4. MVLN Assay
Assay procedure for the MVLN assay was carried out according to Pons et al., 1990 [8] with the following deviations. Cells were seeded at 5 × 105 cells/ml in growth medium for 24 h. Cells were washed and resuspended in experimental medium for 48 h. Test-compounds were added in experimental medium for 24 h. Firefly luminescence was measured immediately.
2.5. Yeast Oestrogen Screen (YES) Assay
The yeast oestrogen screen, previously described by Routledge and Sumpter, 1996 [7] was used to test the environmental samples for oestrogenic activity.
2.6. Ames Standard Plate Incorporation Assay
The procedure of bacterial cultivation, verification of genetic markers and incubation with microsome fraction from rat liver were preformed following standard procedures [16].
2.7. The Micronucleus Assay
Chinese Hamster Ovary (CHO) cells (ECACC, UK) (2 × 104/ml) were seeded onto cell culture discs in 6 well tissue culture plates. Test chemical was added for 24 h. The slides were washed twice with 0.1 M phosphate buffer pH 6.45 and cytochalasin B (3 µg/ml) was added for 12 h. The slides were fixed in ice cold methanol:acetic acid (3:1) containing 0.74% formaldehyde. The fixed cells were washed in 0.1 M phosphate buffer pH 6.45 and stained in acridine orange (12.5 mg/100ml in 0.1 M phosphate buffer pH 6.45) for 1 min. The discs were rinsed in phosphate buffer for 10 min and rinsed in fresh phosphate buffer for 15 min. Cells were examined for micronuclei using fluorescent microscopy. The criteria for identifying micronuclei were performed according to Fenech, 1993 [13].
2.8. UDS Assay
HepG2 cells (5 × 105/ml) were seeded in 12 well cell culture dishes for 24 h in low serum medium (0.5%) for 4 days. 1 ml medium containing 10 mM hydroxyurea was added and incubated for 1 h. Test chemical was added in fresh culture containing 0.005 mCi/ml 3H thymidine in the presence of 10 mM hydroxyurea and incubated for 3 h. The cells were collected onto glass microfibre filter discs using a cell scraper and were washed with 6 ml PBS, 10 ml of 5% TCA and 5 ml of absolute ethanol. The filters were placed in 10 ml of scintillation cocktail and analysed in the liquid scintillation counter for radioactivity due to thymidine uptake in the cells.
2.9. Comet Assay
Assay procedure for the comet assay was carried out according to Singh et al., 1988 [11]. Tail moment [21] was chosen as a measure of DNA damage and was obtained using a computerized image analysis system—Comet assay IV perceptive instruments.
2.10. Sample Collection and Preparation for YES Assay
Influent and effluent samples were collected from inlet and outlet pipes within each STP. River sampling locations were representative of control sites (upstream) and sites receiving effluent (downstream). Samples were taken from the Hind River 30 metres upstream and 70 metres downstream of Roscommon STP. Samples from the Camlin River were collected 2400 metres upstream and 800 metres downstream of Longford STP. Grab sampling was employed. A stainless steel bucket was immersed 0.6 meters below the water level at a midway point across the river, facing upstream. All samples were stored at 4˚C and were prepared for testing within 48 h. Adsorbed compounds were eluted with 5% methanol and filtered through 0.8 μM and 0.45 μM Nalgene filters (AGB, Ireland).
2.11. Statistical Analysis
The EC20 values were calculated using the LevenbergMarquardt fit model (Xlfit2, Microsoft Excel, ID Business Solutions, UK). The EEq of each sample was calculated using the EC20 value of the sample.
Each experiment was tested for normality using the Anderson-Darling test. Differences between the equality of population medians and diverse treatment groups were assessed using the Kruskal-Wallis test and the MannWhitney test. One-way analysis of variance (ANOVA) was used for normally distributed data and the 2-sample t test was used to compute the difference between the means of the diverse treatment groups. A p value of ≤0.05 was regarded as significant.
3. Results
3.1. Oestrogenic Potential of EDCs in the YES Assay
The potency of the EDCs in the YES assay was characterised by the lowest observed effect concentration (LOEC) determined by statistical analysis (Table 1). Statistically significant induction in reporter gene activity was observed after treatment with 1 × 10–7 M DINP, 1.57 × 10–6
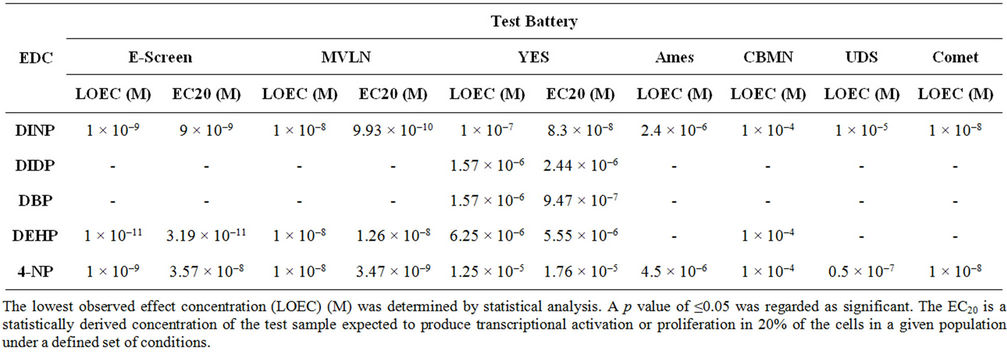
Table 1. Summary table of proliferative, transactivational and genotoxic ability of EDCs.
M DIDP and DBP 6.25 × 10–6 M DEHP and 1.25 × 10–5 M 4-NP (Figure 1, Table 2). A comparison of LOECs showed considerable differences in potency between the EDCs tested (Figure 1).
3.2. Oestrogenic Potential of EDCS in the MVLN and E-Screen Assays
Statistically significant induction in reporter gene activity, in the MVLN assay, was observed after treatment with 1 × 10–8 M DINP, DEHP and 4-NP (Table 1). The maximum amplitudes obtained with DINP, DEHP and 4-NP were 79.9%, 80.1% and 71.5% respectively and were close to the positive control, 10 nM 17β-oestradiol (100%) (Table 2). MCF-7 BOS cells exhibited a good response after treatment with DINP, DEHP and 4-NP inducing statistically significant proliferation (Table 1) at 81.7%, 36.7% and 23.3% (Table 2) respectively. The maximum amplitude obtained with DINP was close to 10 nM 17β- oestradiol (100%) used as a positive control (Table 2). A comparison of the relative proliferative effect (RPE) (Table 2) obtained with DINP, DEHP and 4-NP in the E-SCREEN assay showed considerable differences in potency between the three chemicals.
3.3. Mutagenic and Genotoxic Potential of EDCs in the Ames, Micronucleus, UDS and Comet Assays
DINP induced a significant level of frameshift mutations in TA98 in the presence of an exogenous metabolic system (S9) producing a mutagenic index of 26.55 (Table 2). This indicates that DINP requires bioactivation for mutagenicity to occur. 4-NP induced a significant doserelated response in mutagenic index in TA100 in the absence of S9. Alkylphenol also induced a significant doserelated increase in mutagenic index in TA98 in the presence of S9. A mutagenic index of 34.63 was achieved in the absence of S9 (Table 2). Mutagenicity was observed for 4-NP in TA97a, in the presence and absence of S9 with the level of mutation higher in the presence of S9. 4-NP induced a significant dose-related increase in mutagenicity in TA1535 in the absence of S9.
DINP, DEHP and 4-NP induced statistically significant clastogenicity in the CBMN assay (Table 1). DINP and DEHP induced micronuclei at 19.5% and 18.2% respectively while 4-NP induced micronulei formation at 19.4% (Table 2). Significant induction in the percentage of cells containing UDS was observed after treatment with 1 × 10–5 M DINP and 0.5 × 10–7 M 4-NP indicating that these EDCs are capable of DNA damage (Table 1). Both DINP and 4-NP induced DNA repair at low levels (Table 2). The EDCs differed to some extent with regard to the level of DNA repair induced at a particular dose level.
Statistically significant induction in DNA damage was observed in the comet assay after treatment with 1 × 10–8 M DINP and 1 × 10–8 M 4-NP (Table 1) in the HepG-2 cell line. DINP and 4-NP induced low level DNA strand breakage (Table 2).
3.4. Oestrogenicity of Environmental Samples from the BMW Region of Ireland
Roscommon STP effluent is discharged into the Hind River. The upstream sample (control site) taken in the first year of sampling was more oestrogenic than the downstream sample in the same year (Table 3). The effluent discharged from Roscommon STP in the second year of sampling had EEq value of 9.53 ng/L but dilution of the effluent in receiving waters caused a reduction in this level by approximately 70% (70 metres downstream). Samples from the Hind River both upstream and downstream increased in potency within a 12 month period.
Longford STP plant effluent is discharged into the
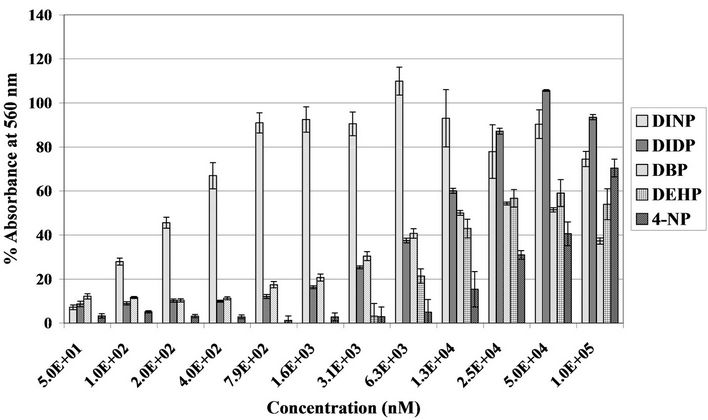
Figure 1. Transcriptional activation induced by EDCs demonstrated by the YES reporter gene assay. Values respresent the mean +/– S.E.M., where n = 4. Graph depicts oestrogenicity induced by diisononylphthalate (DINP), diisododecylphthalate (DIDP), dibutylphthalate (DBP), diethylhexylphthalate (DEHP) and 4-nonylphanol (4-NP) (50 nM to 1.57 × 1 × 105 nM {5.0E + 01 = 50 nM}). Chemical concentrations were plotted against absorbance at 560 nM achieved as a percentage of the positive control, 10 nM 17β-oestradiol. Ethanol was used as the negative control.
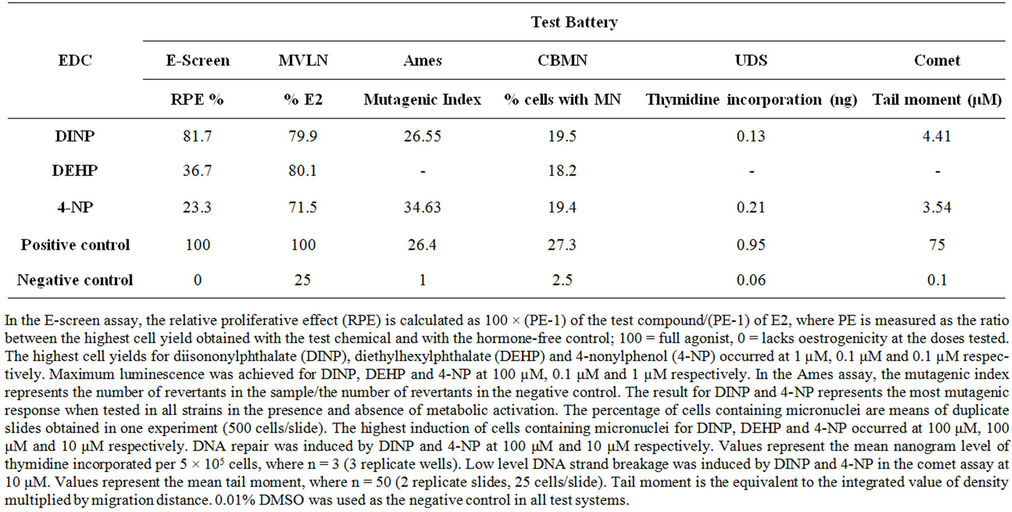
Table 2. The oestrogenic and genotoxic potential of EDCs.
Camlin River. A comparison of the EEq values of the influent and effluent samples procured in the second year of sampling show that the STP is highly efficient in removing oestrogen contamination with approximately 92% reduction in oestrogen load (Table 3). The Camlin River upstream (control, year 2) was potently oestrogenic in the YES assay (EC20 value not determined due to a lack of dose-response). The Camlin River downstream (800 meters) of the STP (year 2) was almost six times more oestrogenic than the effluent discharged.
The EEq values for the influent and effluent samples from Athlone procured in the second year of sampling were of similar potency. A comparison of the influent samples form Athlone STP with those from the other major STPs in the study suggest that Athlone STP does not receive heavy loads of EDCs (91% less than Longford
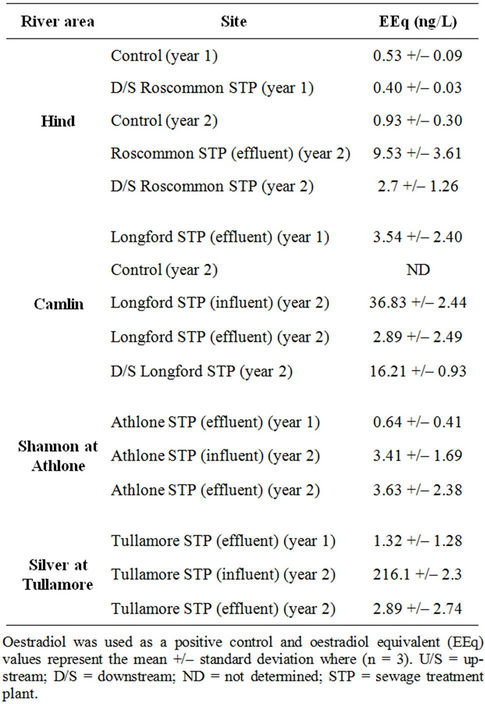
Table 3. Oestrogen levels in environmental samples from the BMW region of Ireland.
STP and 98% less than Tullamore STP).
Tullamore STP is highly efficient in removing oestrogenic contamination with an EEq reduction of approximately 98% (year 2).
3.5. Response of YES Assay to Levels EDCs Found in River Samples in the BMW Region in Ireland
Levels of EDCs (ethinylestradiol, 17β-oestradiol, oestrone, dibutylphthalate, diethylhexylphthalate, diisononylphthalate and diisododecylphthalate) have been found in river samples from the BMW region (Reid et al., 2007; Reid et al., 2008). The chemical combination for each river was denoted as 100% (Figure 2). In order of ascending potency, the Hind River upstream, River Shannon at Athlone/Lanesborough and the Hind River downstream produced LOECs of 12.5%, 0.79% and 0.1% respectively.
4. Discussion
High sensitivity is an essential prerequisite for screening assays to facilitate detection of compounds of low potency that might be of biological significance through chronic exposure and high profusion in the environment. The determination of whether EDCs interact directly with the ER is essential for understanding the risk associated with exposure. Luciferase expression in the MVLN cell line and β-galactosidase expression in the YES assay mimics a natural hormonal response. The ability of a chemical to activate oestrogen-responsive genes through the ER may indicate that the chemical will be oestrogenic in vivo.
The potencies of EDCs in the YES assay when compared to 17β-oestradiol are underestimated due to the fact that circulating endogenous oestrogen is bound to plasma proteins and only a small percentage has the ability to infiltrate cells and activate the ER. It has also been suggested that EDCs may displace endogenous sex steroid hormones from human sex hormone-binding globulin binding sites and disrupt the androgen-to-oestrogen balance and perhaps lead to elevated levels of endogenous hormones [22].
Responses to oestrogen which are too rapid to be mediated by activation of RNA and protein synthesis are thought to be non-genomic. The hallmark of oestrogen activity is its ability to elicit the mitotic stimulation of the tissues of the female genital tract, a non-genomic event, therefore measuring cell proliferation is of key importance in assessing oestrogenicity. The oestrogen induced increase in the number of MCF-7 breast cancer cells (E-SCREEN) is recognised as biologically equivalent to the increase of mitotic activity in the rodent endometrium [10]. The potencies observed in the E-SCREEN assay for the EDCs in this study are in close agreement with those obtained in the MVLN assay and maybe due to the fact that both are mammalian based. However, the potency of some of the chemicals in the E-SCREEN assay was slightly higher than those achieved for the MVLN assay. The E-SCREEN was more sensitive to the oestrogenic potencies of phthalates and alkylphenols than the MVLN assay. This difference is maybe due to the sensitivity of the E-SCREEN assay or to fundamental differences in the complexity of the two natural responses, making the E-Screen a very sensitive assay for screening environmental samples.
Many of the chemicals shown to mimic the action of endogenous oestrogens are reaching freshwater environments and water supplies. Increased cell proliferation, as demonstrated by the E-SCREEN assay, can have a significant input to the process of carcinogenesis. Fixation of genotoxic damage may occur as enhanced cell turnover increases the chance that genotoxic damage will not be repaired resulting in clonal expansion of preneoplastic cells. Therefore human exposure to these EDCs may lead

Figure 2. Response of the yeast oestrogen screen to levels of EDCs found in the river samples in the BMW region of Ireland. Values respresent the mean +/– S.E.M., where n = 4. Graph depicts oestrogenicity induced by the combination of oestrogen mimicking chemicals found in the River Shannon at Athlone and Lanesborough and the Hind River upstream and downstream of Roscommon STP (0.05% to 100%) when compared to 17β-oestradiol, the positive control (5 × 10–3 nM to 10 nM).
to the development of various cancers. The activities of DINP, DEHP and 4-NP in the YES assay were reproduced in the mammalian assays, the MVLN assay and the E-SCREEN assay, implying real oestrogenic effects. Wilkinson and Lamb, 1999 [23] in accordance with Waterman et al., 1999 [24] concluded after a review on toxicological data that DINP in children’s products pose no significant risk to health stating that it was neither teratogenic or a selective developmental toxicant. These findings were also supported by Kavlock et al., 2000 [25] following an extensive review of the potential health effects of DINP to human populations. However, it has been established in this report through the E-SCREEN, MVLN and YES assays that DINP mimics oestrogen action in vitro and may also initiate proliferation thereby enabling the process of carcinogenesis. Moreover, a recent survey by Kelly et al., 2010 [26] reported on levels of phthalates and alkylphenols at various river locations in the Shannon International River Basin District if Ireland. Levels of DINP, DEHP and 4-NP were found at concentrations from 0.52 μg/L (1.2 × 10–8 M) and upwards. The concentrations of DINP found at the various locations were higher than those required to elicit a response in the in vitro E-SCREEN assay reported here. Similarly, the levels of DEHP recovered were higher than the LOEC required to induce a response in the E-SCREEN assay and levels of DEHP recovered at Athlone Lock, Banagher and the Hind River were higher than the LOEC detected in the in vitro MVLN assay. 4-NP was also recovered at all locations [26] and at levels higher than the LOEC needed to induce a proliferative response in the E-SCREEN assay.
Contaminants released into the aquatic environment have the potential to damage the genetic material of exposed organisms resulting in genetic disorders and carcinogenesis. The comet assay provides a useful tool for simultaneous comparison of effects of environmental contaminants in human cell lines. DINP and 4-NP induced DNA strand breaks in this study. The reviews reported by Haighton et al., 2002 [27] and Wilkinson and Lamb, 1999 [23] are not in accordance with results reported here where DINP did induce single strand breakage of the DNA albeit at low levels. Cotelle and Ferard, 1999 [28] have demonstrated that the assay when applied to plants, worms, molluscs, fish, amphibians and mammals provides a sensitive and rapid system for the study of environmental genotoxicity. 4-Nonylphenol has been shown to be positive in the comet assay using human sperm and human peripheral lymphocytes in male and female donors [29].
As most chemical carcinogens in their ultimate reactive form are electrophiles that react with DNA and thereby may result in DNA repair, the measurement of DNA repair is a reliable determination of carcinogenic potential. Martin et al., 1978 [30] have shown a positive correlation between the ability of chemicals to induce non-semiconservative DNA synthesis and their ability to induce cancer in animals. The in vitro UDS was used to assess the cytogenetic potency of the EDCs in HepG2 cells. DINP and 4-NP induced significant levels of DNA repair in the HepG2 cell line. The level of UDS induced by 4-NP was significantly higher than that induced by DINP. This difference is perhaps due to small patch repair by DINP and large patch repair produced by 4-NP. It has also been noted that the amount of repair induced in a given time is sometimes greater with weak carcinogens than with potent ones. Certain carcinogens may have different effects on repair enzyme activity or stimulation of different repair processes depending on the lesion. The relative average amount of UDS elicited by carcinogens in this system is probably more a function of the type of DNA damage and repair provoked than of the potency of the carcinogen.
DINP and 4-NP induced frameshift mutations in one strain or more in the Ames assay. The mutagenicity observed for the EDCs is an indication of possible tumourigenic properties of the EDCs. The presence of micronuclei in somatic cells is recognised as a cytogenetic indicator of genotoxicity and the in vitro micronucleus test with human lymphocytes is used for human monitoring. Furthermore, fish micronuclei have been used to assess genotoxicity in water [31]. The in vitro cytokinesis-block micronucleus technique was used here to assess the cytogenetic damaging potency of DINP, DEHP and 4-NP on the CHO cell line. An increase in the percentage of cells containing micronuclei was observed after exposure to both EDCs indicating that these chemicals are capable of DNA damage.
The biochemical mechanisms underlying the clastogenicity or aneugenic potential of the EDCs reported here are not yet known. Further molecular screening would need to be carried out to classify the contents of the micronuclei and determine clastogenic or aneugenic potential. Fenech and Morley, 1989 [32] have described a method for recognising whole chromosomes and centric fragments within micronuclei in cytokinesis-blocked human lymphocytes using anti-kinetochore antibodies. The results recorded here for the micronuclei forming ability of DINP, DEHP and 4-NP along with the fact that the chemicals induce DNA strand breakage in the comet assay and unscheduled DNA synthesis provides further evidence for the clastogenicity of the environmental oestrogens. Chromosome breakage and aneuploidy is an indication of exposure to genotoxic compounds which may increase the risk of cancer [33].
The comet, UDS, micronucleus and Ames assays can be used as indicators for chromosomal and DNA damaging effects of environmental contaminants. Aberrations in the genome can ultimately lead to the development of cancer therefore exposure to EDCs reported here may have serious implications for reproductive integrity and tumourigenesis. The majority of known carcinogens have initiating and promoting activity and can as a consequence induce neoplasms swiftly and in high yield when administered repetitively. DINP, DEHP and 4-NP also induced proliferation of breast cancer cells in the E-SCREEN assay and therefore have both initiating and proliferative ability.
Many of the chemicals shown to mimic the action of endogenous oestrogens are reaching freshwater environments and water supplies in Ireland [18,19,34], the United Kingdom [35,36], the Netherlands [37], Germany [38,39], the USA [40] and China [41]. Bioaccumulation of these compounds should be taken into account for appropriate risk assessment. EDCs have a much lower affinity for plasma proteins than the endogenous hormone 17β-oestradiol and as a result these chemicals are unbound in the blood and possibly available for oestrogenic activity [42].
It is evident from the data reported here, on the environmental samples from the BMW region of Ireland, that STPs in the Irish Midland region obtain very heavy loads of EDCs. This concurs with analysis carried out in SW Germany [39] where analysis on the effluents of 18 STPs showed that most were found to be positive for a number of EDCs. Efforts should be made to improve the treatment process in Irish STPs so that minimal concentrations of hormone modulating substances are discharged in effluent. Korner et al., 2001 [39] have reported on very low concentrations of EDCs in the effluent of a STP in SW Germany where an activated sludge process is in operation with an additional decolourisation and filtration step using activated charcoal. Activated charcoal filtration appears to be responsible for the reduced concentrations of these compounds. Activated charcoal filtration systems were not in operation in Irish STPs during this study. Moreover, laboratory trials using coconut based granular activated carbon have demonstrated significant removal of the persistent synthetic hormone ethinylestradiol from effluent [43].
The results obtained from the influent, effluent and river samples in the BMW area clearly indicate that they contain oestrogenic compounds. The oestrogenic compounds found in the wastewater samples could be due to EDCs entering these waterways from industrial waste or domestic sewage or natural oestrogens. The amount of effluent entering a river, the size of the receiving water, the time of year and the type of treatment system used all influence the level of oestrogenicity. Jonkers et al., 2009 [44] have reported that higher water flows do not necessarily lead to a proportional dilution of the EDC content. The Camlin River upstream and downstream of Longford STP (year 2) were potently oestrogenic in fact they were more oestrogenic than the effluent being discharged. This suggests that oestrogenic contamination of the river is occurring from unregulated sources other than the STP. The upstream sampling location for the Camlin River is situated in Longford town and the downstream sampling location is situated in an intensely farmed region which may explain the high oestrogenic activity in these two areas. Most studies of oestrogen activity in rivers focus on inputs from STPs, however livestock may contribute to elevated levels of oestrogenicity and this may be the case with the Camlin River at Longford. A recent study in the UK has confirmed that rivers upstream of livestock farms have less hormonal activity than those downstream [45]. The combined livestock population is considerably larger in Ireland than the human population and may contribute a major percentage of environmental load of steroid hormones entering water. It has been suggested that direct excretion of steroid hormones by animals into water courses is likely to be an important source of contamination [46]. It has also been suggested that domestic septic systems may be a significant source of contaminants to groundwater in the USA [40]. It is therefore paramount to research all potential sources of contamination. Contaminant levels in effluent may not be indicative of levels reaching the aquatic environment as some STPs are capable of reducing oestrogenic activity by up to 90% [47].
In humans and mammals steroid hormones undergo biotranformation in the liver. In STPs there is a high population of micro organisms that substantially remove organic material converting ammonia to nitrite and nitrate and ultimately nitrogen gas. Bacteria such as Escherichia coli possess glucuronidase and sulphatase activity and separation of conjugated oestrogens may occur giving rise to elevated environmental concentrations. In the river samples reported in this study it was found that EDCs have been detected in their original unconjugated form. Studies in the Netherlands have also shown that most surface water and effluent samples do not contain hormone glucuronides [37]. One of the first studies to demonstrate the transformation of steroids from oestrogenically inactive to active products by sewage microorganisms involved measurement of plasma vitellogenin in male fathead minnows (Pimephales promelas) [48]. It has been reported, in aerobic batch experiments, that two glucuronides of 17β-oestradiol were cleaved in contact with diluted activated sludge releasing 17β-oestradiol [49]. The similarity between the influent and effluent samples from Athlone STP may be due to this phenomenon of bioactivation within the STP.
In addition to testing pure chemicals in the in vitro assays discussed previously, analysis of levels of EDCs (ethinylestradiol, 17β-oestradiol, oestrone, dibutylphthalate, diethylhexylphthalate, diisononylphthalate and diisododecylphthalate) found in river samples from the BMW region was performed using the YES assay. The chemicals were found at levels capable of inducing transcriptional activation in the YES assay even at high dilutions. This proves that levels of EDCs found in rivers in the Irish midlands are capable of activating the ER and human exposure to these levels may lead to undesirable consequences.
5. Conclusions
EDCs are ubiquitous in nature. There is increasing concern that these compounds could interfere with oestrogen action in humans, resulting in developmental problems, infertility, and cancers of reproductive tissues.
The phthalates, DINP, DEHP and alkylphenol, 4-NP are present in rivers in the BMW region of Ireland. We have established that they are oestrogenic capable of inducing transcriptional activation through the ER using both the YES and MVLN assays and proliferation through non-genomic mechanisms using the E-SCREEN assay, at environmentally relevant concentrations. These chemicals may mimic the endogenous hormone, 17β-oestradiol within the body interfering with endogenous oestrogens. They have the potential to alter the physiological patterns of target tissue function, proliferation and development normally regulated by ovarian oestrogens such as 17β- oestradiol. Exposure to these chemicals may have the same effect as being exposed to excessive amounts of 17β-oestradiol produced by the ovary. A common feature of breast cancer is longer exposure to biologically active oestrogen in a lifetime either due to early menstruation or late menopause. It is therefore possible that prolonged exposure to EDCs may have a similar effect. The mutagenic, clastogenic and genotoxic potential of the phthalates, DINP, DEHP and the alkylphenol 4-NP, established in this report, is cause for concern. These EDCs pose potential problems for wildlife and human populations and may play a role in the etiology of breast and reproductive cancers [50-53].
Human and wildlife exposure to effluent containing EDCs may contribute to the development of carcinogenesis through bioactivation and through proliferation, the hallmark of oestrogen action. Chemical cocktails occur in the environment and dramatically increase the risk factors associated with EDC exposure [34,35,54, 55].
We have established that effluent discharges in the Irish midlands are oestrogenic. It is therefore possible that the effluent discharged from these STPs is oestrogenic to freshwater organisms in Ireland. Kelly et al., 2010 [26] have reported on raised vitellogenin levels in feral male brown trout (Salmo trutta) in the Shannon International River Basin District of Ireland. Human exposure could occur through direct contact with water and through consumption of freshwater fish. Elevated levels of environmental oestrogens in vivo could lead to over expression of oestrogen-regulated genes. Enhanced expression of proto-oncogenes could lead to modifications in growth and differentiation and as a result contribute to neoplasia.
While STPs support the majority of waste entering the waterways in Ireland the burden of EDCs entering these waterways needs to be addressed. Results from Korner et al., 2001 [39] reported substantial removal of EDCs from STPs using activated charcoal filtration. This could also be used to frame legislation in an Irish context.
6. Acknowledgements
This work was funded by the Strand III Core Research Strengths Enhancement Scheme. The MCF-7 cells, for the detection of proliferation, were kindly provided by Dr. A.M. Soto, Tufts University, School of Medicine, Boston, Massacheusetts, U.S.A. The recombinant yeast strain for the detection of oestrogenicity was kindly provided by J.P. Sumpter, Brunel University, UK. The MVLN cell line was kindly provided by Prof. Michael Pons, INSERM, France.
REFERENCES
- S. Jobling, T. Reynolds, R. White, M. G. Parker and J. P. Sumpter, “A variety of Environmentally Persistent Chemicals, Including Some Phthalate Plasticisers Are Weakly Estrogenic,” Environmental Health Perspectives, Vol. 103, No. 6, 1995, pp. 582-587. doi:10.1289/ehp.95103582
- C. E. Purdom, P. A. Hardiman, V. J. Bye, N. C. Eno, C. R. Tyler and J. P. Sumpter, “Estrogenic effects of effluents from Sewage Treatment Works,” Chemistry and Ecology, Vol. 8, No. 4, 1994, pp. 275-285. doi:10.1080/02757549408038554
- K. L. Thorpe, G. Maack, R. Benstead and C. R. Tyler, “Estrogenic Wastewater Treatment Works Effluents Reduce Egg Production in fish,” Environmental Science and Technology, Vol. 43, No. 8, 2009, pp. 2976-2982. doi:10.1021/es803103c
- K. E. Liney, S. Jobling, J. A. Shears, P. Simpson and C. R. Tyler, “Assessing the sensitivity of Different Life Stages for sexual disruption in Roach (Rutilus rutilus) exposed to effluents from Wastewater Treatment Works,” Environmental Health Perspectives, Vol. 113, No. 10, 2005, pp. 1299-1307. doi:10.1289/ehp.7921
- M. Y. Gross-Sorokin, S. D. Roast and G. C. Brighty, “Assessment of feminisation of male fish in English rivers by the Environemnt Agency of England and Wales,” Environmental Health Perspectives, Vol. 114, No. 1, 2006, pp. 147-151. doi:10.1289/ehp.8068
- S. Markman, I. A. Guschina, S. Barnsley, K. L. Buchanan, D. Pascoe and C. T. Muller, “Endocrine Disrupting Chemicals Accumulate in Earthworms Exposed to Sewage Effluent,” Chemosphere, Vol. 70, No. 1, 2007, pp. 119-125. doi:10.1016/j.chemosphere.2007.06.045
- E. J. Routledge and J. P. Sumpter, “Estrogenic activity of surfactants and some of their Degradation Products Assessed Using a Recombinant Yeast Screen,” Environmental Toxicology and Chemistry, Vol. 15, No. 3, 1996, pp. 241-248. doi:10.1002/etc.5620150303
- M. Pons, D. Gagne, J. C. Nicolas and M. Mehtali, “A New Cellular Model of Response to Estrogens: A Bioluminescent Test to Characterise (Anti) Estrogen Molecules,” Biotechniques, Vol. 9, No. 4, 1990, pp. 450-459.
- A. M. Soto, K. L. Chung and C. Sonnenschein, “The Pesticides Endosulfan, Toxaphene, and Dieldrin Have Estrogenic Effects on Human Estrogen-Sensitive Cells,” Environmental Health Perspectives, Vol. 102, No. 4, 1994, pp. 380-383. doi:10.1289/ehp.94102380
- A. M. Soto, T. M. Lin, H. Justicia, R. M. Silvia and C. Sonnenschein, “An ‘in culture” bioassay to assess the estrogenicity of xenobiotics (E-SCREEN),’ In: T. Colburn and C. Clement, eds., Chemically induced Alterations in Sexual and Functional Development: The Wildlife/Human Connection, Princeton Scientific Publishing, Princeton, 1992, pp. 295-309.
- N. P. Singh, M. T. McCoy, R. R. Tice and E. L. Schneider, “A Simple Technique for Quantitation of Low Levels of DNA Damage in Individual Cells,” Experimental Cell Research, Vol. 175, No. 1, 1988, pp. 184-191. doi:10.1016/0014-4827(88)90265-0
- F. Naji-Ali, B. M. Hasspieler, D. Haffner and K. Adeli, “Human bioassays to Assess Environmental Genotoxicity: Development of a DNA Repair Assay in HepG2 cells,” Clinical Biochemistry, Vol. 27, No. 6, 1994, pp. 441-448. doi:10.1016/0009-9120(94)00050-6
- M. Fenech, “The Cytokinesis-Block Micronucleus Technique: A Detailed Description of the method and its application to Genotoxicity Studies in Human Populations,” Mutation research, Vol. 285, No. 1, 1993, pp. 35-44. doi:10.1016/0027-5107(93)90049-L
- B. N. Ames, J. McCann and E. Yamaski, “Methods for Detecting Carcinogens and Mutagens with the Salmonella/Mammalian-Microsome Mutagenicity Test,” Mutation Research, Vol. 31, No. 1, 1975, pp. 347-364.
- B. N. Ames, W. E. Durston, E. Yamasaki and F. D. Lee, “Carcinogens Are Mutagens: A Simple Test System Combining Liver Homogenates for activation and bacteria for detection,” Proceedings of the National Academy of Sciences of USA, Vol. 70, No. 8, 1973, pp. 2281-2285. doi:10.1073/pnas.70.8.2281
- D. M. Maron and B. N. Ames, “Revised methods for the Salmonella mutagenicity test,” Mutation Research, Vol. 113, No. 3-4, 1983, pp. 173-215. doi:10.1016/0165-1161(83)90010-9
- P. Gee, D. M. Maron and B. N. Ames, “Detection and classification of mutagens: A set of Base-Specific Salmonella Tester Strains,” Proceedings of the National Academy of Sciences of USA, Vol. 91, No. 24, 1994, pp. 11606-11610. doi:10.1073/pnas.91.24.11606
- A. M. Reid, C. A. Brougham, A. M. Fogarty and J. J. Roche, “Isocratic LC methods for the Trace Analysis of phthalates and 4-nonylphenol in varying Types of landfill and Adjacent Run-Offs,” Toxicological and Environmental Chemistry, Vol. 89, No. 3, 2007, pp. 399-410. doi:10.1080/02772240601116613
- A. M. Reid, C. A. Brougham, A. M. Fogarty and J. J. Roche, “Accelerated Solvent-Based Extraction and enrichment of Selected Plasticisers and 4-Nonylphenol, and extraction of tin from Organotin Sources in sediments, sludges and Leachate Soils,” Analytica Chimica Acta, Vol. 634, No. 2, 2008, pp. 197-204. doi:10.1016/j.aca.2008.12.032
- A. M. Soto, C. Sonnenschein, K. L. Chung, M. F. Fernandez, N. Olea and F. Serrano, “The E-SCREEN assay as a tool to identify Estrogens: An Update on estrogenic Environmental Pollutants,” Environmental Health Perspectives, Vol. 103, No. 7, 1995, pp. 113-122.
- C. R. Kent, J. J. Eady, G. M. Ross and G. G. Steel, “The Comet Moment as a measure of DNA damage in the Comet Assay,” International Journal of Radiation Biology, Vol. 67, No. 6, 1995, pp. 655-660.
- H. Dechaud, C. Ravard, F. Claustrat, A. B. de la Pierriere and M. Pugeat, “Xenestrogen interaction with human Sex Hormone-Binding Globulin (hSHBG),” Steroids, Vol. 64, No. 5, 1999, pp. 328-334. doi:10.1016/S0039-128X(98)00114-7
- C. F. Wilkinson and J. C. Lamb IV, “The Potential Health Effects of Phthalate Esters in Children’s Toys: A Review and Risk Assessment,” Regulatory Toxicology and Pharmacology, Vol. 30, No. 2, 1999, pp. 140-155. doi:10.1006/rtph.1999.1338
- S. J. Waterman, J. L. Ambroso, L. H. Keller, G. W. Trimmer, A. I. Nikiforov and S. B. Harris, “Developmental toxicity of di-isodecyl and di-isononyl phthalates in rats,” Reproductive Toxicology, Vol. 13, No. 2, 1999, pp. 131-136. doi:10.1016/S0890-6238(99)00002-7
- R. Kavlock, K. Boekelheide, R. Chapin, M. Cunningham, E. Faustman, P. Foster, M. Golub, R. Henderson, I. Hinberg, R. Little, J. Seed, K. Shea, S. Tabacova, R. Tyl, P. Williams, T. Zacharewski, M. Shelby, C. Portier, G. Jahnke, L. Goldman, J. Moore, A. Iannucci and A. Walker, “NTP Centre for the evaluation of risks to human reproduction: Phthalates Expert Panel Report on the reproductive and Developmental Toxicity of Di-Isononyl Phthalate,” Reproductive Toxicology, Vol. 16, No. 5, 2000, pp. 679-708. doi:10.1016/S0890-6238(02)00034-5
- M. A. Kelly, A. M. Reid, K. M. Quinn-Hosey, A. M. Fogarty, J. J. Roche and C. A. Brougham, “Investigation of the Estrogenic Risk to Feral Male Brown Trout (Salmo trutta) in the Shannon International River Basin District of Ireland,” Ecotoxicology and Environemntal Safety, Vol. 73, No. 7, 2010, pp. 1658-1665. doi:10.1016/j.ecoenv.2010.08.018
- L. A. Haighton, J. J. Hlywka, J. Doull, R. Kroes, B. S. Lynch and I. C. Munro, “An evaluation of the possible carcinogenicity of bisphenol A to humans,” Regulatory Toxicology and Pharmamcology, Vol. 35, No. 2, 2002, pp. 238-254. doi:10.1006/rtph.2001.1525
- S. Cotelle and J. F. Ferard, “Comet assay in Genetic Ecotoxicology: A Review,” Environmental and Molecular Mutagenesis, Vol. 34, No. 4, 1999, pp. 246-255. doi:10.1002/(SICI)1098-2280(1999)34:4<246::AID-EM4>3.0.CO;2-V
- D. Anderson, M. M. Dobrzynska and N. Basaran, “Effect of Various Genotoxins and Reproductive Toxins in Human Lymphocytes and Sperm in the Comet Assay,” Teratogenesis, Carcinogenesis, and Mutagenesis, Vol. 17, No. 1, 1997, pp. 29-43. doi:10.1002/(SICI)1520-6866(1997)17:1<29::AID-TCM5>3.0.CO;2-H
- C. N. Martin, A. C. McDermid and R. C. Garner, “Testing of Known Carcinogens and Noncarcinogens for Their Ability to Induce Unscheduled DNA aynthesis in HeLa cells,” Cancer Research, Vol. 38, No. 8, 1978, pp. 2621- 2627.
- K. Al-Sabti and C. D. Metcalfe, “Fish micronuclei for Assessing Genotoxicity in Water,” Mutation Research, Vol. 343, No. 2-3, 1995, pp. 121-135. doi:10.1016/0165-1218(95)90078-0
- M. Fenech and A. A. Morley, “Kinetochore detection in Micronuclei: An Alternative Method for Measuring Chromosome Loss,” Mutagenesis, Vol. 4, No. 2, 1989, pp. 98- 104. doi:10.1093/mutage/4.2.98
- J. D. Tucker and R. J. Preston, “Chromosome Aberrations, Micronuclei, Aneuploidy, Sister Chromatid Exchanges, and Cancer Risk Assessment,” Mutation research, Vol. 365, No. 1-3, 1996, pp. 147-159.
- H. Tarrant, N. Llewellyn, A. Lyons, N. Tattersall, S. Wylde, G. Mouzakitis, M. Maloney and C. McKenzie, “Endocrine disruptors in the Irish Aquatic Environment. Environmental RTDI programme 2000-2006,” Johnstown Castle, Wexford, 2005.
- C. Desbrow, E. J. Routledge, G. C. Brighty, J. P. Sumpter and M. Waldock, “Identification of Estrogenic Chemicals in STW effluent. 1. Chemical fractionation and in vitro Biological Screening,” Environmental Science and Technology, Vol. 32, No. 11, 1998, pp. 1549-1558. doi:10.1021/es9707973
- S. Jobling, R. Williams, A. Johnson, A. Taylor, M. GrossSorokin, M. Nolan, C. R. Tyler, R. van Aerle, E. Santos and G. Brighty, “Predicted exposures to Steroid Estrogens in U.K. Rivers Correlate With Widespread Sexual Disruption in Wild Fish Populations,” Environmental Health Perspectives, Vol. 114, No. 1, 2006, pp. 32-39. doi:10.1289/ehp.8050
- A. C. Belfroid, A. Van der Horst, A. D. Vethaak, A. J. Schafer, G. B. J. Rijs, J. Wegener and W. P. Cofino, “Analysis and occurrence of Estrogenic Hormones and Their Glucuronides in Surface Water and Waste Water in The Netherlands,” The Science of the Total Environment, Vol. 225, No. 1-2, 1999, pp. 101-108. doi:10.1016/S0048-9697(98)00336-2
- P. Spengler, W. Korner and J. W. Metzger, “Substances with Estrogenic Activity in effluents of Sewage Treatment Plants in Southwestern Germany. 1. Chemical analysis,” Environmental Toxicology and Chemistry, Vol. 20, No. 10, 2001, pp. 2133-2141. doi:10.1002/etc.5620201001
- W. Korner, P. Spengler, U. Bolz, W. Schuller, V. Hanf and J. W. Metzger, “Substances with Estrogenic Activity in effluents of Sewage Treatment Plants in Southwestern Germany. 2. Biological analysis,” Environmental Toxicology and Chemistry, Vol. 20, No. 10, 2001, pp. 2142- 2151. doi:10.1002/etc.5620201002
- R. A. Rudel, S. J. Melly, P. W. Geno, G. Sun and J. G. Brody, “Identification of alkylphenols and other phenolic compounds in wastewater, septage, and groundwater on Cape Cod, Massachusetts,” Environmental Science Technology, Vol. 32, No. 7, 1998, pp. 861-869. doi:10.1021/es970723r
- J. H. Shen, B. Gutendorf, H. H. Vahl, L. Shen and J. Westendorf, “Toxicological profile of pollutants in surface water from an area in Taihu Lake, Yangtze Delta,” Toxicology, Vol. 166, No. 1-2, 2001, pp. 71-78. doi:10.1016/S0300-483X(01)00439-5
- J. H. Hodgert, T. R. Zacharewski and G. L. Hammond, “Interactions between human plasma Sex Hormone-Binding Globulin and Xenobiotic Ligands,” The Journal of Steroid Biochemistry and Molecular Biology, Vol. 75, No. 2-3, 2000, pp. 167-176. doi:10.1016/S0960-0760(00)00168-0
- A. O. Ifelebuegu, J. N. Lester, J. Churchley and E. Cartmell, “Removal of an Endocrine Disrupting Chemical (17 Alpha-Ethinylestradiol) from Wastewater Effluent by Activated Carbon Adsorption: Effects of Activated Carbon type and Competitive Adsorption,” Environmental Technology, Vol. 27, No. 12, 2006, pp. 1343-1349. doi:10.1080/09593332708618748
- N. Jonkers, H. P. Kohler, A. Dammshauser and W. Giger, “Mass flows of Endocrine Disruptors in the Glatt River during Varying Weather Conditions,” Environmental Pollution, Vol. 157, No. 3, 2009, pp. 714-723. doi:10.1016/j.envpol.2008.11.029
- P. Matthiessen, D. Arnold, A. C. Johnson, T. J. Pepper, T. G. Pottinger and K. G. Pulman, “Contamination of headwater streams in the United Kingdom by oestrogenic hormones from Livestock Farms,” The Science of the Total Environment, Vol. 367, No. 2-3, 2006, pp. 616-630. doi:10.1016/j.scitotenv.2006.02.007
- A. C. Johnson, R. J. Williams and P. Matthiessen, “The Potential Steroid Hormone Contribution of Farm Animals to freshwaters, the United Kingdom as a Case Study,” The Science of the Total Environment, Vol. 362, No. 1-3, 2006, pp. 166-178. doi:10.1016/j.scitotenv.2005.06.014
- W. Korner, U. Bolz, W. Submuth, G. Hiller, W. Schuller, V. Hanf and H. Hagenmaier, “Input/Output Balance of Estrogenic Active Compounds in a major municipal Sewage Plant in Germany,” Chemosphere, Vol. 40, No. 9-11, 2000, pp. 1131-1142. doi:10.1016/S0045-6535(99)00362-8
- G. H. Panter, R. S. Thompson, N. Beresford and J. P. Sumpter, “Transformation of a Non-Estrogenic Steroid Metabolite to an Estrogenically Active Substance by Minimal Bacterial Activity,” Chemosphere, Vol. 38, No. 15, 1999, pp. 3579-3596. doi:10.1016/S0045-6535(98)00572-4
- T. A. Ternes, P. Kreckel and J. Mueller, “Behaviour and occurrence of estrogens in Municipal Sewage Treatment Plants—II. Aerobic Batch Experiments with Activated Sludge,” The Science of the Total Environment, Vol. 225 No. 1-2, 1999, pp. 91-99. doi:10.1016/S0048-9697(98)00335-0
- D. L. Davis and H. L. Bradlow “Can environmental Estrogens Cause Breast Cancer?” Scientific American, 1995, pp. 144-149.
- T. J. oppari, J. C. Larsen, P. Christiansen, A. Giwercman, P. Grandjean, L. J. Guilette, B. Jegou, T. K. Jensen, P. Jouannet, N. Keiding, H. Leffers, J. A. McLachlan, O. Meyer, J. Muller, E. R.-D. Meyts, T. Scheike, R. Sharpe, J. Sumpter and N. E. Skakkebaek, “Male reproductive health and environmental xenestrogens,” Environmental Health Perspectives, Vol. 104, No. 4, 1996, pp. 741-803.
- R. M. Sharpe and N. E. Skakkebaek, “Are Oestrogens Involved in Falling Sperm Counts and disorders of the Male Reproductive Tract?” The Lancet, Vol. 341, No. 8857, 1993, pp. 1392-1395. doi:10.1016/0140-6736(93)90953-E
- E. Carlsen, A. Giwercman, N. Keiding and N. E. Skakkebaek, “Declining Semen Quality and Increasing Incidence of Testicular Cancer: Is there a Common Cause?” Environmental Health Perspectives, Vol. 103, No. 7, 1995, pp. 137-139.
- S. F. Arnold, D. M. Klotz, B. M. Collins, P. M. Vonier, L. J. Guillette and J. A. McLachlan, “Synergistic activation of Estrogen Receptor with Combinations of Environmental Chemicals,” Science, Vol. 272, No. 5267, 1996, pp. 1489- 1492. doi:10.1126/science.272.5267.1489
- S. M. Dempsey and M. J. Costello, “A Review of Estrogen Mimicking Chemicals in relation to Water Quality in Ireland,” Johnstown Castle, Wexford, 1998.
NOTES
*Corresponding author.

